bc21d8650affb47b74053f0fb225f094.ppt
- Количество слайдов: 12

Viral Hepatitis Prevention Board Geneve, March 14, 2003 Hepatitis B Vaccination: Impact of Safety Issues Israel Daniel Shouval Liver Unit Hadassah – Hebrew University Hospital Jerusalem

*Israel: Population Profile • Population: 6, 609, 000 • Jews and others 5, 281, 000, Arabs 1, 227, 000 • Annual live births (2001) 136, 700 • 68% Jews, 26% Muslims, 2% Christian, 2% Druzes, 2% others. • GNP: $ 16, 310 • Health Expenditure per capita - $1, 500 *Ref: 53 rd annual statistical report.
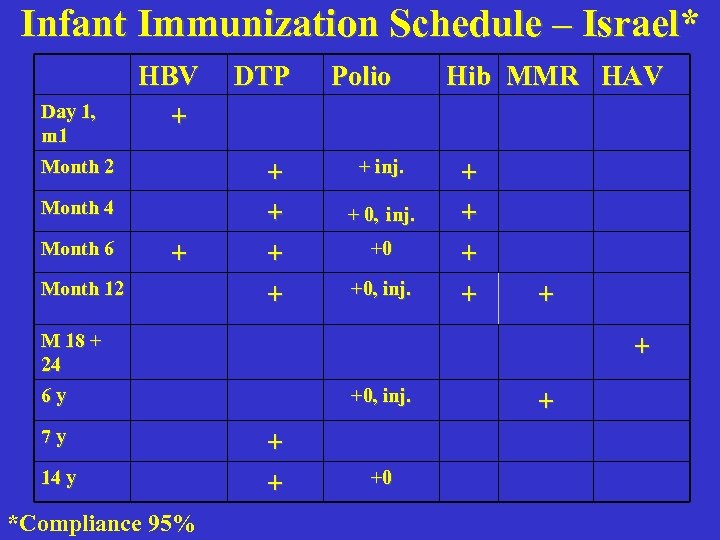
Infant Immunization Schedule – Israel* Day 1, m 1 HBV + Month 2 Month 4 Month 6 + Month 12 DTP + + Polio + inj. + 0, inj. +0 +0, inj. Hib MMR HAV + + + M 18 + 24 + 6 y 7 y 14 y *Compliance 95% +0, inj. + + +0 +

