a6ab4b32a60e117fb0fc1a2ede40c06a.ppt
- Количество слайдов: 34
 YUASA AND HARA LAW, PATENT, TRADEMARK & DESIGN and ACCOUNTING Oct 27 -28, 2009 WIPO Pacific Conference on National IP Strategies for Development Manila Topic 2: Formulation and Development of a National IP Strategy: Needs Assessment VS. Needs Identification, Formulating the Strategy Document: Protection of Cutting-Edge Medical Technology in Japan Shinjiro Ono Yuasa and Hara (Former Deputy Commissioner Japan Patent Office)
YUASA AND HARA LAW, PATENT, TRADEMARK & DESIGN and ACCOUNTING Oct 27 -28, 2009 WIPO Pacific Conference on National IP Strategies for Development Manila Topic 2: Formulation and Development of a National IP Strategy: Needs Assessment VS. Needs Identification, Formulating the Strategy Document: Protection of Cutting-Edge Medical Technology in Japan Shinjiro Ono Yuasa and Hara (Former Deputy Commissioner Japan Patent Office)
 Outline l Phase-1 Oct, 2003 – April, 2005 Task Force on the protection of Medical-Related Activities – IP Policy Headquarters of the Cabinet Office Revision of Examination Guidelines – 2005 l Phase-2 Nov, 2008 – May, 2009 Task Force on the protection of cutting-edge medical technology Final Report – May 29, 2009 Draft Revision of Examination Guidelines – 2009 August 6, 2009 Invitation for Public Comments 1
Outline l Phase-1 Oct, 2003 – April, 2005 Task Force on the protection of Medical-Related Activities – IP Policy Headquarters of the Cabinet Office Revision of Examination Guidelines – 2005 l Phase-2 Nov, 2008 – May, 2009 Task Force on the protection of cutting-edge medical technology Final Report – May 29, 2009 Draft Revision of Examination Guidelines – 2009 August 6, 2009 Invitation for Public Comments 1
 Phase-1 Outline l IP Strategy Formulation in Japan l Related Policies (Needs Assessment) l Historical Background l Tokyo High Court Decision l Cabinet Office Task Force Discussions Major Debates – Conclusion of the Task Force 2005 – l Examination Guidelines – 2005 2
Phase-1 Outline l IP Strategy Formulation in Japan l Related Policies (Needs Assessment) l Historical Background l Tokyo High Court Decision l Cabinet Office Task Force Discussions Major Debates – Conclusion of the Task Force 2005 – l Examination Guidelines – 2005 2
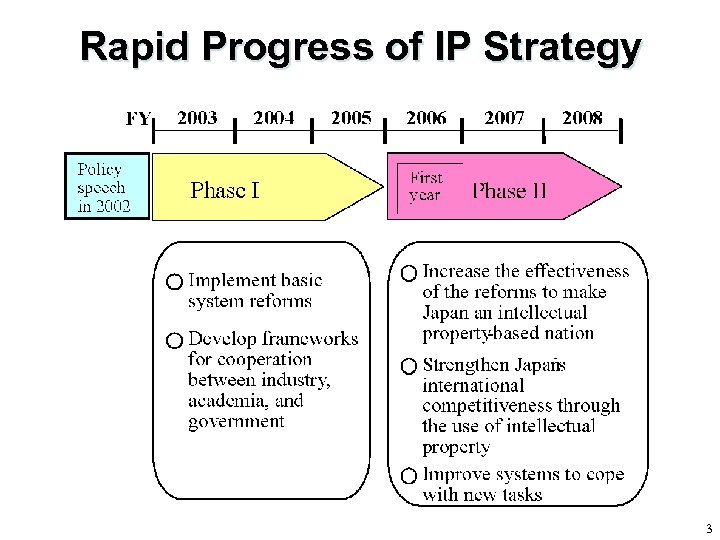 Rapid Progress of IP Strategy 3
Rapid Progress of IP Strategy 3
 Intellectual Property Strategy Headquarters Members --- All Ministers and 10 Experts from Academic and Industrial Sectors Chairman --- Prime Minister KOIZUMI Vice-Chairman --- Chief Cabinet Secretary Minister of State for Science and Technology Policy Minister of Education, Cultures, Sports, Science and Technology Minister of Economy, Trade and Industry 4
Intellectual Property Strategy Headquarters Members --- All Ministers and 10 Experts from Academic and Industrial Sectors Chairman --- Prime Minister KOIZUMI Vice-Chairman --- Chief Cabinet Secretary Minister of State for Science and Technology Policy Minister of Education, Cultures, Sports, Science and Technology Minister of Economy, Trade and Industry 4
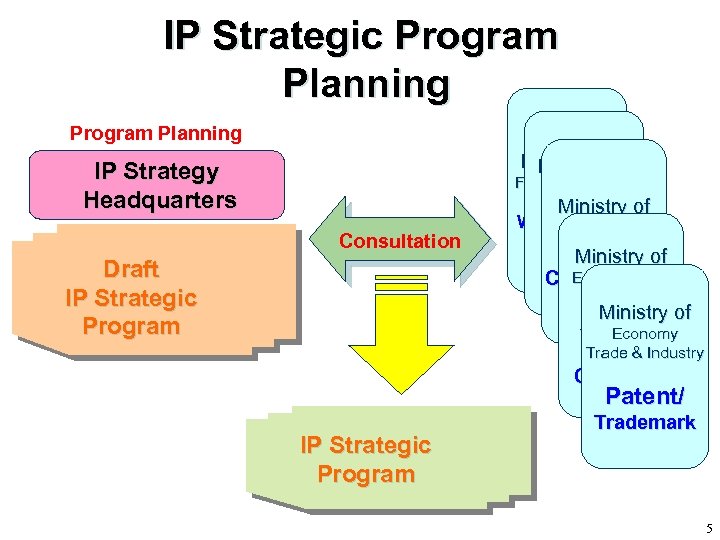 IP Strategic Program Planning IP Strategy Headquarters Consultation Draft IP Strategic Program Ministry ofof Ministry Foreign Affairs Health Ministry of WTO, Finance WIPO Medical, Ministry of DNA… Edu, Culture, Counterfeits Sports, Ministry Science & of Economy Technology Trade & Industry Copyright Patent/ IP Strategic Program Trademark 5
IP Strategic Program Planning IP Strategy Headquarters Consultation Draft IP Strategic Program Ministry ofof Ministry Foreign Affairs Health Ministry of WTO, Finance WIPO Medical, Ministry of DNA… Edu, Culture, Counterfeits Sports, Ministry Science & of Economy Technology Trade & Industry Copyright Patent/ IP Strategic Program Trademark 5
 Patenting Inventions Relating to Medical Activities (Prime minister cabinet office Task Force) IP Strategic Program 2004 Needs identification (1) Studying the Desirable Way of Providing Patent Protection for Medical Treatment i) While giving sufficient consideration so as not to have a bad influence on medical activities that should be equally performed under trustful relationships between patients and medical practitioners, the GOJ will set up a forum for extensively discussing the treatment of medical activities under the Patent Law, from the viewpoint of driving the progress of useful and safe medical technologies that will contribute to raising the level of citizens’ health care, such as allowing patients to receive more advanced medical treatment. The GOJ will draw a conclusion early in FY 2004. (Council for Science and Technology Policy, Ministry of Health, Labor and Welfare, and Ministry of Economy, Trade and Industry) 6
