dd20b6b31409b421f541c29d8c578a37.ppt
- Количество слайдов: 62
 Viral Hemorrhagic Fever
Viral Hemorrhagic Fever
 Overview n n n n Organism History Epidemiology Transmission Disease in Humans Disease in Animals Prevention and Control
Overview n n n n Organism History Epidemiology Transmission Disease in Humans Disease in Animals Prevention and Control
 What is Viral Hemorrhagic Fever? n n n Severe multisystem syndrome Diffuse Damage to overall vascular system Symptoms often accompanied by hemorrhage Rarely life threatening in itself n Includes conjunctivitis, petechia, echymosis n n Relatively high mortality
What is Viral Hemorrhagic Fever? n n n Severe multisystem syndrome Diffuse Damage to overall vascular system Symptoms often accompanied by hemorrhage Rarely life threatening in itself n Includes conjunctivitis, petechia, echymosis n n Relatively high mortality
 Quick Overview: Who are they? n VHFs are: Enveloped Lipid-encapsulated n Single-strand RNA n Zoonotic (animal-borne) n Geographically restricted by host n Persistent in nature (rodents, bats, mosquitoes, ticks, livestock, monkeys, and primates) n n Survival dependent on an animal or insect host, for the natural reservoir
Quick Overview: Who are they? n VHFs are: Enveloped Lipid-encapsulated n Single-strand RNA n Zoonotic (animal-borne) n Geographically restricted by host n Persistent in nature (rodents, bats, mosquitoes, ticks, livestock, monkeys, and primates) n n Survival dependent on an animal or insect host, for the natural reservoir
 Quick Overview: Who are they? n Arenaviridae n n n Lassa Fever Argentine HF (Junin) Bolivian HF (Machupo) Brazilian HF (Sabia) Venezuelan HF (Guanarito) n Filoviridae n n n Flaviviridae n n n Bunyaviridae n n Rift Valley Fever (RVF) Crimean Congo HF (CCHF) Hantavirus (Hemorrhagic Fever with Renal Syndrome (HFRS)) Hantavirus Pulmonary Syndrome (HPS) Marburg Ebola n n Yellow Fever Dengue Fever Omsk HF Kyasanur Forest Disease
Quick Overview: Who are they? n Arenaviridae n n n Lassa Fever Argentine HF (Junin) Bolivian HF (Machupo) Brazilian HF (Sabia) Venezuelan HF (Guanarito) n Filoviridae n n n Flaviviridae n n n Bunyaviridae n n Rift Valley Fever (RVF) Crimean Congo HF (CCHF) Hantavirus (Hemorrhagic Fever with Renal Syndrome (HFRS)) Hantavirus Pulmonary Syndrome (HPS) Marburg Ebola n n Yellow Fever Dengue Fever Omsk HF Kyasanur Forest Disease
 Quick Overview: How do we get infected? n Rodents & Arthropods, both reservoir & vector Bites of infected mosquito or tick n Inhalation of rodent excreta n Infected animal product exposure n n Person-to-Person Blood/body fluid exposure n Airborne potential for some arenaviridae, filoviridae n
Quick Overview: How do we get infected? n Rodents & Arthropods, both reservoir & vector Bites of infected mosquito or tick n Inhalation of rodent excreta n Infected animal product exposure n n Person-to-Person Blood/body fluid exposure n Airborne potential for some arenaviridae, filoviridae n
 Arenaviridae • • • Junin virus Machupo virus Guanarito virus Lassa virus Sabia virus
Arenaviridae • • • Junin virus Machupo virus Guanarito virus Lassa virus Sabia virus
 Arenaviridae History n n First isolated in 1933 1958: Junin virus - Argentina First to cause hemorrhagic fever n Argentine hemorrhagic fever n n 1963: Machupo virus – Bolivia n n Bolivian hemorrhagic fever Guanarito (Venezuela) Sabia (Brazil) 1969: Lassa virus – Nigeria n Lassa fever
