e5b60882fc5d12a137823027ffdcd857.ppt
- Количество слайдов: 55

Women’s issues in neurology Dr. Amy Yu November 10 th 2010 Mc. Gill University

Outline Epilepsy n Multiple sclerosis n Headache n

FDA safety rating system n >60% of drugs are risk Category C

FDA safety rating system n A – Adequate, well-controlled human studies have not shown an risk of fetal abnormalities to the fetus in any trimester of pregnancy. n B – Animal studies have revealed no evidence of harm to the fetus, no adequate human studies OR Animal studies have shown an adverse effect, but adequate human studies have failed to demonstrate a risk to the fetus in any trimester. n C – Animal studies have shown an adverse effect without adequate human studies OR Inadequate studies n D – Adequate well-controlled or observational studies in pregnant women have demonstrated a risk to the fetus, but benefits may outweigh the potential risk n X – Adequate well-controlled or observational studies in animals or pregnant women have demonstrated positive evidence of fetal abnormalities or risks. The use of the product is contraindicated in women who are or may become pregnant.

Epilepsy & Pregnancy 24 -year-old woman, diagnosed with a seizure disorder since 13 -year-old, treated with valproic acid and lamotrigine, comes to your office for pregnancy counselling. She currently uses oral contraceptive pills, but wishes to become pregnant. Last seizure was of GTCS type, occurred 18 months ago. She asks: 1. Should I continue taking AED? 2. What are the risks to my baby? 3. What will happen if I have a seizure? n

Women with epilepsy (WWE) ü AAN/AES Practice Parameter 2009 ü What I am not going to talk about ü Catamenial seizure exacerbation ü Menstrual disorders and infertility ü Osteoporosis

Women with epilepsy (WWE) n Over 1 million WWE in the active reproductive years (US data) ¨ 24, 000 n Actual in utero exposure to AED may be two times higher ¨ n births per year H/a, chronic pain, mood disorder Balancing act ¨ Teratogenic risks versus seizure control ¨ For most WWE, withdrawal of AEDs prior to pregnancy is not a realistic option

Pregnancy & seizure control n Physiologic and psychosocial changes ¨ Sex hormones, AED metabolism n clearance, elimination, ∆ free available drug n LTG and OXC d/t glucuronidation ¨ Sleep deprivation, new stressors ¨ Noncompliance to medication n Variable impact on seizures ¨ 20 -33% increase in seizures ¨ 50 -83% no significant change ¨ 7 -25% decrease in seizures

Risks of seizures n GTCS: Hypoxia & acidosis ¨ Fetal ICH, miscarriages, stillbirths ¨ 1 brief GTCS FHR for 20 minutes ¨ SE occurs in <2%, but high mortality rate n Nonconvulsive seizures, effect unclear ¨ CPS during labor caused prolonged uterine contraction with FHR deceleration n Trauma-related complications ¨ ROM, infection, premature labor, abruptio ¨ Enforce seizure precautions (driving, heights)

Teratogenicity by AED Infants of untreated WWE have similar frequency of MCM as general population n Anti-folate effects n Free radicals and oxidative metabolites n ¨ Polytherapy ¨ MCM promotes epoxide production rates n General population 1. 6 - 3. 2% n Monotherapy 2. 3 - 7. 8% n Polytherapy 6. 5 - 18. 8%

AED impact on fetus n Minor anomalies (6 -20%, RR: 2. 5) ¨ Structural deviation w/o threat to health ¨ Distal digital and nail hypoplasia ¨ Midline craniofacial anomaly (broad nasal bridge, ocular hypertelorism, upturned nose, altered lips, etc…) n Major congenital malformation (3. 1 -9%, RR: 2 -3) ¨ Interferes significantly with function and/or requires major intervention ¨ Most common MCM: CHD, cleft lip/palate, urogenital defects, and neural tube defects

AED impact on fetus n Prenatal screening ¨ NTD: maternal serum AFP 15 -22 wks + structural U/S 1620 wks (95% sensitive) n Amniocentesis if above equivocal (99% sensitive) heart detailed U/S 18 -20 wks fetal echocardiography (85% sensitive) ¨ Fetal n Perinatal complications ¨ SGA & 1 -minute Apgar < 7 (RR: ¨ No substantially perinatal death n 2) Neurodevelopment and risk for cognition ¨ Insufficient evidence if children of treated WWE in general are at risk ¨ Monotherapy with VPA/PHT/PB cognition ¨ Polytherapy cognition ¨ CBZ probably does not poor cognitive outcome

