Вирусные гепатиты.ppt
- Количество слайдов: 49
 ВИРУСНЫЕ ГЕПАТИТЫ Этиология, патогенез, иммунитет, профилактика гепатита А. Вирусы гепатитов В, С, Д, Е, G. Систематическое положение. Характеристика вирусов и вызываемых ими заболеваний. Профилактика парентеральных вирусных гепатитов.
ВИРУСНЫЕ ГЕПАТИТЫ Этиология, патогенез, иммунитет, профилактика гепатита А. Вирусы гепатитов В, С, Д, Е, G. Систематическое положение. Характеристика вирусов и вызываемых ими заболеваний. Профилактика парентеральных вирусных гепатитов.
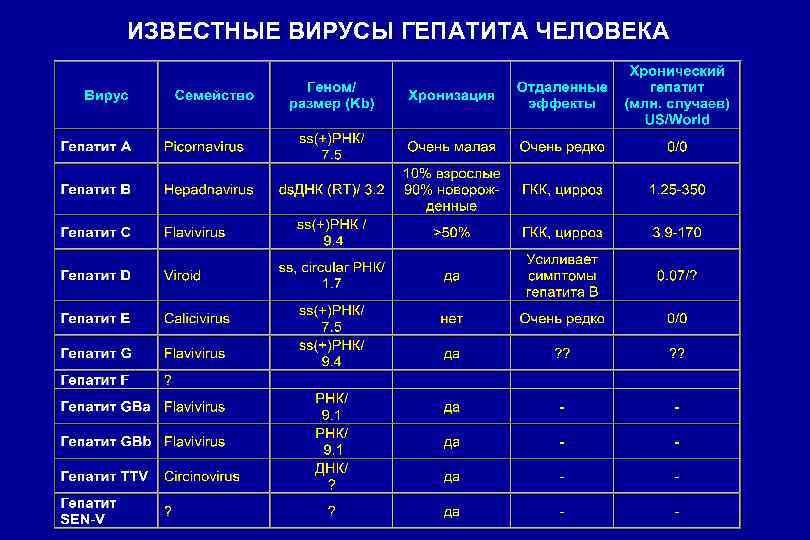 ИЗВЕСТНЫЕ ВИРУСЫ ГЕПАТИТА ЧЕЛОВЕКА
ИЗВЕСТНЫЕ ВИРУСЫ ГЕПАТИТА ЧЕЛОВЕКА
 ГЕНОТИПИРОВАНИЕ НЕКОТОРЫХ ВИРУСОВ Гепатит А 7 генотипов (1, 2, 3 и 7 - инфекционные для человека) Гепатит В 6 -7 генотипов, 4 субтипа и мутантные формы ВГВ (мутации в S-, С-, Х- и Р-генах генома HBV) Гепатит С 1 -11 генотипов, >100 субтипов (наиболее распространенные 1 а, 1 b, 2 a, 2 b и 3 а)
ГЕНОТИПИРОВАНИЕ НЕКОТОРЫХ ВИРУСОВ Гепатит А 7 генотипов (1, 2, 3 и 7 - инфекционные для человека) Гепатит В 6 -7 генотипов, 4 субтипа и мутантные формы ВГВ (мутации в S-, С-, Х- и Р-генах генома HBV) Гепатит С 1 -11 генотипов, >100 субтипов (наиболее распространенные 1 а, 1 b, 2 a, 2 b и 3 а)
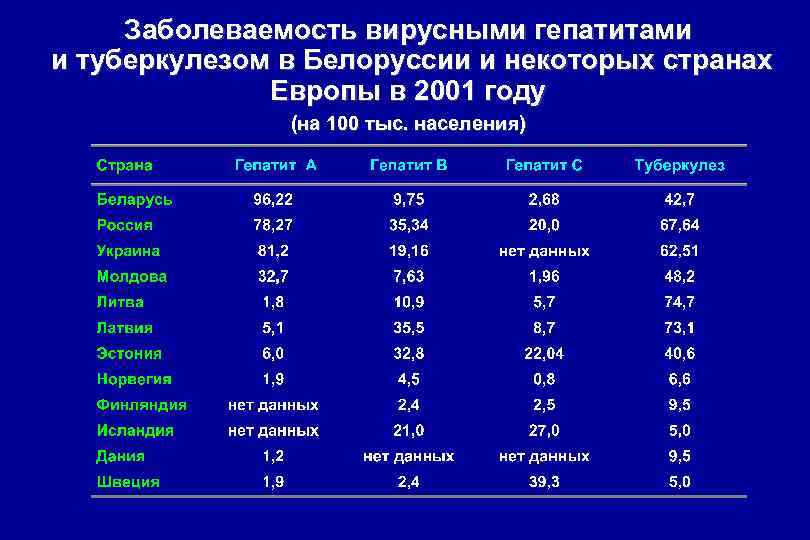 Заболеваемость вирусными гепатитами и туберкулезом в Белоруссии и некоторых странах Европы в 2001 году (на 100 тыс. населения)
Заболеваемость вирусными гепатитами и туберкулезом в Белоруссии и некоторых странах Европы в 2001 году (на 100 тыс. населения)
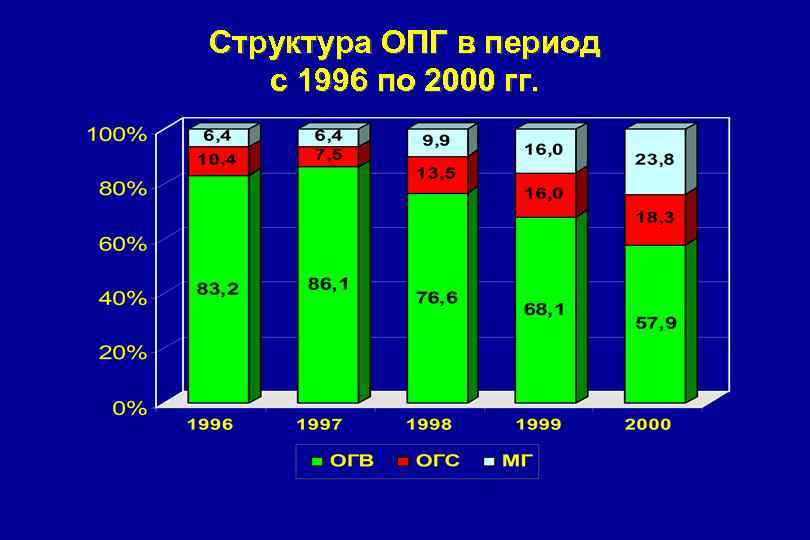 Структура ОПГ в период с 1996 по 2000 гг.