Arenaviridae History n n First isolated in 1933 1958: Junin virus - Argentina First to cause hemorrhagic fever n Argentine hemorrhagic fever n n 1963: Machupo virus – Bolivia n n Bolivian hemorrhagic fever Guanarito (Venezuela) Sabia (Brazil) 1969: Lassa virus – Nigeria n Lassa fever
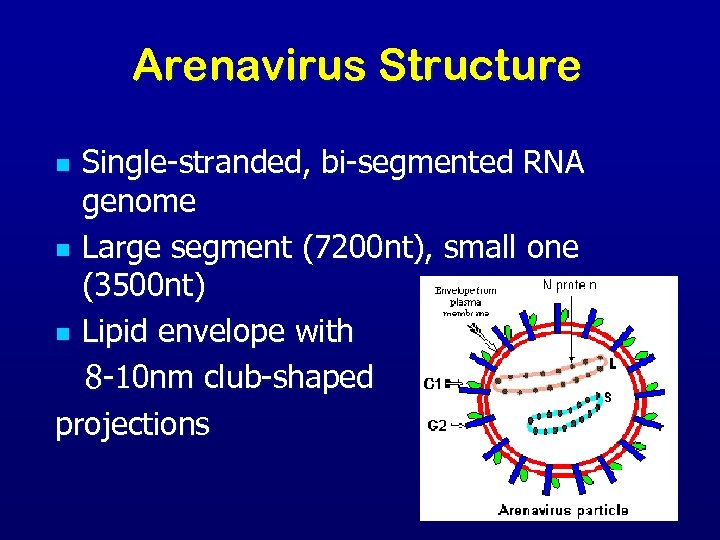 Arenavirus Structure Single-stranded, bi-segmented RNA genome n Large segment (7200 nt), small one (3500 nt) n Lipid envelope with 8 -10 nm club-shaped projections n
Arenavirus Structure Single-stranded, bi-segmented RNA genome n Large segment (7200 nt), small one (3500 nt) n Lipid envelope with 8 -10 nm club-shaped projections n
 Arenaviridae Transmission n Virus transmission and amplification occurs in rodents Shed virus through urine, feces, and other excreta Human infection n n Contact with excreta Contaminated materials Aerosol transmission Person-to-person transmission
Arenaviridae Transmission n Virus transmission and amplification occurs in rodents Shed virus through urine, feces, and other excreta Human infection n n Contact with excreta Contaminated materials Aerosol transmission Person-to-person transmission
 Arenaviridae in Humans n n n Incubation period 10– 14 days Fever and malaise 2– 4 days Hemorrhagic stage Hemorrhage, leukopenia, thrombocytopenia n Neurologic signs n
Arenaviridae in Humans n n n Incubation period 10– 14 days Fever and malaise 2– 4 days Hemorrhagic stage Hemorrhage, leukopenia, thrombocytopenia n Neurologic signs n
 Arenaviridae: Lassa Fever n n First seen in Lassa, Nigeria in 1969. Now in all countries of West Africa n n n 5 -14% of all hospitalized febrile illness Rodent-borne (Mastomys natalensis) Interpersonal transmission Direct Contact n Sex n Breast Feeding n
Arenaviridae: Lassa Fever n n First seen in Lassa, Nigeria in 1969. Now in all countries of West Africa n n n 5 -14% of all hospitalized febrile illness Rodent-borne (Mastomys natalensis) Interpersonal transmission Direct Contact n Sex n Breast Feeding n
 Lassa Fever Virus n Background n Discovered in 1969 when two missionary nurses died in Lassa, Nigeria, W. Africa n It expands to Guinea, Liberia, Sierra Leone n 100 to 300 thousand cases per year with approx. 5, 000 deaths
Lassa Fever Virus n Background n Discovered in 1969 when two missionary nurses died in Lassa, Nigeria, W. Africa n It expands to Guinea, Liberia, Sierra Leone n 100 to 300 thousand cases per year with approx. 5, 000 deaths
 Lassa Fever n n n Distinguishing Features n Gradual onset n Retro-sternal pain n Exudative pharyngitis n Hearing loss in 25% may be persistent n Spontaneous abortion Mortality 1 -3% overall (up to 50% in epidemics) Therapy: Ribavirin
Lassa Fever n n n Distinguishing Features n Gradual onset n Retro-sternal pain n Exudative pharyngitis n Hearing loss in 25% may be persistent n Spontaneous abortion Mortality 1 -3% overall (up to 50% in epidemics) Therapy: Ribavirin
 • • • Bunyaviridae Rift Valley Fever virus Crimean-Congo Hemorrhagic Fever virus Hantavirus L-segment codes for an Lprotein (the RNA dependent RNA polymerase); M segment codes for two surface glycoproteins G 1 and G 2 which form the envelope spikes; S segment codes for an Nprotein (nucleocapsid protein).
• • • Bunyaviridae Rift Valley Fever virus Crimean-Congo Hemorrhagic Fever virus Hantavirus L-segment codes for an Lprotein (the RNA dependent RNA polymerase); M segment codes for two surface glycoproteins G 1 and G 2 which form the envelope spikes; S segment codes for an Nprotein (nucleocapsid protein).
