a3d9b5f9d650abed5cda893f51cdb5fe.ppt
- Количество слайдов: 109
 VIRAL INFECTIONS Dr. ALAA HUSSAIN A. AWN
VIRAL INFECTIONS Dr. ALAA HUSSAIN A. AWN
 u Viruses are simple infectious agents consisting of a portion of genetic material, RNA or DNA, enclosed in a protein coat which is antigenically unique for that species. u They are essentially inert and cannot exist in a free-living state, needing to infect host cells to survive.
u Viruses are simple infectious agents consisting of a portion of genetic material, RNA or DNA, enclosed in a protein coat which is antigenically unique for that species. u They are essentially inert and cannot exist in a free-living state, needing to infect host cells to survive.
 u Once it is in the intracellular environment, they utilise host material for protein synthesis and genetic reproduction. u All viral infections must therefore originate from an infected source by either direct or vector-mediated spread.
u Once it is in the intracellular environment, they utilise host material for protein synthesis and genetic reproduction. u All viral infections must therefore originate from an infected source by either direct or vector-mediated spread.
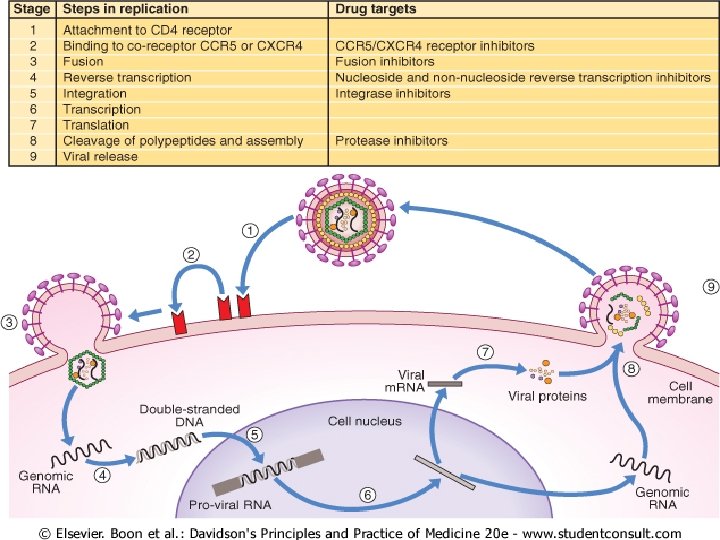
 CLASSIFICATION OF VIRAL INFECTIONS u The classification of viral infections in humans is shown in the fallowing box (13. 25) dav. Pg 299 20 th ed.
CLASSIFICATION OF VIRAL INFECTIONS u The classification of viral infections in humans is shown in the fallowing box (13. 25) dav. Pg 299 20 th ed.
 VIRUSES INVOLVED IN HUMAN DISEASE
VIRUSES INVOLVED IN HUMAN DISEASE
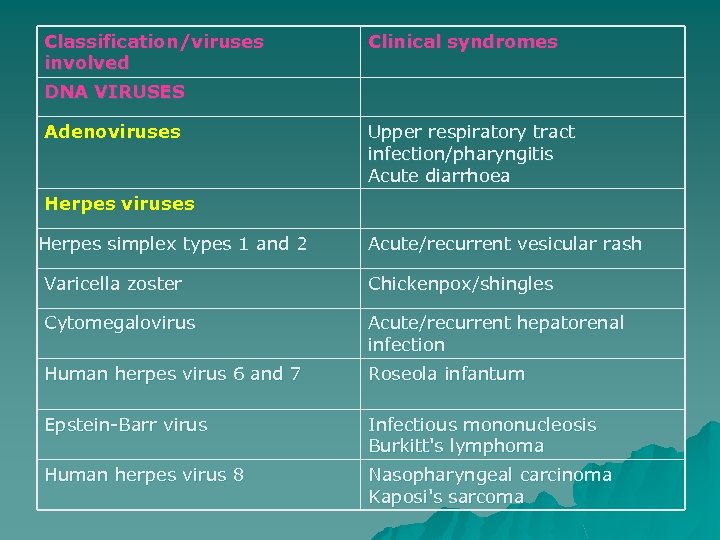 Classification/viruses involved Clinical syndromes DNA VIRUSES Adenoviruses Upper respiratory tract infection/pharyngitis Acute diarrhoea Herpes viruses Herpes simplex types 1 and 2 Acute/recurrent vesicular rash Varicella zoster Chickenpox/shingles Cytomegalovirus Acute/recurrent hepatorenal infection Human herpes virus 6 and 7 Roseola infantum Epstein-Barr virus Infectious mononucleosis Burkitt's lymphoma Human herpes virus 8 Nasopharyngeal carcinoma Kaposi's sarcoma
Classification/viruses involved Clinical syndromes DNA VIRUSES Adenoviruses Upper respiratory tract infection/pharyngitis Acute diarrhoea Herpes viruses Herpes simplex types 1 and 2 Acute/recurrent vesicular rash Varicella zoster Chickenpox/shingles Cytomegalovirus Acute/recurrent hepatorenal infection Human herpes virus 6 and 7 Roseola infantum Epstein-Barr virus Infectious mononucleosis Burkitt's lymphoma Human herpes virus 8 Nasopharyngeal carcinoma Kaposi's sarcoma
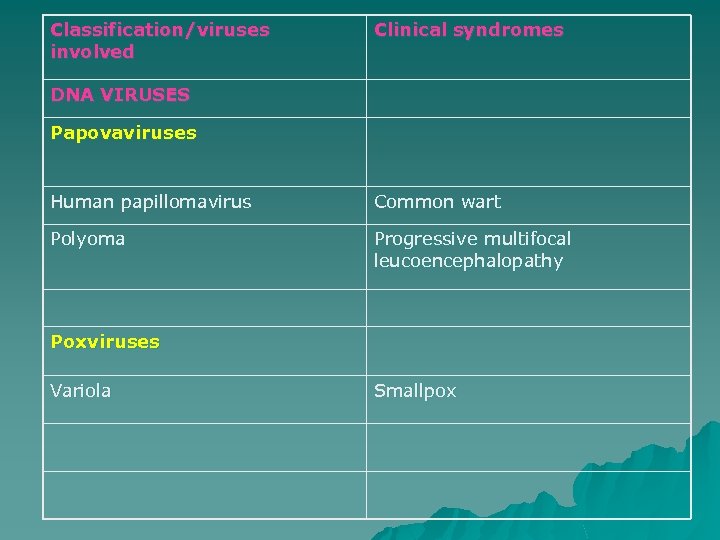 Classification/viruses involved Clinical syndromes DNA VIRUSES Papovaviruses Human papillomavirus Common wart Polyoma Progressive multifocal leucoencephalopathy Poxviruses Variola Smallpox
Classification/viruses involved Clinical syndromes DNA VIRUSES Papovaviruses Human papillomavirus Common wart Polyoma Progressive multifocal leucoencephalopathy Poxviruses Variola Smallpox
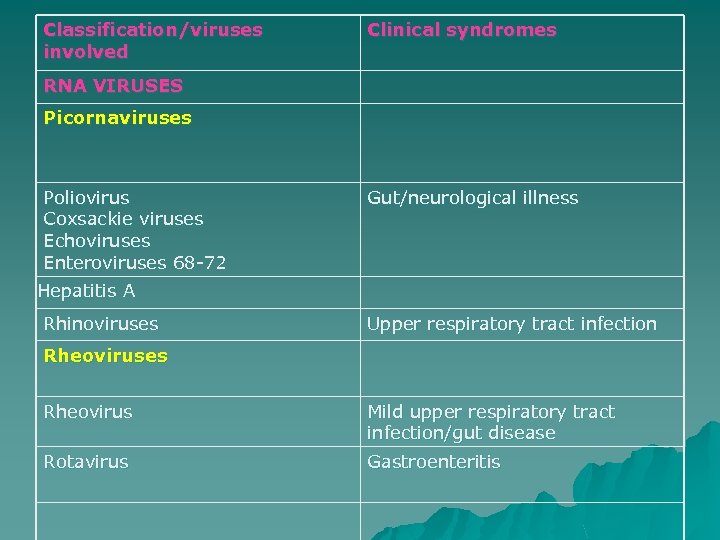 Classification/viruses involved Clinical syndromes RNA VIRUSES Picornaviruses Poliovirus Coxsackie viruses Echoviruses Enteroviruses 68 -72 Gut/neurological illness Hepatitis A Rhinoviruses Upper respiratory tract infection Rheoviruses Rheovirus Mild upper respiratory tract infection/gut disease Rotavirus Gastroenteritis
Classification/viruses involved Clinical syndromes RNA VIRUSES Picornaviruses Poliovirus Coxsackie viruses Echoviruses Enteroviruses 68 -72 Gut/neurological illness Hepatitis A Rhinoviruses Upper respiratory tract infection Rheoviruses Rheovirus Mild upper respiratory tract infection/gut disease Rotavirus Gastroenteritis
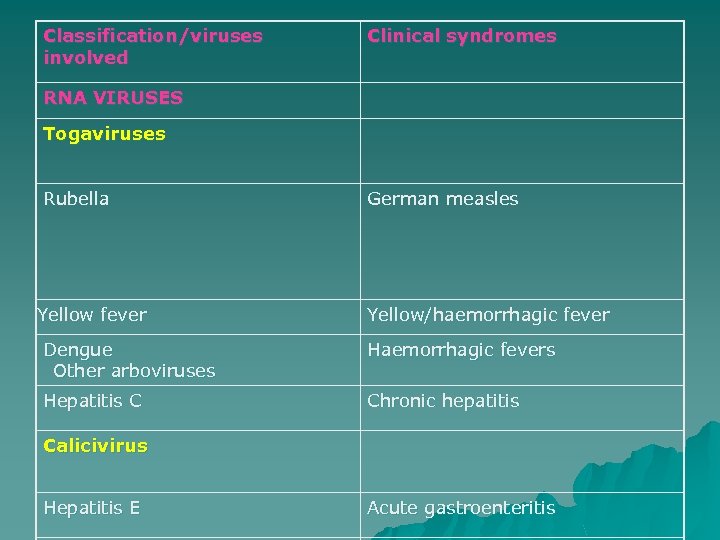 Classification/viruses involved Clinical syndromes RNA VIRUSES Togaviruses Rubella Yellow fever German measles Yellow/haemorrhagic fever Dengue Other arboviruses Haemorrhagic fevers Hepatitis C Chronic hepatitis Calicivirus Hepatitis E Acute gastroenteritis
Classification/viruses involved Clinical syndromes RNA VIRUSES Togaviruses Rubella Yellow fever German measles Yellow/haemorrhagic fever Dengue Other arboviruses Haemorrhagic fevers Hepatitis C Chronic hepatitis Calicivirus Hepatitis E Acute gastroenteritis
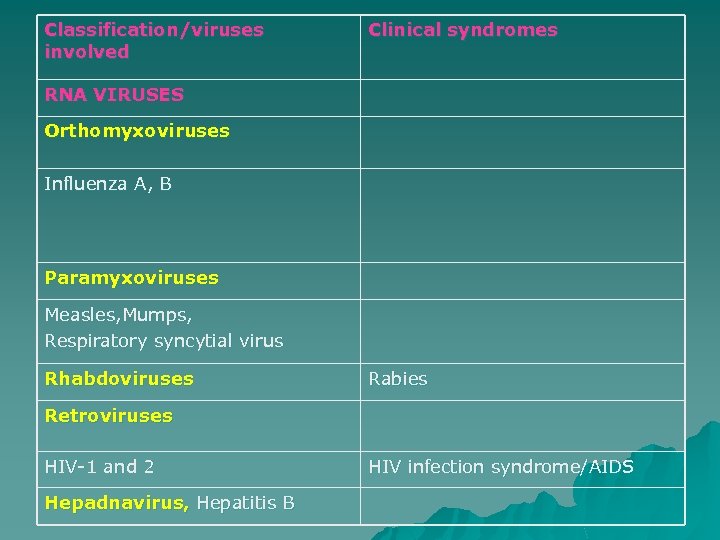 Classification/viruses involved Clinical syndromes RNA VIRUSES Orthomyxoviruses Influenza A, B Paramyxoviruses Measles, Mumps, Respiratory syncytial virus Rhabdoviruses Rabies Retroviruses HIV-1 and 2 Hepadnavirus, Hepatitis B HIV infection syndrome/AIDS
Classification/viruses involved Clinical syndromes RNA VIRUSES Orthomyxoviruses Influenza A, B Paramyxoviruses Measles, Mumps, Respiratory syncytial virus Rhabdoviruses Rabies Retroviruses HIV-1 and 2 Hepadnavirus, Hepatitis B HIV infection syndrome/AIDS
 CLASSIFICATION ACCORDING TO THE HOST AND ORGANS INVOLVED u COMMON VIRAL INFECTIONS AND CHILDHOOD EXANTHEMS. u VIRAL INFECTIONS OF THE SKIN. u SYSTEMIC VIRAL INFECTIONS. u GASTRO-INTESTINAL VIRAL INFECTIONS. u RESPIRATORY VIRAL INFECTIONS. u VIRAL INFECTIONS WITH NEUROLOGICAL INVOLVEMENT.