Структура ОПГ в период с 1996 по 2000 гг.
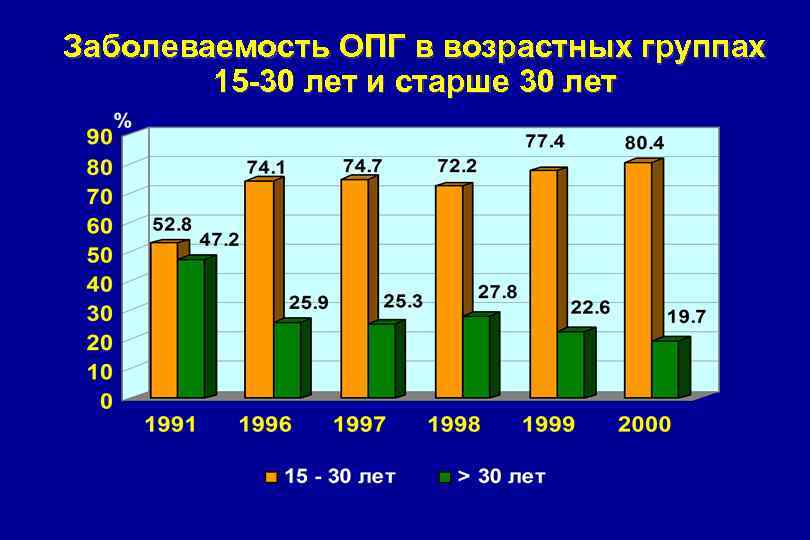 Заболеваемость ОПГ в возрастных группах 15 -30 лет и старше 30 лет
Заболеваемость ОПГ в возрастных группах 15 -30 лет и старше 30 лет
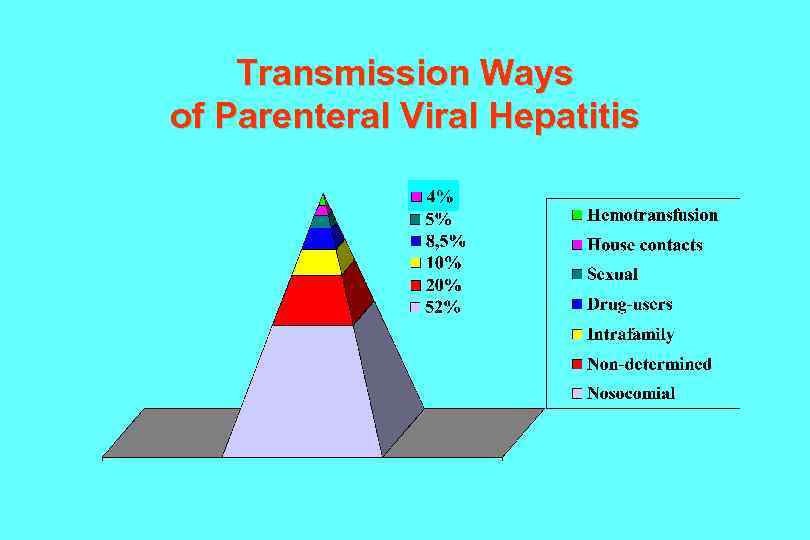 Transmission Ways of Parenteral Viral Hepatitis
Transmission Ways of Parenteral Viral Hepatitis
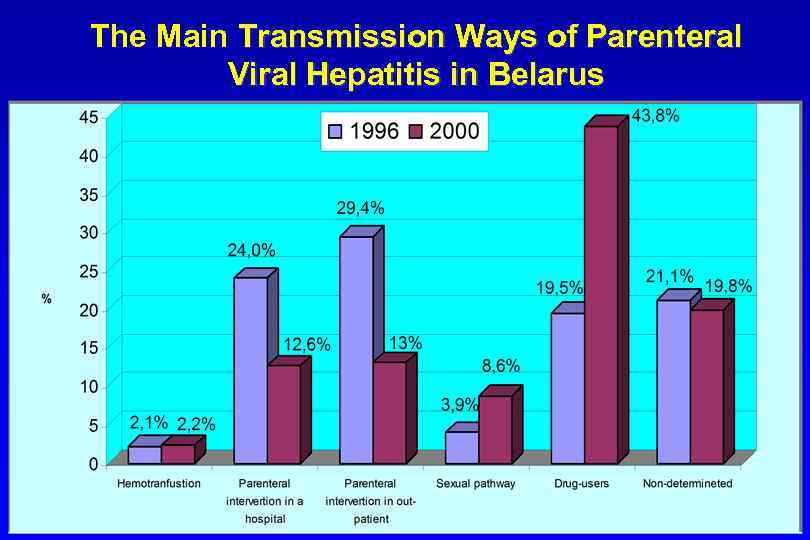 The Main Transmission Ways of Parenteral Viral Hepatitis in Belarus
The Main Transmission Ways of Parenteral Viral Hepatitis in Belarus
 ВИРУСНЫЙ ГЕПАТИТ А
ВИРУСНЫЙ ГЕПАТИТ А
 HEPATITIS A VIRUS
HEPATITIS A VIRUS
 HEPATITIS A VIRUS • RNA Picornavirus · Single serotype worldwide · Acute disease and asymptomatic infection • No chronic infection · Protective antibodies develop in response to infection - confers lifelong immunity
HEPATITIS A VIRUS • RNA Picornavirus · Single serotype worldwide · Acute disease and asymptomatic infection • No chronic infection · Protective antibodies develop in response to infection - confers lifelong immunity
 HEPATITIS A - CLINICAL FEATURES • Jaundice by age group: <6 yrs 6 -14 yrs >14 yrs <10% 40%-50% 70%-80% • Rare complications: Fulminant hepatitis Cholestatic hepatitis Relapsing hepatitis • Incubation period: Average 30 days Range 15 -50 days • Chronic sequelae: None
HEPATITIS A - CLINICAL FEATURES • Jaundice by age group: <6 yrs 6 -14 yrs >14 yrs <10% 40%-50% 70%-80% • Rare complications: Fulminant hepatitis Cholestatic hepatitis Relapsing hepatitis • Incubation period: Average 30 days Range 15 -50 days • Chronic sequelae: None
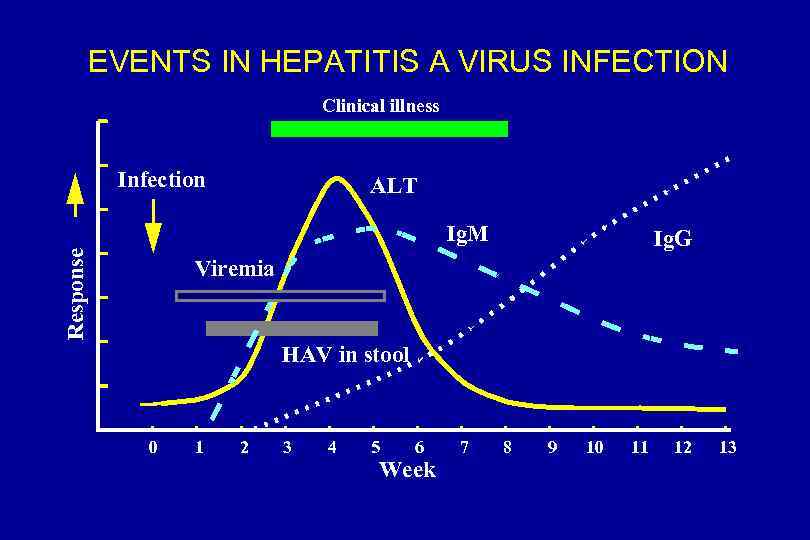 EVENTS IN HEPATITIS A VIRUS INFECTION Clinical illness Infection ALT Response Ig. M Ig. G Viremia HAV in stool 0 1 2 3 4 5 6 Week 7 8 9 10 11 12 13
EVENTS IN HEPATITIS A VIRUS INFECTION Clinical illness Infection ALT Response Ig. M Ig. G Viremia HAV in stool 0 1 2 3 4 5 6 Week 7 8 9 10 11 12 13
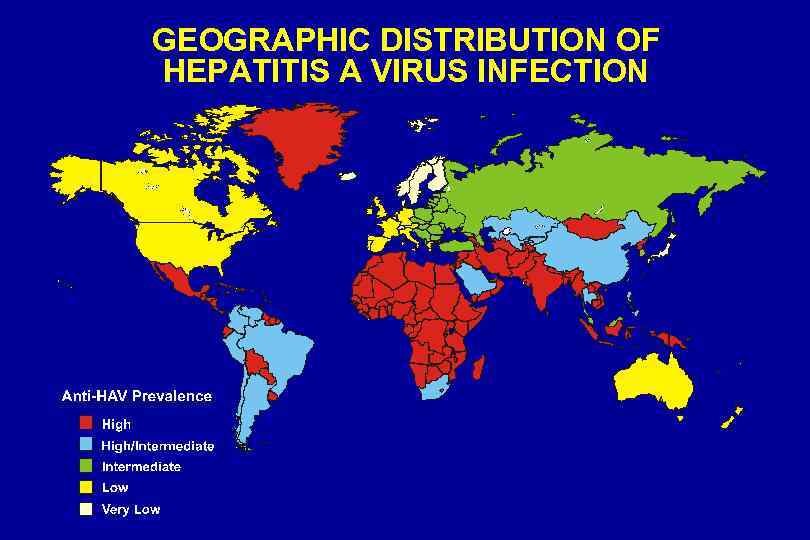 GEOGRAPHIC DISTRIBUTION OF HEPATITIS A VIRUS INFECTION
GEOGRAPHIC DISTRIBUTION OF HEPATITIS A VIRUS INFECTION
