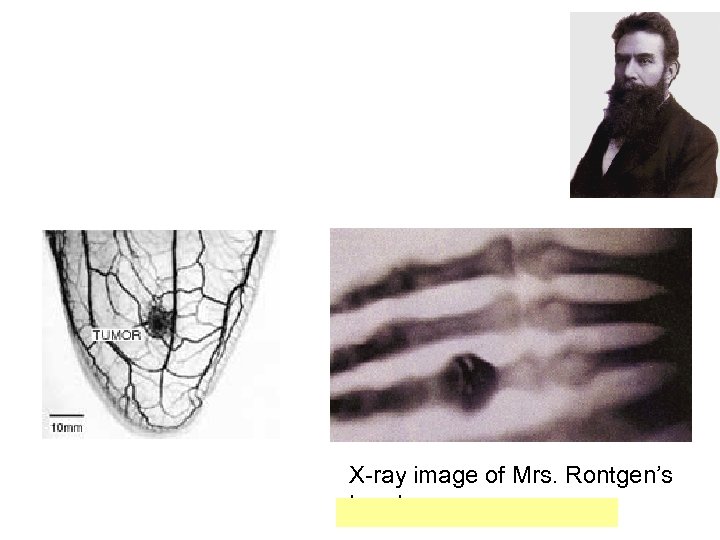
 X-ray image of Mrs. Rontgen’s hand
X-ray image of Mrs. Rontgen’s hand
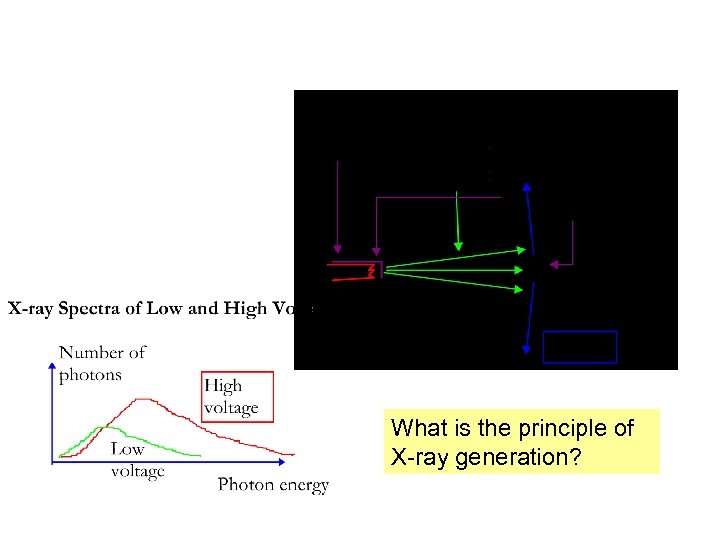 What is the principle of X-ray generation?
What is the principle of X-ray generation?
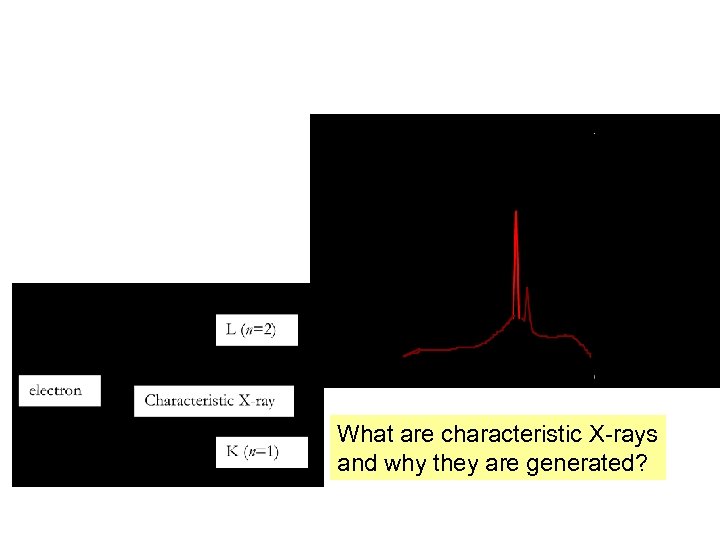 What are characteristic X-rays and why they are generated?