CLASSIFICATION ACCORDING TO THE HOST AND ORGANS INVOLVED u COMMON VIRAL INFECTIONS AND CHILDHOOD EXANTHEMS. u VIRAL INFECTIONS OF THE SKIN. u SYSTEMIC VIRAL INFECTIONS. u GASTRO-INTESTINAL VIRAL INFECTIONS. u RESPIRATORY VIRAL INFECTIONS. u VIRAL INFECTIONS WITH NEUROLOGICAL INVOLVEMENT.
 COMMON VIRAL INFECTONS AND CHILDHOOD EXANTHEMS u MEASELS u RUBELLA u MUMPS (GERMAN MEASELS)
COMMON VIRAL INFECTONS AND CHILDHOOD EXANTHEMS u MEASELS u RUBELLA u MUMPS (GERMAN MEASELS)
 VIRAL INFECTIONS OF THE SKIN u THE HERPES VIRUS GROUP
VIRAL INFECTIONS OF THE SKIN u THE HERPES VIRUS GROUP
 13. 29 HERPES VIRUS INFECTIONS Virus Infection Herpesvirus hominis (herpes simplex, HSV) Type 1 Herpes labialis ('cold sores') Keratoconjunctivitis Finger infections ('whitlows') Encephalitis Primary stomatitis Genital infections Type 2 Genital infections Neonatal infection (acquired during vaginal delivery) Varicella zoster virus Chickenpox , Shingles (herpes zoster) Cytomegalovirus (CMV) Congenital infection Disease in immunocompromised patients Pneumonitis Retinitis Enteritis Generalised infection Epstein-Barr virus (EBV) Infectious mononucleosis Burkitt's lymphoma Nasopharyngeal carcinoma Oral hairy leucoplakia (AIDS patients) Human herpes virus 8 Associated with Kaposi's sarcoma
13. 29 HERPES VIRUS INFECTIONS Virus Infection Herpesvirus hominis (herpes simplex, HSV) Type 1 Herpes labialis ('cold sores') Keratoconjunctivitis Finger infections ('whitlows') Encephalitis Primary stomatitis Genital infections Type 2 Genital infections Neonatal infection (acquired during vaginal delivery) Varicella zoster virus Chickenpox , Shingles (herpes zoster) Cytomegalovirus (CMV) Congenital infection Disease in immunocompromised patients Pneumonitis Retinitis Enteritis Generalised infection Epstein-Barr virus (EBV) Infectious mononucleosis Burkitt's lymphoma Nasopharyngeal carcinoma Oral hairy leucoplakia (AIDS patients) Human herpes virus 8 Associated with Kaposi's sarcoma
 HERPES SIMPLEX VIRUS (HSV) u u u Types 1 and 2 of this common virus affect humans. Type 1 HSV produces mucocutaneous lesions, predominantly of the head and neck type 2 disease is a sexually transmitted anogenital infection
HERPES SIMPLEX VIRUS (HSV) u u u Types 1 and 2 of this common virus affect humans. Type 1 HSV produces mucocutaneous lesions, predominantly of the head and neck type 2 disease is a sexually transmitted anogenital infection
 u The source of infection is a case of primary or active recurrent disease. Primary infection normally occurs as a gingivostomatitis in infancy and may be subclinical or mistaken for 'teething'. u It may present as a keratitis (dendritic ulcer), viral paronychia ('whitlow'), u vulvovaginitis, cervicitis (often unrecognised), balanitis u or rarely as encephalitis. u
u The source of infection is a case of primary or active recurrent disease. Primary infection normally occurs as a gingivostomatitis in infancy and may be subclinical or mistaken for 'teething'. u It may present as a keratitis (dendritic ulcer), viral paronychia ('whitlow'), u vulvovaginitis, cervicitis (often unrecognised), balanitis u or rarely as encephalitis. u
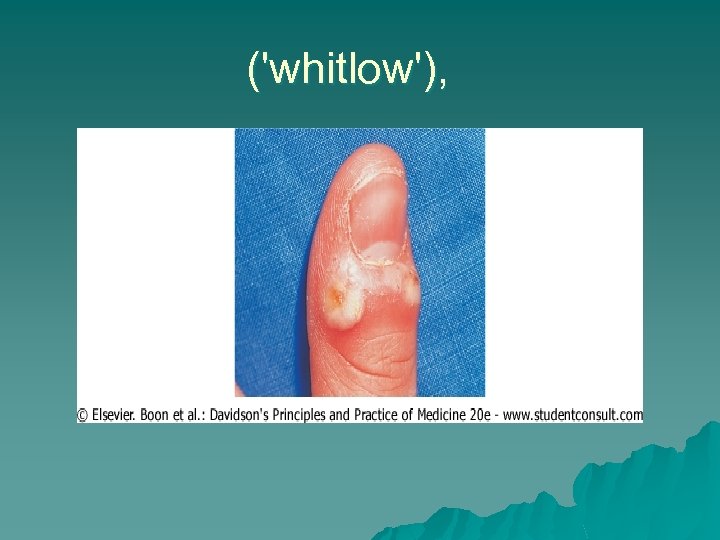 ('whitlow'),
('whitlow'),
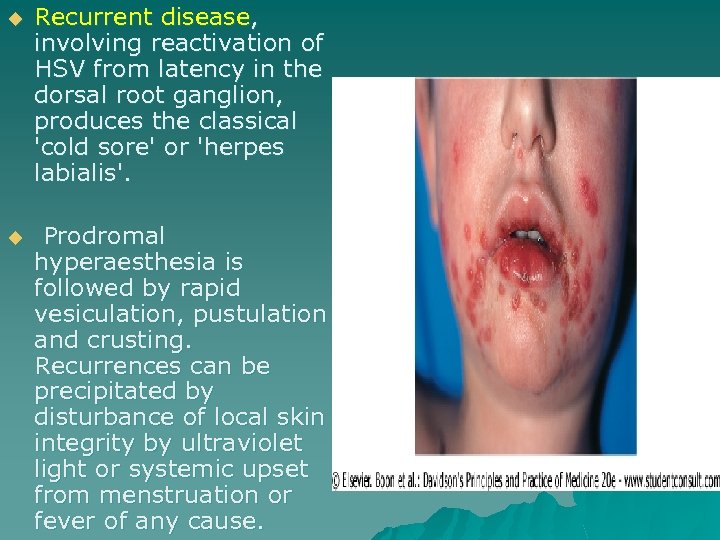 u Recurrent disease, involving reactivation of HSV from latency in the dorsal root ganglion, produces the classical 'cold sore' or 'herpes labialis'. u Prodromal hyperaesthesia is followed by rapid vesiculation, pustulation and crusting. Recurrences can be precipitated by disturbance of local skin integrity by ultraviolet light or systemic upset from menstruation or fever of any cause.
u Recurrent disease, involving reactivation of HSV from latency in the dorsal root ganglion, produces the classical 'cold sore' or 'herpes labialis'. u Prodromal hyperaesthesia is followed by rapid vesiculation, pustulation and crusting. Recurrences can be precipitated by disturbance of local skin integrity by ultraviolet light or systemic upset from menstruation or fever of any cause.
 u Type 2 (genital) disease is a common cause of recurrent painful genital ulceration
u Type 2 (genital) disease is a common cause of recurrent painful genital ulceration
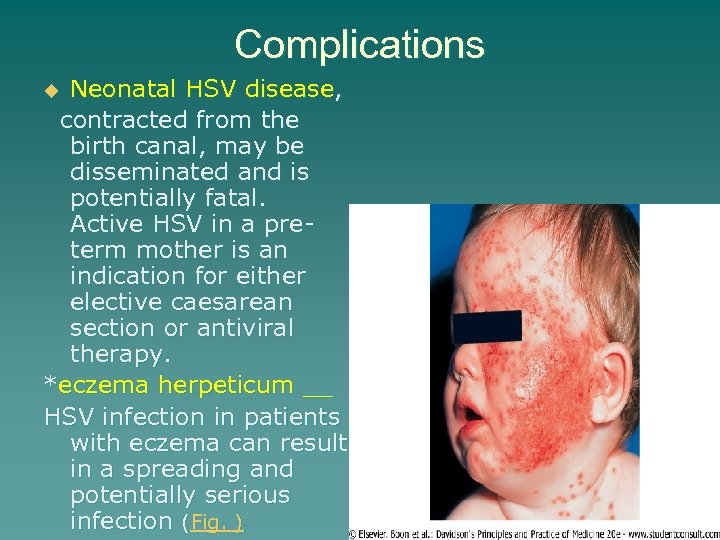 Complications Neonatal HSV disease, contracted from the birth canal, may be disseminated and is potentially fatal. Active HSV in a preterm mother is an indication for either elective caesarean section or antiviral therapy. *eczema herpeticum __ HSV infection in patients with eczema can result in a spreading and potentially serious infection (Fig. ) u
Complications Neonatal HSV disease, contracted from the birth canal, may be disseminated and is potentially fatal. Active HSV in a preterm mother is an indication for either elective caesarean section or antiviral therapy. *eczema herpeticum __ HSV infection in patients with eczema can result in a spreading and potentially serious infection (Fig. ) u
 u u Dendritic ulcers may produce corneal scarring and permanently damage eyesight. These require aggressive antiviral therapy. Encephalitis, the most serious complication of HSV disease, may occur following either primary or secondary disease. A haemorrhagic necrotising temporal lobe cerebritis produces temporal lobe epilepsy and decreasing conscious level/coma. Without treatment, mortality is 80%. Any suggestion of HSV encephalitis is an indication for immediate empirical systemic antiviral therapy.
u u Dendritic ulcers may produce corneal scarring and permanently damage eyesight. These require aggressive antiviral therapy. Encephalitis, the most serious complication of HSV disease, may occur following either primary or secondary disease. A haemorrhagic necrotising temporal lobe cerebritis produces temporal lobe epilepsy and decreasing conscious level/coma. Without treatment, mortality is 80%. Any suggestion of HSV encephalitis is an indication for immediate empirical systemic antiviral therapy.