 HEPATITIS A, UNITED STATES l Most disease occurs in the context of community-wide outbreaks l Infection transmitted from person to person in households and extended family settings - facilitated by asymptomatic infection among children Some groups at increased risk – specific factor varies – do not account for majority of cases l l No risk factor identified for 40%-50% of cases
HEPATITIS A, UNITED STATES l Most disease occurs in the context of community-wide outbreaks l Infection transmitted from person to person in households and extended family settings - facilitated by asymptomatic infection among children Some groups at increased risk – specific factor varies – do not account for majority of cases l l No risk factor identified for 40%-50% of cases
 ACUTE HEPATITIS A CASE DEFINITION FOR SURVEILLANCE – Clinical criteria An acute illness with: • discrete onset of symptoms (e. g. fatigue, abdominal pain, loss of appetite, intermittent nausea, vomiting), and • jaundice or elevated serum aminotransferase levels – Laboratory criteria • Ig. M antibody to hepatitis A virus (anti-HAV) positive – Case Classification • Confirmed. A case that meets the clinical case definition and is laboratory confirmed or a case that meets the clinical case definition and occurs in a person who has an epidemiologic link with a person who has laboratory-confirmed hepatitis A (i. e. , household or sexual contact with an infected person during the 15 -50 days before the onset of symptoms).
ACUTE HEPATITIS A CASE DEFINITION FOR SURVEILLANCE – Clinical criteria An acute illness with: • discrete onset of symptoms (e. g. fatigue, abdominal pain, loss of appetite, intermittent nausea, vomiting), and • jaundice or elevated serum aminotransferase levels – Laboratory criteria • Ig. M antibody to hepatitis A virus (anti-HAV) positive – Case Classification • Confirmed. A case that meets the clinical case definition and is laboratory confirmed or a case that meets the clinical case definition and occurs in a person who has an epidemiologic link with a person who has laboratory-confirmed hepatitis A (i. e. , household or sexual contact with an infected person during the 15 -50 days before the onset of symptoms).
