a4a5aa5830443fe99b380105d40fba07.ppt
- Количество слайдов: 74

What’s New with Disease? Jane Seward, MBBS, MPH Centers for Disease Control and Prevention National Immunization Conference Atlanta, USA, March 20 th, 2008

Surveillance for Vaccine Preventable Diseases • Tremendous successes • Challenges • Mature vaccine programs – Pertussis, Measles, mumps, Hib, Hepatitis B • Intermediate vaccine programs – Varicella and herpes zoster, Hepatitis A, Pneumococcal vaccine for children • New vaccine programs – Pediatric influenza (deaths), HPV, rotavirus
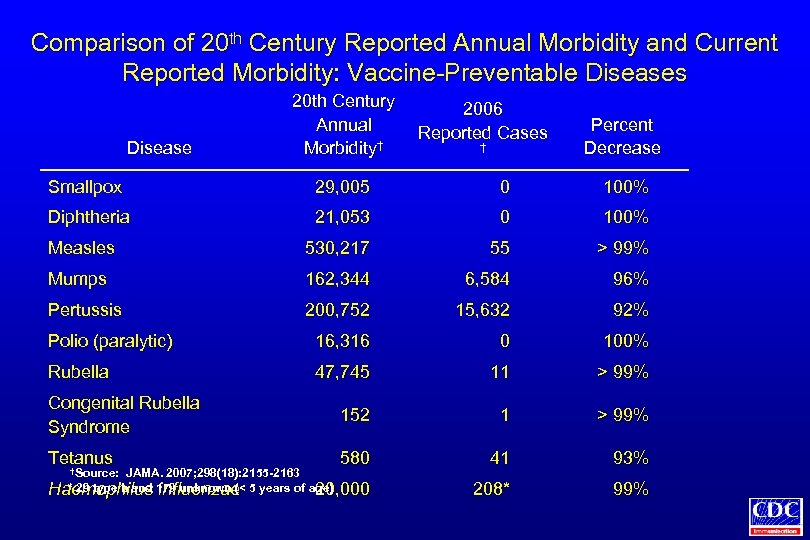
Comparison of 20 th Century Reported Annual Morbidity and Current Reported Morbidity: Vaccine-Preventable Diseases Disease 20 th Century Annual Morbidity† 2006 Reported Cases † Percent Decrease Smallpox 29, 005 0 100% Diphtheria 21, 053 0 100% Measles 530, 217 55 > 99% Mumps 162, 344 6, 584 96% Pertussis 200, 752 15, 632 92% Polio (paralytic) 16, 316 0 100% Rubella 47, 745 11 > 99% Congenital Rubella Syndrome 152 1 > 99% Tetanus † 580 41 93% 208* 99% Source: JAMA. 2007; 298(18): 2155 -2163 * 29 type b and influenzae Haemophilus 179 unknown (< 5 years of age) 20, 000
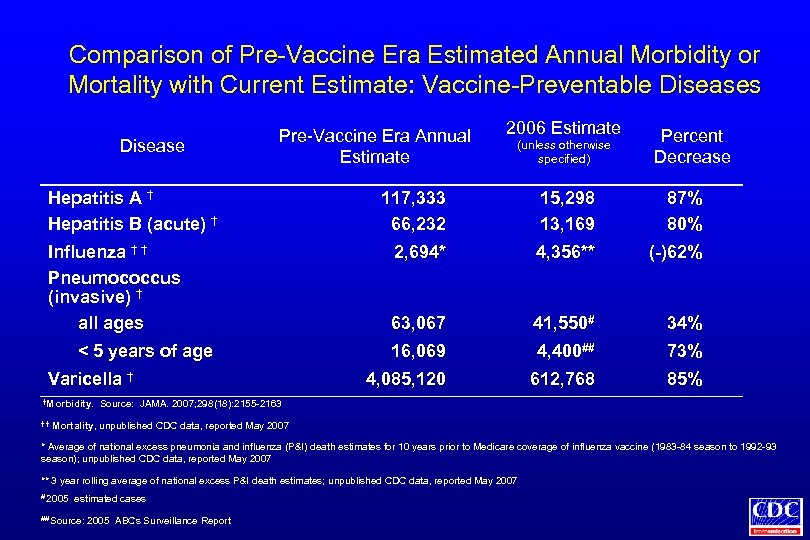
Comparison of Pre-Vaccine Era Estimated Annual Morbidity or Mortality with Current Estimate: Vaccine-Preventable Diseases Disease Pre-Vaccine Era Annual Estimate Hepatitis A † Hepatitis B (acute) † Influenza † † Pneumococcus (invasive) † all ages < 5 years of age Varicella † †Morbidity. (unless otherwise specified) Percent Decrease 117, 333 66, 232 15, 298 13, 169 87% 80% 2, 694* 4, 356** (-)62% 63, 067 41, 550# 34% 16, 069 4, 400## 73% 4, 085, 120 612, 768 85% Source: JAMA. 2007; 298(18): 2155 -2163 † † Mortality, 2006 Estimate unpublished CDC data, reported May 2007 * Average of national excess pneumonia and influenza (P&I) death estimates for 10 years prior to Medicare coverage of influenza vaccine (1983 -84 season to 1992 -93 season); unpublished CDC data, reported May 2007 ** 3 year rolling average of national excess P&I death estimates; unpublished CDC data, reported May 2007 # 2005 estimated cases ## Source: 2005 ABCs Surveillance Report

Mature Vaccine Programs What’s happening with disease? New surveillance priorities? • • • Pertussis Measles Mumps Hib Hepatitis B

Pertussis
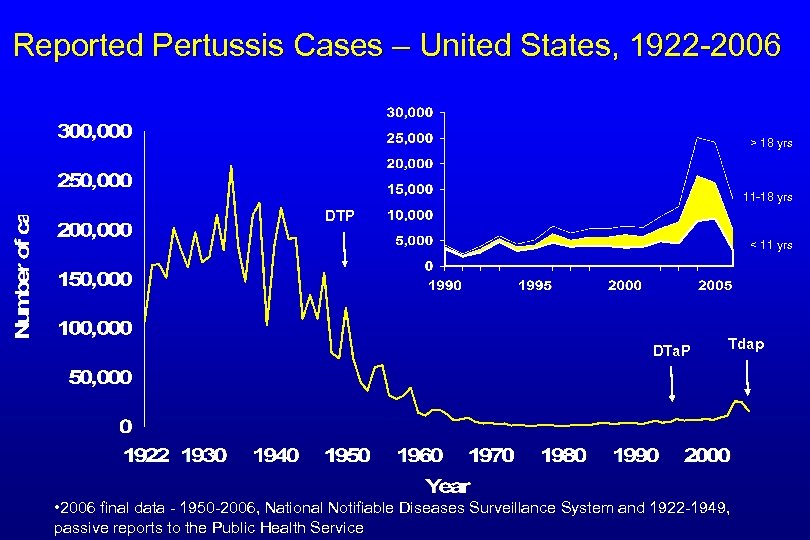
Reported Pertussis Cases – United States, 1922 -2006 > 18 yrs 11 -18 yrs DTP < 11 yrs DTa. P Tdap • 2006 final data - 1950 -2006, National Notifiable Diseases Surveillance System and 1922 -1949, passive reports to the Public Health Service
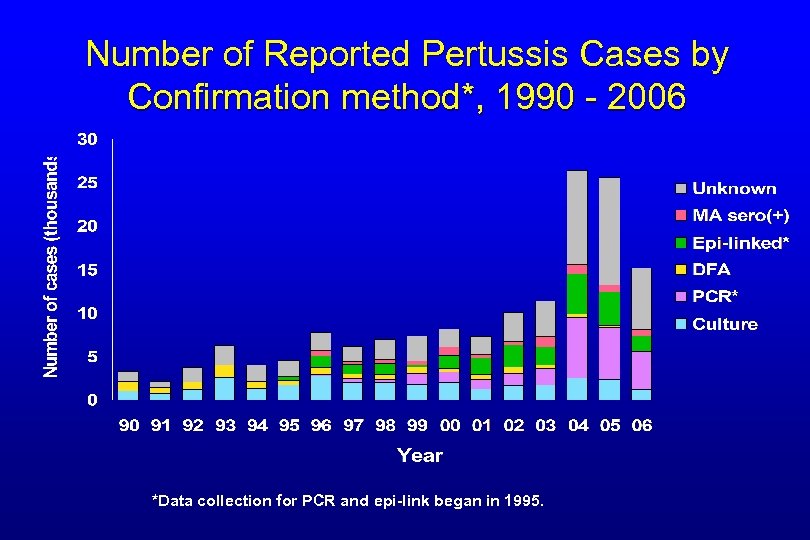
Number of Reported Pertussis Cases by Confirmation method*, 1990 - 2006 *Data collection for PCR and epi-link began in 1995.
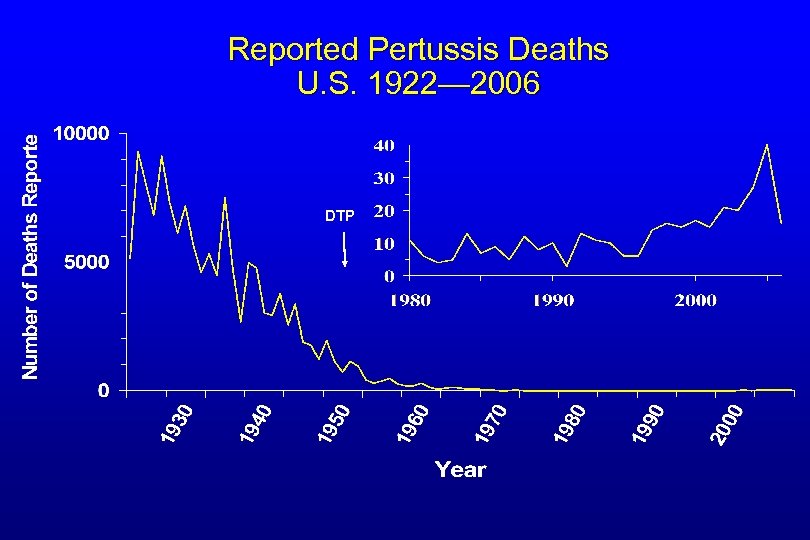
Reported Pertussis Deaths U. S. 1922— 2006 DTP
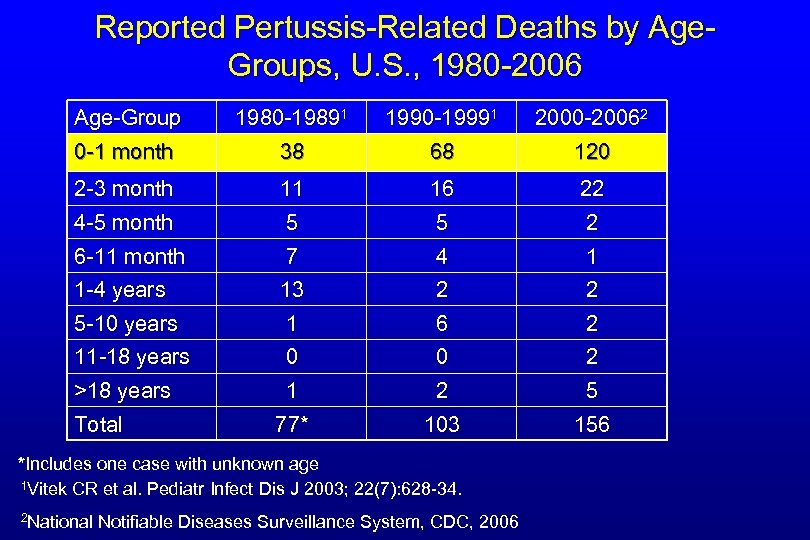
Reported Pertussis-Related Deaths by Age. Groups, U. S. , 1980 -2006 Age-Group 0 -1 month 1980 -19891 38 1990 -19991 68 2000 -20062 120 2 -3 month 4 -5 month 11 5 16 5 22 2 6 -11 month 1 -4 years 7 13 4 2 1 2 5 -10 years 11 -18 years 1 0 6 0 2 2 1 77* 2 103 5 156 >18 years Total *Includes one case with unknown age 1 Vitek CR et al. Pediatr Infect Dis J 2003; 22(7): 628 -34. 2 National Notifiable Diseases Surveillance System, CDC, 2006

