7297ee154035d48ada6edb582ab2af18.ppt
- Количество слайдов: 17
Whole Blood Thrombin Generation Monitored with a Calibrated Automated Thrombogram Based Assay M. Ninivaggi, R. Apitz-Castro, Y. Dargaud, B. de Laat, H. C. Hemker, and T. Lindhout August 2012 www. clinchem. org/cgi/content/article/58/8/1252. full © Copyright 2012 by the American Association for Clinical Chemistry
Introduction Ø A balanced interaction between vasculature, blood cells and plasma proteins prevents bleeding on the one hand thrombosis on the other Hemostasis: no bleeding no thrombosis © Copyright 2009 by the American Association for Clinical Chemistry
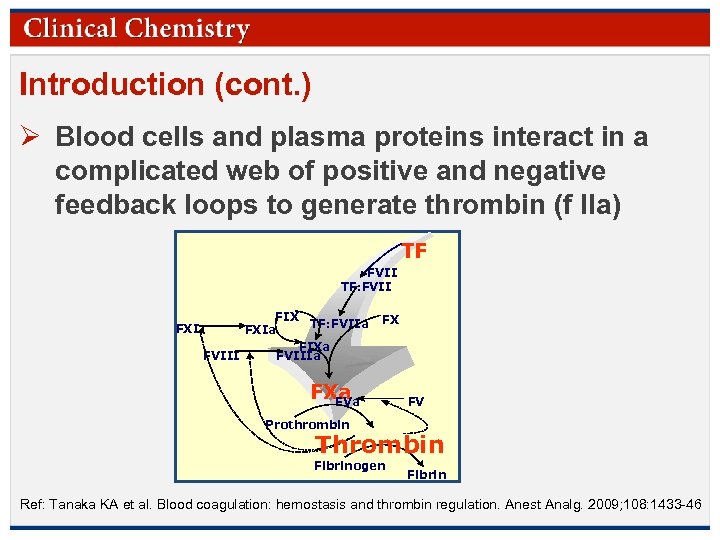
Introduction (cont. ) Ø Blood cells and plasma proteins interact in a complicated web of positive and negative feedback loops to generate thrombin (f IIa) TF FVII TF: FVII FXI FIX TF: FVIIa FXIa FIXa FVIIIa FX FXa FV Prothrombin Thrombin Fibrinogen Fibrin Ref: Tanaka KA et al. Blood coagulation: hemostasis and thrombin regulation. Anest Analg. 2009; 108: 1433 -46 © Copyright 2009 by the American Association for Clinical Chemistry

Introduction (cont. ) Ø Thrombin is a multifunctional protease > Plays a crucial role in hemostasis: the more thrombin the less bleeding but the more thrombosis; the less thrombin the more bleeding but the less thrombosis > Also has other important “non-hemostatic” functions Ø Calibrated Automated Thrombogram (CAT) assay > Allows quantitative assessment of thrombin formation in plateletrich and platelet poor plasma > Can distinguish between normal, hypo-, and hypercoagulable states Thrombosis thrombin Normal Bleeding time © Copyright 2009 by the American Association for Clinical Chemistry Ref: Tripodi A. The long-awaited whole-blood thrombin generation test. Clin Chem 2012; 58: xxx-xxx.
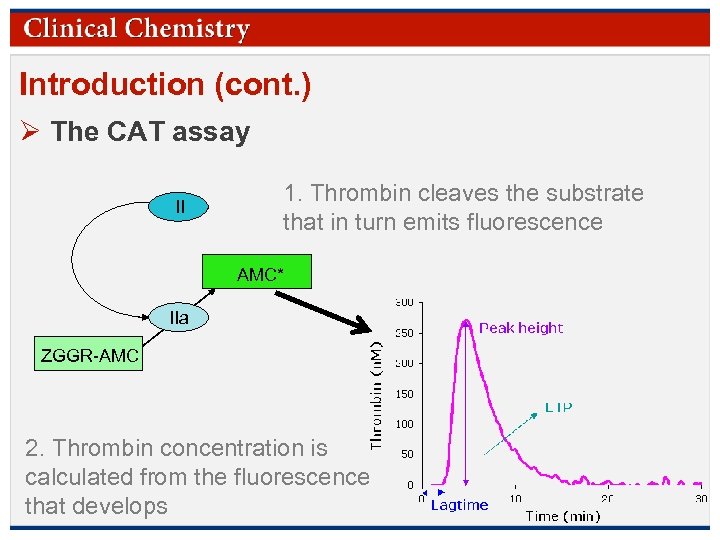
Introduction (cont. ) Ø The CAT assay II 1. Thrombin cleaves the substrate that in turn emits fluorescence AMC* IIa ZGGR-AMC 2. Thrombin concentration is calculated from the fluorescence that develops © Copyright 2009 by the American Association for Clinical Chemistry