Patenting Inventions Relating to Medical Activities (Prime minister cabinet office Task Force) IP Strategic Program 2004 Needs identification (1) Studying the Desirable Way of Providing Patent Protection for Medical Treatment i) While giving sufficient consideration so as not to have a bad influence on medical activities that should be equally performed under trustful relationships between patients and medical practitioners, the GOJ will set up a forum for extensively discussing the treatment of medical activities under the Patent Law, from the viewpoint of driving the progress of useful and safe medical technologies that will contribute to raising the level of citizens’ health care, such as allowing patients to receive more advanced medical treatment. The GOJ will draw a conclusion early in FY 2004. (Council for Science and Technology Policy, Ministry of Health, Labor and Welfare, and Ministry of Economy, Trade and Industry) 6
 A List of Members of the Task Force 2005 on the Protection of Patents of Medical-Related Activities Member of the Council for Science and Technology Policy President, National Cardiovascular Center Chairman of board of directors, Kyowa Hakko Kogyo Co. ltd. Professor, Graduate School of Medicine, Nagoya University Attorney, Founding Partner Professor, Aomori University, Faculty of Sociology and a commentator Executive member of Japan Medical Association Professor, Chiba University, Faculty of Law and Economics Professor, Hokkaido University, Graduate School of Law Director, An. Ges MG, Inc. and a member of Intellectual Property Headquarters Managing Director, Takeda Pharmaceutical Co. ltd. (Japan Pharmaceutical Manufactures Association) The “Task Force on the Protection of Patents of Medical-Related Activities” was established in Oct. 2003 under the Intellectual Property Policy Headquarters of the Cabinet Office (from Oct. 2003 to Nov. 2004. ) Experts from Ministry of Health, Labor and Welfare as well as Japan Patent Office attended as witness 7
A List of Members of the Task Force 2005 on the Protection of Patents of Medical-Related Activities Member of the Council for Science and Technology Policy President, National Cardiovascular Center Chairman of board of directors, Kyowa Hakko Kogyo Co. ltd. Professor, Graduate School of Medicine, Nagoya University Attorney, Founding Partner Professor, Aomori University, Faculty of Sociology and a commentator Executive member of Japan Medical Association Professor, Chiba University, Faculty of Law and Economics Professor, Hokkaido University, Graduate School of Law Director, An. Ges MG, Inc. and a member of Intellectual Property Headquarters Managing Director, Takeda Pharmaceutical Co. ltd. (Japan Pharmaceutical Manufactures Association) The “Task Force on the Protection of Patents of Medical-Related Activities” was established in Oct. 2003 under the Intellectual Property Policy Headquarters of the Cabinet Office (from Oct. 2003 to Nov. 2004. ) Experts from Ministry of Health, Labor and Welfare as well as Japan Patent Office attended as witness 7
 Related Policies l Forward-Looking Policies (Needs) Patent encourages innovations and investments in R&D – Failure of patent protection discourages Japanese industry from investing money to develop inventions relating to medical activities – l Backward-Looking – Policies Patent is a monopoly preventing access to medical activities 8
Related Policies l Forward-Looking Policies (Needs) Patent encourages innovations and investments in R&D – Failure of patent protection discourages Japanese industry from investing money to develop inventions relating to medical activities – l Backward-Looking – Policies Patent is a monopoly preventing access to medical activities 8
 Historical Background l Patent Act of 1959 (JPL) Art. 32 Excluded from Patentability – – – l 1975 Revision – l (1) foods or non essential grocery items (2) medicines and processes for mixing two or more medicines (3) chemical substances (4) substances to be produced by nuclear transformation and (5) inventions liable to contravene public order, morality or public health. (1) (2) (3) removed from the exclusion 1995 Revision – (4) removed from the exclusion 9