Obstetrical complications n Seizure risk during labor & delivery ¨ ¨ ¨ n n 1 -2% GTCS during labor & an additional 1 -2% in 1 st 24 hrs Risk highest in primary generalized epilepsy (lowest in partial) Avoid meperidine as anesthesia choice IV PB, PHT, VA, BZD (neonatal respiratory/cardiac depression) Consider C/S if repeated GTCS, LOC interfering in labor C/S or late pregnancy bleeding (RR<2, probably no substantially risk) Premature contractions, labor, delivery (RR<1. 5, probably no moderately risk) WWE who smoke: possibly substantially risk of premature contractions, labor, delivery Inadequate evidence to determine if the newborns have a substantially risk of hemorrhagic complications

Which poison to give? n n All AED have been associated with features of fetal anticonvulsant syndrome Valproic acid (VPA) ¨ ¨ ¨ n Carbamazepine (CBZ) ¨ n “Probably” contribute to MCM as part of polytherapy “Possibly” with monotherapy Compared to CBZ: “Highly probable” associated with er MCM Compared to PHT or LTG: “Possibly” associated with er MCM Probable relationship between dose and risk of MCM “Probably” does not substantially increase the risk of MCM Insufficient evidence on LTG or other specific AEDs on risk of MCM ¨ Probable relationship between dose of LTG and risk of MCM

Which poison to give? n Notable findings for specific types of MCM: ¨ PHT possibly contributes to the risk of cleft palate. ¨ CBZ possibly contributes to the risk of posterior cleft palate. ¨ VPA probably contributes to neural tube defects and facial clefts & possibly contributes to hypospadias. ¨ PB possibly contributes to cardiac malformations

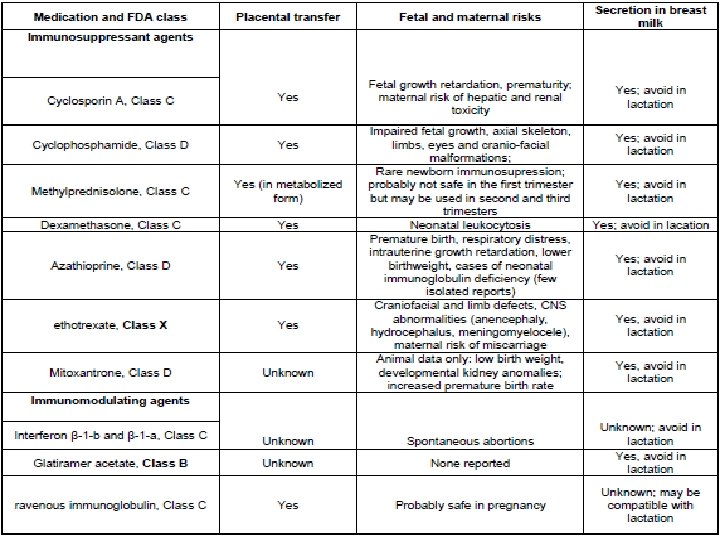
Postpartum care n AED levels (plateau by 10 wks PP) ¨ LTG n may need anticipatory adjustment Breast feeding is supported ¨ PRM & LEV probably transfer into breast milk in amounts that may be clinically important (cf VA, PB, PHT, CBZ are not) ¨ NEAD study: Mental developmental index scores higher in breastfed children at 2 yrs (nonsignificant after maternal IQ adjustment) n Vulnerable period extra precautions ¨ Harness when carrying BB ¨ Stroller in house ¨ Diaper and clothes changes on floor ¨ Never perform bathing alone ¨ Sharing night-time feedings and daytime naps

Epilepsy & Pregnancy 24 -year-old woman, diagnosed with a seizure disorder since 13 -year-old, treated with valproic acid and lamotrigine, comes to your office for pregnancy counselling. She currently uses oral contraceptive pills, but wishes to become pregnant. Last seizure was of GTCS type, occurred 18 months ago. She asks: 1. Should I continue taking AED? 2. What are the risks to my baby? 3. What will happen if I have a seizure? n