 HEPATITIS A VIRUS TRANSMISSION • Close personal contact (e. g. , household contact, sex contact, child daycare centers) • Contaminated food, water (e. g. , infected food handlers) • Blood exposure (rare) (e. g. , injection drug use, rarely by transfusion)
HEPATITIS A VIRUS TRANSMISSION • Close personal contact (e. g. , household contact, sex contact, child daycare centers) • Contaminated food, water (e. g. , infected food handlers) • Blood exposure (rare) (e. g. , injection drug use, rarely by transfusion)
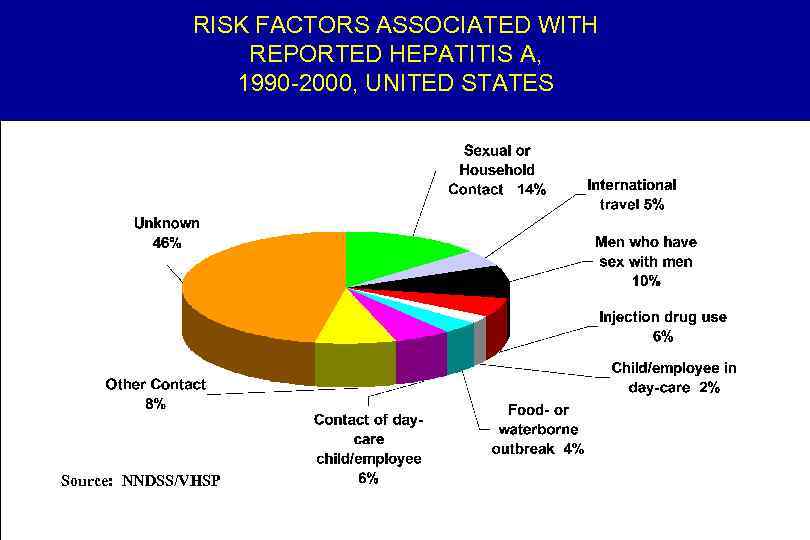 RISK FACTORS ASSOCIATED WITH REPORTED HEPATITIS A, 1990 -2000, UNITED STATES Source: NNDSS/VHSP
RISK FACTORS ASSOCIATED WITH REPORTED HEPATITIS A, 1990 -2000, UNITED STATES Source: NNDSS/VHSP
 PREVENTING HEPATITIS A • Hygiene (e. g. , hand washing) • Sanitation (e. g. , clean water sources) • Hepatitis A vaccine (pre-exposure) • Immune globulin (pre- and post-exposure)
PREVENTING HEPATITIS A • Hygiene (e. g. , hand washing) • Sanitation (e. g. , clean water sources) • Hepatitis A vaccine (pre-exposure) • Immune globulin (pre- and post-exposure)
 PREPARATION OF INACTIVATED HEPATITIS A VACCINES • Cell culture adapted virus grown in human fibroblasts • Purified product inactivated with formalin • Adsorbed to aluminum hydroxide adjuvant
PREPARATION OF INACTIVATED HEPATITIS A VACCINES • Cell culture adapted virus grown in human fibroblasts • Purified product inactivated with formalin • Adsorbed to aluminum hydroxide adjuvant
 HEPATITIS A VACCINES • Highly immunogenic • 97%-100% of children, adolescents, and adults have protective levels of antibody within 1 month of receiving first dose; essentially 100% have protective levels after second dose • Highly efficacious • In published studies, 94%-100% of children protected against clinical hepatitis A after equivalent of one dose
HEPATITIS A VACCINES • Highly immunogenic • 97%-100% of children, adolescents, and adults have protective levels of antibody within 1 month of receiving first dose; essentially 100% have protective levels after second dose • Highly efficacious • In published studies, 94%-100% of children protected against clinical hepatitis A after equivalent of one dose
 ВИРУСНЫЙ ГЕПАТИТ С
ВИРУСНЫЙ ГЕПАТИТ С
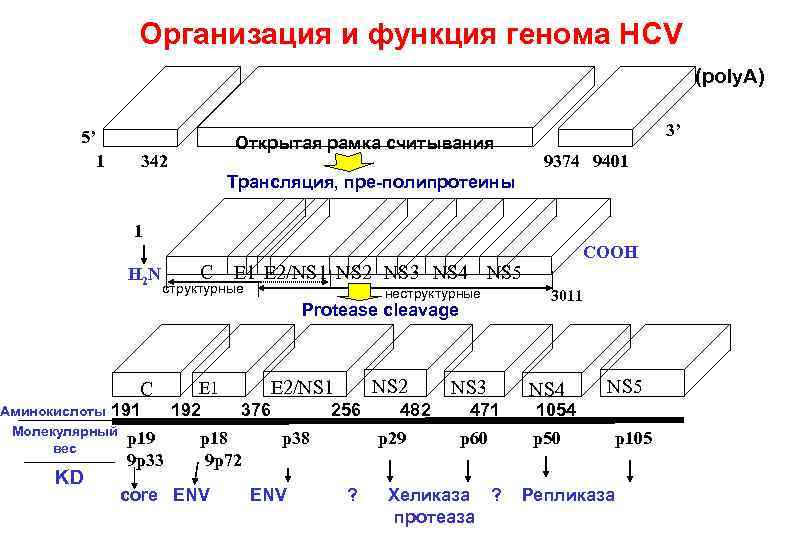 Организация и функция генома HCV (poly. A) 5’ 1 3’ Открытая рамка считывания 342 9374 9401 Трансляция, пре-полипротеины 1 H 2 N C E 1 E 2/NS 1 NS 2 NS 3 NS 4 NS 5 структурные неструктурные Protease cleavage C Аминокислоты 191 Молекулярный p 19 вес KD 9 p 33 E 1 192 NS 2 E 2/NS 1 376 p 18 9 p 72 core ENV COOH 256 p 38 ENV 482 p 29 ? NS 3 471 p 60 Хеликаза ? протеаза 3011 NS 4 NS 5 1054 p 50 p 105 Репликаза