Historical Background l Patent Act of 1959 (JPL) Art. 32 Excluded from Patentability – – – l 1975 Revision – l (1) foods or non essential grocery items (2) medicines and processes for mixing two or more medicines (3) chemical substances (4) substances to be produced by nuclear transformation and (5) inventions liable to contravene public order, morality or public health. (1) (2) (3) removed from the exclusion 1995 Revision – (4) removed from the exclusion 9
 The effects of “Substance patent System” in 1976 on Pharmaceutical Industry 1960 1976. 1 1980 “Substance Patent System” came into effect R&D Expenditure 95. 2 billion yen, 4. 91%/sales in 1975 Number of Novel Drug 6 drugs 1988. 1 2000 “Term extensions for Pharmaceuticals Patent” came into effect R&D Expenditure 746. 2 billion yen, 8. 60%/sales in 2000 Number of Novel Drug 32 drugs 10
The effects of “Substance patent System” in 1976 on Pharmaceutical Industry 1960 1976. 1 1980 “Substance Patent System” came into effect R&D Expenditure 95. 2 billion yen, 4. 91%/sales in 1975 Number of Novel Drug 6 drugs 1988. 1 2000 “Term extensions for Pharmaceuticals Patent” came into effect R&D Expenditure 746. 2 billion yen, 8. 60%/sales in 2000 Number of Novel Drug 32 drugs 10
 Medical Activities l JPL Art. 29: Industrial Applicability Case Law JPO Examination Guidelines – An invention of a method for treating the human body or therapy and diagnostic method is not considered as being taken to be capable of industrial application. EPC Art. 52(4) Industrially inapplicable: EPC 2000 (Dec, 2007); Art. 53(c) Exceptions to Patentability (Up-stream limitation) l US Patentable Subject matter Section. 287(c): to immunize medical practitioners from an infringement suit (Down -stream limitation) l 11
Medical Activities l JPL Art. 29: Industrial Applicability Case Law JPO Examination Guidelines – An invention of a method for treating the human body or therapy and diagnostic method is not considered as being taken to be capable of industrial application. EPC Art. 52(4) Industrially inapplicable: EPC 2000 (Dec, 2007); Art. 53(c) Exceptions to Patentability (Up-stream limitation) l US Patentable Subject matter Section. 287(c): to immunize medical practitioners from an infringement suit (Down -stream limitation) l 11
 Tokyo High Court Decision in the suits against the JPO’s Board decision for rejection Judgment of April 11, 2002 l Case regarding the invention of “Process and device for optical representation of surgical operations” l Tokyo High Court upheld the JPO’s Board decision for rejection of patentability of an invention of medical method for not being capable of industrial application l 12
Tokyo High Court Decision in the suits against the JPO’s Board decision for rejection Judgment of April 11, 2002 l Case regarding the invention of “Process and device for optical representation of surgical operations” l Tokyo High Court upheld the JPO’s Board decision for rejection of patentability of an invention of medical method for not being capable of industrial application l 12
 Tokyo High Court Decision-2 Reasons Distinction between medicines/medical devices and medical treatment l Possible risks for medical practitioners to be subjected to patent infringement liability without any statutory measures to immunize them from suit (The Patent Law does not provide such preventive measures) l No choice but to consider that the Patent Law does not recognize patentability of medical treatment and to deem the inventions involving medical treatment to be industrially inapplicable l 13
Tokyo High Court Decision-2 Reasons Distinction between medicines/medical devices and medical treatment l Possible risks for medical practitioners to be subjected to patent infringement liability without any statutory measures to immunize them from suit (The Patent Law does not provide such preventive measures) l No choice but to consider that the Patent Law does not recognize patentability of medical treatment and to deem the inventions involving medical treatment to be industrially inapplicable l 13
 Cabinet Office Task Force 2005 Discussions Needs assessment l Agenda (Needs Identification) – l Patentability of methods relating to medical activities in general Issues taken into consideration: Redefining the scope of medical methods, reflecting on the progress made in the areas of regenerative medical techniques, gene therapy, diagnostic methods, surgical robots, delivery systems, etc. – Balancing the interests of the health science industry against the public interests (medical practitioners, patients, consumers, etc. ) – 14