Measles
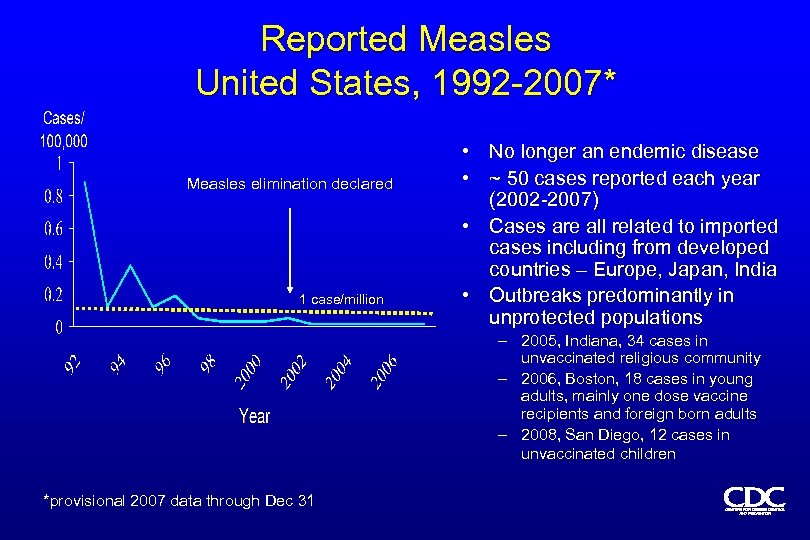
Reported Measles United States, 1992 -2007* Measles elimination declared 1 case/million • No longer an endemic disease • ~ 50 cases reported each year (2002 -2007) • Cases are all related to imported cases including from developed countries – Europe, Japan, India • Outbreaks predominantly in unprotected populations – 2005, Indiana, 34 cases in unvaccinated religious community – 2006, Boston, 18 cases in young adults, mainly one dose vaccine recipients and foreign born adults – 2008, San Diego, 12 cases in unvaccinated children *provisional 2007 data through Dec 31
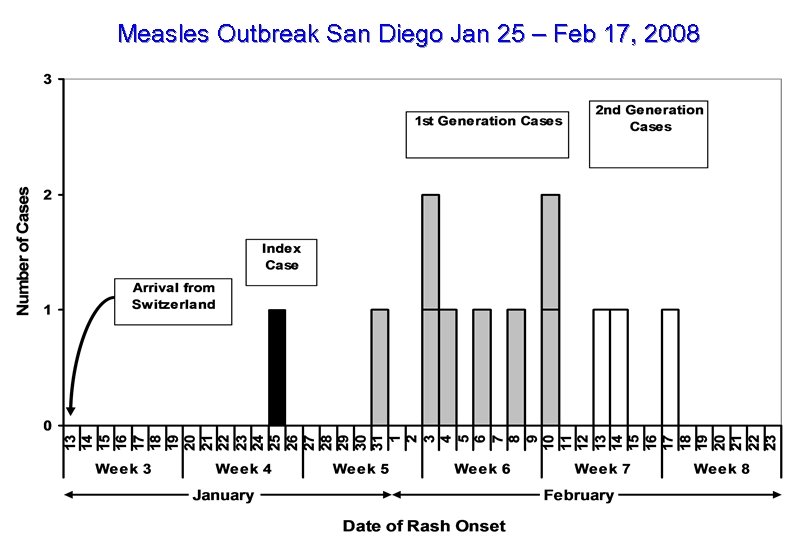
Figure Measles Outbreak San Diego Jan 25 – Feb 17, 2008

Measles 2008 • 21 confirmed cases reported so far to NCIRD – – San Diego Arizona NYC Virginia 12 case outbreak 6 case outbreak 2 cases 1 case Switzerland, D 5 Switzerland D 4 ? Israel India • 17 > 12 months, all unvaccinated • High index of suspicion in persons who travel or in contact with travelers
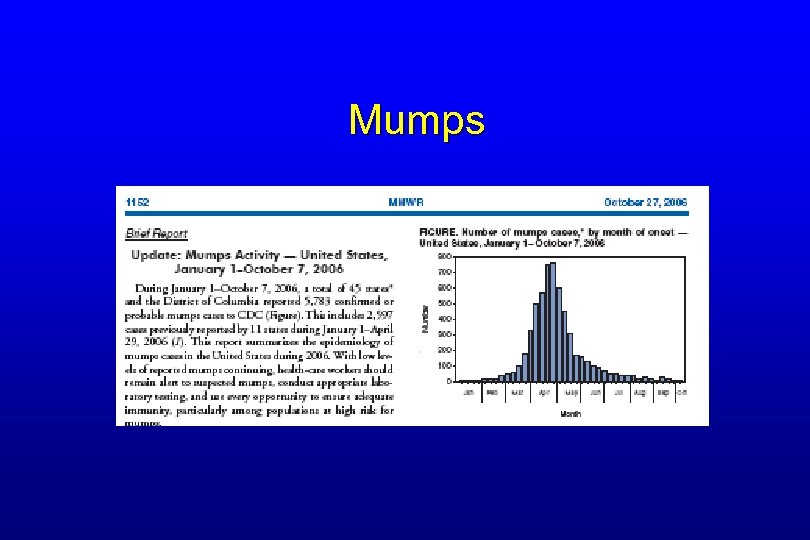
Mumps
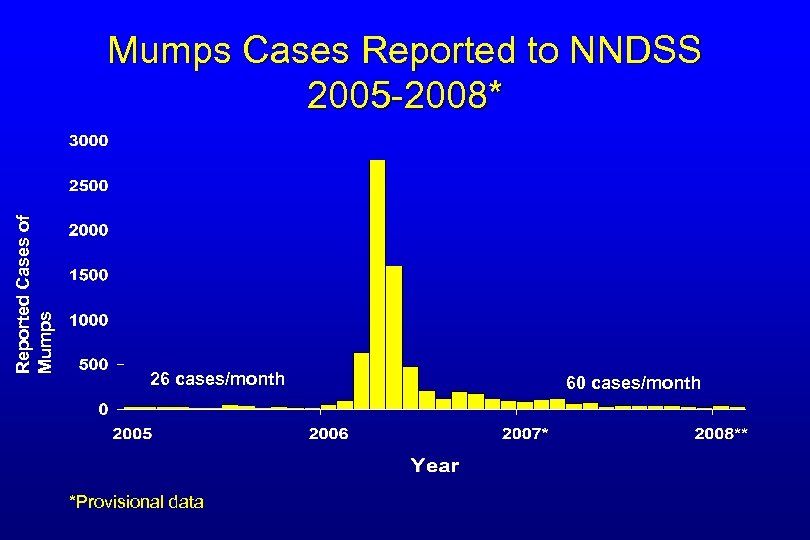
Reported Cases of Mumps Cases Reported to NNDSS 2005 -2008* 26 cases/month *Provisional data 60 cases/month
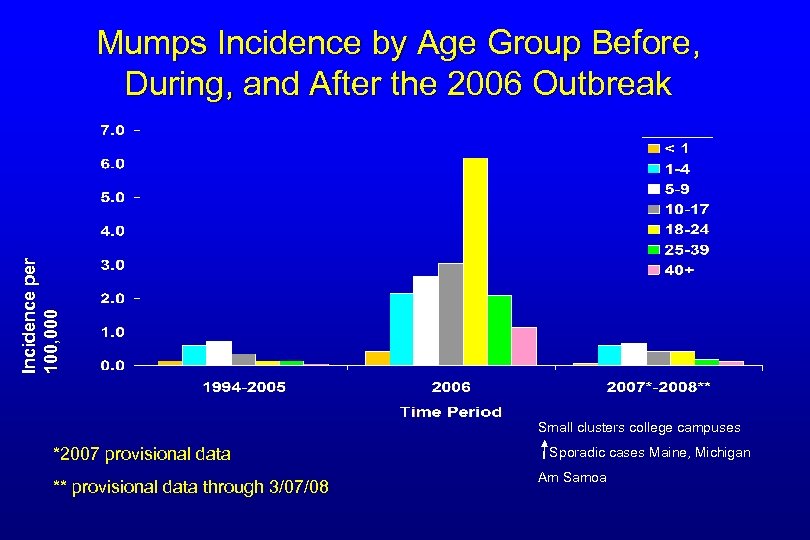
Incidence per 100, 000 Mumps Incidence by Age Group Before, During, and After the 2006 Outbreak Small clusters college campuses *2007 provisional data ** provisional data through 3/07/08 Sporadic cases Maine, Michigan Am Samoa