 Bunyaviridae n n n Rift Valley Fever (RVF) Crimean-Congo Hemorrhagic Fever (CCHF) Hantavirus Old World: Hemorrhagic fever with renal syndrome (HFRS) n New World: Hantavirus pulmonary syndrome (HPS) n n 5 genera with over 350 viruses
Bunyaviridae n n n Rift Valley Fever (RVF) Crimean-Congo Hemorrhagic Fever (CCHF) Hantavirus Old World: Hemorrhagic fever with renal syndrome (HFRS) n New World: Hantavirus pulmonary syndrome (HPS) n n 5 genera with over 350 viruses
 Bunyaviridae Transmission n Arthropod vector n n n Exception – Hantaviruses RVF – Aedes mosquito CCHF – Ixodid tick Hantavirus – Rodents Less common Aerosol n Exposure to infected animal tissue n
Bunyaviridae Transmission n Arthropod vector n n n Exception – Hantaviruses RVF – Aedes mosquito CCHF – Ixodid tick Hantavirus – Rodents Less common Aerosol n Exposure to infected animal tissue n
 Bunyaviridae n Transmission to humans n Arthropod vector (RVF, CCHF) n Contact with animal blood or products of infected livestock n Rodents (Hantavirus) n Laboratory aerosol n Person-to-person transmission with CCHF
Bunyaviridae n Transmission to humans n Arthropod vector (RVF, CCHF) n Contact with animal blood or products of infected livestock n Rodents (Hantavirus) n Laboratory aerosol n Person-to-person transmission with CCHF
 Rift Valley Fever n n n Predominantly a disease of sheep and cattle 1930: First identified in an infected newborn lamb in Egypt In livestock: ~100% abortion n 90% mortality in young n 5 -60% mortality in adults n
Rift Valley Fever n n n Predominantly a disease of sheep and cattle 1930: First identified in an infected newborn lamb in Egypt In livestock: ~100% abortion n 90% mortality in young n 5 -60% mortality in adults n
 Rift Valley Fever n n Asymptomatic or mild illness in humans Distinguishing Characteristics n Hemorrhagic complications rare (<5%) n Vision loss (retinal hemorrhage, vasculitis) in 1 -10% Overall mortality 1% Therapy: Ribavirin?
Rift Valley Fever n n Asymptomatic or mild illness in humans Distinguishing Characteristics n Hemorrhagic complications rare (<5%) n Vision loss (retinal hemorrhage, vasculitis) in 1 -10% Overall mortality 1% Therapy: Ribavirin?
 Crimean-Congo Hemorrhagic Fever n Distinguishing features n Abrupt onset n Most humans infected will develop hemorrhagic fever n Profuse hemorrhage Mortality 15 -40% n Therapy: Ribavirin n
Crimean-Congo Hemorrhagic Fever n Distinguishing features n Abrupt onset n Most humans infected will develop hemorrhagic fever n Profuse hemorrhage Mortality 15 -40% n Therapy: Ribavirin n
 Bunyaviridae: Crimean-Congo HF § Transmission to humans: n Ixodid, Hyalomma spp. ticks n Contact with animal blood/products n Person-to-person n Laboratory aerosols n Extensive geographical distribution
Bunyaviridae: Crimean-Congo HF § Transmission to humans: n Ixodid, Hyalomma spp. ticks n Contact with animal blood/products n Person-to-person n Laboratory aerosols n Extensive geographical distribution
 Bunyaviridae: Hantaviruses n Transmission to humans: n Exposure to rodent saliva and excreta n n Inhalation Bites Ingestion in contaminated food/water (? ) Person-to-person (Andes virus in Argentina)
Bunyaviridae: Hantaviruses n Transmission to humans: n Exposure to rodent saliva and excreta n n Inhalation Bites Ingestion in contaminated food/water (? ) Person-to-person (Andes virus in Argentina)
 Hemorrhagic Fever with Renal Syndrome (HFRS) n Distinguishing Features Insidious onset n Intense headaches, n Blurred vision n kidney failure n n n (causing severe fluid overload) Mortality: 1 -15%
Hemorrhagic Fever with Renal Syndrome (HFRS) n Distinguishing Features Insidious onset n Intense headaches, n Blurred vision n kidney failure n n n (causing severe fluid overload) Mortality: 1 -15%