Question Ø What makes the CAT assay superior to conventional tests such as the PT and a. PTT? © Copyright 2009 by the American Association for Clinical Chemistry

Introduction (cont. ) Ø Why perform thrombin generation (TG) assays in whole blood? > Closer to physiology > Less laboratory manipulation (no centrifugation, thereby reducing experimental errors and time needed to perform the assay) > Possibility of direct measurement from non-anticoagulated blood (fingerprick) Ø Impediments to performing the CAT assay with whole blood > The fluorescent signal of the cleaved substrate is quenched by hemoglobin > The red blood cells sediment, cluster, and retract with the formed clot, leading to highly erratic signals © Copyright 2009 by the American Association for Clinical Chemistry

Introduction (cont. ) Ø Whole blood (WB) CAT-based assay To overcome the problems of performing TG assays in WB, the following adaptations were made: - The use of a rhodamine-based thrombin substrate for which the excitation and emission wavelength are less affected by hemoglobin than an AMC-based substrate - The use of a porous filter paper to create a thin layer of blood, resulting in the entrapment of the red blood cells © Copyright 2009 by the American Association for Clinical Chemistry

Materials & Methods Ø Whole blood (WB) drawn on citrate Ø 5µl of activated blood (50% WB + 50% reagents) > TG: 0. 3 m. M P 2 Rho, 0. 5 p. M tissue factor (TF), 16. 7 m. M Ca. Cl 2 > Calibration: 0. 3 m. M P 2 Rho, 100 n. M calibrator Ø 40µl of mineral oil Filter paper 40µl oil filter paper with 5µl sample Filter paper + sample + oil © Copyright 2009 by the American Association for Clinical Chemistry
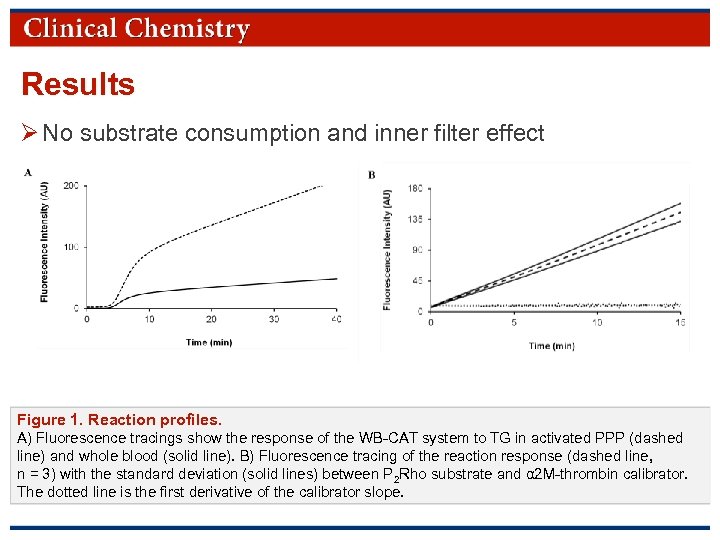