Организация и функция генома HCV (poly. A) 5’ 1 3’ Открытая рамка считывания 342 9374 9401 Трансляция, пре-полипротеины 1 H 2 N C E 1 E 2/NS 1 NS 2 NS 3 NS 4 NS 5 структурные неструктурные Protease cleavage C Аминокислоты 191 Молекулярный p 19 вес KD 9 p 33 E 1 192 NS 2 E 2/NS 1 376 p 18 9 p 72 core ENV COOH 256 p 38 ENV 482 p 29 ? NS 3 471 p 60 Хеликаза ? протеаза 3011 NS 4 NS 5 1054 p 50 p 105 Репликаза
 Liver is the Main Site for Hepatitis С Virus Reproduction
Liver is the Main Site for Hepatitis С Virus Reproduction
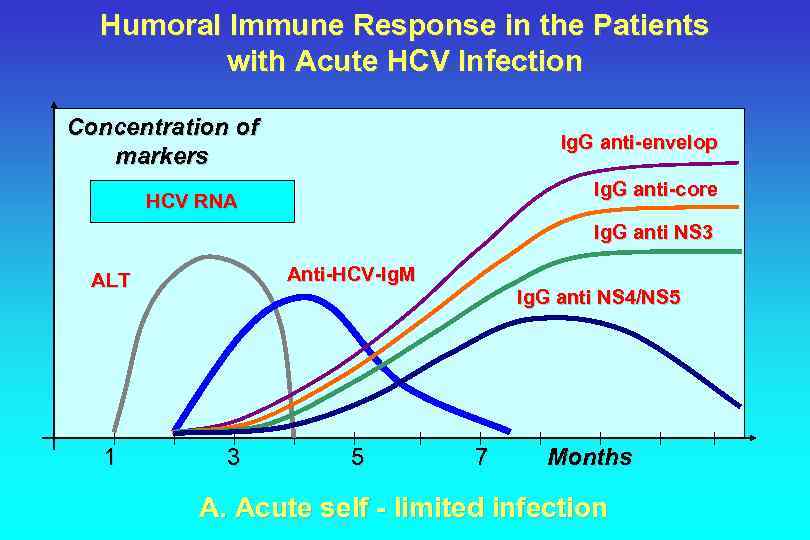 Humoral Immune Response in the Patients with Acute HCV Infection Concentration of markers Ig. G anti-envelop Ig. G anti-core HCV RNA Ig. G anti NS 3 Anti-HCV-Ig. M ALT 1 3 5 Ig. G anti NS 4/NS 5 7 Months A. Acute self - limited infection
Humoral Immune Response in the Patients with Acute HCV Infection Concentration of markers Ig. G anti-envelop Ig. G anti-core HCV RNA Ig. G anti NS 3 Anti-HCV-Ig. M ALT 1 3 5 Ig. G anti NS 4/NS 5 7 Months A. Acute self - limited infection
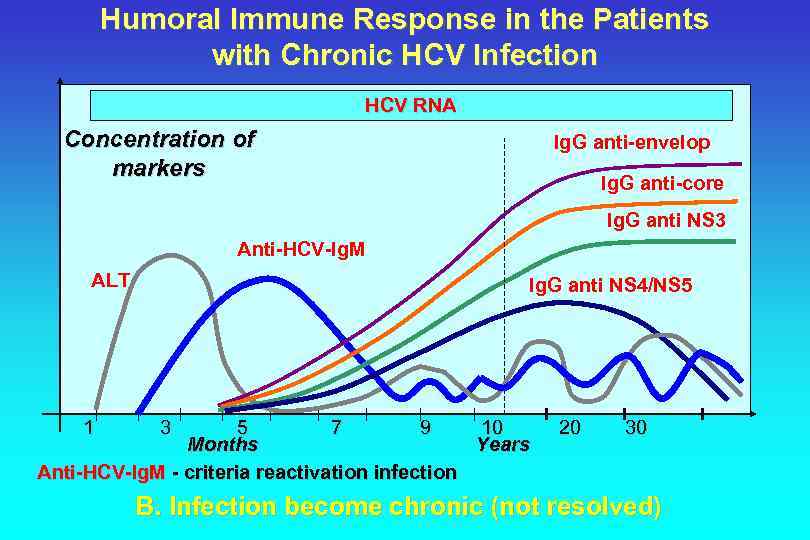 Humoral Immune Response in the Patients with Chronic HCV Infection HCV RNA Concentration of markers Ig. G anti-envelop Ig. G anti-core Ig. G anti NS 3 Anti-HCV-Ig. M ALT 1 Ig. G anti NS 4/NS 5 3 5 7 9 10 Months Years Anti-HCV-Ig. M - criteria reactivation infection 20 30 B. Infection become chronic (not resolved)
Humoral Immune Response in the Patients with Chronic HCV Infection HCV RNA Concentration of markers Ig. G anti-envelop Ig. G anti-core Ig. G anti NS 3 Anti-HCV-Ig. M ALT 1 Ig. G anti NS 4/NS 5 3 5 7 9 10 Months Years Anti-HCV-Ig. M - criteria reactivation infection 20 30 B. Infection become chronic (not resolved)
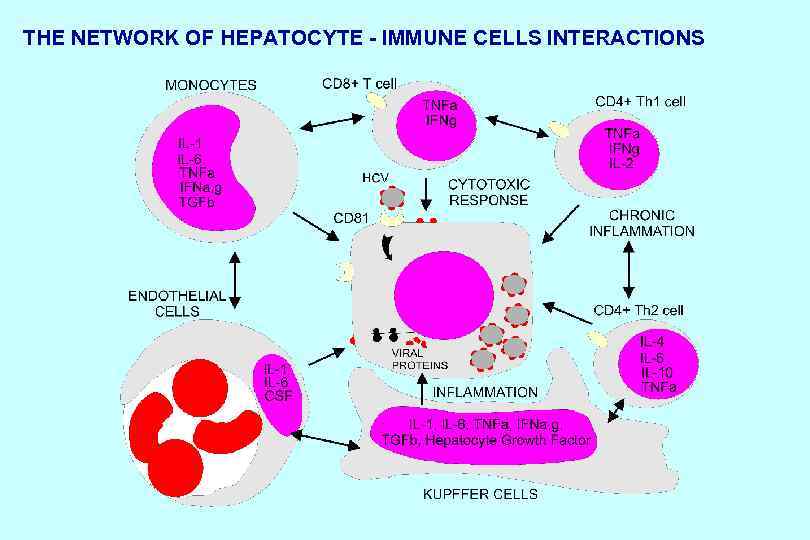 THE NETWORK OF HEPATOCYTE - IMMUNE CELLS INTERACTIONS
THE NETWORK OF HEPATOCYTE - IMMUNE CELLS INTERACTIONS
 Intrahepatic CD 4+T- Lymphocytes and Cytokines Production in Patients with Viral Hepatitis C
Intrahepatic CD 4+T- Lymphocytes and Cytokines Production in Patients with Viral Hepatitis C
 INTRAHEPATIC CD 8+ T-LYMPHOCYTES IN PATIENTS WITH HEPATITIS
INTRAHEPATIC CD 8+ T-LYMPHOCYTES IN PATIENTS WITH HEPATITIS
 ROLE OF THE DENDRITIC CELLS IN VIRAL HEPATITIS C
ROLE OF THE DENDRITIC CELLS IN VIRAL HEPATITIS C
 Apoptosis of Intrahepatic T-Lymphocytes in the Patients with Chronic Hepatitis C
Apoptosis of Intrahepatic T-Lymphocytes in the Patients with Chronic Hepatitis C
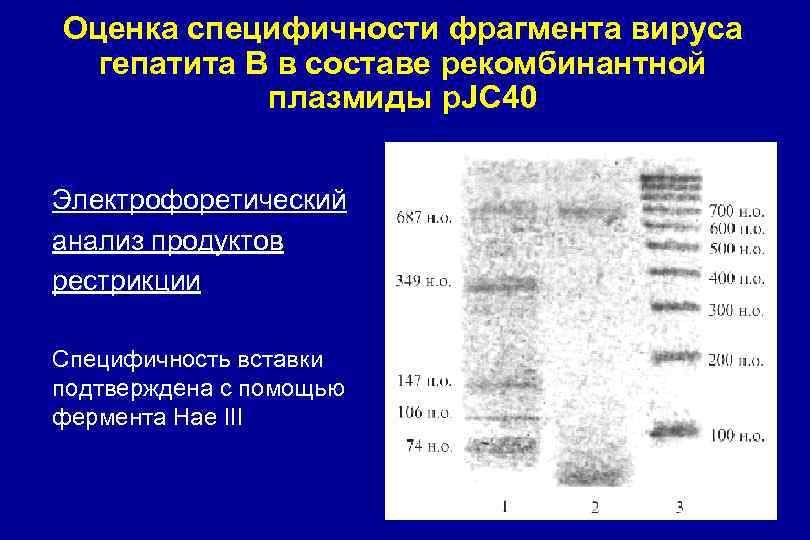 Оценка специфичности фрагмента вируса гепатита В в составе рекомбинантной плазмиды p. JC 40 Электрофоретический анализ продуктов рестрикции Специфичность вставки подтверждена с помощью фермента Hae III
Оценка специфичности фрагмента вируса гепатита В в составе рекомбинантной плазмиды p. JC 40 Электрофоретический анализ продуктов рестрикции Специфичность вставки подтверждена с помощью фермента Hae III
 Индикация гепатита С методом ПЦР • Проведен молекулярнобиологический анализ изолятов вируса гепатита С, циркулирующих на территории РБ. • В Республике Беларусь циркулируют 1 b и 3 а генотипы.
