bc4076ea271fe61bbf0f7c9960ee3d50.ppt
- Количество слайдов: 42
 Welcome to: CHEMISTRY The Study of the Universe!
Welcome to: CHEMISTRY The Study of the Universe!
 When Doing Labs • Use the Scientific Method – accepted method for scientists to explain how things work • Steps: 1. State Problem and Collect Data 2. Formulate Hypothesis 3. Perform Experiments
When Doing Labs • Use the Scientific Method – accepted method for scientists to explain how things work • Steps: 1. State Problem and Collect Data 2. Formulate Hypothesis 3. Perform Experiments
![Step 1: Collecting Data [Two Types] Quality vs. Quantity? Qualitative (Quality) Quantitative (Quantity) NO Step 1: Collecting Data [Two Types] Quality vs. Quantity? Qualitative (Quality) Quantitative (Quantity) NO](https://present5.com/presentation/bc4076ea271fe61bbf0f7c9960ee3d50/image-3.jpg) Step 1: Collecting Data [Two Types] Quality vs. Quantity? Qualitative (Quality) Quantitative (Quantity) NO NUMBERS involved Ex. The sky is blue Ex. The solution is 34. 50 grams Ex. The solution is cloudy Ex. Water boils at 100 degrees
Step 1: Collecting Data [Two Types] Quality vs. Quantity? Qualitative (Quality) Quantitative (Quantity) NO NUMBERS involved Ex. The sky is blue Ex. The solution is 34. 50 grams Ex. The solution is cloudy Ex. Water boils at 100 degrees
 Step 2: Hypothesis • Hypothesis – a POSSIBLE explanation for why something happens • Observations are NOT hypotheses • Ex. the solution is cloudy (observation) • Ex. the solution is cloudy because it is contaminated (hypothesis)
Step 2: Hypothesis • Hypothesis – a POSSIBLE explanation for why something happens • Observations are NOT hypotheses • Ex. the solution is cloudy (observation) • Ex. the solution is cloudy because it is contaminated (hypothesis)
 Step 3: Experiments Use controls and variables Control – the constant Variable – the thing that changes in the experiment Lake vs. Ocean Perform multiple Trials – attempts at experiment
Step 3: Experiments Use controls and variables Control – the constant Variable – the thing that changes in the experiment Lake vs. Ocean Perform multiple Trials – attempts at experiment
 When Measuring Liquids… Meniscus – curve at upper surface of liquid Measure liquids HERE
When Measuring Liquids… Meniscus – curve at upper surface of liquid Measure liquids HERE
 Quick Quiz #1 The scientist notes that there are 42 eggs in the basket. Is this a qualitative or quantitative measurement? What is a variable? When measuring volumes of liquids, what is the curved line that you use?
Quick Quiz #1 The scientist notes that there are 42 eggs in the basket. Is this a qualitative or quantitative measurement? What is a variable? When measuring volumes of liquids, what is the curved line that you use?
 Lab Safety
Lab Safety
 Types of Chemicals Flammables and Combustibles – chemicals that can catch on fire Corrosives – things that damage/burn through skin tissue Pressure – chemicals that are dangerous at different pressures
Types of Chemicals Flammables and Combustibles – chemicals that can catch on fire Corrosives – things that damage/burn through skin tissue Pressure – chemicals that are dangerous at different pressures
 Flammables Ex. Bunsen Burners (used in this lab!) Precaution: WEAR GOGGLES! Similar but NOT the same as COMBUSTABLES – catch fire at HIGH temperatures
Flammables Ex. Bunsen Burners (used in this lab!) Precaution: WEAR GOGGLES! Similar but NOT the same as COMBUSTABLES – catch fire at HIGH temperatures
 Handling Boiling/Hot Chemicals DON’T TOUCH WITH BARE HANDS! USE TONGS or “Hot Hands”
Handling Boiling/Hot Chemicals DON’T TOUCH WITH BARE HANDS! USE TONGS or “Hot Hands”
 Corrosives = acids and bases that can damage skin Precautions: WEAR GLOVES, PROTECTIVE CLOTHING (longsleeves) AND GOGGLES
Corrosives = acids and bases that can damage skin Precautions: WEAR GLOVES, PROTECTIVE CLOTHING (longsleeves) AND GOGGLES
 If Corrosive Chemicals Get On Skin… RINSE IMMEDIATELY continuous running water (NOT SOAP) Eyes – RINSE IMMEDIATELY for at least 5 minutes under eye wash.
If Corrosive Chemicals Get On Skin… RINSE IMMEDIATELY continuous running water (NOT SOAP) Eyes – RINSE IMMEDIATELY for at least 5 minutes under eye wash.