 DIAGNOSIS PCR, u Electron microscopy or culture from vesicular fluid. u CSF PCR is very useful in HSV encephalitis. u Serology is of limited value, only confirming primary infection. u
DIAGNOSIS PCR, u Electron microscopy or culture from vesicular fluid. u CSF PCR is very useful in HSV encephalitis. u Serology is of limited value, only confirming primary infection. u
 Management The acyclic antivirals are the treatment of choice for HSV infection. u Therapy must commence in the first 48 hours of clinical disease (primary or recurrent); thereafter it is unlikely to influence clinical outcome or modify the disease process. u Severe manifestations should be treated regardless of the time of presentation (Box 13. 30). u
Management The acyclic antivirals are the treatment of choice for HSV infection. u Therapy must commence in the first 48 hours of clinical disease (primary or recurrent); thereafter it is unlikely to influence clinical outcome or modify the disease process. u Severe manifestations should be treated regardless of the time of presentation (Box 13. 30). u
 Rx. Primary HSV -Famciclovir 250 mg 8 -hourly -Valaciclovir 500 mg 12 -hourly -Aciclovir 200 mg 5 times daily u * Severe and preventing oral intake Aciclovir 5 mg/kg 8 -hourly i. v. *Recurrent HSV-1 or 2 - Aciclovir ointment 3 -5 times daily -Oral aciclovir 200 mg 6 -hourly -Famciclovir 250 mg 12 -hourly -Valaciclovir 500 mg daily * In immunocompromised -Aciclovir 400 mg 6 -hourly -Famciclovir 500 mg 12 -hourly -Valaciclovir 1 g 12 -hourly
Rx. Primary HSV -Famciclovir 250 mg 8 -hourly -Valaciclovir 500 mg 12 -hourly -Aciclovir 200 mg 5 times daily u * Severe and preventing oral intake Aciclovir 5 mg/kg 8 -hourly i. v. *Recurrent HSV-1 or 2 - Aciclovir ointment 3 -5 times daily -Oral aciclovir 200 mg 6 -hourly -Famciclovir 250 mg 12 -hourly -Valaciclovir 500 mg daily * In immunocompromised -Aciclovir 400 mg 6 -hourly -Famciclovir 500 mg 12 -hourly -Valaciclovir 1 g 12 -hourly
 u Severe complications - Aciclovir i. v. 10 mg/kg 8 -hourly (up to 20 mg/kg in severe encephalitis)
u Severe complications - Aciclovir i. v. 10 mg/kg 8 -hourly (up to 20 mg/kg in severe encephalitis)
 CHICKENPOX u u u Varicella zoster virus (VZV) is dermo- and neurotropic infection. Spread by the aerosol route, it is highly infectious to susceptible individuals. Disease in children is usually well tolerated. It is more severe in adults, pregnant women and the immunocompromised. Pneumonitis can be fatal and is more likely in smokers, pregnant women and the immunocompromised.
CHICKENPOX u u u Varicella zoster virus (VZV) is dermo- and neurotropic infection. Spread by the aerosol route, it is highly infectious to susceptible individuals. Disease in children is usually well tolerated. It is more severe in adults, pregnant women and the immunocompromised. Pneumonitis can be fatal and is more likely in smokers, pregnant women and the immunocompromised.
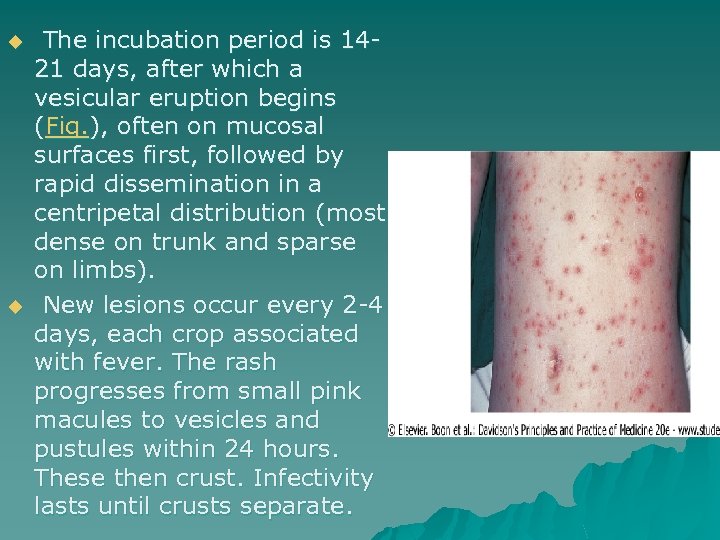 u u The incubation period is 1421 days, after which a vesicular eruption begins (Fig. ), often on mucosal surfaces first, followed by rapid dissemination in a centripetal distribution (most dense on trunk and sparse on limbs). New lesions occur every 2 -4 days, each crop associated with fever. The rash progresses from small pink macules to vesicles and pustules within 24 hours. These then crust. Infectivity lasts until crusts separate.
u u The incubation period is 1421 days, after which a vesicular eruption begins (Fig. ), often on mucosal surfaces first, followed by rapid dissemination in a centripetal distribution (most dense on trunk and sparse on limbs). New lesions occur every 2 -4 days, each crop associated with fever. The rash progresses from small pink macules to vesicles and pustules within 24 hours. These then crust. Infectivity lasts until crusts separate.
 complications u u u . Due to intense itch secondary bacterial infection from scratching is the most common complication of primary chickenpox. Self-limiting cerebellar ataxia may rarely occur 7 -10 days after recovery from the rash. Maternal infection in early pregnancy carries a 3% risk of neonatal damage, and disease within 5 days of delivery can lead to severe neonatal varicella.
complications u u u . Due to intense itch secondary bacterial infection from scratching is the most common complication of primary chickenpox. Self-limiting cerebellar ataxia may rarely occur 7 -10 days after recovery from the rash. Maternal infection in early pregnancy carries a 3% risk of neonatal damage, and disease within 5 days of delivery can lead to severe neonatal varicella.
 Diagnosis Usually this is clinically obvious from the classical appearance of the rash. u Aspiration of vesicular fluid and PCR or tissue culture will confirm the diagnosis. u Electron microscopy cannot distinguish HSV from VZV. u Serological examination for rising titres of antibody is only useful in primary infection. u u Chickenpox can recur as a subclinical infection following primary disease.
Diagnosis Usually this is clinically obvious from the classical appearance of the rash. u Aspiration of vesicular fluid and PCR or tissue culture will confirm the diagnosis. u Electron microscopy cannot distinguish HSV from VZV. u Serological examination for rising titres of antibody is only useful in primary infection. u u Chickenpox can recur as a subclinical infection following primary disease.
 Management Aciclovir, valaciclovir and famciclovir, effective if commenced within 48 hours of rash appearance. u They are required in the management of the immunocompromised or any case of pneumonitis. u Note Aciclovir shortens symptoms in chickenpox by an average of 1 day. In shingles aciclovir reduces pain by 10 days and the risk of postherpetic neuralgia by 8%. Aciclovir is therefore cost-effective in shingles but not chickenpox. u
Management Aciclovir, valaciclovir and famciclovir, effective if commenced within 48 hours of rash appearance. u They are required in the management of the immunocompromised or any case of pneumonitis. u Note Aciclovir shortens symptoms in chickenpox by an average of 1 day. In shingles aciclovir reduces pain by 10 days and the risk of postherpetic neuralgia by 8%. Aciclovir is therefore cost-effective in shingles but not chickenpox. u
 Human VZV immunoglobulin may be used to attenuate infection in highly susceptible contacts of chickenpox such as: u u u u bone marrow recipients patients with debilitating disease HIV-positive contacts without VZV immunity pregnant women with no known VZV antibody (screen for antibody if in doubt) immunosuppressed contacts who have received highdose corticosteroids in the previous 3 months neonates whose mothers develop chickenpox between 1 week before and 4 weeks after delivery neonates in contact with chickenpox/shingles whose mothers have no history of chickenpox or any demonstrable antibody premature infants of less than 30 weeks' gestation, or weighing less than 1 kg at birth who contact chickenpox or shingles.
Human VZV immunoglobulin may be used to attenuate infection in highly susceptible contacts of chickenpox such as: u u u u bone marrow recipients patients with debilitating disease HIV-positive contacts without VZV immunity pregnant women with no known VZV antibody (screen for antibody if in doubt) immunosuppressed contacts who have received highdose corticosteroids in the previous 3 months neonates whose mothers develop chickenpox between 1 week before and 4 weeks after delivery neonates in contact with chickenpox/shingles whose mothers have no history of chickenpox or any demonstrable antibody premature infants of less than 30 weeks' gestation, or weighing less than 1 kg at birth who contact chickenpox or shingles.
 SHINGLES (HERPES ZOSTER) u This is produced by reactivation of latent VZV from the dorsal root ganglion of sensory nerves. u Commonly seen in the elderly, u It may present in younger patients with immune deficiency or after intra-uterine infection.
SHINGLES (HERPES ZOSTER) u This is produced by reactivation of latent VZV from the dorsal root ganglion of sensory nerves. u Commonly seen in the elderly, u It may present in younger patients with immune deficiency or after intra-uterine infection.
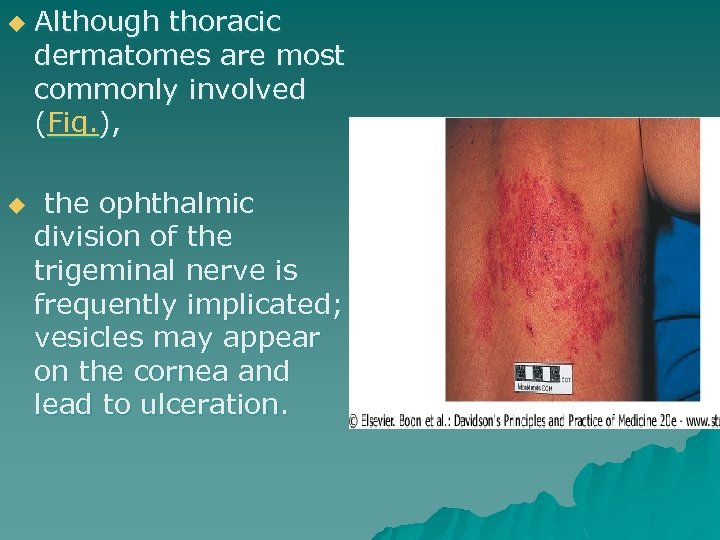 u u Although thoracic dermatomes are most commonly involved (Fig. ), the ophthalmic division of the trigeminal nerve is frequently implicated; vesicles may appear on the cornea and lead to ulceration.
u u Although thoracic dermatomes are most commonly involved (Fig. ), the ophthalmic division of the trigeminal nerve is frequently implicated; vesicles may appear on the cornea and lead to ulceration.
 u Geniculate ganglion involvement causes the Ramsay Hunt syndrome of facial palsy, ipsilateral loss of taste and buccal ulceration, plus a rash in the external auditory canal. This may be mistaken for Bell's palsy. u Bowel and bladder dysfunction occurs with sacral nerve root involvement. u The virus occasionally causes myelitis or encephalitis.
u Geniculate ganglion involvement causes the Ramsay Hunt syndrome of facial palsy, ipsilateral loss of taste and buccal ulceration, plus a rash in the external auditory canal. This may be mistaken for Bell's palsy. u Bowel and bladder dysfunction occurs with sacral nerve root involvement. u The virus occasionally causes myelitis or encephalitis.
 u Burning Clinical features discomfort in the affected dermatome progresses to frank neuralgia. Discrete vesicles appear in the dermatome 3 -4 days later and often coalesce. u Severe disease, multiple dermatomal involvement or recurrence suggests underlying immune deficiency.
u Burning Clinical features discomfort in the affected dermatome progresses to frank neuralgia. Discrete vesicles appear in the dermatome 3 -4 days later and often coalesce. u Severe disease, multiple dermatomal involvement or recurrence suggests underlying immune deficiency.
 Complications u The most common and troublesome complication is post-herpetic neuralgia: persistence of pain for 1 -6 months or more following healing of the rash.
Complications u The most common and troublesome complication is post-herpetic neuralgia: persistence of pain for 1 -6 months or more following healing of the rash.
 Management u Early therapy with aciclovir 800 mg 5 times daily or valaciclovir 1 g 8 -hourly, or aciclovir 10 mg/kg i. v. 8 -hourly in severe infection and in the immunocompromised has been shown to reduce both early- and late-onset pain, especially in patients over 65. Post-herpetic neuralgia requires u aggressive analgesia and u the use of transcutaneous nerve stimulation (a 'TENS' machine), along with u neurotransmitter modification with agents such as amitriptyline 25 -100 mg daily or gabapentin (commencing at 300 mg daily and building slowly to 300 mg 12 -hourly or more.