Take home message Confirm the presence of seizures n Confirm the need for an antiepileptic drug n Confirm the need to continue an antiepileptic drug n VA has been associated with a higher rate of malformations and lowers verbal IQ n ¨ Consider n transition to LTG or LVT AED monotherapy ¨ Aim to achieve lowest effective dose ¨ Establish baseline “ideal” AED level

Take home message n Presentation after conception ¨ Still aim for monotherapy ¨ Avoid new medication trials & cross-over ¨ teratogenic and seizure risk n Monitoring AED level during & after concentration of LTG, PHT, +/CBZ (to a lesser extent) ¨ Possibly PB, LEV, OXC ¨ Individual variability consider monthly monitoring ¨ Probable

Take home message n Supplemental folic acid 4 -5 mg/d ¨ Prior to conception and during pregnancy ¨ Folate 0. 4 mg/d for all WWE of childbearing age (50% of all pregnancy is unplanned) No strong evidence for Vitamin K supplementation n ? Selenium supplementation 200 mcg/d n

Take home message n 2 -3 times higher MCM ¨ Most infants exposed in utero are healthy ¨ Most MCM can be detected by prenatal ultrasound and some are treatable No substantial increased risk of obstetrical complications (< 2 times expected) n Breastfeeding is safe n Continue AED level monitoring postpartum n Reinforce seizure precautions n

Birth control and AED n Inducers of hepatic cytochrome P-450 patch and vaginal ring also failure rate ¨ Medroxyprogesterone IM q 8 -10 wks (usually q 12 wks) ¨ *** OCP significantly in lamotrigine levels ¨ Transdermal n 1998 AAN guideline ¨ High dose estradiol 50 mcg (no supportive studies) ¨ Backup barrier method, IUD (Mirena has local effect)

Multiple sclerosis & Pregnancy 17 -year-old girl, new diagnosis MS after 2 optic neuritis within 1 year and moderate burden of disease on MRI. You discuss with her interferon therapy. n 1. Will I ever become pregnant? n 2. Will my children have MS? n 3. How will having kids affect my disease? n

Multiple sclerosis n #1 neurologic disease affecting people in their productive years of young adulthood ¨ High incidence in ♀ in childbearing age T-cell mediated autoimmune disease n Lifetime risk 0. 1% n

Genetics and MS n 15% of MS pt have ≥ 1 family mb with MS ¨ Close or distant ¨ Co-occurrence of disease in the family is most likely due to genetic factors n n Higher concordance in mono vs. dizygotic twins 1 parent with MS: 4% risk in child ¨ 40% increase ¨ 2 parents with MS: 20% risk in child n No current specific gene tests available

Pregnancy in MS n Pregnancy and MS (PRIMS), Brain 2004 ¨ 254 women (269 pregnancies, 2 years f/u) ¨ 72% no relapse for the entire study period ¨ Relapse rates (relapses/year): n n Pre-pregnancy 0. 7 3 rd trimester 0. 2 1 st 3 months postpartum 1. 2 Unchanged annualized RR in 21 mths postpartum ¨ Postpartum n n n relapse associated with: Relapse rate in year preceding pregnancy Relapse during pregnancy EDSS at the beginning of pregnancy ¨ Disability at 2 years not related to pregnancy

Pregnancy in MS n Conversion to progressive disease ¨ Risk is 3. 2 times higher in non-pregnant ♀ ¨ Rate of progression in disability is most rapid in nulliparous women ¨ Cannot R/O bias that women with more severe disease have less children ¨ Some studies show no association

Obstetrical outcomes in MS No CI to C/S or vaginal delivery n Variable reports with regards to n ¨ Risk of malformations, fetal BW, duration of pregnancy n Neurology 2009 ¨ 2003 -2005 study time ¨ 10, 000 MS obstetric hospitalization ¨ 30% risk for C/S, 70% rate of IUGR ¨ Adjusted for maternal race and age ¨ Similar rate of HTN disorder and PROM

Take home message n Most patients undergo pregnancy without relapses ¨ Relapse rates decrease in 3 rd trimester ¨ Higher in the 1 st 3 months postpartum n Pregnancy not been shown to be harmful ¨ n Not associated with relapses or disability Effects of MS on pregnancy outcomes do not appear to adversely affect the child’s health or directly influence the mother’s health.