 Broken Glass Accidents happen (no, really) Broken glass (beakers, test tubes, thermometers, etc) LET ME KNOW ASAP! Move away from station until glass is cleared
Broken Glass Accidents happen (no, really) Broken glass (beakers, test tubes, thermometers, etc) LET ME KNOW ASAP! Move away from station until glass is cleared
 Warm Up #5
Warm Up #5
 The Metric System Converting Units
The Metric System Converting Units
 Amurrica vs. The World Amurrica The Rest of the World Distance: Inches, Feet, Miles Distance: Millimeters, Meters, Kilometers Volume: Cups, Quarts, Gallons Volume: Milliliters, Liters, Kilometers Mass: Ounces, Pounds, Tons Mass: Milligrams, Grams, Kilograms WHY DOES THIS STILL EXIST? THIS IS WAY EASIER.
Amurrica vs. The World Amurrica The Rest of the World Distance: Inches, Feet, Miles Distance: Millimeters, Meters, Kilometers Volume: Cups, Quarts, Gallons Volume: Milliliters, Liters, Kilometers Mass: Ounces, Pounds, Tons Mass: Milligrams, Grams, Kilograms WHY DOES THIS STILL EXIST? THIS IS WAY EASIER.
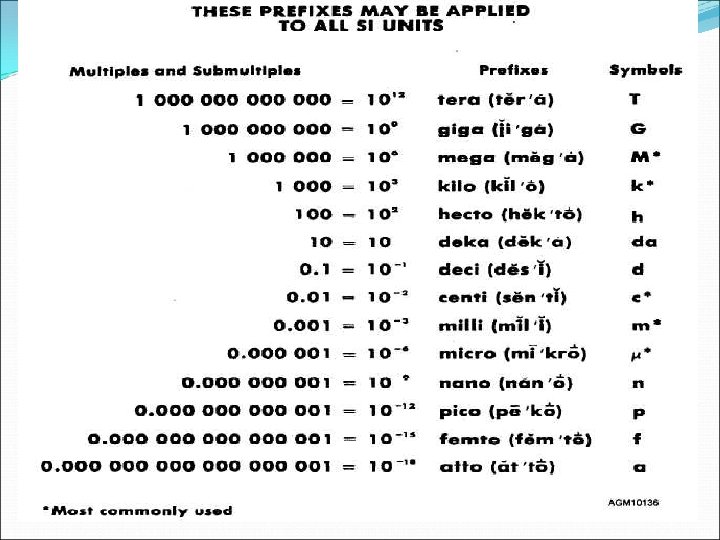 The Metric System Prefixes
The Metric System Prefixes
 Going from BIG to small Ex: 145. 8 Decimeters (dm) into Kilometers (km) FOUR decimal places from DECI to KILO Move Decimal FOUR PLACES LEFT 145. 8 dm = 0. 01458
Going from BIG to small Ex: 145. 8 Decimeters (dm) into Kilometers (km) FOUR decimal places from DECI to KILO Move Decimal FOUR PLACES LEFT 145. 8 dm = 0. 01458
 Quick Quiz #3 Convert the following: 34. 67 m. L = ____ L? 4. 903 hg = _____ dg? John travelled a distance of 290480000 millimeters. How many kilometers is that?
Quick Quiz #3 Convert the following: 34. 67 m. L = ____ L? 4. 903 hg = _____ dg? John travelled a distance of 290480000 millimeters. How many kilometers is that?
 Density and Speed Density: Concentration of a substance in a given space Mass over Volume (m/v) Ex. g/m. L, kg/L, etc. Speed: rate at which an object moves Distance over Time (d/t) Ex. m/s, km/hr, etc.
Density and Speed Density: Concentration of a substance in a given space Mass over Volume (m/v) Ex. g/m. L, kg/L, etc. Speed: rate at which an object moves Distance over Time (d/t) Ex. m/s, km/hr, etc.
 Dimensional Analysis Intro. Problem #1: Karla has a substance with a density of 4. 68 g/m. L. How many g/c. L is that? Problem #2: James was measuring the speed of a car in meters per second. If he found that a car was moving at the speed of 5. 50 m/s, how many km/hr is that?
Dimensional Analysis Intro. Problem #1: Karla has a substance with a density of 4. 68 g/m. L. How many g/c. L is that? Problem #2: James was measuring the speed of a car in meters per second. If he found that a car was moving at the speed of 5. 50 m/s, how many km/hr is that?
 On your Calculator: 500 x 500 = 250000. (that’s a big number!) . 005 x. 005 = 2. 5 -05 (WTF? ) Why does the second answer look like this?
On your Calculator: 500 x 500 = 250000. (that’s a big number!) . 005 x. 005 = 2. 5 -05 (WTF? ) Why does the second answer look like this?