Management u Early therapy with aciclovir 800 mg 5 times daily or valaciclovir 1 g 8 -hourly, or aciclovir 10 mg/kg i. v. 8 -hourly in severe infection and in the immunocompromised has been shown to reduce both early- and late-onset pain, especially in patients over 65. Post-herpetic neuralgia requires u aggressive analgesia and u the use of transcutaneous nerve stimulation (a 'TENS' machine), along with u neurotransmitter modification with agents such as amitriptyline 25 -100 mg daily or gabapentin (commencing at 300 mg daily and building slowly to 300 mg 12 -hourly or more.
 Smallpox (variola) u u This severe disease, with a 30% mortality in the unvaccinated patient and no current effective therapy, was eradicated world-wide in 1980 by a successful international vaccination campaign coordinated by the WHO. Interest in the disease has re-emerged due to its potential as a bioterrorist weapon. In view of this threat some developed countries have reintroduced vaccination for key healthcare personnel and re-evaluated national plans for the containment of disease.
Smallpox (variola) u u This severe disease, with a 30% mortality in the unvaccinated patient and no current effective therapy, was eradicated world-wide in 1980 by a successful international vaccination campaign coordinated by the WHO. Interest in the disease has re-emerged due to its potential as a bioterrorist weapon. In view of this threat some developed countries have reintroduced vaccination for key healthcare personnel and re-evaluated national plans for the containment of disease.
 Clinical features u The classical form is characterised by a typical deep-seated centrifugal vesicular/pustular rash, worst on the face and extremities, with no cropping (i. e. unlike chickenpox); u the rash is accompanied by fever, severe myalgia.
Clinical features u The classical form is characterised by a typical deep-seated centrifugal vesicular/pustular rash, worst on the face and extremities, with no cropping (i. e. unlike chickenpox); u the rash is accompanied by fever, severe myalgia.
 SYSTEMIC VIRAL INFECTIONS u INFLUENZA EBV -Infectios mononucleosus u CMV u VIRAL HEMORAGIC FEVER u HIV u
SYSTEMIC VIRAL INFECTIONS u INFLUENZA EBV -Infectios mononucleosus u CMV u VIRAL HEMORAGIC FEVER u HIV u
 INFECTIOUS MONONUCLEOSIS (IM) u - Virology and epidemiology The disease is caused by the Epstein-Barr virus (EBV), a gamma herpes virus. . - In developing countries and poorer societies in developed nations subclinical infection in childhood is virtually universal. In richer communities, particularly among upper socioeconomic groups, primary infection may be delayed until adolescence or early adult life. - About 50% of infections result in typical IM. The virus is usually acquired from asymptomatic excreters. . - Saliva is the main means of spread, either by droplet infection or environmental contamination in childhood, or by kissing among adolescents and adults. . - IM is not highly contagious, isolation is unnecessary and documented outbreaks seldom occur.
INFECTIOUS MONONUCLEOSIS (IM) u - Virology and epidemiology The disease is caused by the Epstein-Barr virus (EBV), a gamma herpes virus. . - In developing countries and poorer societies in developed nations subclinical infection in childhood is virtually universal. In richer communities, particularly among upper socioeconomic groups, primary infection may be delayed until adolescence or early adult life. - About 50% of infections result in typical IM. The virus is usually acquired from asymptomatic excreters. . - Saliva is the main means of spread, either by droplet infection or environmental contamination in childhood, or by kissing among adolescents and adults. . - IM is not highly contagious, isolation is unnecessary and documented outbreaks seldom occur.
 Clinical features (IM) u lymphadenopathy, especially posterior cervical, u pharyngeal inflammation or exudates, u fever, u splenomegaly, u palatal petechiae, u periorbital oedema, u clinical or biochemical evidence of hepatitis, u And a non-specific rash.
Clinical features (IM) u lymphadenopathy, especially posterior cervical, u pharyngeal inflammation or exudates, u fever, u splenomegaly, u palatal petechiae, u periorbital oedema, u clinical or biochemical evidence of hepatitis, u And a non-specific rash.
 u 20% or more of peripheral lymphocytes must have an atypical morphology (Fig. ) u characteristic heterophile antibody. (classical Paul. Bunnell titration or by slide test 'Monospot‘)). u Specific EBV serology (immunofluoresence)
u 20% or more of peripheral lymphocytes must have an atypical morphology (Fig. ) u characteristic heterophile antibody. (classical Paul. Bunnell titration or by slide test 'Monospot‘)). u Specific EBV serology (immunofluoresence)
 COMPLICATIONS OF INFECTIOUS MONONUCLEOSIS Common u Severe pharyngeal oedema u Antibiotic-induced rash u Chronic fatigue syndrome (10%) u u u Rare Ruptured spleen Respiratory obstruction Arthritis Agranulocytosis Agammaglobulinaemia Uncommon Neurological u Cranial nerve palsies u Polyneuritis u Transverse myelitis u Meningoencephalitis Haematological u Haemolytic anaemia u Thrombocytopenia Renal u Glomerulonephritis u Interstitial nephritis Cardiac u Myocarditis u Pericarditis Pulmonary u Interstitial pneumonitis
COMPLICATIONS OF INFECTIOUS MONONUCLEOSIS Common u Severe pharyngeal oedema u Antibiotic-induced rash u Chronic fatigue syndrome (10%) u u u Rare Ruptured spleen Respiratory obstruction Arthritis Agranulocytosis Agammaglobulinaemia Uncommon Neurological u Cranial nerve palsies u Polyneuritis u Transverse myelitis u Meningoencephalitis Haematological u Haemolytic anaemia u Thrombocytopenia Renal u Glomerulonephritis u Interstitial nephritis Cardiac u Myocarditis u Pericarditis Pulmonary u Interstitial pneumonitis
 Management u u u Treatment is largely symptomatic: aspirin gargles to relieve a sore throat. If a throat culture yields a β-haemolytic streptococcus, a course of erythromycin should be prescribed. Amoxicillin and similar semi-synthetic penicillins should be avoided. When pharyngeal oedema is severe a short course of corticosteroids, e. g. prednisolone 30 mg daily for 5 days, may help to relieve the swelling. contact sports should be avoided until splenomegaly has completely resolved because of the danger of splenic rupture. Unfortunately, about 10% of patients with IM suffer a chronic relapsing syndrome.
Management u u u Treatment is largely symptomatic: aspirin gargles to relieve a sore throat. If a throat culture yields a β-haemolytic streptococcus, a course of erythromycin should be prescribed. Amoxicillin and similar semi-synthetic penicillins should be avoided. When pharyngeal oedema is severe a short course of corticosteroids, e. g. prednisolone 30 mg daily for 5 days, may help to relieve the swelling. contact sports should be avoided until splenomegaly has completely resolved because of the danger of splenic rupture. Unfortunately, about 10% of patients with IM suffer a chronic relapsing syndrome.
 ACQUIRED CYTOMEGALOVIRUS INFECTION Virology and epidemiology - Cytomegalovirus (CMV) is a beta herpes virus. u - It circulates readily among children, especially in crowded communities. - Although most primary infections are asymptomatic, many children continue to excrete virus for months or years.
ACQUIRED CYTOMEGALOVIRUS INFECTION Virology and epidemiology - Cytomegalovirus (CMV) is a beta herpes virus. u - It circulates readily among children, especially in crowded communities. - Although most primary infections are asymptomatic, many children continue to excrete virus for months or years.
 u u u A second peak in virus acquisition occurs among teenagers and young adults. CMV infection is persistent, and is characterised by subclinical cycles of active virus replication and by persistent low-level virus shedding. Sexual transmission and oral spread are common among adults, but infection may also be acquired by women caring for children with asymptomatic infections. The peak incidence occurs between the ages of 25 and 35, rather later than with EBV-related mononucleosis.
u u u A second peak in virus acquisition occurs among teenagers and young adults. CMV infection is persistent, and is characterised by subclinical cycles of active virus replication and by persistent low-level virus shedding. Sexual transmission and oral spread are common among adults, but infection may also be acquired by women caring for children with asymptomatic infections. The peak incidence occurs between the ages of 25 and 35, rather later than with EBV-related mononucleosis.
 Clinical features u u u Most post-childhood CMV infections are subclinical, although some young adults develop a mononucleosis-like syndrome. Some patients have a prolonged influenzalike illness lasting 2 weeks or more. Physical signs such as a palpable liver and spleen resemble those of IM, but in CMV mononucleosis hepatomegaly is relatively more common, while lymphadenopathy, pharyngitis and tonsillitis are found less often. Jaundice is uncommon and usually mild.
Clinical features u u u Most post-childhood CMV infections are subclinical, although some young adults develop a mononucleosis-like syndrome. Some patients have a prolonged influenzalike illness lasting 2 weeks or more. Physical signs such as a palpable liver and spleen resemble those of IM, but in CMV mononucleosis hepatomegaly is relatively more common, while lymphadenopathy, pharyngitis and tonsillitis are found less often. Jaundice is uncommon and usually mild.
 complications u neurological involvement, u autoimmune haemolytic anaemia, u pericarditis, u pneumonitis. u arthropathy.
complications u neurological involvement, u autoimmune haemolytic anaemia, u pericarditis, u pneumonitis. u arthropathy.
 Investigations u Atypical lymphocytosis is not as prominent as in IM u heterophile u u antibody tests are negative. LFTs are often abnormal, with an alkaline phosphatase level raised out of proportion to transaminases. Serological diagnosis depends on the detection of CMV-specific Ig. M antibody.
Investigations u Atypical lymphocytosis is not as prominent as in IM u heterophile u u antibody tests are negative. LFTs are often abnormal, with an alkaline phosphatase level raised out of proportion to transaminases. Serological diagnosis depends on the detection of CMV-specific Ig. M antibody.
 u Only Management symptomatic treatment. u Amoxicillin and similar antibiotics should not be prescribed because of the risk of a skin reaction. u Since CMV infection in immunocompetent subjects is selflimiting, the use of potentially toxic antiviral agents is usually inappropriate In immunosupp. Pt. , u Ganciclover injection u valganciclover tab. (valcyte) 450 mg 2 tab. 12 hourly for 4 -6 weeks
u Only Management symptomatic treatment. u Amoxicillin and similar antibiotics should not be prescribed because of the risk of a skin reaction. u Since CMV infection in immunocompetent subjects is selflimiting, the use of potentially toxic antiviral agents is usually inappropriate In immunosupp. Pt. , u Ganciclover injection u valganciclover tab. (valcyte) 450 mg 2 tab. 12 hourly for 4 -6 weeks
 u VIRAL HAEMORRHAGIC FEVERS caused by several The viral haemorrhagic fevers are zoonoses different viruses. They are endemic world-wide, examples. : Yellow feveru Reservoir ; Monkeys u Transmission; Mosquitoes u Geography ; Tropical Africa, South and Central Americau Mortality rate ; 10 -60% death u Clinical features ; Hepatic failure Blood oozing : * Dengue u Reservoir ; Humans u Transmission; Aedes aegyptiu Geography ; tropical and subtropical coasts u Mortality rate ; Nil-10% u Clinical features - Joint and bone pain Petechiae
u VIRAL HAEMORRHAGIC FEVERS caused by several The viral haemorrhagic fevers are zoonoses different viruses. They are endemic world-wide, examples. : Yellow feveru Reservoir ; Monkeys u Transmission; Mosquitoes u Geography ; Tropical Africa, South and Central Americau Mortality rate ; 10 -60% death u Clinical features ; Hepatic failure Blood oozing : * Dengue u Reservoir ; Humans u Transmission; Aedes aegyptiu Geography ; tropical and subtropical coasts u Mortality rate ; Nil-10% u Clinical features - Joint and bone pain Petechiae
 Pathogenesis u These viruses cause endothelial dysfunction with the development of leaky capillary syndrome. u Bleeding is due to this and associated platelet dysfunction. u Hypovolaemic shock and acute respiratory distress syndrome develop
Pathogenesis u These viruses cause endothelial dysfunction with the development of leaky capillary syndrome. u Bleeding is due to this and associated platelet dysfunction. u Hypovolaemic shock and acute respiratory distress syndrome develop
 Clinical features u All viral haemorrhagic fevers have similar non-specific presentations with fever, malaise, body pains, sore throat and headache. u On examination conjunctivitis, throat congestion, an erythematous or petechial rash, haemorrhage, lymphadenopathy and bradycardia may be noted.