Индикация гепатита С методом ПЦР • Проведен молекулярнобиологический анализ изолятов вируса гепатита С, циркулирующих на территории РБ. • В Республике Беларусь циркулируют 1 b и 3 а генотипы.
 Особенности течения вирусного гепатита С Инкубационный в среднем 6 -7 недель период диапазон 2 -26 недель Острая болезнь (желтуха) умеренная (<20%) Летальность низкая Хроническая инфекция 60%-85% Хронический гепатит Возраст 10%-70% зависимые Цирроз <5%-20% Смертность от CLD 1%-5% (most asx)
Особенности течения вирусного гепатита С Инкубационный в среднем 6 -7 недель период диапазон 2 -26 недель Острая болезнь (желтуха) умеренная (<20%) Летальность низкая Хроническая инфекция 60%-85% Хронический гепатит Возраст 10%-70% зависимые Цирроз <5%-20% Смертность от CLD 1%-5% (most asx)
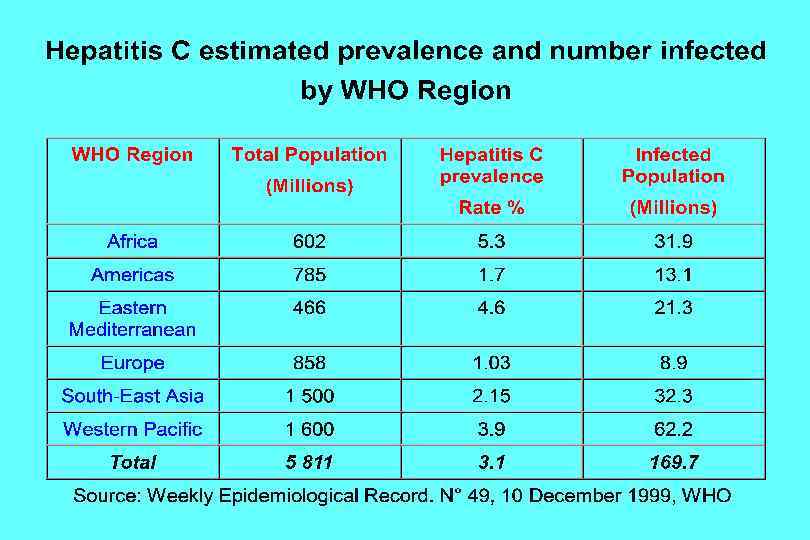
 Hepatitis C Virus Infection, United States New infections per year 1985 -89 2001 25, 000 242, 000 Deaths from acute liver failure Rare Persons ever infected (1. 8%) 3. 9 million (3. 1 -4. 8)* Persons with chronic infection 2. 7 million (2. 4 -3. 0)* HCV-related chronic liver disease 40% - 60% Deaths from chronic disease/year 8, 000 -10, 000 *95% Confidence Interval
Hepatitis C Virus Infection, United States New infections per year 1985 -89 2001 25, 000 242, 000 Deaths from acute liver failure Rare Persons ever infected (1. 8%) 3. 9 million (3. 1 -4. 8)* Persons with chronic infection 2. 7 million (2. 4 -3. 0)* HCV-related chronic liver disease 40% - 60% Deaths from chronic disease/year 8, 000 -10, 000 *95% Confidence Interval
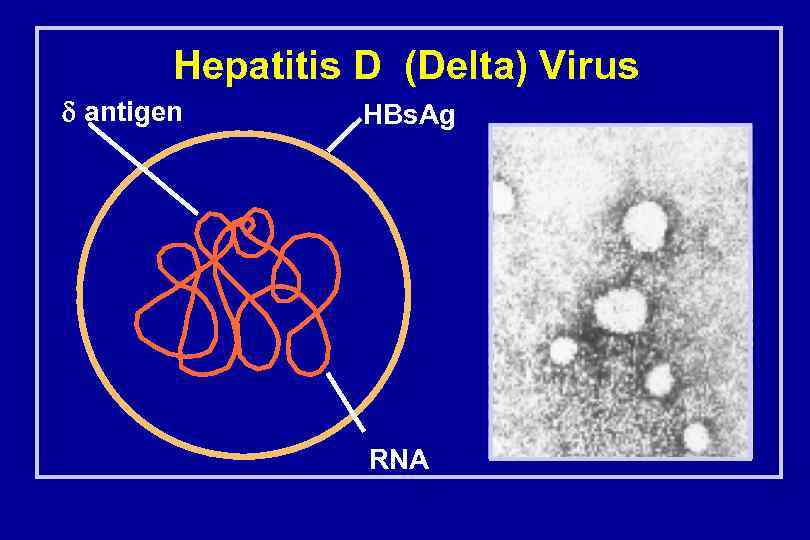 Hepatitis D (Delta) Virus antigen HBs. Ag RNA
Hepatitis D (Delta) Virus antigen HBs. Ag RNA
 Hepatitis D - Clinical Features • Coinfection – severe acute disease – low risk of chronic infection • Superinfection – usually develop chronic HDV infection – high risk of severe chronic liver disease
Hepatitis D - Clinical Features • Coinfection – severe acute disease – low risk of chronic infection • Superinfection – usually develop chronic HDV infection – high risk of severe chronic liver disease