Mumps Surveillance • January 2008: changes to case definition and case classifications • Probable case now requires epi link • Reporting to NNDSS of some key surveillance variables incomplete – Vaccination status and # doses – Transmission setting – Lab testing • Vaccinated cases – lower confirmation rate using Ig. M, PCR, viral isolation – Can’t rule out a case if lab testing negative • Testing for other viral causes parotitis may help rule in other diagnoses if mumps testing negative
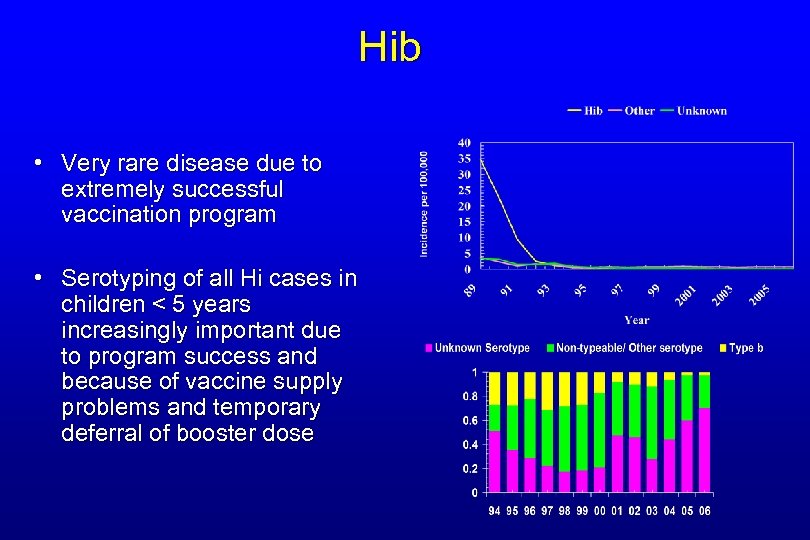
Hib • Very rare disease due to extremely successful vaccination program • Serotyping of all Hi cases in children < 5 years increasingly important due to program success and because of vaccine supply problems and temporary deferral of booster dose

Hepatitis B
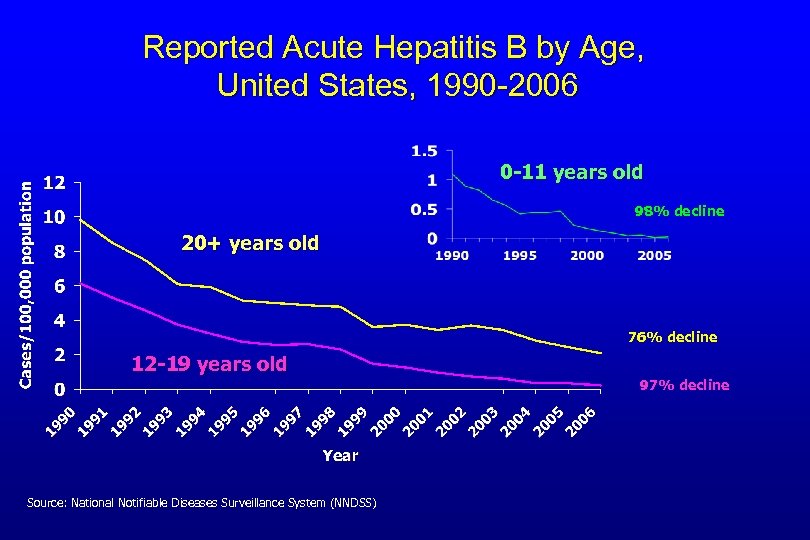
Reported Acute Hepatitis B by Age, United States, 1990 -2006 0 -11 years old 98% decline 20+ years old 76% decline 12 -19 years old Source: National Notifiable Diseases Surveillance System (NNDSS) 97% decline
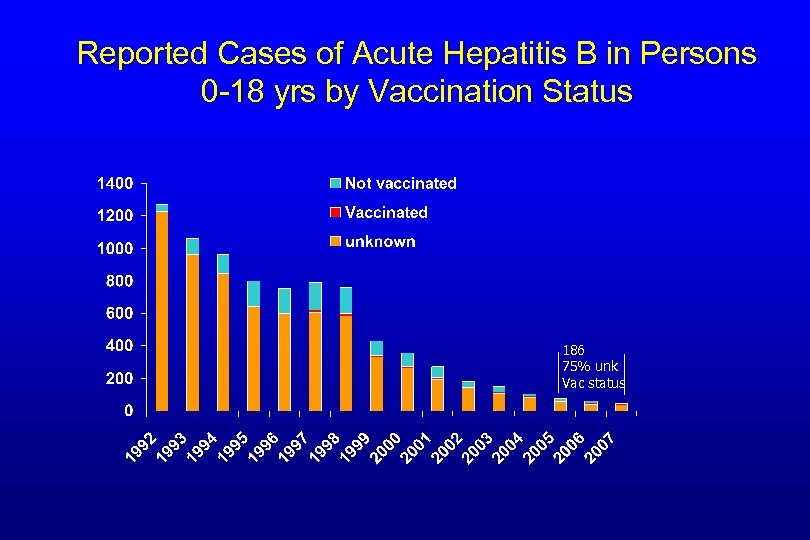
Reported Cases of Acute Hepatitis B in Persons 0 -18 yrs by Vaccination Status 186 75% unk Vac status

Viral Hepatitis in Children: What is their Vaccination status? • Need to differentiate “vaccine failure” from “program failure” • If child is vaccinated, – Did the child receive all of the required doses according to recommended schedule? – At what age were they vaccinated? • If child is not vaccinated – What are characteristics of the child and were there opportunities to vaccinate that were missed?