Cabinet Office Task Force 2005 Discussions Needs assessment l Agenda (Needs Identification) – l Patentability of methods relating to medical activities in general Issues taken into consideration: Redefining the scope of medical methods, reflecting on the progress made in the areas of regenerative medical techniques, gene therapy, diagnostic methods, surgical robots, delivery systems, etc. – Balancing the interests of the health science industry against the public interests (medical practitioners, patients, consumers, etc. ) – 14
 Major Debates (Needs Assessment) l Argument of the Opposition – An incentive by patent protection is not required for promoting medical technology. - “Medicine is a benevolent art. ” Medical activities are performed for relieving patients from diseases based on mutual trust between a doctor and a patient. – – – l Restriction on access to medical activities Possible increase in medical expenses More frequent litigation Conflict of interest Safety concerns Counter Arguments 15
Major Debates (Needs Assessment) l Argument of the Opposition – An incentive by patent protection is not required for promoting medical technology. - “Medicine is a benevolent art. ” Medical activities are performed for relieving patients from diseases based on mutual trust between a doctor and a patient. – – – l Restriction on access to medical activities Possible increase in medical expenses More frequent litigation Conflict of interest Safety concerns Counter Arguments 15
 Why are process claims of medical activities really needed? What is the difference in the scope of claim relating to invention, between “Medical devices or Medicines” and “Process of medical treatment”? l Can we expect “Indirect Infringement” in case of no “Direct Infringement” by immunization (Down-stream limitation)? l 16
Why are process claims of medical activities really needed? What is the difference in the scope of claim relating to invention, between “Medical devices or Medicines” and “Process of medical treatment”? l Can we expect “Indirect Infringement” in case of no “Direct Infringement” by immunization (Down-stream limitation)? l 16
 Conclusion of the Task Force 2005 (Final Report; Formulating the Strategy Document) l Basic concept: Patent protection of medical activities appears to be premature. – Patent protection, as an incentive to industries, is important, particularly in the fields of medical devices and medicines. – l Consensus: Techniques relating to an action of a medical doctor should be industrially inapplicable inventions (i. e. maintaining of the status quo). – The patent protection of the following inventions should be made clear in the Examination Guidelines: – methods for controlling the operation of a medical device (2) techniques related to a method of generating new effects of medicines (1) 17
Conclusion of the Task Force 2005 (Final Report; Formulating the Strategy Document) l Basic concept: Patent protection of medical activities appears to be premature. – Patent protection, as an incentive to industries, is important, particularly in the fields of medical devices and medicines. – l Consensus: Techniques relating to an action of a medical doctor should be industrially inapplicable inventions (i. e. maintaining of the status quo). – The patent protection of the following inventions should be made clear in the Examination Guidelines: – methods for controlling the operation of a medical device (2) techniques related to a method of generating new effects of medicines (1) 17
 Examination Guidelines 2005 Gist of Examination Guidelines (April 15 th 2005) A method for controlling the operation of a medical device 1. A method for controlling the operation of a medical device is industrially applicable as long as the function of the medical device is represented as a method. 2. The method including an action 2) of a medical doctor and/or an influence 3) on the human body by a device is not considered to be a method for controlling the operation of the medical device. Medicinal Inventions 1. A medicinal invention means “an invention of a product” of use inventions. 4) It is, therefore, considered to be industrially applicable. 2. A medicinal invention having a medicinal use specified by combination of medicines or by dosing interval and/or given dose is also industrially applicable, and its patentability is examined from two viewpoints: a compound (group of compound) and a medicinal use. 1) New Guidelines were made available to the public on April 15 th 2005 2) an action by a medical doctor to operate a device in order to provide medical treatment in accordance with a symptom, etc 3) an incision and/or excision of a specific part of a patient by a device, etc 4) In Japan, “a use invention" is an invention based on a discovery of a new method of using a product focused on a particular property of the product. As for a use invention in a medicinal field, it must be claimed as “an invention of a product. ” 18