Management of MS in pregnancy D/C immunomodulating therapy 1 -4 weeks prior to attempts to conceive n Stable disease (no attacks, no new MRI lesion, no disability progression for 1 year) n ¨ Conception attempts for 6 months off meds ¨ MRI/clinical review of disease activity n If inactive continue for another 6 months n If active, consider during the next 6 months: ¨ Return to regular therapy ¨ Solumedrol 1 gm IV q 1 mth, immediately after each menstrual cycle if pregnancy test negative

Management of MS in pregnancy n Active disease w/in preceding year despite therapy with good compliance ¨ Discuss changing platform therapy for 3 -6 months prior to conception attempts ¨ If not, consider monthly Solumedrol regimen No immuno-modulating/-suppressive therapy should be used in pregnancy n IV steroids can be used in major exacerbations n ¨ Consult obstetrician ¨ More acceptable after 1 st trimester

Postpartum management Follow-up 2 -3 months after delivery n MRI within that time frame n Stable disease prior and during pregnancy n ¨ Breastfeeding n off MS medications is possible Active/aggressive disease ¨ Return to MS treatments immediately ¨ If breastfeeding: consider steroids monthly ¨ Resume breastfeeding 24 hrs after steroids infusion

Management in NMO n IVIG monthly is safe in pregnancy ¨ Consider in active NMO to stabilize disease ¨ Can be continued until 12 weeks postpartum n Shown to lower postpartum relapse

A few more things to consider… Discuss reproductive wishes in choosing choices of therapy n Chemotherapy treatment n ¨ Pregnancy test prior to each treatment ¨ Counsel to use 2 methods of birth control

Question 3: Headaches n n n 34 -year-old woman, smoker, known migraines with aura, presents at 34 weeks GA. Since the onset of pregnancy, she had been free of headache until 2 days ago when she developed severe headaches much like her usual migraines, without preceding aura. Due to high levels of stress at work, she had been unable to quit smoking. Examination is normal. 1. Is improvement in migraines expected in pregnancy? 2. What is your differential diagnosis in this case? 3. Is it safe for this patient to undergo CT/MRI? 4. What are the treatment options during pregnancy?

Headache & Pregnancy n Headache is #1 reason for neurology referral ¨ Migraines affects 1/5 women in the reproductive years Menstrual migraine: h/a -2 to +3 days after the onset of menstrual flow (day 1) n Menstrually related migraine: some headache menstrually related but headache present at other times of the month n
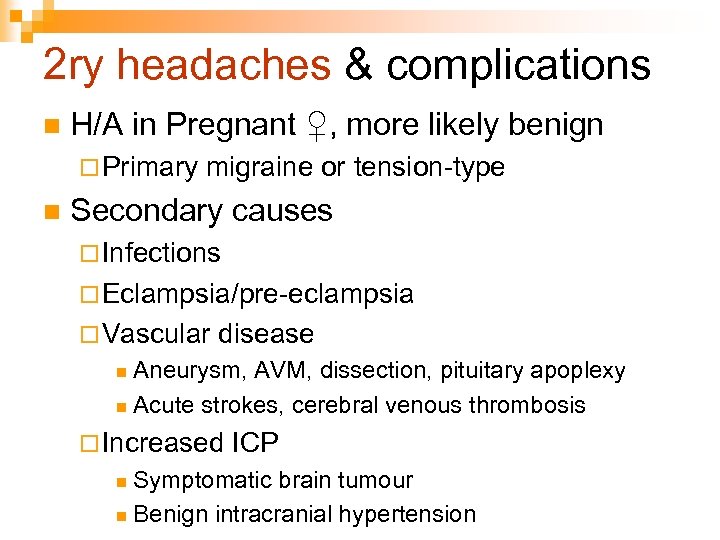
2 ry headaches & complications n H/A in Pregnant ♀, more likely benign ¨ Primary n migraine or tension-type Secondary causes ¨ Infections ¨ Eclampsia/pre-eclampsia ¨ Vascular disease Aneurysm, AVM, dissection, pituitary apoplexy n Acute strokes, cerebral venous thrombosis n ¨ Increased ICP Symptomatic brain tumour n Benign intracranial hypertension n