Intermediate Vaccine Programs • Hepatitis A • Varicella (and herpes zoster) • Pneumococcal (PCV 7) disease

Hepatitis A
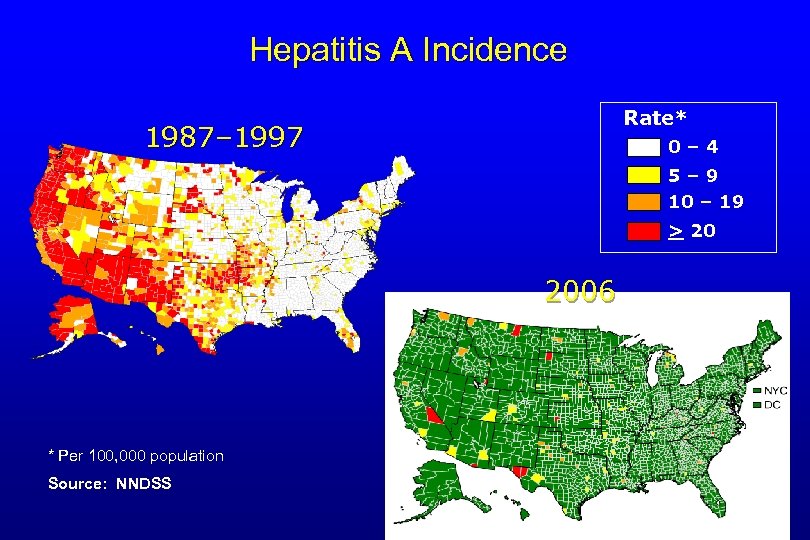
Hepatitis A Incidence Rate* 1987– 1997 0– 4 5– 9 10 – 19 > 20 2006 * Per 100, 000 population Source: NNDSS
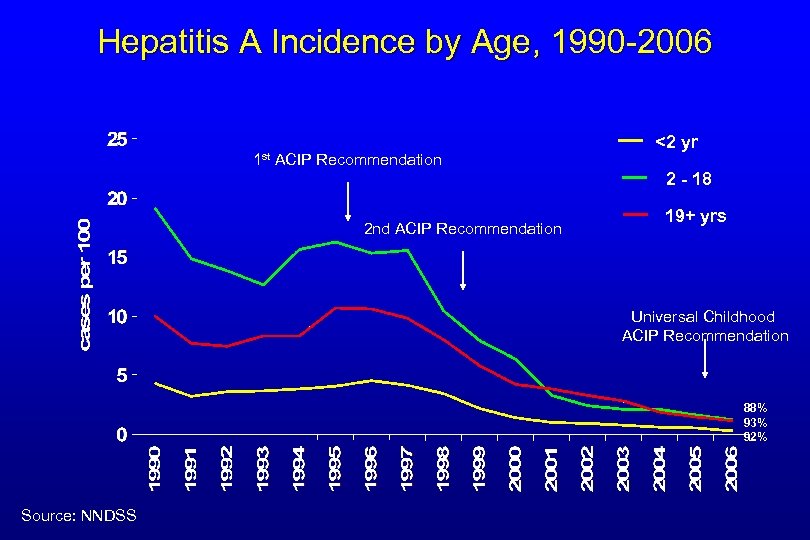
Hepatitis A Incidence by Age, 1990 -2006 1 st ACIP Recommendation <2 yr 2 - 18 2 nd ACIP Recommendation 19+ yrs Universal Childhood ACIP Recommendation 88% 93% 92% Source: NNDSS
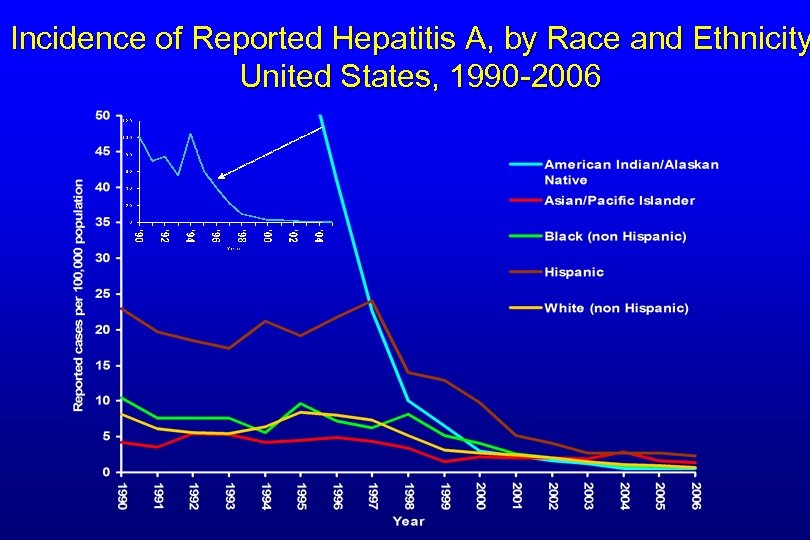
Incidence of Reported Hepatitis A, by Race and Ethnicity United States, 1990 -2006

Varicella and Herpes Zoster

Examples of Recent Varicella Vaccine Preventable Deaths, 2005, 2007 • Healthy 10 -year old female • Unvaccinated, no varicella disease history • Presented with classic symptoms of varicella on 10/20, was hospitalized on 10/28 with encephalitis and died 2 days later • Her likely source of transmission was an ongoing varicella outbreak at her school • • • Healthy 13 month old female Unvaccinated, no prior varicella disease history Presented with classic symptoms of varicella on 4/24, was hospitalized on 4/29 for dehydration, developed septic shock and died the next day • Her likely source of transmission was her father who had varicella 2 weeks prior to her rash onset
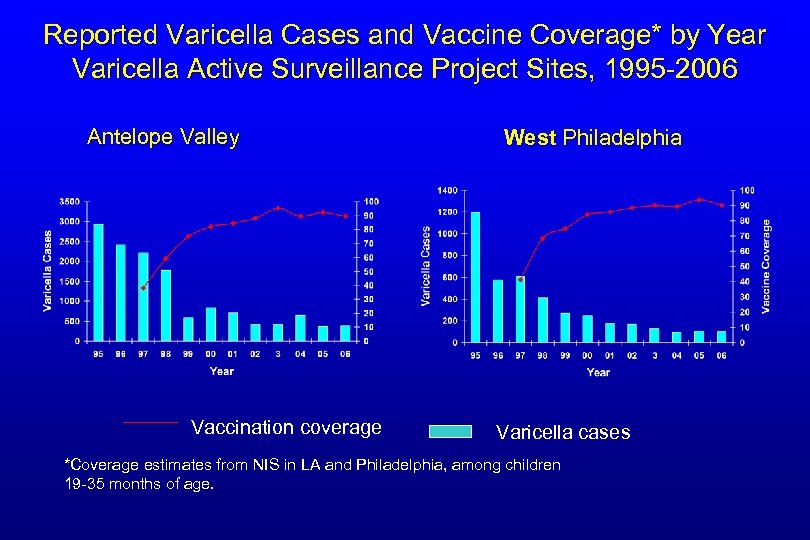
Reported Varicella Cases and Vaccine Coverage* by Year Varicella Active Surveillance Project Sites, 1995 -2006 Antelope Valley Vaccination coverage West Philadelphia Varicella cases *Coverage estimates from NIS in LA and Philadelphia, among children 19 -35 months of age.
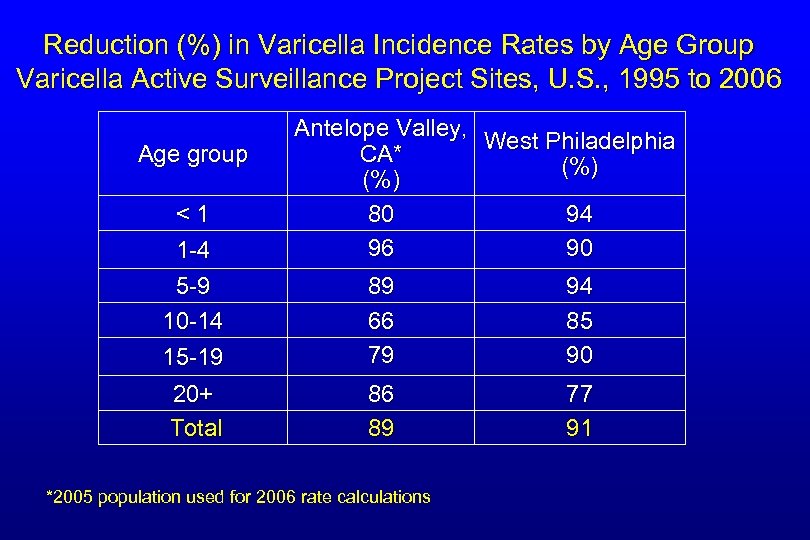
Reduction (%) in Varicella Incidence Rates by Age Group Varicella Active Surveillance Project Sites, U. S. , 1995 to 2006 Age group <1 1 -4 Antelope Valley, West Philadelphia CA* (%) 80 94 96 90 5 -9 10 -14 15 -19 89 66 79 94 85 90 20+ Total 86 89 77 91 *2005 population used for 2006 rate calculations
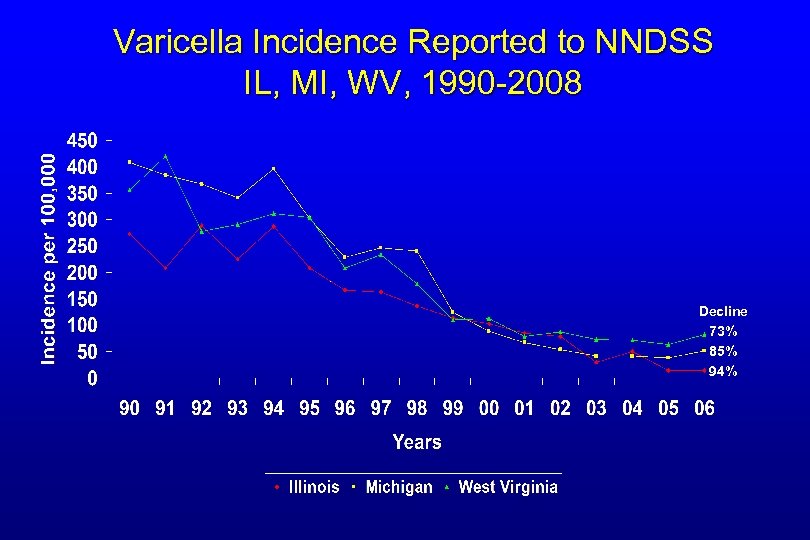
Varicella Incidence Reported to NNDSS IL, MI, WV, 1990 -2008 Decline 73% 85% 94%