 Bunyaviridae Humans n RVF n n n CCHF n n n Incubation period – 2 -5 days 0. 5% - Hemorrhagic Fever Incubation period – 3 -7 days Hemorrhagic Fever - 3– 6 days following clinical signs Hantavirus n n Incubation period – 7– 21 days HPS and HFRS
Bunyaviridae Humans n RVF n n n CCHF n n n Incubation period – 2 -5 days 0. 5% - Hemorrhagic Fever Incubation period – 3 -7 days Hemorrhagic Fever - 3– 6 days following clinical signs Hantavirus n n Incubation period – 7– 21 days HPS and HFRS
 Filoviridae Ebola n Ebola-Zaire n Ebola-Sudan n Ebola-Ivory Coast n Ebola-Bundibugyo n (Ebola-Reston) n Marburg n Ebola Marburg
Filoviridae Ebola n Ebola-Zaire n Ebola-Sudan n Ebola-Ivory Coast n Ebola-Bundibugyo n (Ebola-Reston) n Marburg n Ebola Marburg
 Filoviridae History n 1967: Marburg, Frankfurt, Belgrade n n European laboratory workers 1976: Ebola virus Ebola Zaire n Ebola Sudan n n 1989 and 1992: Ebola Reston USA and Italy n Imported macaques from Philippines n n 1994: Ebola Côte d'Ivoire
Filoviridae History n 1967: Marburg, Frankfurt, Belgrade n n European laboratory workers 1976: Ebola virus Ebola Zaire n Ebola Sudan n n 1989 and 1992: Ebola Reston USA and Italy n Imported macaques from Philippines n n 1994: Ebola Côte d'Ivoire
 Filoviridae Transmission n Reservoir is UNKNOWN n n n Bats implicated with Marburg Intimate contact Nosicomial transmission Reuse of needles and syringes n Exposure to infectious tissues, excretions, and hospital wastes n n Aerosol transmission n Primates
Filoviridae Transmission n Reservoir is UNKNOWN n n n Bats implicated with Marburg Intimate contact Nosicomial transmission Reuse of needles and syringes n Exposure to infectious tissues, excretions, and hospital wastes n n Aerosol transmission n Primates
 Filoviridae: Ebola n n Rapidly fatal febrile hemorrhagic illness Transmission: n bats implicated as reservoir n Person-to-person n Nosocomial Five subtypes n Ebola-Zaire, Ebola-Sudan, Ebola-Ivory Coast, Ebola-Bundibugyo, Ebola-Reston n Ebola-Reston imported to US, but only causes illness in non-human primates Human-infectious subtypes found only in Africa
Filoviridae: Ebola n n Rapidly fatal febrile hemorrhagic illness Transmission: n bats implicated as reservoir n Person-to-person n Nosocomial Five subtypes n Ebola-Zaire, Ebola-Sudan, Ebola-Ivory Coast, Ebola-Bundibugyo, Ebola-Reston n Ebola-Reston imported to US, but only causes illness in non-human primates Human-infectious subtypes found only in Africa
 Filoviridae: Ebola n Distinguishing features: n Acute onset n Weight loss/protration n 25 -90% case-fatality
Filoviridae: Ebola n Distinguishing features: n Acute onset n Weight loss/protration n 25 -90% case-fatality
 Filoviridae: Marburg n Transmission: n n Animal host unknown Person-to-person infected animal blood/fluid exposure Indigenous to Africa Uganda n Western Kenya n Zimbabwe n Democratic Republic of Congo n Angola n
Filoviridae: Marburg n Transmission: n n Animal host unknown Person-to-person infected animal blood/fluid exposure Indigenous to Africa Uganda n Western Kenya n Zimbabwe n Democratic Republic of Congo n Angola n
 Filoviridae: Marburg n n Distinguising features n Sudden onset n Chest pain n Maculopapular rash on trunk n Pancreatitis n Jaundice 21 -90% mortality
Filoviridae: Marburg n n Distinguising features n Sudden onset n Chest pain n Maculopapular rash on trunk n Pancreatitis n Jaundice 21 -90% mortality
 Filoviridae Humans n n n Most severe hemorrhagic fever Incubation period: 4– 10 days Abrupt onset n n Fever, chills, malaise, and myalgia Hemorrhage and DIC Death around day 7– 11 Painful recovery
Filoviridae Humans n n n Most severe hemorrhagic fever Incubation period: 4– 10 days Abrupt onset n n Fever, chills, malaise, and myalgia Hemorrhage and DIC Death around day 7– 11 Painful recovery