 Scientific Notation – way of making REALLY big or REALLY small numbers easy to read. General Form: Mx n 10 M = number between 1 and 10 n = number of decimal places you move
Scientific Notation – way of making REALLY big or REALLY small numbers easy to read. General Form: Mx n 10 M = number between 1 and 10 n = number of decimal places you move


 Quick Quiz #1 Multiply 123456 x 7890 on your calculator 1. What was your answer? 2. Write your answer in scientific notation. Do the same for the following problems: 1. 98765 x 43210 = ? 2. 10000 x 892800 = ? 3. 90 x 9 = ?
Quick Quiz #1 Multiply 123456 x 7890 on your calculator 1. What was your answer? 2. Write your answer in scientific notation. Do the same for the following problems: 1. 98765 x 43210 = ? 2. 10000 x 892800 = ? 3. 90 x 9 = ?
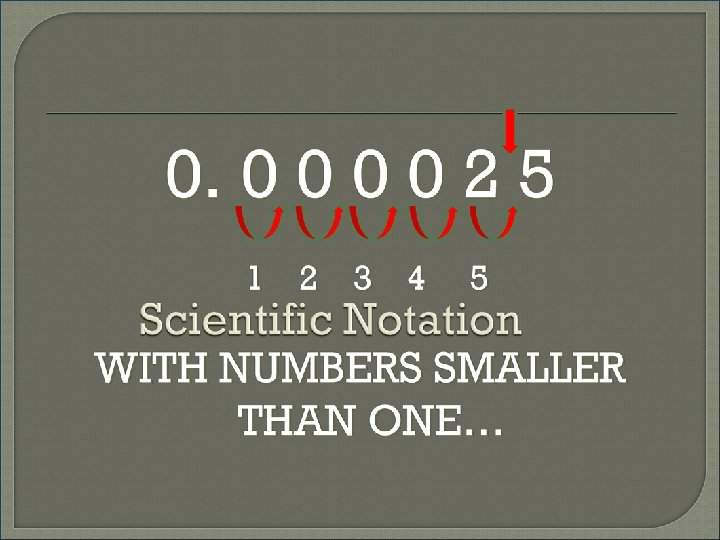

 Review: WITH NUMBERS WAY BIGGER THAN 1 MOVE DECIMAL TO THE LEFT n VALUE WILL BE A POSITIVE NUMBER WITH NUMBERS WAY SMALLER THAN 1 MOVE DECIMAL TO THE RIGHT n VALUE WILL BE A NEGATIVE NUMBER
Review: WITH NUMBERS WAY BIGGER THAN 1 MOVE DECIMAL TO THE LEFT n VALUE WILL BE A POSITIVE NUMBER WITH NUMBERS WAY SMALLER THAN 1 MOVE DECIMAL TO THE RIGHT n VALUE WILL BE A NEGATIVE NUMBER
 Dimensional Analysis Part 1
Dimensional Analysis Part 1
 What does that mean? Dimensional Analysis – Converting one thing to another Used in chemistry (converting units of mass/distance/volume) BUT YOU DO THIS IN REAL LIFE ALL THE TIME
What does that mean? Dimensional Analysis – Converting one thing to another Used in chemistry (converting units of mass/distance/volume) BUT YOU DO THIS IN REAL LIFE ALL THE TIME
 Convert the following: How many seconds in a minute? 1 minute = ___ seconds How many quarters in a dollar? 1 dollar = ___ quarters How many points do you score in a touchdown? 1 touchdown = ___ points 1 min. 60 sec. $1 OR OR OR 60 sec. 1 min. $1
Convert the following: How many seconds in a minute? 1 minute = ___ seconds How many quarters in a dollar? 1 dollar = ___ quarters How many points do you score in a touchdown? 1 touchdown = ___ points 1 min. 60 sec. $1 OR OR OR 60 sec. 1 min. $1
 Example Problem Julia measured the length of Catherine’s hair. She found out that her hair was 24 inches long. How many feet is that? First ask yourself: What is your starting amount? What are you converting to? How many inches are in a foot?
Example Problem Julia measured the length of Catherine’s hair. She found out that her hair was 24 inches long. How many feet is that? First ask yourself: What is your starting amount? What are you converting to? How many inches are in a foot?