Herpes Zoster Surveillance 1. Surveillance for monitoring impact of varicella vaccine on HZ epidemiology • No consistent trends since 1995 • • In vaccinated cohorts (children < 10 years), HZ appears to be decreasing In cohorts with varicella history, inconsistent findings: stable or increasing incidence 2. • Surveillance for zoster vaccine (> 60 years) • • HZ not likely to become reportable • Administrative databases: Medicare, VSD, Med. Stat, other To monitor effectiveness of vaccine program against HZ and complications Active surveillance (varicella sites), surveys (BRFSS, NHIS, Adult NIS, HRS
Influenza-associated pediatric mortality surveillance • Nationally Notifiable Diseases Surveillance System • Surveillance implemented following severe influenza season of 2003 -2004
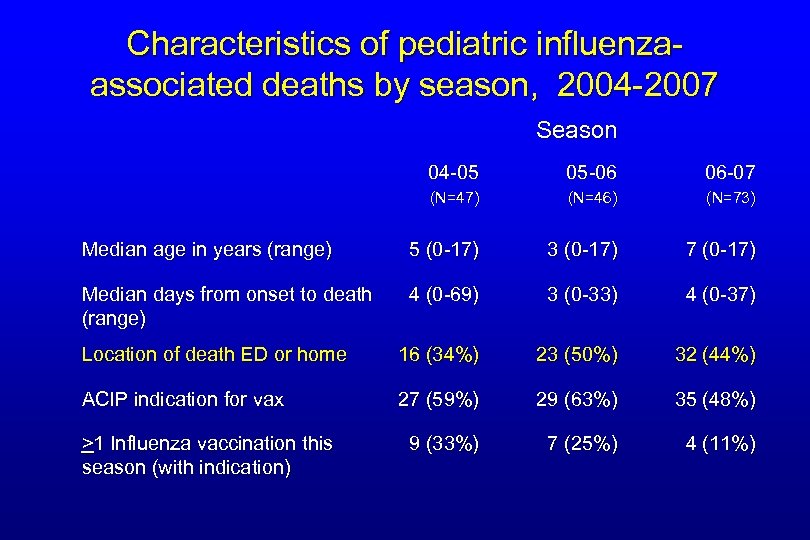
Characteristics of pediatric influenzaassociated deaths by season, 2004 -2007 Season 04 -05 05 -06 06 -07 (N=47) (N=46) (N=73) Median age in years (range) 5 (0 -17) 3 (0 -17) 7 (0 -17) Median days from onset to death (range) 4 (0 -69) 3 (0 -33) 4 (0 -37) Location of death ED or home 16 (34%) 23 (50%) 32 (44%) ACIP indication for vax 27 (59%) 29 (63%) 35 (48%) 9 (33%) 7 (25%) 4 (11%) >1 Influenza vaccination this season (with indication)
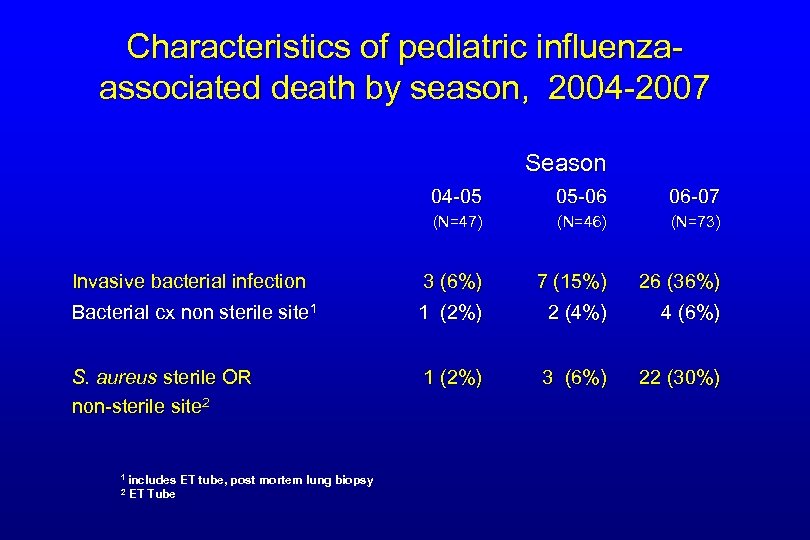
Characteristics of pediatric influenzaassociated death by season, 2004 -2007 Season 04 -05 05 -06 06 -07 (N=47) (N=46) (N=73) Invasive bacterial infection 3 (6%) 7 (15%) 26 (36%) Bacterial cx non sterile site 1 1 (2%) 2 (4%) 4 (6%) S. aureus sterile OR non-sterile site 2 1 (2%) 3 (6%) 22 (30%) 1 includes 2 ET Tube ET tube, post mortem lung biopsy

Summary of Influenza-Associated Pediatric Mortality for 2004 -07* • Influenza was rapidly fatal – almost half died within 3 days of onset – almost half died at home or in ED • Vaccination rates low in those with ACIP indication • Co-infection with S. aureus increased dramatically over 3 years – Older median age and more likely to have ARDS and radiologically confirmed pneumonia – Factors that may have contributed to the increase • Predominance of USA 300 strain in community • MRSA colonization rates increasing especially in school- aged children – Artifact? *2004 -05 (n=47), 2005 -06 (n=46), and 2006 -07 (n=74)

Invasive Pneumococcal Disease MCV 7 Vaccine
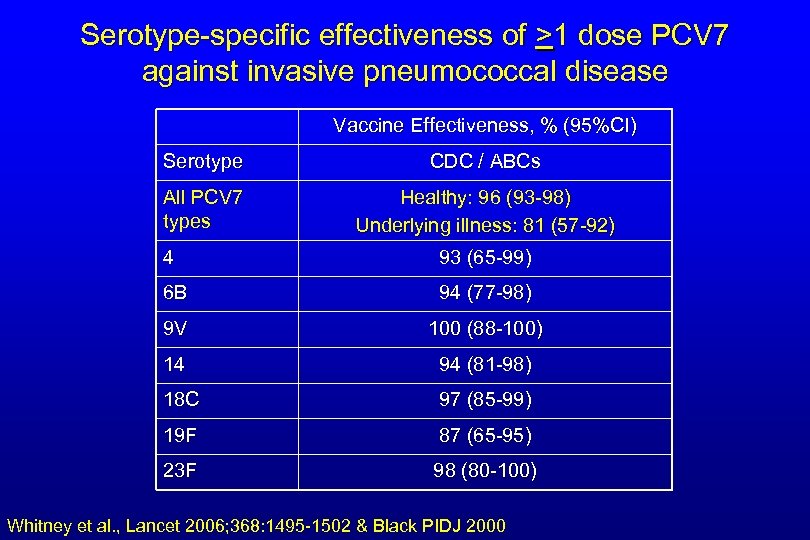
Serotype-specific effectiveness of >1 dose PCV 7 against invasive pneumococcal disease Vaccine Effectiveness, % (95%CI) Serotype CDC / ABCs All PCV 7 types Healthy: 96 (93 -98) Underlying illness: 81 (57 -92) 4 93 (65 -99) 6 B 94 (77 -98) 9 V 100 (88 -100) 14 94 (81 -98) 18 C 97 (85 -99) 19 F 87 (65 -95) 23 F 98 (80 -100) Whitney et al. , Lancet 2006; 368: 1495 -1502 & Black PIDJ 2000
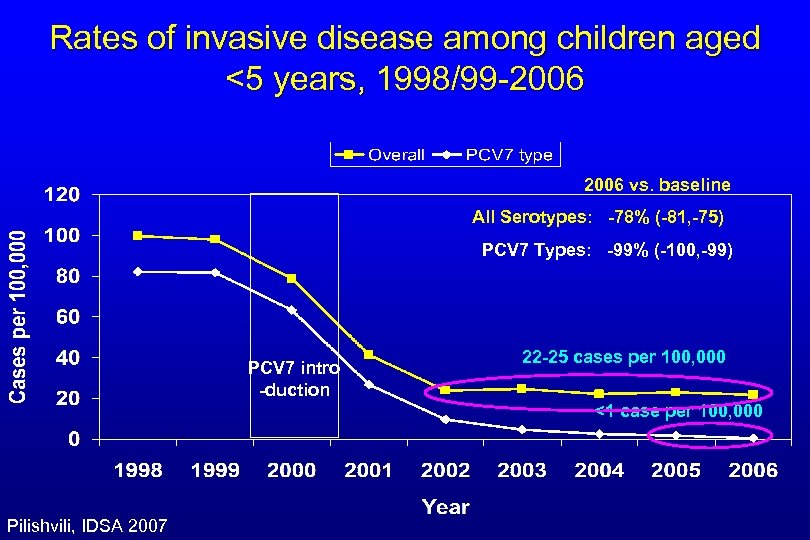
Rates of invasive disease among children aged <5 years, 1998/99 -2006 vs. baseline All Serotypes: -78% (-81, -75) PCV 7 Types: -99% (-100, -99) PCV 7 intro -duction 22 -25 cases per 100, 000 <1 case per 100, 000 Pilishvili, IDSA 2007
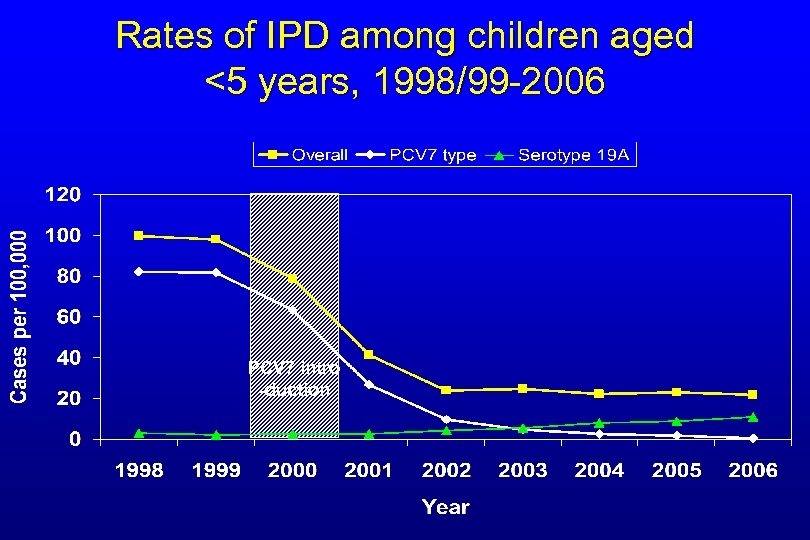
Rates of IPD among children aged <5 years, 1998/99 -2006 PCV 7 intro -duction
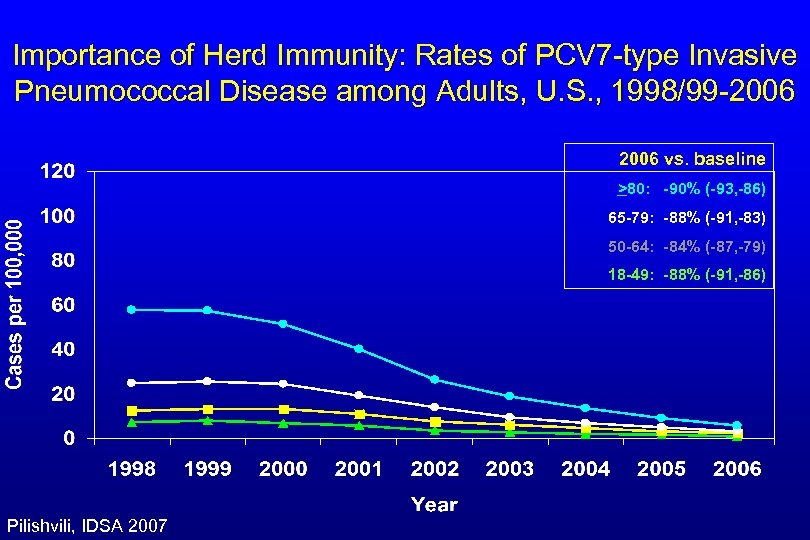
Importance of Herd Immunity: Rates of PCV 7 -type Invasive Pneumococcal Disease among Adults, U. S. , 1998/99 -2006 vs. baseline >80: -90% (-93, -86) 65 -79: -88% (-91, -83) 50 -64: -84% (-87, -79) 18 -49: -88% (-91, -86) Pilishvili, IDSA 2007