 Flaviviridae • • Dengue virus Yellow Fever virus Omsk Hemorrhagic Fever virus Kyassnur Forest Disease virus
Flaviviridae • • Dengue virus Yellow Fever virus Omsk Hemorrhagic Fever virus Kyassnur Forest Disease virus
 Flaviviridae History n n 1648 : Yellow Fever described 17 th– 20 th century n n n Yellow Fever and Dengue outbreaks 1927: Yellow Fever virus isolated 1943: Dengue virus isolated 1947 Omsk Hemorrhagic Fever virus isolated 1957: Kyasanur Forest virus isolated
Flaviviridae History n n 1648 : Yellow Fever described 17 th– 20 th century n n n Yellow Fever and Dengue outbreaks 1927: Yellow Fever virus isolated 1943: Dengue virus isolated 1947 Omsk Hemorrhagic Fever virus isolated 1957: Kyasanur Forest virus isolated
 Flaviviridae Transmission n n Arthropod vector Yellow Fever and Dengue viruses n Aedes aegypti Sylvatic cycle n Urban cycle n n Kasanur Forest Virus n n Ixodid tick Omsk Hemorrhagic Fever virus n Muskrat urine, feces, or blood
Flaviviridae Transmission n n Arthropod vector Yellow Fever and Dengue viruses n Aedes aegypti Sylvatic cycle n Urban cycle n n Kasanur Forest Virus n n Ixodid tick Omsk Hemorrhagic Fever virus n Muskrat urine, feces, or blood
 Flaviviridae Epidemiology n Yellow Fever Virus – Africa and Americas n n Dengue Virus – Asia, Africa, Australia, and Americas n n Case fatality rate – 1 -10% Kyasanur Forest virus – India n n Case fatality rate – varies Case fatality rate – 3– 5% Omsk Hemorrhagic Fever virus – Europe n Case fatlity rate – 0. 5– 3%
Flaviviridae Epidemiology n Yellow Fever Virus – Africa and Americas n n Dengue Virus – Asia, Africa, Australia, and Americas n n Case fatality rate – 1 -10% Kyasanur Forest virus – India n n Case fatality rate – varies Case fatality rate – 3– 5% Omsk Hemorrhagic Fever virus – Europe n Case fatlity rate – 0. 5– 3%
 Flaviviridae Humans n Yellow Fever n n Incubation period – 3– 6 days Short remission Dengue Hemorrhagic Fever Incubation period – 2– 5 days n Infection with different serotype n n n Kyasanur Forest Disease Omsk Hemorrhagic Fever Lasting sequela
Flaviviridae Humans n Yellow Fever n n Incubation period – 3– 6 days Short remission Dengue Hemorrhagic Fever Incubation period – 2– 5 days n Infection with different serotype n n n Kyasanur Forest Disease Omsk Hemorrhagic Fever Lasting sequela
 Yellow Fever n Distinguishing features n Biphasic infection n Common hepatic involvement & jaundice n Mortality: 15 -50%
Yellow Fever n Distinguishing features n Biphasic infection n Common hepatic involvement & jaundice n Mortality: 15 -50%
 Flaviviridae: Dengue n n Dengue Fever (DF) /Fatality: <1% Dengue Hemorrhagic Fever (DHF)/ Fatality: 5 -6% Dengue Shock Syndrome (DSS) /Fatality 12 -44% Four distinct serotypes n n Distinguishing Features n n n DEN-1, DEN-2, DEN-3, DEN-4 Sudden onset Eye pain Rash Complications/sequelae uncommon Illness less severe in younger children
Flaviviridae: Dengue n n Dengue Fever (DF) /Fatality: <1% Dengue Hemorrhagic Fever (DHF)/ Fatality: 5 -6% Dengue Shock Syndrome (DSS) /Fatality 12 -44% Four distinct serotypes n n Distinguishing Features n n n DEN-1, DEN-2, DEN-3, DEN-4 Sudden onset Eye pain Rash Complications/sequelae uncommon Illness less severe in younger children
 Omsk Hemorrhagic Fever n Distinguishing Features Acute Onset n Biphasic infection n Complications n Hearing loss n Hair loss n Psycho-behavioral difficulties n n Mortality: 0. 5 – 3%
Omsk Hemorrhagic Fever n Distinguishing Features Acute Onset n Biphasic infection n Complications n Hearing loss n Hair loss n Psycho-behavioral difficulties n n Mortality: 0. 5 – 3%
 Flaviviridae: Kyanasur Forest n n n Distribution: limited to Karnataka State, India Distinguishing Features n Acute onset n Biphasic Case-fatality: 3 -5% (400 -500 cases annually)