What are characteristic X-rays and why they are generated?
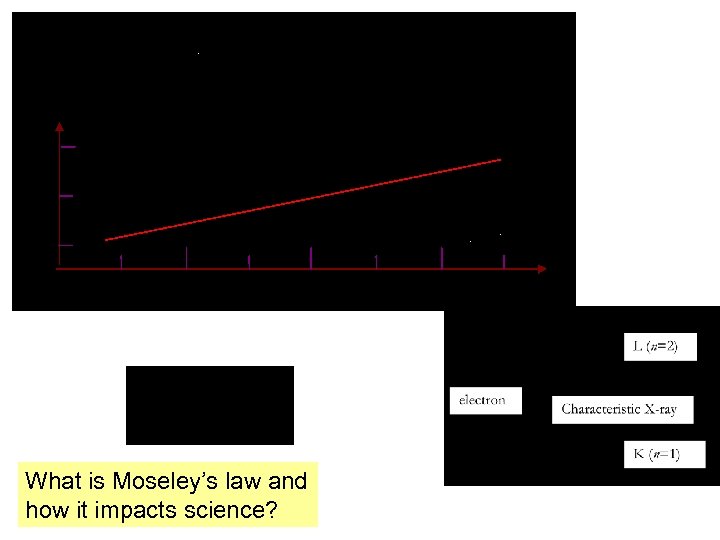 What is Moseley’s law and how it impacts science?
What is Moseley’s law and how it impacts science?
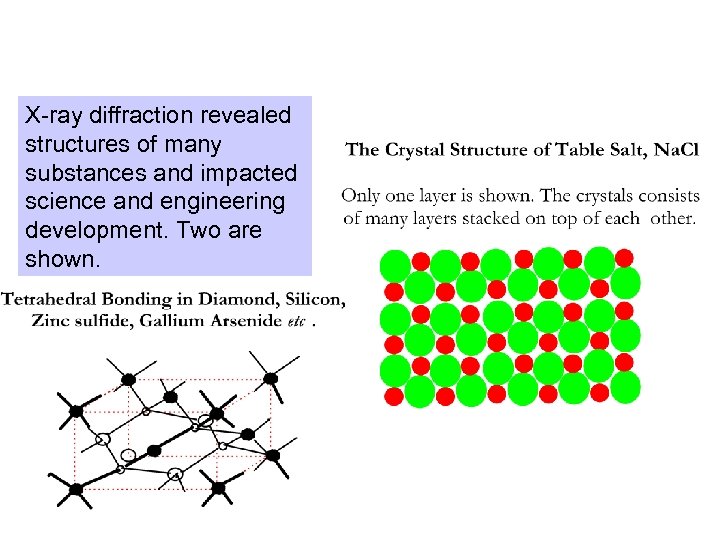 X-ray diffraction revealed structures of many substances and impacted science and engineering development. Two are shown.
X-ray diffraction revealed structures of many substances and impacted science and engineering development. Two are shown.
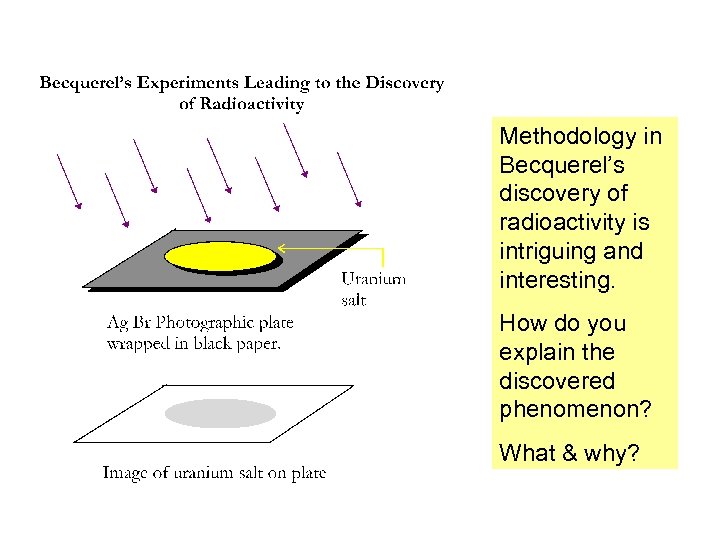 Methodology in Becquerel’s discovery of radioactivity is intriguing and interesting. How do you explain the discovered phenomenon? What & why?