Examination Guidelines 2005 Gist of Examination Guidelines (April 15 th 2005) A method for controlling the operation of a medical device 1. A method for controlling the operation of a medical device is industrially applicable as long as the function of the medical device is represented as a method. 2. The method including an action 2) of a medical doctor and/or an influence 3) on the human body by a device is not considered to be a method for controlling the operation of the medical device. Medicinal Inventions 1. A medicinal invention means “an invention of a product” of use inventions. 4) It is, therefore, considered to be industrially applicable. 2. A medicinal invention having a medicinal use specified by combination of medicines or by dosing interval and/or given dose is also industrially applicable, and its patentability is examined from two viewpoints: a compound (group of compound) and a medicinal use. 1) New Guidelines were made available to the public on April 15 th 2005 2) an action by a medical doctor to operate a device in order to provide medical treatment in accordance with a symptom, etc 3) an incision and/or excision of a specific part of a patient by a device, etc 4) In Japan, “a use invention" is an invention based on a discovery of a new method of using a product focused on a particular property of the product. As for a use invention in a medicinal field, it must be claimed as “an invention of a product. ” 18
 Medical Use Claim of substances or compositions in Japan First and second medical use “An invention of a product” of use inventions (product type claim: medicine) l l US: Process Claim except new compounds l EPC: Swiss type claim (Revision of EPC shall permit to protect a new medical use of a compound already known as a medicine without claiming of Swiss type claim); EPC 2000 (Dec, 2007) same as Japan 19
Medical Use Claim of substances or compositions in Japan First and second medical use “An invention of a product” of use inventions (product type claim: medicine) l l US: Process Claim except new compounds l EPC: Swiss type claim (Revision of EPC shall permit to protect a new medical use of a compound already known as a medicine without claiming of Swiss type claim); EPC 2000 (Dec, 2007) same as Japan 19
 Examination Guidelines 2005 Comparison among the Trilateral Offices Method for treatment of human body by surgery -method for anesthesia Method for treatment of human body by therapy -method for gene therapy -method for dialysis treatment -method for controlling the operation of a micro operation robot system -method of stimulating heart by pacemaker -method for controlling the operation of a pacemaker Medicines Medical devices (method for inspection/measurement ) -medical -gene diagnosis devices themselves -endoscopy -method for radiotherapy - method of dosing -method for surgery Diagnostic method -method for NMR inspection, X-ray inspection, or blood-pressure check -method of producing medical devices -chemical substance -medicinal composition for cancer therapy -dosage form *a medicinal invention comprising; (i) a compound, and (ii) a medicinal use -kit -method of producing medicines (getting only interim results for diagnosis) -method for controlling the operation of a MRI device JP -method for controlling the operation of a medical device + *a medicinal invention having a medicinal use specified by; (i) combination of medicines, or (ii) dosing interval and/or given dose JP EP US 20
Examination Guidelines 2005 Comparison among the Trilateral Offices Method for treatment of human body by surgery -method for anesthesia Method for treatment of human body by therapy -method for gene therapy -method for dialysis treatment -method for controlling the operation of a micro operation robot system -method of stimulating heart by pacemaker -method for controlling the operation of a pacemaker Medicines Medical devices (method for inspection/measurement ) -medical -gene diagnosis devices themselves -endoscopy -method for radiotherapy - method of dosing -method for surgery Diagnostic method -method for NMR inspection, X-ray inspection, or blood-pressure check -method of producing medical devices -chemical substance -medicinal composition for cancer therapy -dosage form *a medicinal invention comprising; (i) a compound, and (ii) a medicinal use -kit -method of producing medicines (getting only interim results for diagnosis) -method for controlling the operation of a MRI device JP -method for controlling the operation of a medical device + *a medicinal invention having a medicinal use specified by; (i) combination of medicines, or (ii) dosing interval and/or given dose JP EP US 20