Headache changes in pregnancy n 50% of migraineurs will improve during pregnancy (up to 80%) ¨ Estrogen increases pain threshold ¨ More common in migraine without aura and menstrual migraines ¨ If ongoing h/a by the end of 1 st trimester unlikely to have significant improvement later ¨ Return 2 -4 weeks postpartum

Migraines & complications n n n Pregnancy-related HTN (OR=2. 85) LBW infant (OR=1. 97) Migraine with aura ¨ ischemic stroke (OR=16. 9) n NOT associated with malformations

Postpartum period & headache n 39% of ♀ dvlp h/a postpartum (often benign) ¨ Study n followed 985 women over 3 months PP 2/3 postpartum headache is caused by migraine or tension-type ¨ MSK/cervicogenic, n spinal low pressure Beware of 2 ry causes in severe h/a occurring >24 hrs postpartum ¨ Eclampsia (late-onset) n 4 x more likely to be associated with headache ¨ Pituitary mass/hemorrhage (3%) ¨ Cerebral venous thrombosis (3%) ¨ Cerebral vasculopathy (2%) ¨ Thalamic lesion (1%) ¨ Subarachnoid hemorrhage (1%)
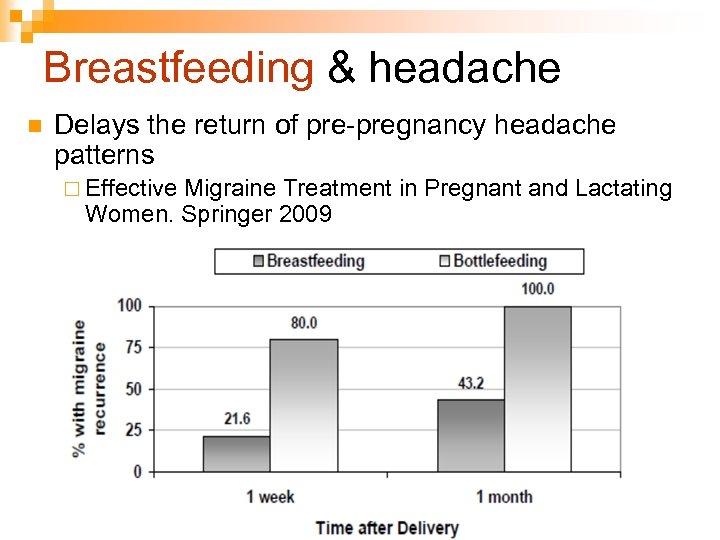
Breastfeeding & headache n Delays the return of pre-pregnancy headache patterns ¨ Effective Migraine Treatment in Pregnant and Lactating Women. Springer 2009

H/A evaluation in pregnancy History & examination as usual n CSF examination is safe and interpretation is same as in non-pregnant (including OP) n MRI preferred (non-emergent/-traumatic) n ¨ Retrospective review, Am J Neuroradiol 2007 ¨ 73% normal or incidental findings ¨ Sinusitis 8%, CVST 6%, PRES/eclampsia 6%, IIH 3%, intracranial hemorrhage 3% ¨ 19% of abnormal scan had initial normal neuro exam
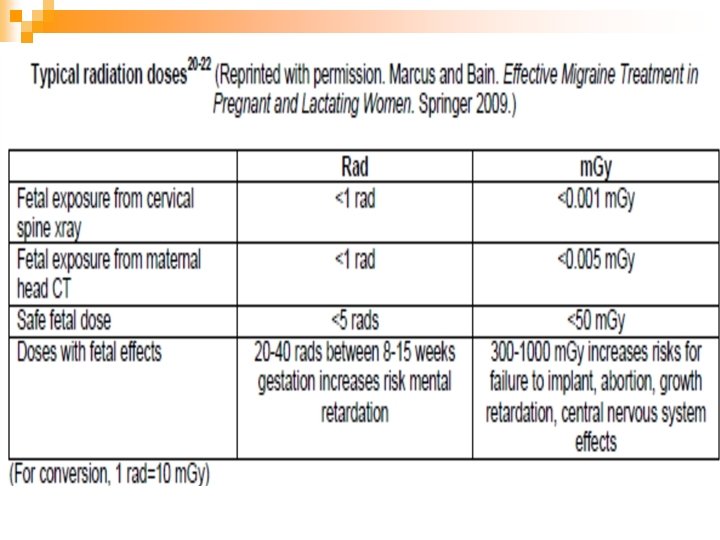