Results Ø No substrate consumption and inner filter effect Figure 1. Reaction profiles. A) Fluorescence tracings show the response of the WB-CAT system to TG in activated PPP (dashed line) and whole blood (solid line). B) Fluorescence tracing of the reaction response (dashed line, n = 3) with the standard deviation (solid lines) between P 2 Rho substrate and α 2 M-thrombin calibrator. The dotted line is the first derivative of the calibrator slope. © Copyright 2009 by the American Association for Clinical Chemistry
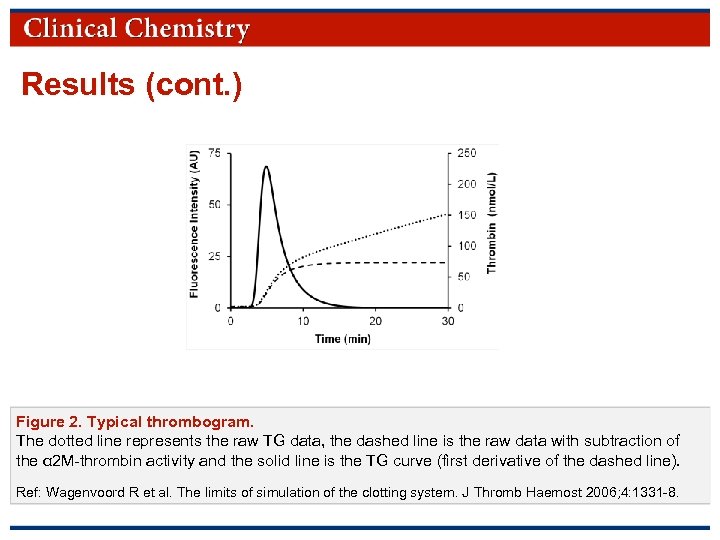
Results (cont. ) Figure 2. Typical thrombogram. The dotted line represents the raw TG data, the dashed line is the raw data with subtraction of the α 2 M-thrombin activity and the solid line is the TG curve (first derivative of the dashed line). Ref: Wagenvoord R et al. The limits of simulation of the clotting system. J Thromb Haemost 2006; 4: 1331 -8. © Copyright 2009 by the American Association for Clinical Chemistry
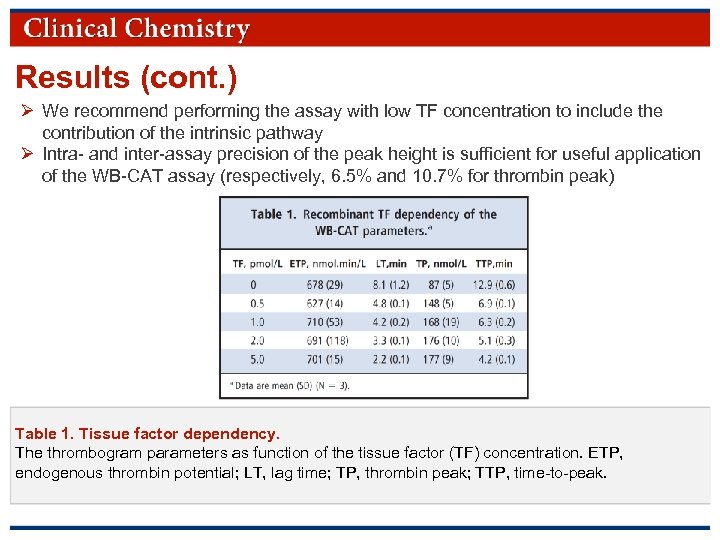
Results (cont. ) Ø We recommend performing the assay with low TF concentration to include the contribution of the intrinsic pathway Ø Intra- and inter-assay precision of the peak height is sufficient for useful application of the WB-CAT assay (respectively, 6. 5% and 10. 7% for thrombin peak) Table 1. Tissue factor dependency. The thrombogram parameters as function of the tissue factor (TF) concentration. ETP, endogenous thrombin potential; LT, lag time; TP, thrombin peak; TTP, time-to-peak. © Copyright 2009 by the American Association for Clinical Chemistry
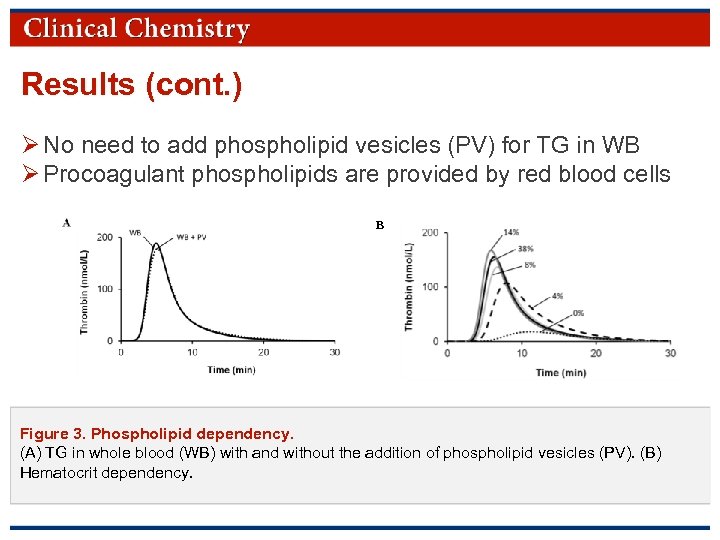
Results (cont. ) Ø No need to add phospholipid vesicles (PV) for TG in WB Ø Procoagulant phospholipids are provided by red blood cells B Figure 3. Phospholipid dependency. (A) TG in whole blood (WB) with and without the addition of phospholipid vesicles (PV). (B) Hematocrit dependency. © Copyright 2009 by the American Association for Clinical Chemistry