State and National Surveillance for IPD • PCV 13 is in development • State surveillance for IPD in children important for monitoring continued impact of PCV 7, replacement serotypes, and impact of new vaccine introduction • PCR-based approach to serotyping available to state health laboratories • Protocol available at http: //www. cdc. gov/ncidod/biotech/strep/prc. htm • CDC Streptococcal lab available to provide technical assistance

Monitoring New Vaccine Programs

Rotavirus

Rotavirus Surveillance • National surveillance for cases impractical – ~ 3 million cases annually – Require lab test to confirm diagnosis • Strain surveillance: National Respiratory and Enteric Virus Surveillance System • Focused efforts to evaluate vaccine impact on severe disease and viral strains at sentinel sites (NVSN)

• Population-based surveillance in 3 counties • Started in Jan 2006 • Children with acute gastroenteritis (AGE) enrolled and stool specimen tested for rotavirus
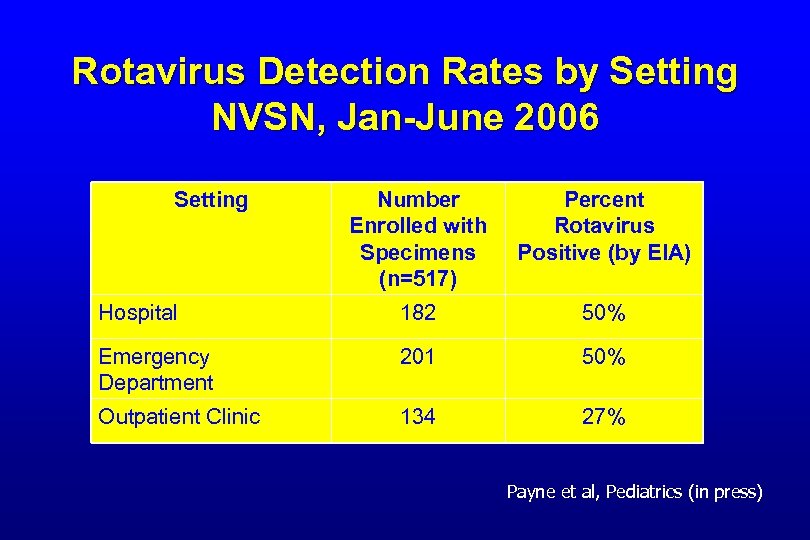
Rotavirus Detection Rates by Setting NVSN, Jan-June 2006 Setting Number Enrolled with Specimens (n=517) Percent Rotavirus Positive (by EIA) Hospital 182 50% Emergency Department 201 50% Outpatient Clinic 134 27% Payne et al, Pediatrics (in press)
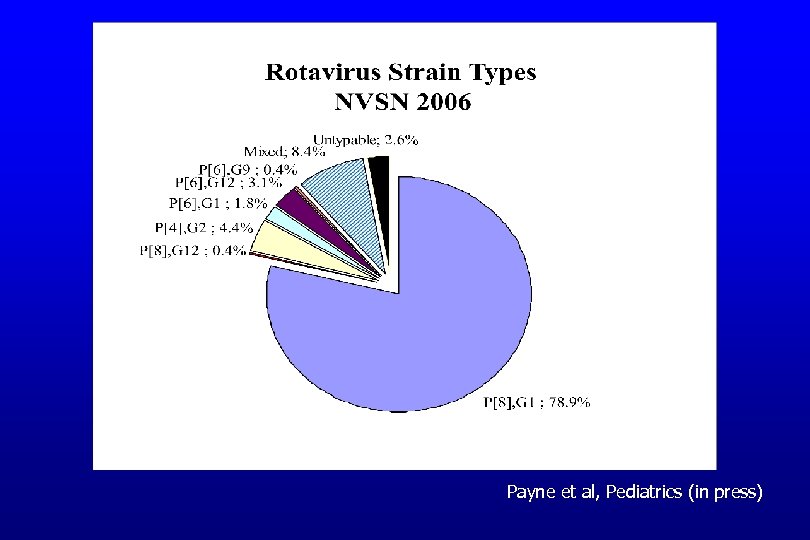
Payne et al, Pediatrics (in press)
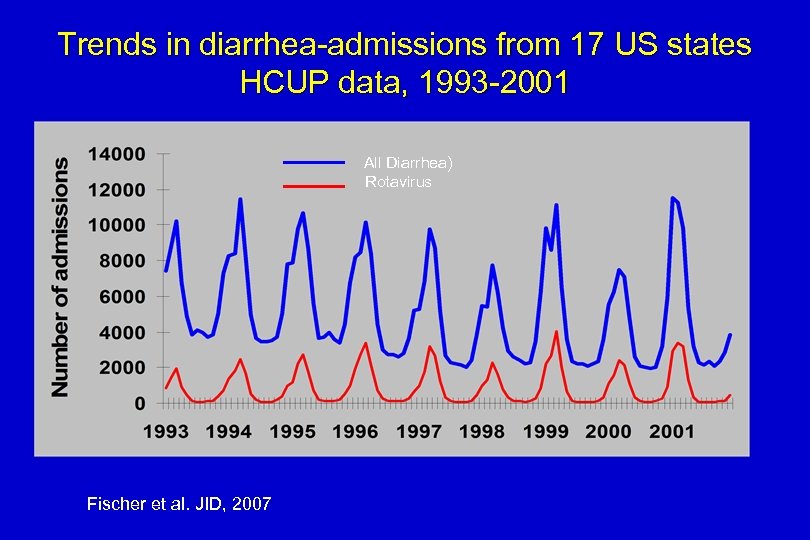
Trends in diarrhea-admissions from 17 US states HCUP data, 1993 -2001 All Diarrhea) Rotavirus Fischer et al. JID, 2007