Clinical features u All viral haemorrhagic fevers have similar non-specific presentations with fever, malaise, body pains, sore throat and headache. u On examination conjunctivitis, throat congestion, an erythematous or petechial rash, haemorrhage, lymphadenopathy and bradycardia may be noted.
 Investigations u There is leucopenia, u thrombocytopenia u proteinuria. u Prolong PT and PTT
Investigations u There is leucopenia, u thrombocytopenia u proteinuria. u Prolong PT and PTT
 Diagnosis u The clue to the viral aetiology will come from the travel and exposure history, so it is important to be aware of the incubation periods for these illnesses. u The causative virus may be isolated, or antigen detected, in maximum security laboratories from serum, pharynx, pleural exudate and urine.
Diagnosis u The clue to the viral aetiology will come from the travel and exposure history, so it is important to be aware of the incubation periods for these illnesses. u The causative virus may be isolated, or antigen detected, in maximum security laboratories from serum, pharynx, pleural exudate and urine.
 Management It is important to exclude other causes of fever, especially malaria, typhoid and respiratory tract infections. u Particular care must be taken with body fluids. Patients returning from endemic area with a fever should be managed in isolation until a diagnosis is made. u General supportive measures, preferably in a special unit, are required. u Ribavirin is given intravenously (100 mg/kg, then 25 mg/kg daily for 3 days and 12. 5 mg/kg daily for 4 days). u Once haemorrhagic fever is confirmed, full pressure isolation is mandatory and good infection control practices will prevent further transmission. u
Management It is important to exclude other causes of fever, especially malaria, typhoid and respiratory tract infections. u Particular care must be taken with body fluids. Patients returning from endemic area with a fever should be managed in isolation until a diagnosis is made. u General supportive measures, preferably in a special unit, are required. u Ribavirin is given intravenously (100 mg/kg, then 25 mg/kg daily for 3 days and 12. 5 mg/kg daily for 4 days). u Once haemorrhagic fever is confirmed, full pressure isolation is mandatory and good infection control practices will prevent further transmission. u
 GASTROINTESTINAL VIRAL INFECTIONS ROTAVIRUS u Rotaviruses are the major cause of diarrhoeal illness in young children, accounting for 30 -50% of cases admitted to hospital in developed countries, and 1020% of deaths due to gastroenteritis in developing countries. u Infection is endemic in developing countries and there are winter epidemics in developed countries. These viruses are easily transmitted and resist alcohol denaturation; person-to-person spread, especially by health-care workers in hospitals, is well documented. u u The virus infects enterocytes, causing decreased surface absorption and loss of enzymes on the brush border.
GASTROINTESTINAL VIRAL INFECTIONS ROTAVIRUS u Rotaviruses are the major cause of diarrhoeal illness in young children, accounting for 30 -50% of cases admitted to hospital in developed countries, and 1020% of deaths due to gastroenteritis in developing countries. u Infection is endemic in developing countries and there are winter epidemics in developed countries. These viruses are easily transmitted and resist alcohol denaturation; person-to-person spread, especially by health-care workers in hospitals, is well documented. u u The virus infects enterocytes, causing decreased surface absorption and loss of enzymes on the brush border.
 Diagnosis u The u incubation period is 48 hours. patients present with watery diarrhoea, vomiting, fever and abdominal pain. u Diagnosis is aided by commercially available enzyme immunoassay kits which simply require fresh or refrigerated stool for effective demonstration of the pathogens.
Diagnosis u The u incubation period is 48 hours. patients present with watery diarrhoea, vomiting, fever and abdominal pain. u Diagnosis is aided by commercially available enzyme immunoassay kits which simply require fresh or refrigerated stool for effective demonstration of the pathogens.
 Treatment u The disease is self-limiting but dehydration needs appropriate management. u Immunity infection. develops to natural
Treatment u The disease is self-limiting but dehydration needs appropriate management. u Immunity infection. develops to natural
 Other GIT viruses u HEPATITUS VIRUSES (A, B, C, D, E, F). Adenoviruses -Are frequently identified from stool culture and implicated as a cause of diarrhoea. u -Two serotypes (40 and 41) appear to be more frequently found in association with diarrhoea rather than the more common upper respiratory types 1 -7.
Other GIT viruses u HEPATITUS VIRUSES (A, B, C, D, E, F). Adenoviruses -Are frequently identified from stool culture and implicated as a cause of diarrhoea. u -Two serotypes (40 and 41) appear to be more frequently found in association with diarrhoea rather than the more common upper respiratory types 1 -7.
 RESPIRATORY VIRAL INFECTIONS u Adenoviruses, rhinoviruses u enteroviruses (Coxsackie viruses and echoviruses) often produce non-specific symptoms. The individual infections each produce lasting specific immunity. ** Influenza viruses u
RESPIRATORY VIRAL INFECTIONS u Adenoviruses, rhinoviruses u enteroviruses (Coxsackie viruses and echoviruses) often produce non-specific symptoms. The individual infections each produce lasting specific immunity. ** Influenza viruses u
 u. Human immunodeficiency virus infection (HIV) and the acquired immunodeficiency syndrome (AIDS)
u. Human immunodeficiency virus infection (HIV) and the acquired immunodeficiency syndrome (AIDS)
 EPIDEMIOLOGY AND BIOLOGY OF HIV u u The acquired immunodeficiency syndrome (AIDS) was first recognized in 1981. It is caused by the human immunodeficiency virus (HIV-1). HIV-2 causes a similar illness to HIV-1 but is less aggressive and restricted mainly to western Africa. Since 1981 AIDS has grown to be the second leading cause of disease burden world-wide and the leading cause of death in Africa, where it accounts for over 20% of deaths.
EPIDEMIOLOGY AND BIOLOGY OF HIV u u The acquired immunodeficiency syndrome (AIDS) was first recognized in 1981. It is caused by the human immunodeficiency virus (HIV-1). HIV-2 causes a similar illness to HIV-1 but is less aggressive and restricted mainly to western Africa. Since 1981 AIDS has grown to be the second leading cause of disease burden world-wide and the leading cause of death in Africa, where it accounts for over 20% of deaths.
 u. Immune deficiency is a consequence of continuous high-level HIV replication leading to immunemediated destruction of the key immune effector cell, the CD 4 lymphocyte.
u. Immune deficiency is a consequence of continuous high-level HIV replication leading to immunemediated destruction of the key immune effector cell, the CD 4 lymphocyte.
 MODES OF TRANSMISSION HIV is present in blood, semen and other body fluids such as breast milk and saliva. u Exposure to infected fluid leads to a risk of contacting infection, which is dependent on the integrity of the exposed site, the type and volume of body fluid, and the viral load. u HIV can enter either as free virus or within cells. u The modes of spread are sexual (man to man, heterosexual and oral), parenteral (blood or blood product recipients, injection drug-users and those experiencing occupational injury) and vertical. u
MODES OF TRANSMISSION HIV is present in blood, semen and other body fluids such as breast milk and saliva. u Exposure to infected fluid leads to a risk of contacting infection, which is dependent on the integrity of the exposed site, the type and volume of body fluid, and the viral load. u HIV can enter either as free virus or within cells. u The modes of spread are sexual (man to man, heterosexual and oral), parenteral (blood or blood product recipients, injection drug-users and those experiencing occupational injury) and vertical. u
 VIROLOGY AND IMMUNOLOGY HIV is a single-stranded RNA retrovirus from the Lentivirus family. u After mucosal exposure, HIV is transported to the lymph nodes via dendritic, CD 4 or Langerhans cells, where infection becomes established. u Free or cell-associated virus is then disseminated widely through the blood with seeding of 'sanctuary' sites (e. g. central nervous system) and latent CD 4 cell reservoirs. u With time, there is gradual attrition of the CD 4 cell population, resulting in increasing impairment of cell-mediated immunity and susceptibility to opportunistic infections. u
VIROLOGY AND IMMUNOLOGY HIV is a single-stranded RNA retrovirus from the Lentivirus family. u After mucosal exposure, HIV is transported to the lymph nodes via dendritic, CD 4 or Langerhans cells, where infection becomes established. u Free or cell-associated virus is then disseminated widely through the blood with seeding of 'sanctuary' sites (e. g. central nervous system) and latent CD 4 cell reservoirs. u With time, there is gradual attrition of the CD 4 cell population, resulting in increasing impairment of cell-mediated immunity and susceptibility to opportunistic infections. u
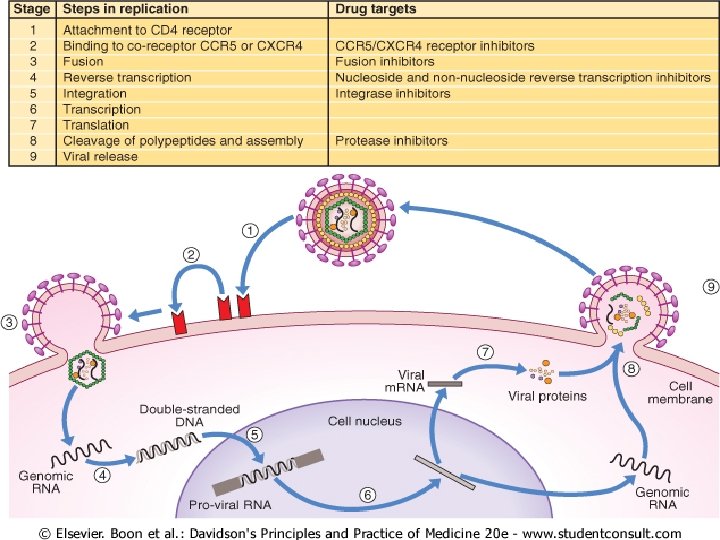
 As CD 4 cells are pivotal in orchestrating the immune response, any depletion in numbers renders the body susceptible to opportunistic infections and oncogenic virus-related tumours. u The predominant opportunist infections seen in HIV disease are intracellular parasites (e. g. Mycobacterium tuberculosis) or pathogens susceptible to cell-mediated rather than antibody-mediated immune responses. u The reduction in the number of CD 4 cells circulating in peripheral blood is tightly correlated with the amount of plasma viral load. u Both are monitored closely in patients and are used as measures of disease progression. u
As CD 4 cells are pivotal in orchestrating the immune response, any depletion in numbers renders the body susceptible to opportunistic infections and oncogenic virus-related tumours. u The predominant opportunist infections seen in HIV disease are intracellular parasites (e. g. Mycobacterium tuberculosis) or pathogens susceptible to cell-mediated rather than antibody-mediated immune responses. u The reduction in the number of CD 4 cells circulating in peripheral blood is tightly correlated with the amount of plasma viral load. u Both are monitored closely in patients and are used as measures of disease progression. u
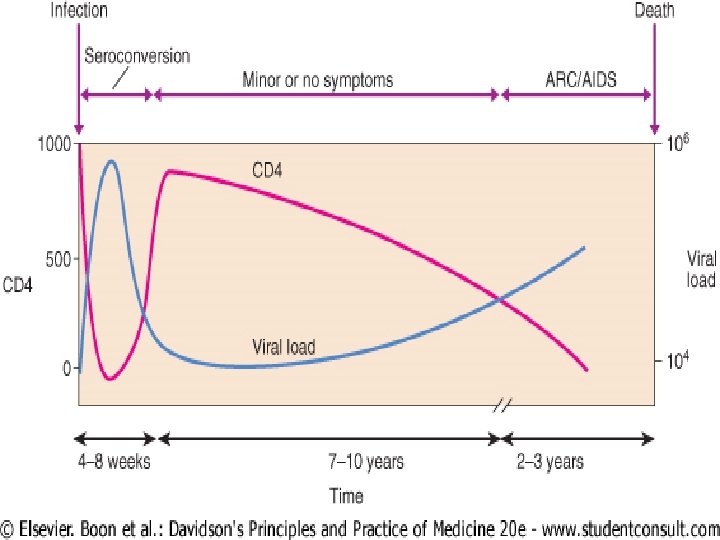
 u Virus-specific CD 8 cytotoxic T-cell lymphocytes develop rapidly after infection and are the most important element in recognising, binding and lysing infected CD 4 cells. u They play a crucial role in controlling HIV replication after infection and determine the viral 'set-point' and subsequent rate of disease progression.
u Virus-specific CD 8 cytotoxic T-cell lymphocytes develop rapidly after infection and are the most important element in recognising, binding and lysing infected CD 4 cells. u They play a crucial role in controlling HIV replication after infection and determine the viral 'set-point' and subsequent rate of disease progression.