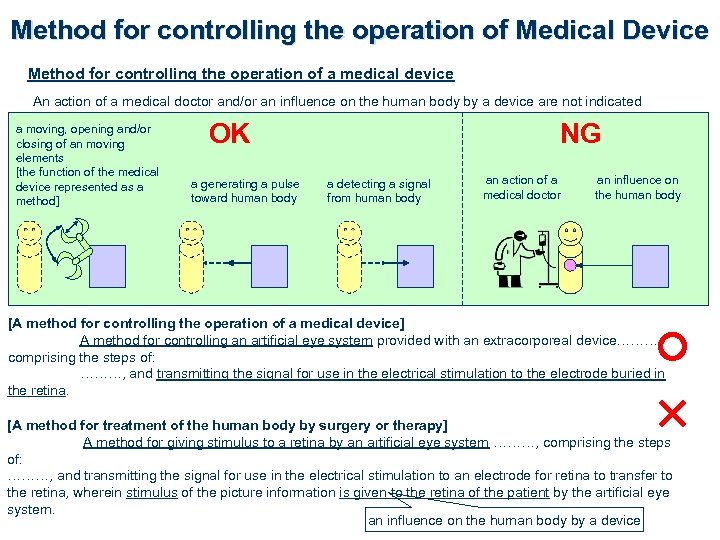 Method for controlling the operation of Medical Device Method for controlling the operation of a medical device An action of a medical doctor and/or an influence on the human body by a device are not indicated a moving, opening and/or closing of an moving elements [the function of the medical device represented as a method] OK a generating a pulse toward human body NG a detecting a signal from human body an action of a medical doctor an influence on the human body [A method for controlling the operation of a medical device] A method for controlling an artificial eye system provided with an extracorporeal device………, comprising the steps of: ………, and transmitting the signal for use in the electrical stimulation to the electrode buried in the retina. [A method for treatment of the human body by surgery or therapy] A method for giving stimulus to a retina by an artificial eye system ………, comprising the steps of: ………, and transmitting the signal for use in the electrical stimulation to an electrode for retina to transfer to the retina, wherein stimulus of the picture information is given to the retina of the patient by the artificial eye system. an influence on the human body by a device
Method for controlling the operation of Medical Device Method for controlling the operation of a medical device An action of a medical doctor and/or an influence on the human body by a device are not indicated a moving, opening and/or closing of an moving elements [the function of the medical device represented as a method] OK a generating a pulse toward human body NG a detecting a signal from human body an action of a medical doctor an influence on the human body [A method for controlling the operation of a medical device] A method for controlling an artificial eye system provided with an extracorporeal device………, comprising the steps of: ………, and transmitting the signal for use in the electrical stimulation to the electrode buried in the retina. [A method for treatment of the human body by surgery or therapy] A method for giving stimulus to a retina by an artificial eye system ………, comprising the steps of: ………, and transmitting the signal for use in the electrical stimulation to an electrode for retina to transfer to the retina, wherein stimulus of the picture information is given to the retina of the patient by the artificial eye system. an influence on the human body by a device
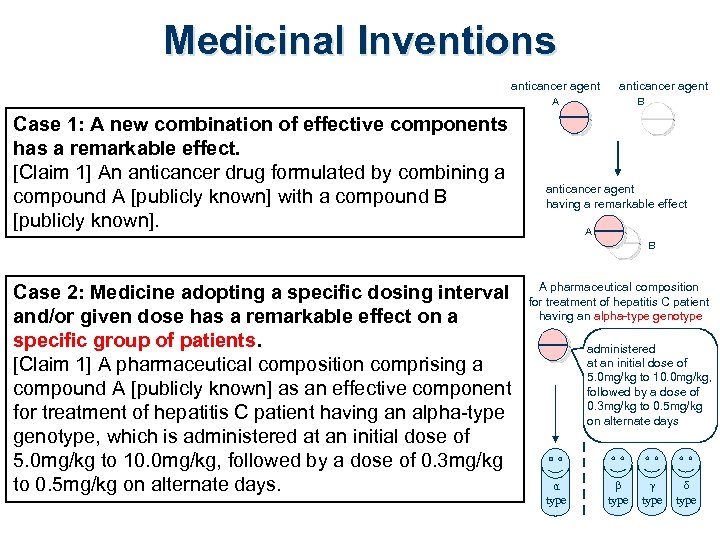 Medicinal Inventions anticancer agent A Case 1: A new combination of effective components has a remarkable effect. [Claim 1] An anticancer drug formulated by combining a compound A [publicly known] with a compound B [publicly known]. B anticancer agent having a remarkable effect A B Case 2: Medicine adopting a specific dosing interval and/or given dose has a remarkable effect on a specific group of patients. [Claim 1] A pharmaceutical composition comprising a compound A [publicly known] as an effective component for treatment of hepatitis C patient having an alpha-type genotype, which is administered at an initial dose of 5. 0 mg/kg to 10. 0 mg/kg, followed by a dose of 0. 3 mg/kg to 0. 5 mg/kg on alternate days. A pharmaceutical composition for treatment of hepatitis C patient having an alpha-type genotype administered at an initial dose of 5. 0 mg/kg to 10. 0 mg/kg, followed by a dose of 0. 3 mg/kg to 0. 5 mg/kg on alternate days α type β type γ type δ type
Medicinal Inventions anticancer agent A Case 1: A new combination of effective components has a remarkable effect. [Claim 1] An anticancer drug formulated by combining a compound A [publicly known] with a compound B [publicly known]. B anticancer agent having a remarkable effect A B Case 2: Medicine adopting a specific dosing interval and/or given dose has a remarkable effect on a specific group of patients. [Claim 1] A pharmaceutical composition comprising a compound A [publicly known] as an effective component for treatment of hepatitis C patient having an alpha-type genotype, which is administered at an initial dose of 5. 0 mg/kg to 10. 0 mg/kg, followed by a dose of 0. 3 mg/kg to 0. 5 mg/kg on alternate days. A pharmaceutical composition for treatment of hepatitis C patient having an alpha-type genotype administered at an initial dose of 5. 0 mg/kg to 10. 0 mg/kg, followed by a dose of 0. 3 mg/kg to 0. 5 mg/kg on alternate days α type β type γ type δ type