A few notes on contrast studies 11 th European Symposium on Urogenital Radiology n Contrast agents can be used when deemed necessary n ¨ Iodinated contrast agents: no animal fetal risk ¨ Can depress fetal thyroid screen for T 4 ¨ Gadolinium: potential animal fetal toxicity ¨ Risk vs “overwhelming” benefit n Safe to continue nursing after exposure

MRI & neonatal hearing loss n Neonatal Cochlear Function: Measurement after Exposure to Acoustic Noise during in Utero MR Imaging (Radiology. 2010 Sep 27) ¨ 96 neonates, 1% prevalence of hearing impairment ¨ Fetal exposure to 1. 5 -T MRI during the 2 nd & 3 rd trimesters of pregnancy is not associated with an increased risk of substantial neonatal hearing impairment

Treatments n Although most women will have a desire to avoid medications, most will continue to use it ¨ Norwegian study ¨ No change in medication use in the 6 months prior to pregnancy vs. the first 5 months of pregnancy

Non-pharmacological Pain management techniques n Exercise n Smoking cessation n ¨ Nicotine n linked to increase h/a activity Sleep management ¨ Evaluate n for obstructive sleep apnea Lifestyle regulation

Pharmacological – Acute Do not medicate mild h/a n Treat nausea and avoid dehydration n ¨ Mild: dietary restrictions, P 6 acupressure point, Vit B 6 30 mg QD +/- ginger 1 gm QD ¨ Mod-Severe: Ondansetron & metoclopramide
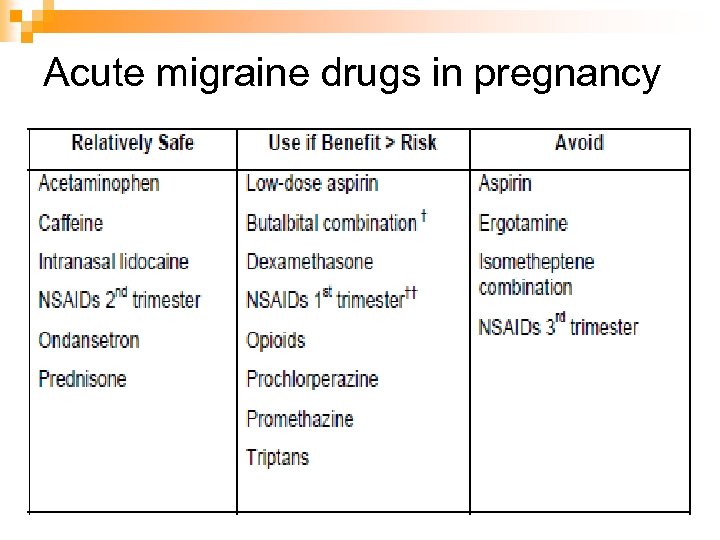
Acute migraine drugs in pregnancy

What about triptans? n Swedish database (n=658) not statistically significant preterm and LBW babies ¨ No change in congenital malformations ¨ Small, n Sumatriptan registry (n=558) ¨ MCM 4. 7%, occurring with 1 st trimester exposure

Acute migraine drugs in lactation

Prophylactic pharmacotherapy n Recommended during pregnancy ¨ Magnesium oxide or Riboflavin (400 mg QD) ¨ Propranolol ¨ Gabapentin in 1 st and 2 nd trimester n Use in T 3 may interfere with bony growth & dvlpmt n Avoid ¨ Atenolol, n Divalproex, Paroxetine Compatible with nursing ¨ Divalproex (with adequate contraception) ¨ Magnesium ¨ Propranolol, Timolol, Verapamil

ED treatment

Take home message n Think about your differential ¨ 50% of migraine patients improve during pregnancy ¨ They are at risk to certain conditions n n If needed, neuroimaging can be safely done Promote non-pharmacological lifestyle modifications Use medications safely: choose wisely, lowest dose, shortest duration, use as late in pregnancy as possible If breastfeeding, use drug after nursing or pump & discard milk after dosing
e5b60882fc5d12a137823027ffdcd857.ppt