 NATURAL HISTORY AND CLASSIFICATION OF HIV u Primary infection u Asymptomatic u HIV infection SYMPTOMATIC DISEASES u Mildly symptomatic disease u Acquired immunodeficiency syndrome (AIDS)
NATURAL HISTORY AND CLASSIFICATION OF HIV u Primary infection u Asymptomatic u HIV infection SYMPTOMATIC DISEASES u Mildly symptomatic disease u Acquired immunodeficiency syndrome (AIDS)
 Primary infection u Fever with rash u Pharyngitis with cervical lymphadenopathy u Myalgia/arthralgia u Headache u Mucosal ulceration
Primary infection u Fever with rash u Pharyngitis with cervical lymphadenopathy u Myalgia/arthralgia u Headache u Mucosal ulceration
 u u u Primary infection is symptomatic in 70 -80% of cases and usually occurs 2 -6 weeks after exposure. Rarely, presentation may be neurological (aseptic meningitis, encephalitis, myelitis, polyneuritis). This coincides with a surge in plasma HIV-RNA levels to > 1 million copies/ml (peak between 4 and 8 weeks), and a fall in the CD 4 count to 300 -400 cells/mm 3, but occasionally to below 200 when opportunistic infections (e. g. oropharyngeal candidiasis, Pneumocystis carinii (jirovecii) pneumonia) may rarely occur. Symptomatic recovery occurs after 1 -2 weeks but occasionally may take up to 10 weeks and parallels the return of the CD 4 count and fall in the viral load. In many patients the illness is mild and only identified by retrospective enquiry at later presentation. However, the CD 4 count rarely recovers to its previous value.
u u u Primary infection is symptomatic in 70 -80% of cases and usually occurs 2 -6 weeks after exposure. Rarely, presentation may be neurological (aseptic meningitis, encephalitis, myelitis, polyneuritis). This coincides with a surge in plasma HIV-RNA levels to > 1 million copies/ml (peak between 4 and 8 weeks), and a fall in the CD 4 count to 300 -400 cells/mm 3, but occasionally to below 200 when opportunistic infections (e. g. oropharyngeal candidiasis, Pneumocystis carinii (jirovecii) pneumonia) may rarely occur. Symptomatic recovery occurs after 1 -2 weeks but occasionally may take up to 10 weeks and parallels the return of the CD 4 count and fall in the viral load. In many patients the illness is mild and only identified by retrospective enquiry at later presentation. However, the CD 4 count rarely recovers to its previous value.
 Diagnosis is made by u 1 - detecting HIV-RNA in the serum or u 2 - immunoblot assay (which shows antibodies developing to early proteins). u The appearance of specific anti-HIV antibodies in serum (seroconversion) takes place later at 3 -12 weeks (median 8 weeks), although very rarely seroconversion may take place after 3 months.
Diagnosis is made by u 1 - detecting HIV-RNA in the serum or u 2 - immunoblot assay (which shows antibodies developing to early proteins). u The appearance of specific anti-HIV antibodies in serum (seroconversion) takes place later at 3 -12 weeks (median 8 weeks), although very rarely seroconversion may take place after 3 months.
 Factors likely to indicate a faster progression of HIV are u 1 -the presence and duration of symptoms, u 2 - evidence of candidiasis, and u 3 - neurological involvement. u 4 - The level of the viral load postseroconversion strongly correlates with subsequent progression of disease.
Factors likely to indicate a faster progression of HIV are u 1 -the presence and duration of symptoms, u 2 - evidence of candidiasis, and u 3 - neurological involvement. u 4 - The level of the viral load postseroconversion strongly correlates with subsequent progression of disease.
 The differential diagnosis of primary HIV includes u acute Epstein-Barr virus (EBV), u cytomegalovirus (CMV), u streptococcal pharyngitis, u toxoplasmosis u secondary syphilis.
The differential diagnosis of primary HIV includes u acute Epstein-Barr virus (EBV), u cytomegalovirus (CMV), u streptococcal pharyngitis, u toxoplasmosis u secondary syphilis.
 Asymptomatic infection category A disease in the Centers for Disease Control (CDC) classification u follows and lasts for a variable period, during which the infected individual remains well with no evidence of disease except for the possible presence of persistent generalised lymphadenopathy (PGL, defined as enlarged glands at ≥ 2 extra-inguinal sites). u At this stage the bulk of virus replication takes place within lymphoid tissue (e. g. follicular dendritic cells). u There is sustained viraemia with a decline in CD 4 count dependent on the height of the viral load but usually between 50 and 150 cells/cc/year u
Asymptomatic infection category A disease in the Centers for Disease Control (CDC) classification u follows and lasts for a variable period, during which the infected individual remains well with no evidence of disease except for the possible presence of persistent generalised lymphadenopathy (PGL, defined as enlarged glands at ≥ 2 extra-inguinal sites). u At this stage the bulk of virus replication takes place within lymphoid tissue (e. g. follicular dendritic cells). u There is sustained viraemia with a decline in CD 4 count dependent on the height of the viral load but usually between 50 and 150 cells/cc/year u
 HIV SYMPTOMATIC DISEASES Oral hairy leucoplakia u Recurrent oropharyngeal candidiasis u Recurrent vaginal candidiasis u Severe pelvic inflammatory disease u Bacillary angiomatosis u Cervical dysplasia u Idiopathic thrombocytopenic purpura u Weight loss* u Chronic diarrhoea* u Herpes zoster u Peripheral neuropathy u Low-grade fever/night sweats* u
HIV SYMPTOMATIC DISEASES Oral hairy leucoplakia u Recurrent oropharyngeal candidiasis u Recurrent vaginal candidiasis u Severe pelvic inflammatory disease u Bacillary angiomatosis u Cervical dysplasia u Idiopathic thrombocytopenic purpura u Weight loss* u Chronic diarrhoea* u Herpes zoster u Peripheral neuropathy u Low-grade fever/night sweats* u
 Mildly symptomatic disease u Centers for Disease Control (CDC) Classification category B disease. u indicating some impairment of the cellular immune system. u These diseases correspond to AIDSrelated complex (ARC) conditions but by definition are not AIDS-defining. u The median interval from infection to the development of symptoms is around 7 -10 years, although subgroups of patients exhibit 'fast' or 'slow' rates of progression.
Mildly symptomatic disease u Centers for Disease Control (CDC) Classification category B disease. u indicating some impairment of the cellular immune system. u These diseases correspond to AIDSrelated complex (ARC) conditions but by definition are not AIDS-defining. u The median interval from infection to the development of symptoms is around 7 -10 years, although subgroups of patients exhibit 'fast' or 'slow' rates of progression.
 Acquired immunodeficiency syndrome (AIDS) u AIDS (CDC Classification category C disease) u is defined by the development of specified opportunistic infections, tumours etc. u There is correlation between CD 4 count and HIV-related diseases (type of the disease). ( box 14. 7)
Acquired immunodeficiency syndrome (AIDS) u AIDS (CDC Classification category C disease) u is defined by the development of specified opportunistic infections, tumours etc. u There is correlation between CD 4 count and HIV-related diseases (type of the disease). ( box 14. 7)
 AIDS-DEFINING DISEASE (box 14. 6 ) Oesophageal candidiasis u Cryptococcal meningitis u Chronic cryptosporidial diarrhoea u CMV retinitis or colitis u Chronic mucocutaneous herpes simplex u Disseminated Mycobacterium avium intracellulare u Pulmonary or extrapulmonary tuberculosis u Pneumocystis carinii (jirovecii) pneumonia u Progressive multifocal leucoencephalopathy u Recurrent non-typhi Salmonella septicaemia u Cerebral toxoplasmosis u
AIDS-DEFINING DISEASE (box 14. 6 ) Oesophageal candidiasis u Cryptococcal meningitis u Chronic cryptosporidial diarrhoea u CMV retinitis or colitis u Chronic mucocutaneous herpes simplex u Disseminated Mycobacterium avium intracellulare u Pulmonary or extrapulmonary tuberculosis u Pneumocystis carinii (jirovecii) pneumonia u Progressive multifocal leucoencephalopathy u Recurrent non-typhi Salmonella septicaemia u Cerebral toxoplasmosis u
 AIDS-DEFINING DISEASE (box 14. 6 ) cont. u Extrapulmonary coccidioidomycosis u Invasive cervical cancer u Extrapulmonary histoplasmosis u Kaposi's sarcoma u Non-Hodgkin lymphoma u Primary cerebral lymphoma u HIV-associated wasting u HIV-associated dementia
AIDS-DEFINING DISEASE (box 14. 6 ) cont. u Extrapulmonary coccidioidomycosis u Invasive cervical cancer u Extrapulmonary histoplasmosis u Kaposi's sarcoma u Non-Hodgkin lymphoma u Primary cerebral lymphoma u HIV-associated wasting u HIV-associated dementia
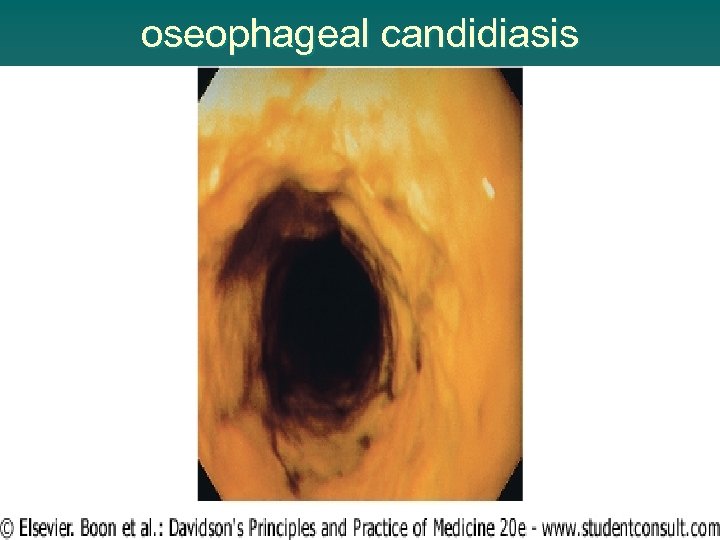 oseophageal candidiasis
oseophageal candidiasis
 DIFFERENTIAL DIAGNOSIS OF HIV-RELATED SKIN DISEASE Early HIV Infection u Herpes simplex u Varicella zoster u Human papillomavirus (HPV) u Impetigo u Dermatophytosis u Scabies u Syphilis u HIV seroconversion u Other u Xeroderma u Pruritus u Seborrhoeic dermatitis u Drug reaction (co-trimoxazole/nevirapine) u Itchy folliculitis u Psoriasis u Acne
DIFFERENTIAL DIAGNOSIS OF HIV-RELATED SKIN DISEASE Early HIV Infection u Herpes simplex u Varicella zoster u Human papillomavirus (HPV) u Impetigo u Dermatophytosis u Scabies u Syphilis u HIV seroconversion u Other u Xeroderma u Pruritus u Seborrhoeic dermatitis u Drug reaction (co-trimoxazole/nevirapine) u Itchy folliculitis u Psoriasis u Acne
 Late HIV Common u Kaposi's sarcoma u Molluscum contagiosum u Chronic mucocutaneous herpes simplex Rare u Bacillary angiomatosis u CMV u Non-Hodgkin lymphoma u Cryptococcus u Histoplasmosis u Mycobacterial (tuberculosis/atypical)
Late HIV Common u Kaposi's sarcoma u Molluscum contagiosum u Chronic mucocutaneous herpes simplex Rare u Bacillary angiomatosis u CMV u Non-Hodgkin lymphoma u Cryptococcus u Histoplasmosis u Mycobacterial (tuberculosis/atypical)
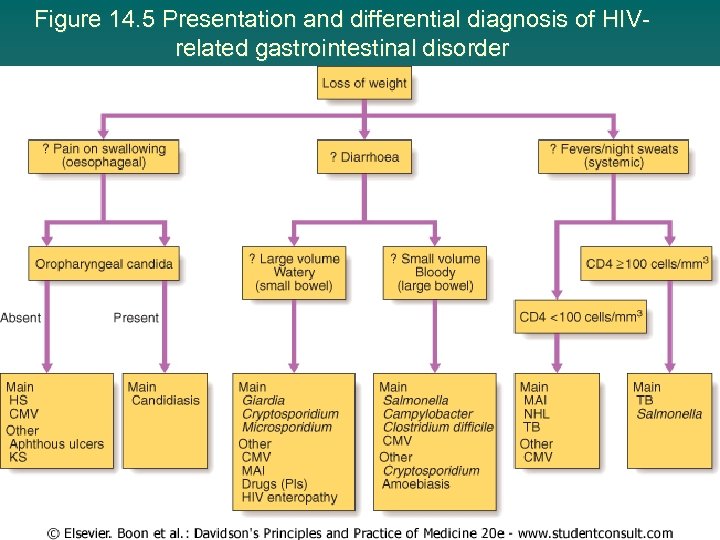 Figure 14. 5 Presentation and differential diagnosis of HIVrelated gastrointestinal disorder
Figure 14. 5 Presentation and differential diagnosis of HIVrelated gastrointestinal disorder
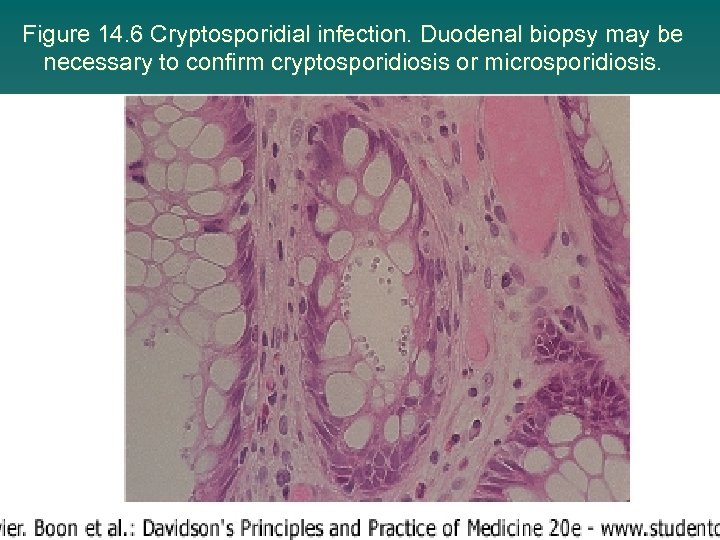 Figure 14. 6 Cryptosporidial infection. Duodenal biopsy may be necessary to confirm cryptosporidiosis or microsporidiosis.