Post Immunization AE Report Form Local Shock Redness swelling >5 cm, pain. Anaphylaxis Nodule (cc) Pallor, cyanosis Cellulitis Sweling of lips, face Abcess Miscellaneous Limping Dyspnea, Loss of consciousness General Restlessness, Screaming >3 h Temperature >38°c apathy, collapse. Allergic reaction (Erythema, rash, urticaria Convulsions, encephalitis, vomitting, diarrea, catarrh conjunctivitis, lymphadentitis, Orchitis, arthralgia, arthritis
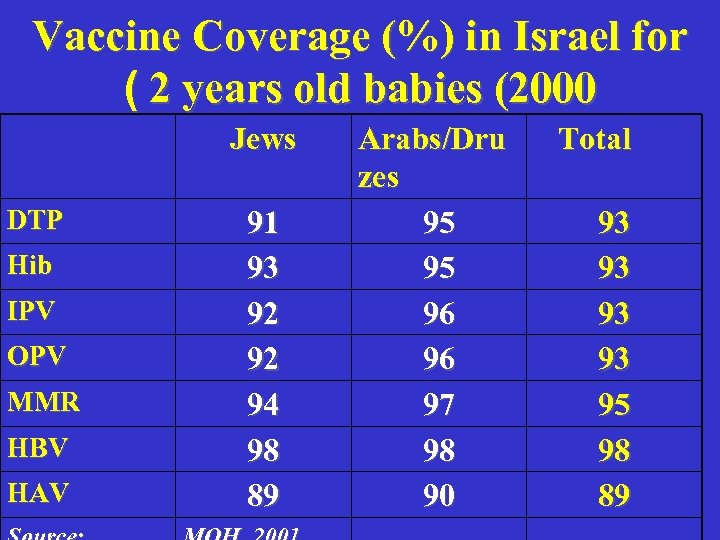
Vaccine Coverage (%) in Israel for ( 2 years old babies (2000 Jews DTP Hib IPV OPV MMR HBV HAV 91 93 92 92 94 98 89 Arabs/Dru zes 95 95 96 96 97 98 90 Total 93 93 95 98 89
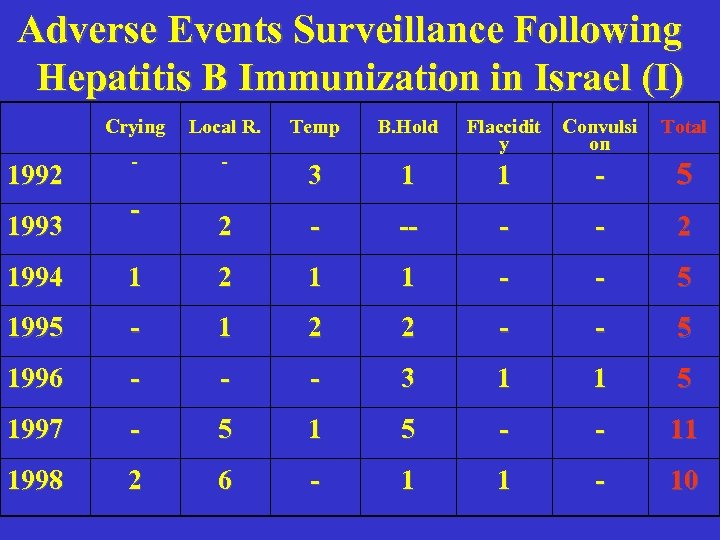
Adverse Events Surveillance Following Hepatitis B Immunization in Israel (I) Crying 1992 1993 Local R. Temp B. Hold - Convulsi on Total - Flaccidit y 3 1 1 - 5 2 - -- - - 2 - 1994 1 2 1 1 - - 5 1995 - 1 2 2 - - 5 1996 - - - 3 1 1 5 1997 - 5 1 5 - - 11 1998 2 6 - 1 1 - 10
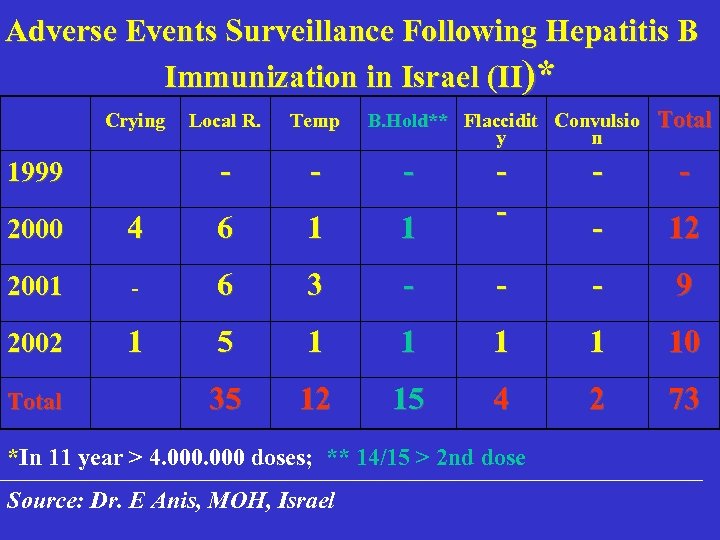
Adverse Events Surveillance Following Hepatitis B Immunization in Israel (II)* Crying Temp - 1999 Local R. B. Hold** Flaccidit Convulsio y n - - Total - - 12 2000 4 6 1 1 2001 - 6 3 - - - 9 2002 1 5 1 1 10 35 12 15 4 2 73 Total *In 11 year > 4. 000 doses; ** 14/15 > 2 nd dose Source: Dr. E Anis, MOH, Israel

*Hepatitis B Vaccines: No of doses Year No 1999 2000 2001 2002 436, 300 432, 422 436, 674 453, 500 * > 4, 000 doses in 10 years.

Litigations Regarding Vaccine AEs Year No 1995 1 1999 1 1996 1 2000 3 1997 1 2001 1 1998 2 2002 3 In past 30 years, 35 litigations concerning polio, 35 litigations concerning DTP, 1 concerning HBV.

Safety Issues and Immunization • Patient bill of rights • How much information should be given to parents prior to immunization of a child (informed consent). • The law does not enable forced vaccination • National compensation law for vaccinees with serious AES (1989).

Arguments of Alternative Medicine and Antivaccine Advocates Against Universal Immunization on Day 1 of Life • Unawareness of parents • Informed consent is given at admission to maternity • • ward (stress) MOH takes advantage of parents fear of “neonatal jaundice” Vaccine contains Thimerosal and formaldehyde which maybe toxic to CNS Immature immune system of the neonate HBV is a sexual transmitted infection and therefore irrelevant to neonates. Vaccination start at birth for technical convenience reasons

Causes of delaying or refusal of vaccination • Negligence of parents • Advocate of alternative medicine (“Natural immunization”, AEs) • Special groups (religious reasons) • Fear of AEs
bc21d8650affb47b74053f0fb225f094.ppt