Human Papillomavirus

Monitoring Impact of HPV Vaccine Challenges/Opportunities • Multiple outcomes/multiple HPV types • HPV 16, 18 - cervical cancer, precancer, abn Paps • HPV 6, 11 - genital warts and recurrent respiratory papillomatosis • Time between infection and outcomes • Months for warts • Years/Decades for cervical cancer • Interface with cervical cancer screening • New collaborations • Immunization, STD, Cancer
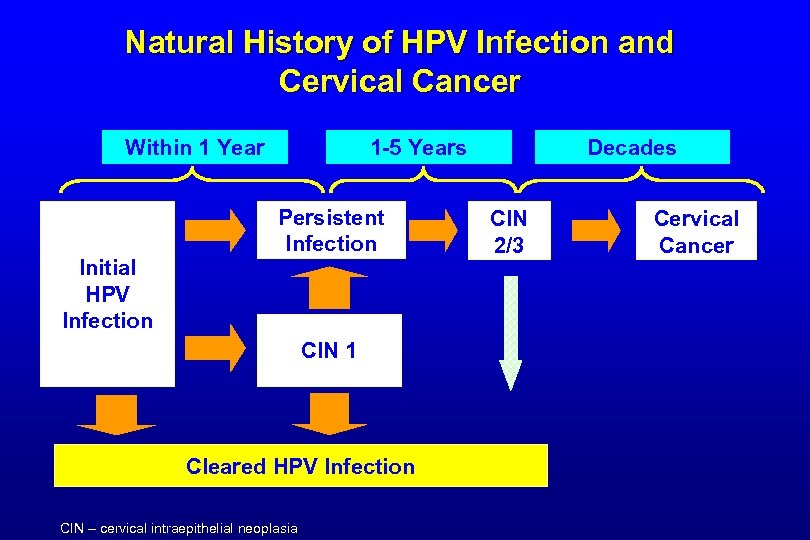
Natural History of HPV Infection and Cervical Cancer Within 1 Year Initial HPV Infection 1 -5 Years Persistent Infection CIN 1 Cleared HPV Infection CIN – cervical intraepithelial neoplasia Decades CIN 2/3 Cervical Cancer

Cancer Surveillance in the US

Monitoring Impact of HPV Vaccination in the United States • Cervical cancer – Established cancer registries – HPV typing to be initiated at several sites • Precancers (CIN 2/3) – Sentinel projects initiated to collect population-based data – Administrative data • Genital warts – STD clinic-based surveillance – Administrative data • Type-specific HPV prevalence – Self collected vaginal swabs - NHANES

Disease Surveillance • Essential for demonstrating impact of vaccines, guiding vaccine policy and programs • Provide critical data for monitoring effectiveness of vaccines • Thank you all for your work

Thank You • • • Sandy Roush Annemarie Wasley Kathy Gallagher Laurie Markowitz Umesh Parashar Amy Parker Al Barskey Susan Redd Adriana Lopez • • • Jessica Leung Rafael Harpaz John Zhang Aisha Jumaan Lynn Finelli Tom Clark Nancy Messonnier Cindy Whitney Matt Moore CDC laboratories supporting disease surveillance

Extras

Public health impact of PCV 7 in the U. S. During each year from 2001 -2006… <5 year-olds: 11, 000 -15, 000 invasive cases prevented + >5 year-olds: 9, 000 -18, 000 invasive cases prevented = 170, 000 cases and 9, 800 deaths prevented Pilishvili, CDC, manuscript in preparation, 2008
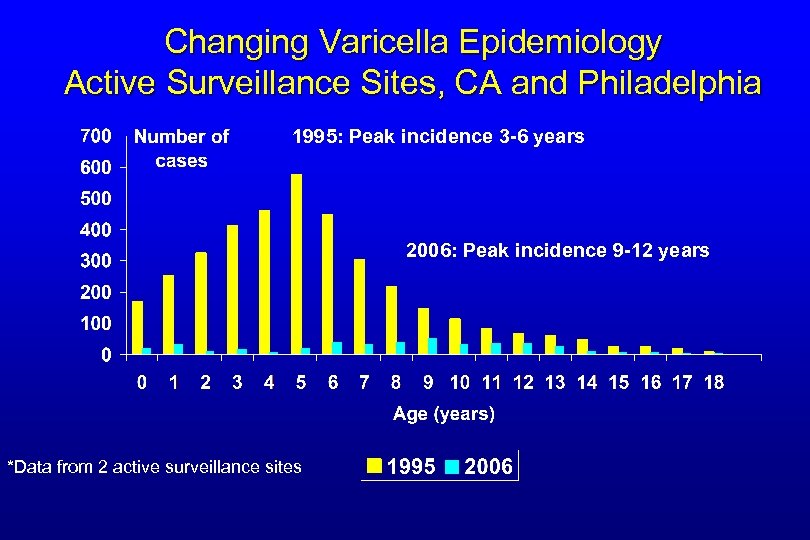
Changing Varicella Epidemiology Active Surveillance Sites, CA and Philadelphia 1995: Peak incidence 3 -6 years 2006: Peak incidence 9 -12 years *Data from 2 active surveillance sites
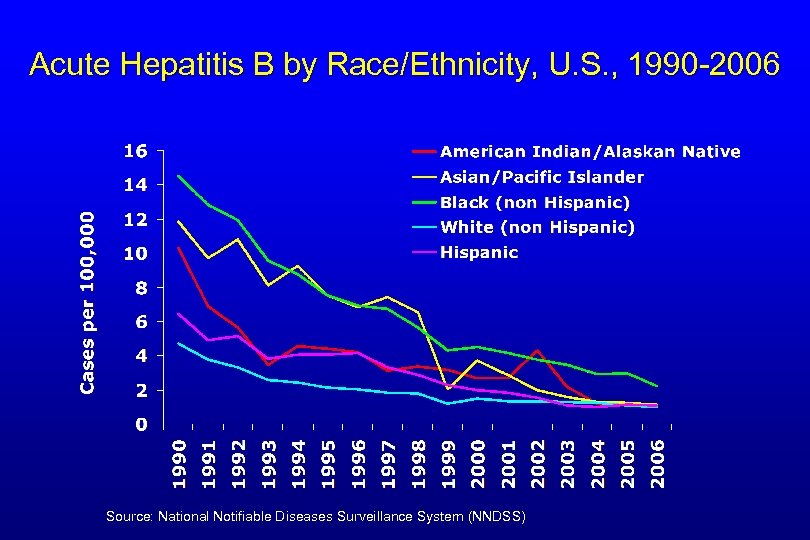
Acute Hepatitis B by Race/Ethnicity, U. S. , 1990 -2006 Source: National Notifiable Diseases Surveillance System (NNDSS)

Pertussis Complications in Infants <12 months 2000 -2006* 18, 564 reported pertussis cases Complication Hospitalization Apnea Pneumonia§ Seizures Deaths Number (% of total)† 9. 078 8. 348 1, 578 186 145 (61. 0) (56. 0) (12. 8) (1. 3) (0. 8) *SOURCE: CDC, National Notifiable Diseases Surveillance System and Supplemental Pertussis Surveillance System, 2000 -2006. † Percentages are based on total number with information. For 20% of infant cases, no information was available on hospitalization or apnea; for 21% no information was available on seizure; and for 33%, no information was available on pneumonia. § Radiographically confirmed.

Herpes Zoster

Herpes Zoster Surveillance: why? Herpes Zoster is now vaccine preventable • Need to monitor the vaccination program’s effectiveness – Against herpes zoster-related outcomes – disease and complications – Gaps in program • Need to monitor vaccine effectiveness – Against various herpes zoster-related outcomes (HZ, PHN etc) – Duration of protection – Effectiveness in specific subgroups (e. g. diabetics) • Need to understand trends in HZ rates (controlled for age) – To interpret surveillance & impact of vaccine program – Understand reasons for apparent increase (& intervene) – Identify risk groups that might be targeted for vaccine