Figure 14. 6 Cryptosporidial infection. Duodenal biopsy may be necessary to confirm cryptosporidiosis or microsporidiosis.
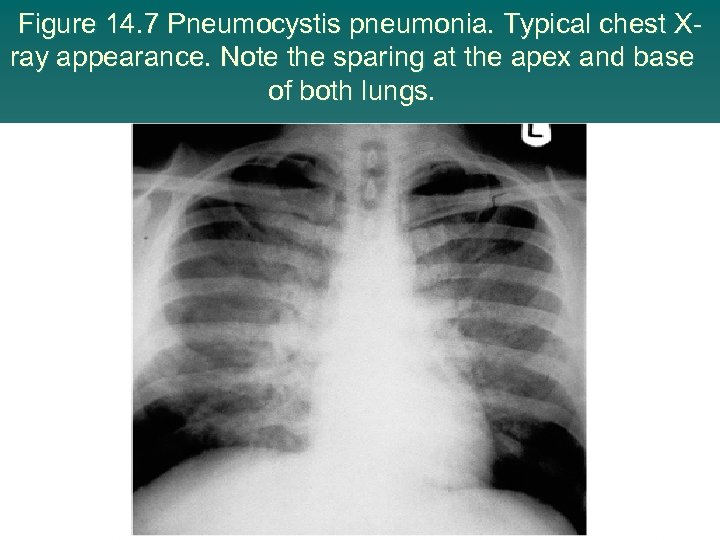 Figure 14. 7 Pneumocystis pneumonia. Typical chest Xray appearance. Note the sparing at the apex and base of both lungs.
Figure 14. 7 Pneumocystis pneumonia. Typical chest Xray appearance. Note the sparing at the apex and base of both lungs.
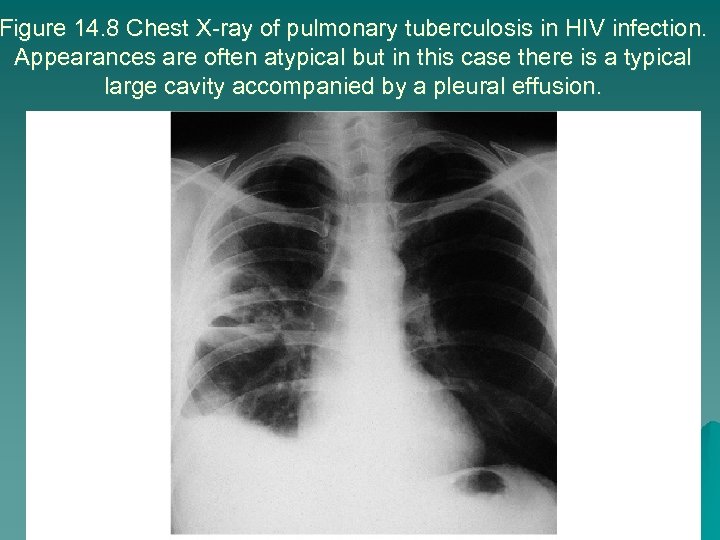 Figure 14. 8 Chest X-ray of pulmonary tuberculosis in HIV infection. Appearances are often atypical but in this case there is a typical large cavity accompanied by a pleural effusion.
Figure 14. 8 Chest X-ray of pulmonary tuberculosis in HIV infection. Appearances are often atypical but in this case there is a typical large cavity accompanied by a pleural effusion.
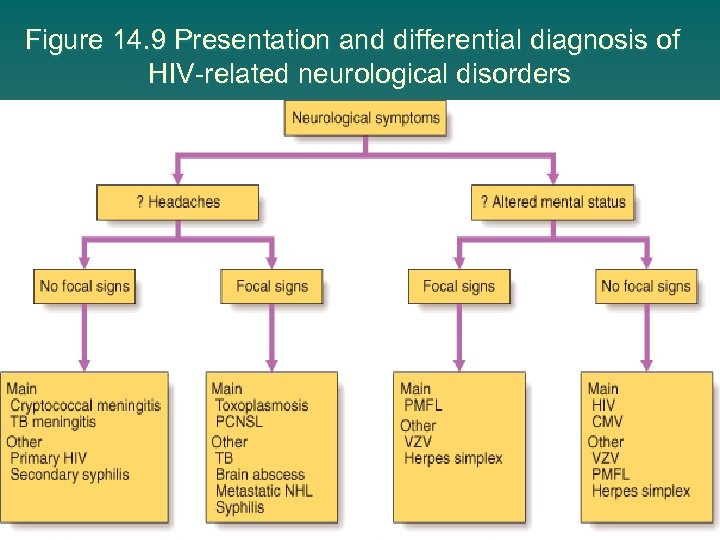 Figure 14. 9 Presentation and differential diagnosis of HIV-related neurological disorders
Figure 14. 9 Presentation and differential diagnosis of HIV-related neurological disorders
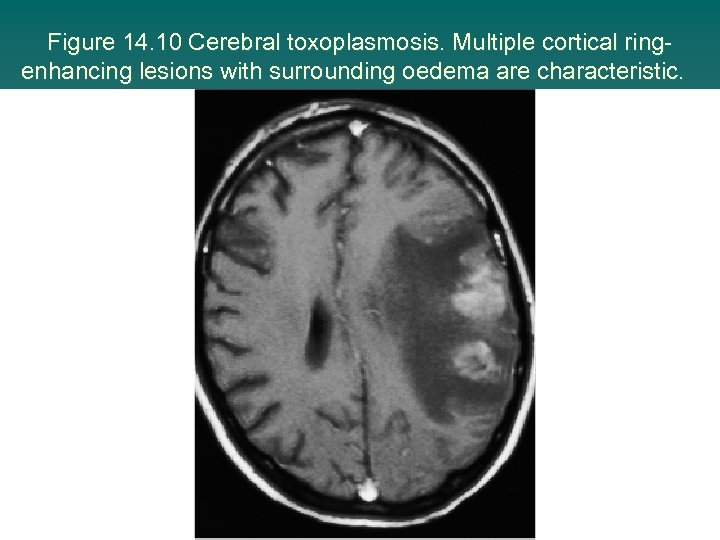 Figure 14. 10 Cerebral toxoplasmosis. Multiple cortical ringenhancing lesions with surrounding oedema are characteristic.
Figure 14. 10 Cerebral toxoplasmosis. Multiple cortical ringenhancing lesions with surrounding oedema are characteristic.
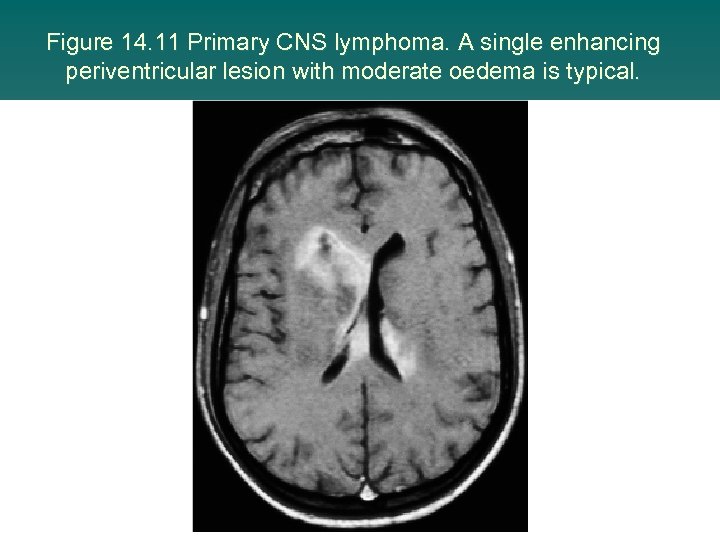 Figure 14. 11 Primary CNS lymphoma. A single enhancing periventricular lesion with moderate oedema is typical.
Figure 14. 11 Primary CNS lymphoma. A single enhancing periventricular lesion with moderate oedema is typical.
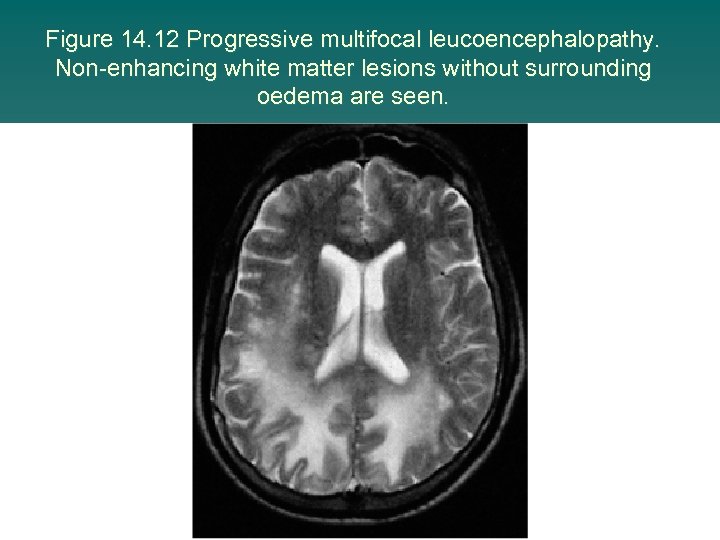 Figure 14. 12 Progressive multifocal leucoencephalopathy. Non-enhancing white matter lesions without surrounding oedema are seen.