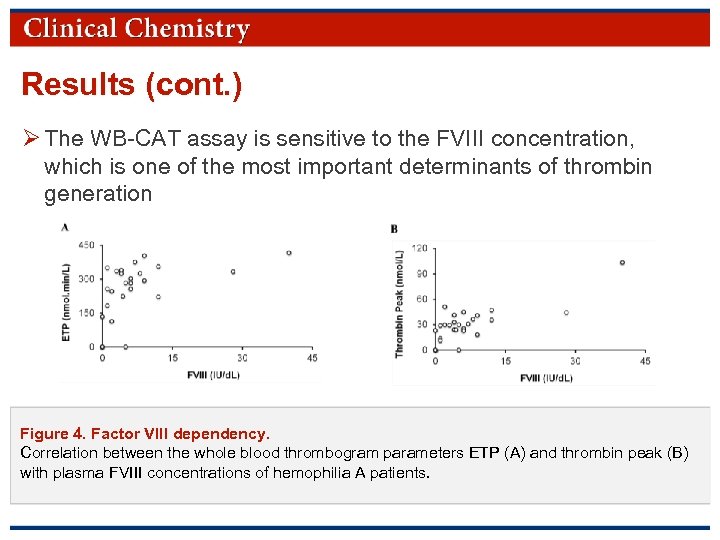
Results (cont. ) Ø The WB-CAT assay is sensitive to the FVIII concentration, which is one of the most important determinants of thrombin generation Figure 4. Factor VIII dependency. Correlation between the whole blood thrombogram parameters ETP (A) and thrombin peak (B) with plasma FVIII concentrations of hemophilia A patients. © Copyright 2009 by the American Association for Clinical Chemistry

Conclusion Ø We developed a method that accurately measures TG in WB by the use of a thin layer of blood in a multiwell microtiter plate Ø Question: The use of filter paper requires some precautions. What are they? Ø Question: What further future work will be need to make the WB-CAT applicable in clinical practice? © Copyright 2009 by the American Association for Clinical Chemistry

Editorial Slide Ø The long-awaited whole blood thrombin generation test (TGT) > TGT is more suitable than the traditional coagulation tests (PT and APTT) to assess the pro- and anticoagulant balance operating in vivo. > Until recently TGT was performed in platelet-poor or platelet-rich plasma. > Ninivaggi et al (Clin Chem, 2012), now describe a new TGT that can be performed in whole-blood, thus mimicking much better than the previous TGT what occurs in vivo. © Copyright 2009 by the American Association for Clinical Chemistry

Thank you for participating in this month’s Clinical Chemistry Journal Club. Additional Journal Clubs are available at www. clinchem. org Follow us © Copyright 2009 by the American Association for Clinical Chemistry
7297ee154035d48ada6edb582ab2af18.ppt