 Phase-2 Outline Nov, 2008 – May, 2009 Task Force 2008 on the protection of cutting-edge medical technology – IP Policy Headquarters of the Cabinet Office Final Report – May 29, 2009 Draft Revision of Examination Guidelines – August 6, 2009 23
Phase-2 Outline Nov, 2008 – May, 2009 Task Force 2008 on the protection of cutting-edge medical technology – IP Policy Headquarters of the Cabinet Office Final Report – May 29, 2009 Draft Revision of Examination Guidelines – August 6, 2009 23
 IP Strategic Program 2008 Considering a Desirable Protective Approach in the field of cutting-Edge Medical Technology, such as i. PSC Technology. Needs identification In FY 2008, the GOJ will promptly launch a discussion on a desirable approach to providing appropriate patent protection in the fields of cutting-edge technology, such as i. PSC technology, which is widely applicable to the medical field and for which international competition in R&D and in obtainment of intellectual property have been intensifying. l From this discussion, the GOJ will draw a conclusion at an early date in light of the application of the Examination Guidelines for patents revised in April 2005 and the trend of discussions on the protection of patents for cutting-edge medical technology in the international community. l 24
IP Strategic Program 2008 Considering a Desirable Protective Approach in the field of cutting-Edge Medical Technology, such as i. PSC Technology. Needs identification In FY 2008, the GOJ will promptly launch a discussion on a desirable approach to providing appropriate patent protection in the fields of cutting-edge technology, such as i. PSC technology, which is widely applicable to the medical field and for which international competition in R&D and in obtainment of intellectual property have been intensifying. l From this discussion, the GOJ will draw a conclusion at an early date in light of the application of the Examination Guidelines for patents revised in April 2005 and the trend of discussions on the protection of patents for cutting-edge medical technology in the international community. l 24
 The application of the Examination Guidelines 2005 for Patents revised in 2005 -1; April, 2005 – June, 2008 l A method for controlling the operation of a medical device; number of patents: 76 l Medicine adopting a specific dosing interval and/or given dose has a remarkable effect on a specific group of patients; number of patents: 0 l A new combination of effective components has a remarkable effect; number of patents: 1 25
The application of the Examination Guidelines 2005 for Patents revised in 2005 -1; April, 2005 – June, 2008 l A method for controlling the operation of a medical device; number of patents: 76 l Medicine adopting a specific dosing interval and/or given dose has a remarkable effect on a specific group of patients; number of patents: 0 l A new combination of effective components has a remarkable effect; number of patents: 1 25
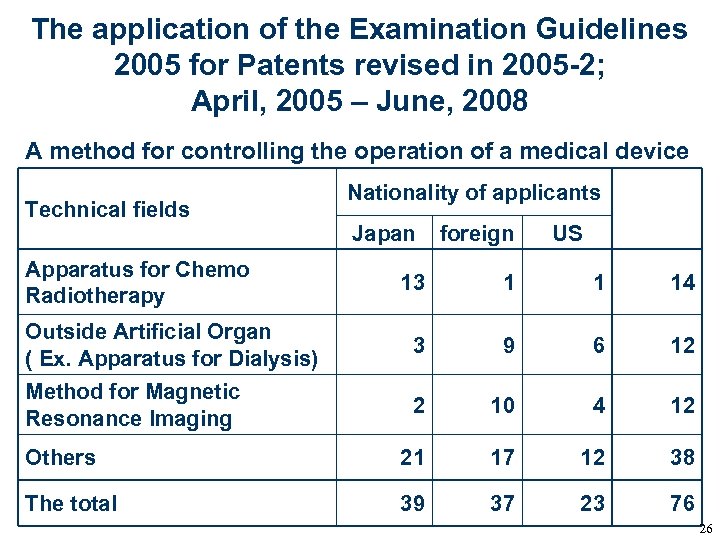 The application of the Examination Guidelines 2005 for Patents revised in 2005 -2; April, 2005 – June, 2008 A method for controlling the operation of a medical device Technical fields Apparatus for Chemo Radiotherapy Nationality of applicants Japan foreign US 13 1 1 14 Outside Artificial Organ ( Ex. Apparatus for Dialysis) 3 9 6 12 Method for Magnetic Resonance Imaging 2 10 4 12 Others 21 17 12 38 The total 39 37 23 76 26
The application of the Examination Guidelines 2005 for Patents revised in 2005 -2; April, 2005 – June, 2008 A method for controlling the operation of a medical device Technical fields Apparatus for Chemo Radiotherapy Nationality of applicants Japan foreign US 13 1 1 14 Outside Artificial Organ ( Ex. Apparatus for Dialysis) 3 9 6 12 Method for Magnetic Resonance Imaging 2 10 4 12 Others 21 17 12 38 The total 39 37 23 76 26
 Proposal of the Report 2009 Products Manufacturi Methods ng a Use Medicine for Surgery or medical invention with a measuring therapy and product by specific structures diagnostic utilizing raw dosing or methods on material interval or functions the human given dose, collected of the from a body having a human remarkable body* being effect Current practice ○ ○ ○ New ○ ○ ○ clarification newly Not appropriate [4] [5] [6] clarificati Newly on on (*) for purposes other than medical ones Example [1] [2] [3] Proposal 27