Figure 14. 12 Progressive multifocal leucoencephalopathy. Non-enhancing white matter lesions without surrounding oedema are seen.
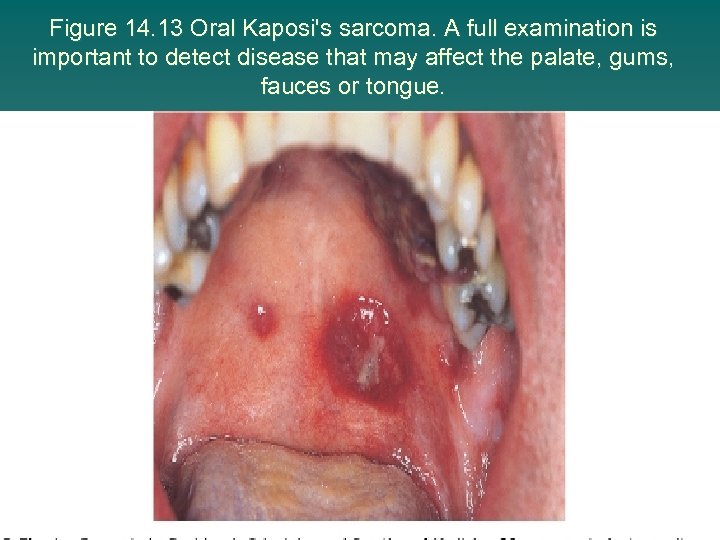 Figure 14. 13 Oral Kaposi's sarcoma. A full examination is important to detect disease that may affect the palate, gums, fauces or tongue.
Figure 14. 13 Oral Kaposi's sarcoma. A full examination is important to detect disease that may affect the palate, gums, fauces or tongue.
 MANAGEMENT OF HIV Management of HIV involves both treatment of the virus and prevention of opportunistic infections. The aims of HIV treatment are to: 1 - reduce the viral load to an undetectable level (< 50 copies/ml) for as long as possible u 2 - improve the CD 4 count (above 200 cells/mm 3 , when the significant HIV-related events rarely occur) u 3 - increase the quantity and improve the quality of life without unacceptable drug-related side-effects or lifestyle alteration u 4 - reduce transmission (mother-to-child and person-to-person). u
MANAGEMENT OF HIV Management of HIV involves both treatment of the virus and prevention of opportunistic infections. The aims of HIV treatment are to: 1 - reduce the viral load to an undetectable level (< 50 copies/ml) for as long as possible u 2 - improve the CD 4 count (above 200 cells/mm 3 , when the significant HIV-related events rarely occur) u 3 - increase the quantity and improve the quality of life without unacceptable drug-related side-effects or lifestyle alteration u 4 - reduce transmission (mother-to-child and person-to-person). u
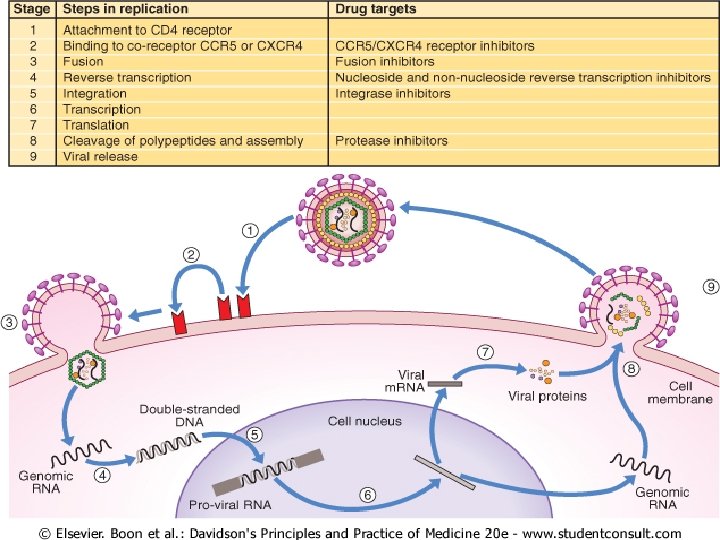
 DRUGS ANTIRETROVIRAL DRUGS u Nucleoside reverse transcriptase inhibitors (NRTIs) u Non-nucleoside reverse transcriptase inhibitors (NNRTIs) u Protease inhibitors (PIs) u Others The drugs that are currently used, their side-effects and a glossary of terms and abbreviations are given in Boxes 14. 22, 14. 23 and 14. 24.
DRUGS ANTIRETROVIRAL DRUGS u Nucleoside reverse transcriptase inhibitors (NRTIs) u Non-nucleoside reverse transcriptase inhibitors (NNRTIs) u Protease inhibitors (PIs) u Others The drugs that are currently used, their side-effects and a glossary of terms and abbreviations are given in Boxes 14. 22, 14. 23 and 14. 24.
 Nucleoside reverse transcriptase inhibitors (NRTIs) u Zalcitabine (dd. C) u Didanosine (dd. I) u Lamivudine (3 TC) u Zidovudine (ZDV) u Stavudine (d 4 T) u Abacavir u Emitricitabine (FTC)
Nucleoside reverse transcriptase inhibitors (NRTIs) u Zalcitabine (dd. C) u Didanosine (dd. I) u Lamivudine (3 TC) u Zidovudine (ZDV) u Stavudine (d 4 T) u Abacavir u Emitricitabine (FTC)
 Non-nucleoside reverse transcriptase inhibitors (NNRTIs) u Nevirapine u Efavirenz u Delavirdine 1
Non-nucleoside reverse transcriptase inhibitors (NNRTIs) u Nevirapine u Efavirenz u Delavirdine 1
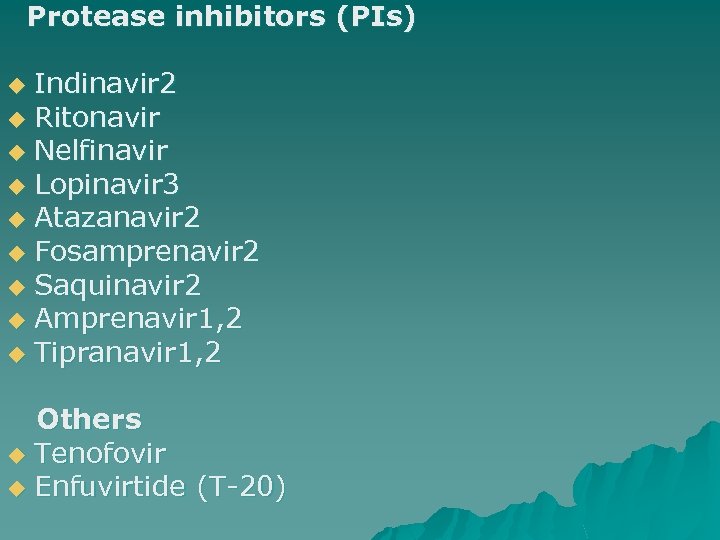 Protease inhibitors (PIs) Indinavir 2 u Ritonavir u Nelfinavir u Lopinavir 3 u Atazanavir 2 u Fosamprenavir 2 u Saquinavir 2 u Amprenavir 1, 2 u Tipranavir 1, 2 u Others u Tenofovir u Enfuvirtide (T-20)
Protease inhibitors (PIs) Indinavir 2 u Ritonavir u Nelfinavir u Lopinavir 3 u Atazanavir 2 u Fosamprenavir 2 u Saquinavir 2 u Amprenavir 1, 2 u Tipranavir 1, 2 u Others u Tenofovir u Enfuvirtide (T-20)
 Notes about HIV drugs u The inclusion of two NRTIs, or one NRTI and tenofovir, remains the cornerstone of HAART. (highly active antiretroviral therapy)=combination treatment u Resistance to all NRTIs will occur unless they are part of a maximally suppressive HAART regimen. u . More recently, two PIs (atazanavir and osamprenavir) have become available; they allow for once-daily administration with fewer tablets. The subsequent fall in morbidity and mortality can be directly linked to the introduction of these drugs and their use in HAART. When they are given with two NRTIs the combination controls viral replication in plasma and tissues, and allows reconstitution of the immune system.
Notes about HIV drugs u The inclusion of two NRTIs, or one NRTI and tenofovir, remains the cornerstone of HAART. (highly active antiretroviral therapy)=combination treatment u Resistance to all NRTIs will occur unless they are part of a maximally suppressive HAART regimen. u . More recently, two PIs (atazanavir and osamprenavir) have become available; they allow for once-daily administration with fewer tablets. The subsequent fall in morbidity and mortality can be directly linked to the introduction of these drugs and their use in HAART. When they are given with two NRTIs the combination controls viral replication in plasma and tissues, and allows reconstitution of the immune system.
 u u Short- and long-term side-effects are not infrequent. Fat redistribution occurred in 2030% of patients treated with HAART including a PI after 2 years. The syndrome is characterised by peripheral fat-wasting (cheeks, temples, limbs and buttocks), localised collections (buffalo hump, peripheral lipomatosis, and breast enlargement in women) and central adiposity. Enfuvirtide (T-20) is a new class of antiretroviral drug which prevents viral entry into cells and preventing fusion. It is highly active but has to be injected subcutaneously 12 -hourly and therefore is reserved for patients with more advanced disease and fewer options.
u u Short- and long-term side-effects are not infrequent. Fat redistribution occurred in 2030% of patients treated with HAART including a PI after 2 years. The syndrome is characterised by peripheral fat-wasting (cheeks, temples, limbs and buttocks), localised collections (buffalo hump, peripheral lipomatosis, and breast enlargement in women) and central adiposity. Enfuvirtide (T-20) is a new class of antiretroviral drug which prevents viral entry into cells and preventing fusion. It is highly active but has to be injected subcutaneously 12 -hourly and therefore is reserved for patients with more advanced disease and fewer options.
 14. 27 FACTORS TO CONSIDER WHEN CHOOSING HAART Ease of compliance u Fit of the drug regimen around the patient's lifestyle u Wishes of the patient u Stage of disease u Coexisting/past medical history u Possibility of additive side-effects (e. g. dd. I and neuropathy) u Potential for drug interactions with non-HIV medications u Antagonistic NRTI combinations (ZDV/d 4 T and dd. C/3 TC) u CNS penetration u Possibility of acquisition of resistant virus u
14. 27 FACTORS TO CONSIDER WHEN CHOOSING HAART Ease of compliance u Fit of the drug regimen around the patient's lifestyle u Wishes of the patient u Stage of disease u Coexisting/past medical history u Possibility of additive side-effects (e. g. dd. I and neuropathy) u Potential for drug interactions with non-HIV medications u Antagonistic NRTI combinations (ZDV/d 4 T and dd. C/3 TC) u CNS penetration u Possibility of acquisition of resistant virus u
 POST-EXPOSURE PROPHYLAXIS u Combination therapy is now recommended for occupational post-exposure prophylaxis (PEP) where the risk is deemed to be significant, although there is no evidence for this practice. The first dose should be given as soon as possible. However, protection is not absolute and health-care workers have been reported to seroconvert despite taking a full course of three drugs started within hours of exposure. Recommended PEP is ZDV, 3 TC and indinavir or nelfinavir for 28 days. u Vaccine development is slow. u
POST-EXPOSURE PROPHYLAXIS u Combination therapy is now recommended for occupational post-exposure prophylaxis (PEP) where the risk is deemed to be significant, although there is no evidence for this practice. The first dose should be given as soon as possible. However, protection is not absolute and health-care workers have been reported to seroconvert despite taking a full course of three drugs started within hours of exposure. Recommended PEP is ZDV, 3 TC and indinavir or nelfinavir for 28 days. u Vaccine development is slow. u