Herpes Zoster Surveillance: how? • We do not anticipate that HZ will become notifiable • Surveys – NHIS, BRFSS, Health & Retirement Survey (HRS), adult module of National Immunization Survey • Active surveillance sites: varicella and herpes zoster: Antelope Valley, W. Philadelphia • Administrative databases – Medicare, Medstat, VSD, other • Need to validate these data for use in tracking HZ and associated outcomes e. g. PHN
Midwest Northeast West South
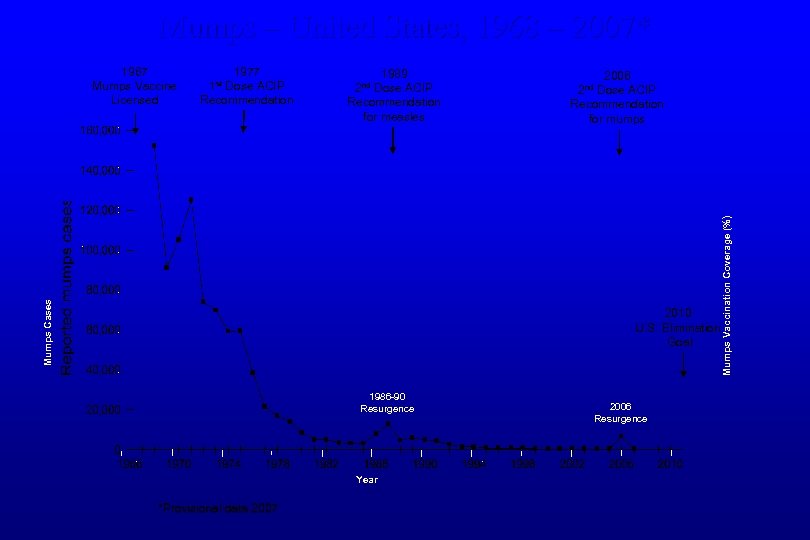
Mumps – United States, 1968 – 2007* 1989 Dose ACIP Recommendation for measles 2 nd 2006 Dose ACIP Recommendation for mumps 2 nd 2010 U. S. Elimination Goal 1986 -90 Resurgence Year *Provisional data 2007 2006 Resurgence Mumps Vaccination Coverage (%) 1977 Dose ACIP Recommendation 1 st Mumps Cases 1967 Mumps Vaccine Licensed
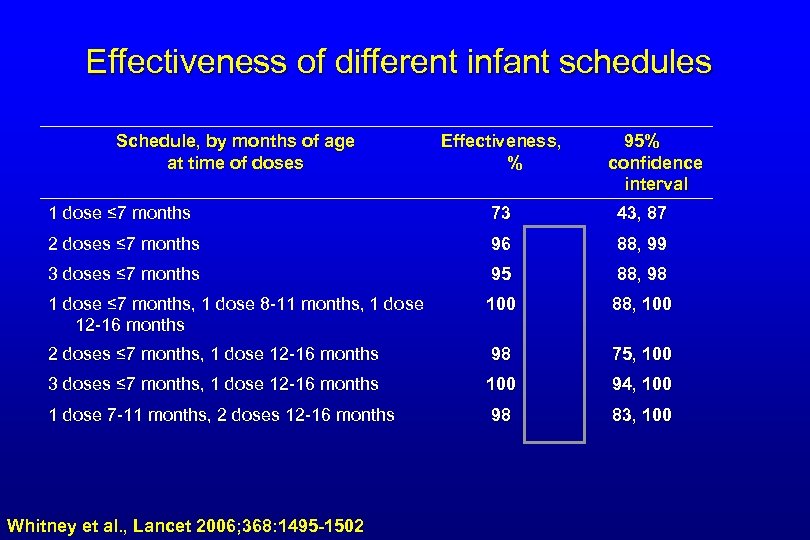
Effectiveness of different infant schedules Schedule, by months of age at time of doses Effectiveness, % 95% confidence interval 1 dose ≤ 7 months 73 43, 87 2 doses ≤ 7 months 96 88, 99 3 doses ≤ 7 months 95 88, 98 1 dose ≤ 7 months, 1 dose 8 -11 months, 1 dose 12 -16 months 100 88, 100 2 doses ≤ 7 months, 1 dose 12 -16 months 98 75, 100 3 doses ≤ 7 months, 1 dose 12 -16 months 100 94, 100 1 dose 7 -11 months, 2 doses 12 -16 months 98 83, 100 Whitney et al. , Lancet 2006; 368: 1495 -1502
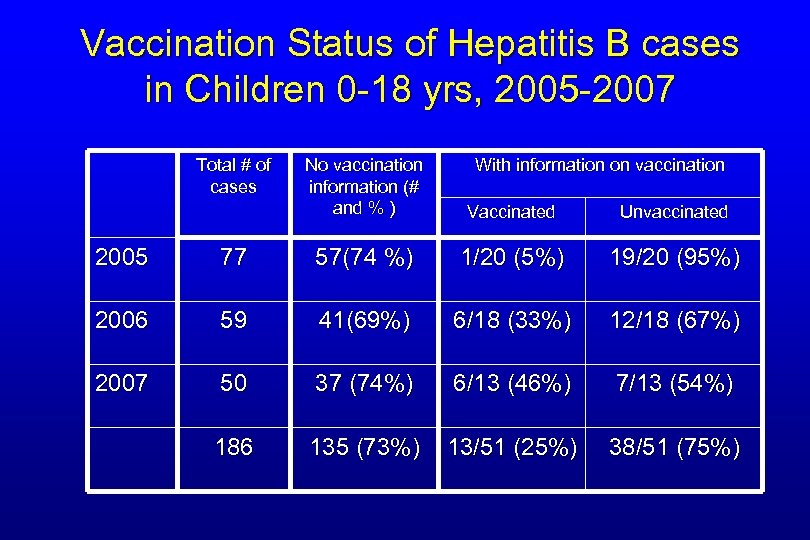
Vaccination Status of Hepatitis B cases in Children 0 -18 yrs, 2005 -2007 Total # of cases No vaccination information (# and % ) With information on vaccination Vaccinated Unvaccinated 2005 77 57(74 %) 1/20 (5%) 19/20 (95%) 2006 59 41(69%) 6/18 (33%) 12/18 (67%) 2007 50 37 (74%) 6/13 (46%) 7/13 (54%) 186 135 (73%) 13/51 (25%) 38/51 (75%)
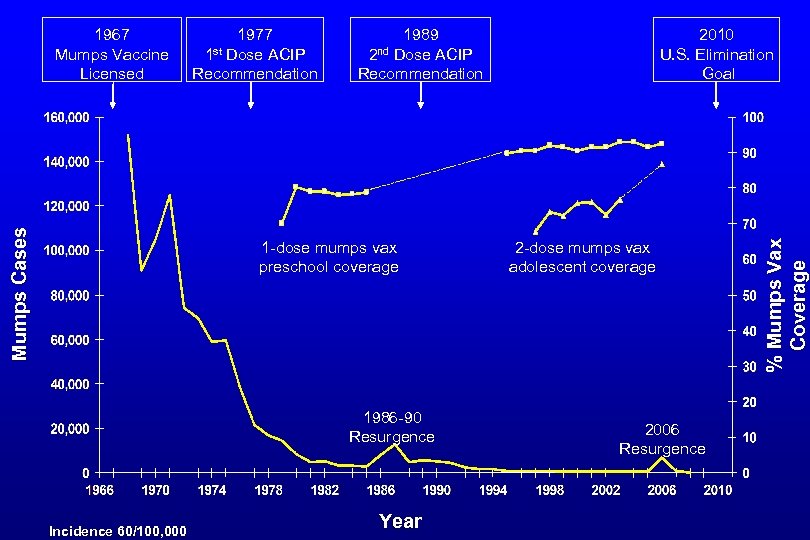
1977 Dose ACIP Recommendation 1 st 1989 Dose ACIP Recommendation 1 -dose mumps vax preschool coverage 1986 -90 Resurgence Incidence 60/100, 000 2010 U. S. Elimination Goal 2 nd Year 2 -dose mumps vax adolescent coverage 2006 Resurgence % Mumps Vax Coverage Mumps Cases 1967 Mumps Vaccine Licensed
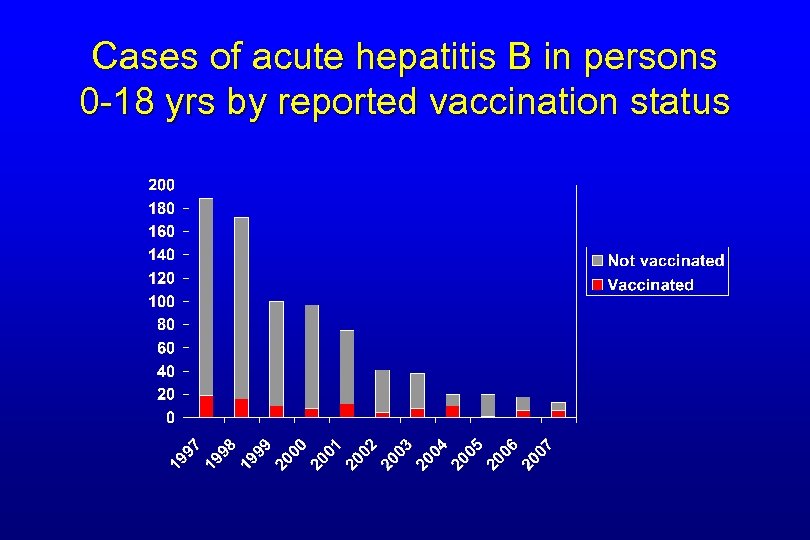
Cases of acute hepatitis B in persons 0 -18 yrs by reported vaccination status

HZ surveillance: how? • We do not anticipate that HZ will become notifiable • Surveys: NHIS, BRFSS, Health & Retirement Survey (HRS), adult module of National Immunization Survey • Administrative databases (Medicare, Medstat, VSD, other) § Enormous catchment for surveillance and analysis § However these data are not validated nor can they readily track impacts such as PHN or pain • Active surveillance sites § Varicella Active Surveillance Program (Antelope Valley, W. Philadelphia)

Herpes Zoster Surveillance: why? • Herpes Zoster is now vaccine preventable § Need to monitor the vaccination program’s effectiveness – Against HZ-related outcomes (trends of HZ, burden of HZ-related outcomes e. g. pain, PHN, hospitalizations) – Gaps in program (e. g. , high rates in oldest old >80) § Need to monitor individual-level vaccine efficacy – Against various HZ-related outcomes – Duration of protection – Effectiveness in specific subgroups (e. g. diabetics) • Need to understand trends in HZ rates (controlled for age) § To interpret surveillance & impact of vaccine program § Understand reasons for apparent increase (& intervene) § Identify risk groups that might be targeted for vaccine
a4a5aa5830443fe99b380105d40fba07.ppt