 Hepatitis D Virus Modes of Transmission • Percutanous exposures –injecting drug use • Permucosal exposures –sex contact
Hepatitis D Virus Modes of Transmission • Percutanous exposures –injecting drug use • Permucosal exposures –sex contact
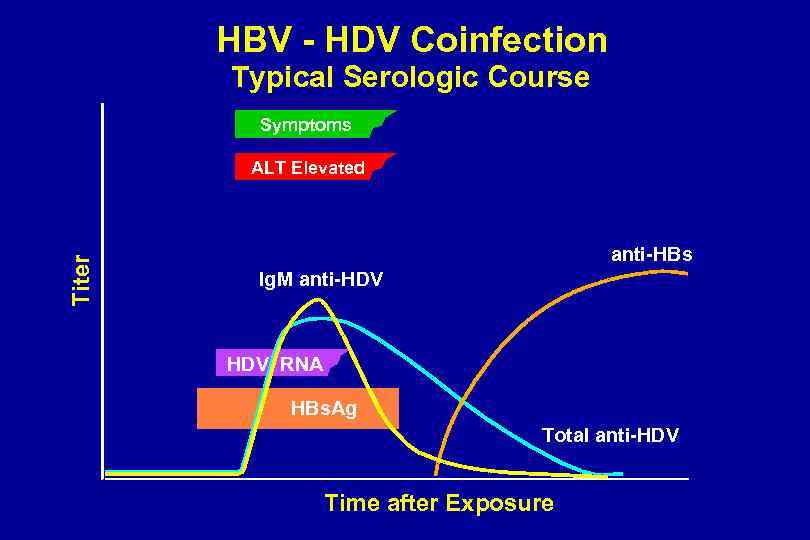 HBV - HDV Coinfection Typical Serologic Course Symptoms Titer ALT Elevated anti-HBs Ig. M anti-HDV RNA HBs. Ag Total anti-HDV Time after Exposure
HBV - HDV Coinfection Typical Serologic Course Symptoms Titer ALT Elevated anti-HBs Ig. M anti-HDV RNA HBs. Ag Total anti-HDV Time after Exposure
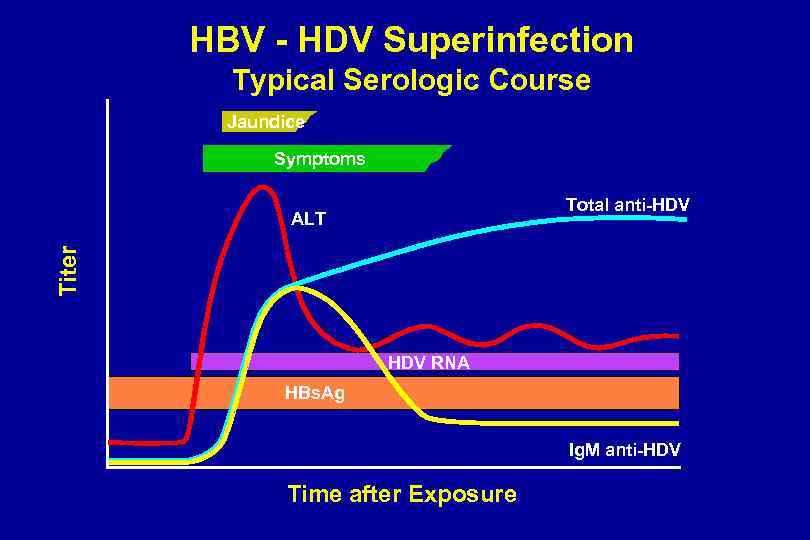 HBV - HDV Superinfection Typical Serologic Course Jaundice Symptoms Total anti-HDV Titer ALT HDV RNA HBs. Ag Ig. M anti-HDV Time after Exposure
HBV - HDV Superinfection Typical Serologic Course Jaundice Symptoms Total anti-HDV Titer ALT HDV RNA HBs. Ag Ig. M anti-HDV Time after Exposure
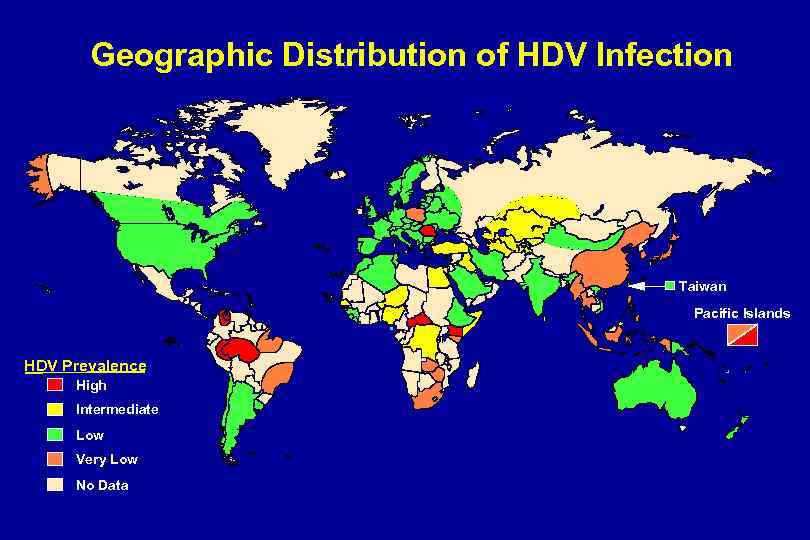 Geographic Distribution of HDV Infection Taiwan Pacific Islands HDV Prevalence High Intermediate Low Very Low No Data
Geographic Distribution of HDV Infection Taiwan Pacific Islands HDV Prevalence High Intermediate Low Very Low No Data
 Hepatitis D - Prevention • HBV-HDV Coinfection Pre or postexposure prophylaxis to prevent HBV infection • HBV-HDV Superinfection Education to reduce risk behaviors among persons with chronic HBV infection
Hepatitis D - Prevention • HBV-HDV Coinfection Pre or postexposure prophylaxis to prevent HBV infection • HBV-HDV Superinfection Education to reduce risk behaviors among persons with chronic HBV infection
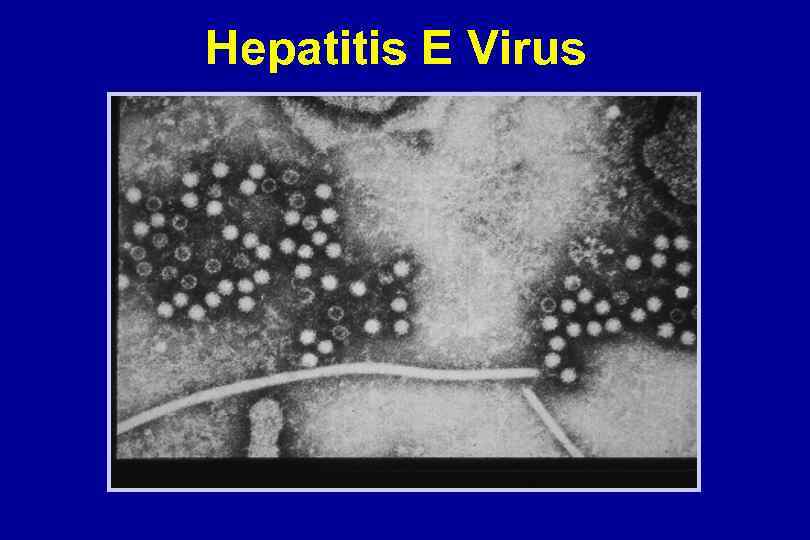 Hepatitis E Virus
Hepatitis E Virus
 Hepatitis E - Clinical Features • Incubation period: Average 40 days Range 15 -60 days • Case-fatality rate: Overall, 1%-3% Pregnant women, 15%-25% • Illness severity: Increased with age • Chronic sequelae: None identified