Proposal of the Report 2009 Products Manufacturi Methods ng a Use Medicine for Surgery or medical invention with a measuring therapy and product by specific structures diagnostic utilizing raw dosing or methods on material interval or functions the human given dose, collected of the from a body having a human remarkable body* being effect Current practice ○ ○ ○ New ○ ○ ○ clarification newly Not appropriate [4] [5] [6] clarificati Newly on on (*) for purposes other than medical ones Example [1] [2] [3] Proposal 27
![[1] (DDS: Drug Delivery System) Thermo sensitive nano-particle anticancer agent Local heating apparatus 28 [1] (DDS: Drug Delivery System) Thermo sensitive nano-particle anticancer agent Local heating apparatus 28](https://present5.com/presentation/a6ab4b32a60e117fb0fc1a2ede40c06a/image-29.jpg) [1] (DDS: Drug Delivery System) Thermo sensitive nano-particle anticancer agent Local heating apparatus 28
[1] (DDS: Drug Delivery System) Thermo sensitive nano-particle anticancer agent Local heating apparatus 28
![[2] Composition comprising cultured mucous membrane cell for generative treatment of a cornea cultured [2] Composition comprising cultured mucous membrane cell for generative treatment of a cornea cultured](https://present5.com/presentation/a6ab4b32a60e117fb0fc1a2ede40c06a/image-30.jpg) [2] Composition comprising cultured mucous membrane cell for generative treatment of a cornea cultured mucous membrane cell Regenerative treatment for a cornea before transplant 1 month after transplant 29
[2] Composition comprising cultured mucous membrane cell for generative treatment of a cornea cultured mucous membrane cell Regenerative treatment for a cornea before transplant 1 month after transplant 29
![[3] Medicine adopting a specific dosing interval and/or given dose, having a remarkable effect [3] Medicine adopting a specific dosing interval and/or given dose, having a remarkable effect](https://present5.com/presentation/a6ab4b32a60e117fb0fc1a2ede40c06a/image-31.jpg) [3] Medicine adopting a specific dosing interval and/or given dose, having a remarkable effect Original dose Sun Every morning → Mon Tue Standing 30 minutes before breakfast to avoid side effect Wed Restrict patient’s life style Thu Fri Sat New dose Once a week Decreases side effect and improves a quality of life 30
[3] Medicine adopting a specific dosing interval and/or given dose, having a remarkable effect Original dose Sun Every morning → Mon Tue Standing 30 minutes before breakfast to avoid side effect Wed Restrict patient’s life style Thu Fri Sat New dose Once a week Decreases side effect and improves a quality of life 30
![[4] Method of Cell Inducing Differentiation and Purification Inducing differentiation Neuron stem cell Hematopoietic [4] Method of Cell Inducing Differentiation and Purification Inducing differentiation Neuron stem cell Hematopoietic](https://present5.com/presentation/a6ab4b32a60e117fb0fc1a2ede40c06a/image-32.jpg) [4] Method of Cell Inducing Differentiation and Purification Inducing differentiation Neuron stem cell Hematopoietic stem cell i. PS cell Mesenchymal stem cell Purification mixture Only neuron stem cell 31
[4] Method of Cell Inducing Differentiation and Purification Inducing differentiation Neuron stem cell Hematopoietic stem cell i. PS cell Mesenchymal stem cell Purification mixture Only neuron stem cell 31
![[5] Method of removing blocked material in a blood vessel (not patentable) * using [5] Method of removing blocked material in a blood vessel (not patentable) * using](https://present5.com/presentation/a6ab4b32a60e117fb0fc1a2ede40c06a/image-33.jpg) [5] Method of removing blocked material in a blood vessel (not patentable) * using a catheter with two cutting blade 32
[5] Method of removing blocked material in a blood vessel (not patentable) * using a catheter with two cutting blade 32
 YUASA AND HARA LAW, PATENT, TRADEMARK & DESIGN and ACCOUNTING Thank you - Thank you for your kind attention - I would be pleased to answer any questions - You may contact me at: ono-ch@yuasa-hara. co. jp URL: http: //www. yuasa-hara. co. jp 33
YUASA AND HARA LAW, PATENT, TRADEMARK & DESIGN and ACCOUNTING Thank you - Thank you for your kind attention - I would be pleased to answer any questions - You may contact me at: ono-ch@yuasa-hara. co. jp URL: http: //www. yuasa-hara. co. jp 33