 Reminder: UNITS DO NOT LEAVE ANY NUMBER NAKED! IT NEEDS A UNIT. GOAL: CANCELLING OUT UNITS One unit label on top, the other on bottom Feet will cancel out (Feet x Feet) = Feet 2 Feet
Reminder: UNITS DO NOT LEAVE ANY NUMBER NAKED! IT NEEDS A UNIT. GOAL: CANCELLING OUT UNITS One unit label on top, the other on bottom Feet will cancel out (Feet x Feet) = Feet 2 Feet
 System to Do This: DIMENSIONAL ANALYSIS Starting amount, draw a line underneath it. Draw a vertical line next to it. One unit on top, other on bottom Multiply stuff on top Multiply stuff on bottom Divide Top and Bottom
System to Do This: DIMENSIONAL ANALYSIS Starting amount, draw a line underneath it. Draw a vertical line next to it. One unit on top, other on bottom Multiply stuff on top Multiply stuff on bottom Divide Top and Bottom
 Quick Quiz #2 Convert the following using Dimensional Analysis: 12 feet = ___ inches? (1 foot = 12 inches) 9 touchdowns = ___ points? (1 TD = 6 points) 4 apples = ___ oranges? (1 apple = 3 oranges)
Quick Quiz #2 Convert the following using Dimensional Analysis: 12 feet = ___ inches? (1 foot = 12 inches) 9 touchdowns = ___ points? (1 TD = 6 points) 4 apples = ___ oranges? (1 apple = 3 oranges)
 Word Problems Read the problems carefully Underline important information (things with numbers) Know what units you start with Know what units you need to end with
Word Problems Read the problems carefully Underline important information (things with numbers) Know what units you start with Know what units you need to end with
 Example Problem Steven was REALLY thirsty, so he wanted to buy some water bottles from Mr. Lockett. He ended up buying 4 water bottles. If each water bottle costs $2, how much money (in dollars) did he spend?
Example Problem Steven was REALLY thirsty, so he wanted to buy some water bottles from Mr. Lockett. He ended up buying 4 water bottles. If each water bottle costs $2, how much money (in dollars) did he spend?
 Underline Important Info. Steven was REALLY thirsty, so he wanted to buy some water bottles from Mr. Lockett. He ended up buying 4 water bottles. If each water bottle costs $2, how much money (in dollars) did he spend?
Underline Important Info. Steven was REALLY thirsty, so he wanted to buy some water bottles from Mr. Lockett. He ended up buying 4 water bottles. If each water bottle costs $2, how much money (in dollars) did he spend?
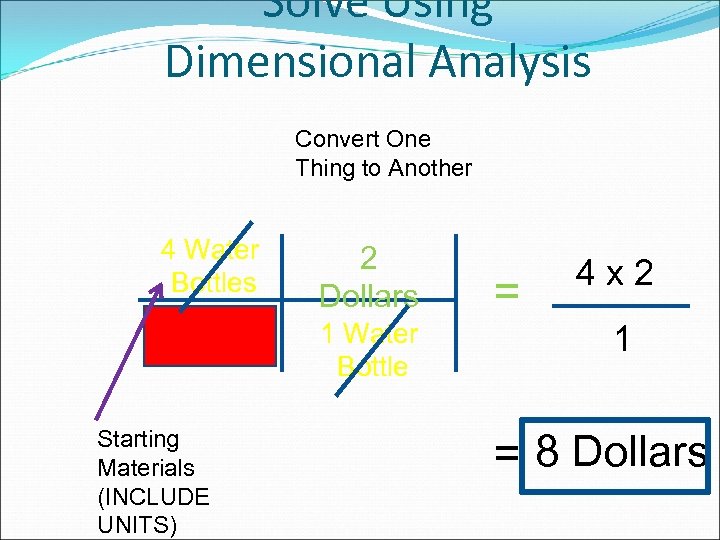 Solve Using Dimensional Analysis Convert One Thing to Another 4 Water Bottles 2 Dollars 1 Water Bottle Starting Materials (INCLUDE UNITS) = 4 x 2 1 = 8 Dollars
Solve Using Dimensional Analysis Convert One Thing to Another 4 Water Bottles 2 Dollars 1 Water Bottle Starting Materials (INCLUDE UNITS) = 4 x 2 1 = 8 Dollars
 Quick Quiz #3: SOLVE USING DIMENSIONAL ANALYSIS 1. Guadalupe was bored and wanted class to be over. Mr. Lockett said class was over in 90 minutes. How many hours is that? (1 hour = 60 minutes) 2. For some reason Esteban really wanted quarters. He gives the cashier a 5 dollar bill. How many quarters will he get from this 5 dollar bill? (1 dollar = 4 quarters)
Quick Quiz #3: SOLVE USING DIMENSIONAL ANALYSIS 1. Guadalupe was bored and wanted class to be over. Mr. Lockett said class was over in 90 minutes. How many hours is that? (1 hour = 60 minutes) 2. For some reason Esteban really wanted quarters. He gives the cashier a 5 dollar bill. How many quarters will he get from this 5 dollar bill? (1 dollar = 4 quarters)