Hepatitis E - Clinical Features • Incubation period: Average 40 days Range 15 -60 days • Case-fatality rate: Overall, 1%-3% Pregnant women, 15%-25% • Illness severity: Increased with age • Chronic sequelae: None identified
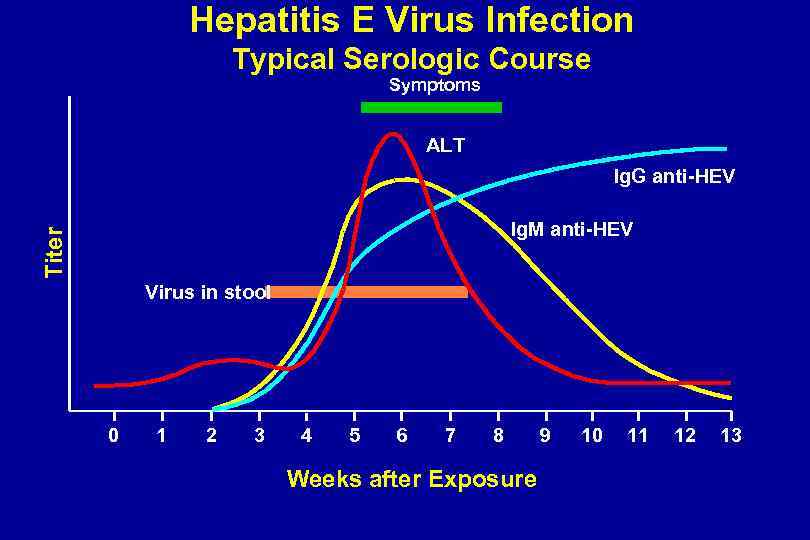 Hepatitis E Virus Infection Typical Serologic Course Symptoms ALT Ig. G anti-HEV Titer Ig. M anti-HEV Virus in stool 0 1 2 3 4 5 6 7 8 Weeks after Exposure 9 10 11 12 13
Hepatitis E Virus Infection Typical Serologic Course Symptoms ALT Ig. G anti-HEV Titer Ig. M anti-HEV Virus in stool 0 1 2 3 4 5 6 7 8 Weeks after Exposure 9 10 11 12 13
 Hepatitis E Epidemiologic Features • Most outbreaks associated with fecally contaminated drinking water • Minimal person-to-person transmission • U. S. cases usually have history of travel to HEV-endemic areas
Hepatitis E Epidemiologic Features • Most outbreaks associated with fecally contaminated drinking water • Minimal person-to-person transmission • U. S. cases usually have history of travel to HEV-endemic areas
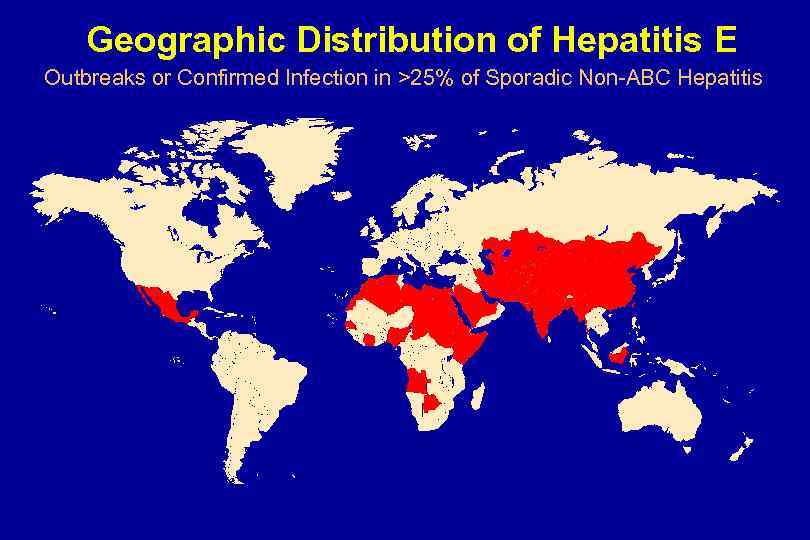 Geographic Distribution of Hepatitis E Outbreaks or Confirmed Infection in >25% of Sporadic Non-ABC Hepatitis
Geographic Distribution of Hepatitis E Outbreaks or Confirmed Infection in >25% of Sporadic Non-ABC Hepatitis
 Prevention and Control Measures for Travelers to HEV-Endemic Regions • Avoid drinking water (and beverages with ice) of unknown purity, uncooked shellfish, and uncooked fruit/vegetables not peeled or prepared by traveler • IG prepared from donors in Western countries does not prevent infection • Unknown efficacy of IG prepared from donors in endemic areas • Vaccine?
Prevention and Control Measures for Travelers to HEV-Endemic Regions • Avoid drinking water (and beverages with ice) of unknown purity, uncooked shellfish, and uncooked fruit/vegetables not peeled or prepared by traveler • IG prepared from donors in Western countries does not prevent infection • Unknown efficacy of IG prepared from donors in endemic areas • Vaccine?