Methodology in Becquerel’s discovery of radioactivity is intriguing and interesting. How do you explain the discovered phenomenon? What & why?
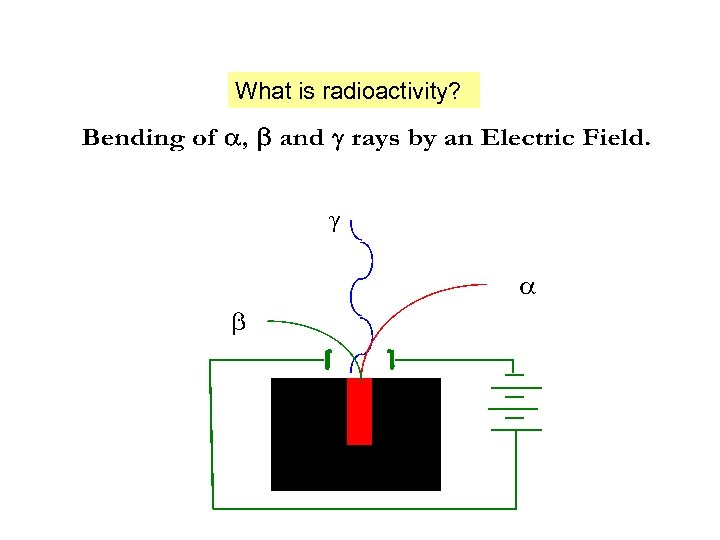 What is radioactivity?
What is radioactivity?
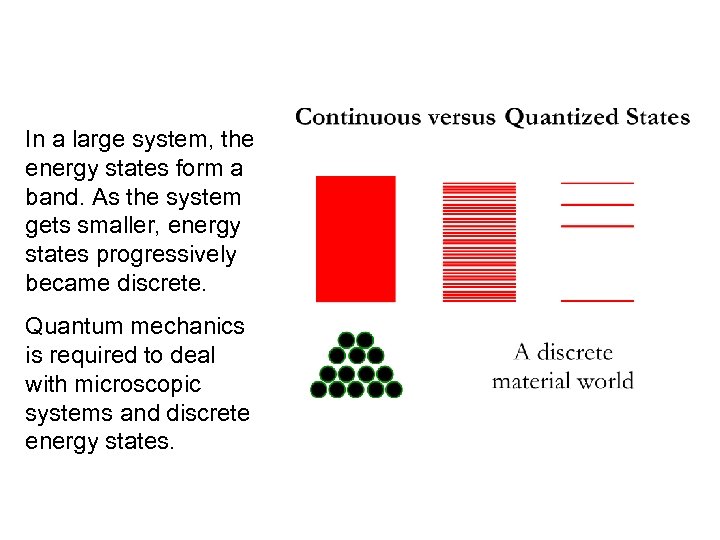 In a large system, the energy states form a band. As the system gets smaller, energy states progressively became discrete. Quantum mechanics is required to deal with microscopic systems and discrete energy states.
In a large system, the energy states form a band. As the system gets smaller, energy states progressively became discrete. Quantum mechanics is required to deal with microscopic systems and discrete energy states.
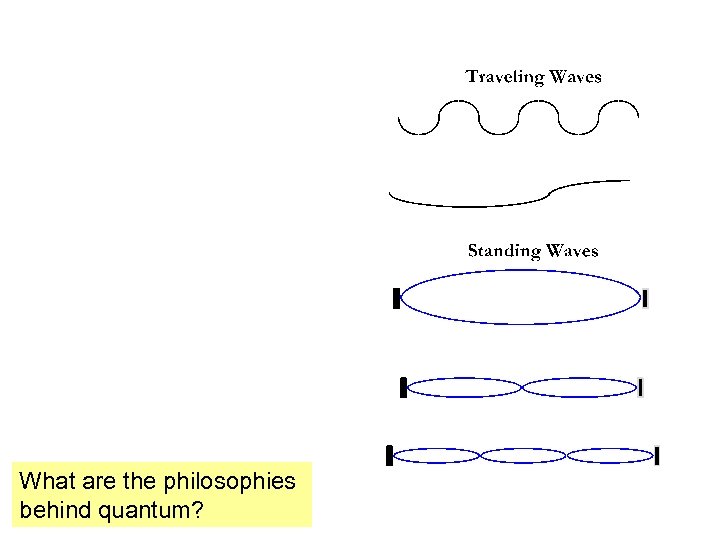 What are the philosophies behind quantum?
What are the philosophies behind quantum?
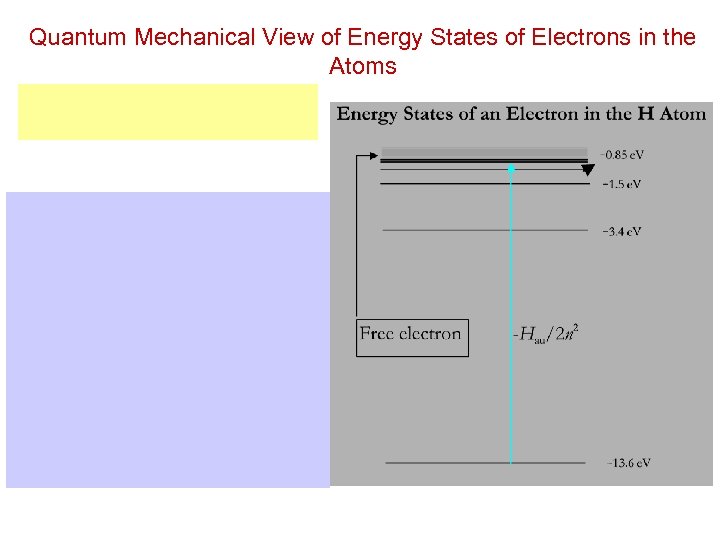 Quantum Mechanical View of Energy States of Electrons in the Atoms
Quantum Mechanical View of Energy States of Electrons in the Atoms
 Filling electrons in energy states gives the electronic configurations of all elements.
Filling electrons in energy states gives the electronic configurations of all elements.
 Some facts about atoms: Radii of atoms ~ 1 e-10 m (Å) most space occupied by electrons Radii of atomic nuclei ~ 1 e-15 m (fm) most mass concentrated in
Some facts about atoms: Radii of atoms ~ 1 e-10 m (Å) most space occupied by electrons Radii of atomic nuclei ~ 1 e-15 m (fm) most mass concentrated in
 1886 Goldstein discovered canal rays 1898 Wien and Thomson identified them as nuclei of H and determined some properties of protons The atomic number used in Moseley’s law turns out to be the number of protons in the nuclei Protons were thought to be fundamental particles What is a proton?
1886 Goldstein discovered canal rays 1898 Wien and Thomson identified them as nuclei of H and determined some properties of protons The atomic number used in Moseley’s law turns out to be the number of protons in the nuclei Protons were thought to be fundamental particles What is a proton?
 Discovery of neutron in atomic nuclei led to the concept of isotopes. Why atomic weights are not all integers? Can atoms of an element have different number of neutrons? What are isotopes? Atomic weight Na Mg Cl Ar K Ca Cu 23 24. 3 35. 5 40. 39 40 63. 5
Discovery of neutron in atomic nuclei led to the concept of isotopes. Why atomic weights are not all integers? Can atoms of an element have different number of neutrons? What are isotopes? Atomic weight Na Mg Cl Ar K Ca Cu 23 24. 3 35. 5 40. 39 40 63. 5
 Does hydrogen have isotopes? How to separate isotopes? How are their properties different?
Does hydrogen have isotopes? How to separate isotopes? How are their properties different?
 Practice the evaluation of atomic weight of an element using the same method as this example shows.
Practice the evaluation of atomic weight of an element using the same method as this example shows.
 Scientific progress often involves establishing conventional wisdom. Other times, it is more a matter of defying it.
Scientific progress often involves establishing conventional wisdom. Other times, it is more a matter of defying it.