Flaviviridae: Kyanasur Forest n n n Distribution: limited to Karnataka State, India Distinguishing Features n Acute onset n Biphasic Case-fatality: 3 -5% (400 -500 cases annually)
 Symptoms/Signs vary with the type of VHF
Symptoms/Signs vary with the type of VHF
 Common Pathophysiology n n Small vessel involvement n Increased vascular permeability n Multiple cytokine activation n Cellular damage n Abnormal vascular regulation: n Early -> mild hypotension n Severe/Advanced -> Shock Viremia n Macrophage involvement n Inadequate/delayed immune response
Common Pathophysiology n n Small vessel involvement n Increased vascular permeability n Multiple cytokine activation n Cellular damage n Abnormal vascular regulation: n Early -> mild hypotension n Severe/Advanced -> Shock Viremia n Macrophage involvement n Inadequate/delayed immune response
 Common Pathophysiology n Multisystem Involvement n n n Hematopoietic Neurologic Pulmonary Hepatic (Ebola, Marburg, RVF, CCHF, Yellow Fever) Renal (Hantavirus) Hemorrhagic complications n n n Hepatic damage Consumptive coagulopathy Primary marrow injury to megakaryocytes
Common Pathophysiology n Multisystem Involvement n n n Hematopoietic Neurologic Pulmonary Hepatic (Ebola, Marburg, RVF, CCHF, Yellow Fever) Renal (Hantavirus) Hemorrhagic complications n n n Hepatic damage Consumptive coagulopathy Primary marrow injury to megakaryocytes
 Common Clinical Features: Early/Prodromal Symptoms n n n Fever Myalgia Malaise Fatigue/weakness Headache n n Dizziness Arthralgia Nausea Non-bloody diarrhea
Common Clinical Features: Early/Prodromal Symptoms n n n Fever Myalgia Malaise Fatigue/weakness Headache n n Dizziness Arthralgia Nausea Non-bloody diarrhea
 Common Clinical Features: Progressive Signs n n n Conjunctivitis Facial & thoracic flushing Pharyngitis Exanthems Periorbital edema Pulmonary edema n Hemorrhage n n Subconjunctival hemorrhage Ecchymosis Petechiae But the hemorrhage itself is rarely lifethreatening.
Common Clinical Features: Progressive Signs n n n Conjunctivitis Facial & thoracic flushing Pharyngitis Exanthems Periorbital edema Pulmonary edema n Hemorrhage n n Subconjunctival hemorrhage Ecchymosis Petechiae But the hemorrhage itself is rarely lifethreatening.
 Symptoms n n n Incubation period of 6 -21 days 80% of human infections are asyptomatic Onset is slow: fever, weakness, & malaise Few days: headache, pharyngitis, muscle pain, retrostinal & abdominal pain, nausea, vomiting, conjunctivitis, diarrhea, cough, & proteinuria Severe cases: n n n facial swelling, lung cavity fluid, hemorrhaging, hyopotension, Neurological problems: tremors, encephalitis, hair loss, gait disturbance, deafness 95% death rate among pregnant women & spontaneous abortion
Symptoms n n n Incubation period of 6 -21 days 80% of human infections are asyptomatic Onset is slow: fever, weakness, & malaise Few days: headache, pharyngitis, muscle pain, retrostinal & abdominal pain, nausea, vomiting, conjunctivitis, diarrhea, cough, & proteinuria Severe cases: n n n facial swelling, lung cavity fluid, hemorrhaging, hyopotension, Neurological problems: tremors, encephalitis, hair loss, gait disturbance, deafness 95% death rate among pregnant women & spontaneous abortion
 Common Clinical Features: Severe/End-stage n n n Multisystem compromise Profuse bleeding Consumptive coagulopathy/DIC Encephalopathy Shock Death
Common Clinical Features: Severe/End-stage n n n Multisystem compromise Profuse bleeding Consumptive coagulopathy/DIC Encephalopathy Shock Death
 Clinical Symptoms n More severe n Bleeding under skin n Petechiae, echymoses, conjunctivitis Bleeding in internal organs n Bleeding from orifices n Blood loss rarely cause of death n
Clinical Symptoms n More severe n Bleeding under skin n Petechiae, echymoses, conjunctivitis Bleeding in internal organs n Bleeding from orifices n Blood loss rarely cause of death n
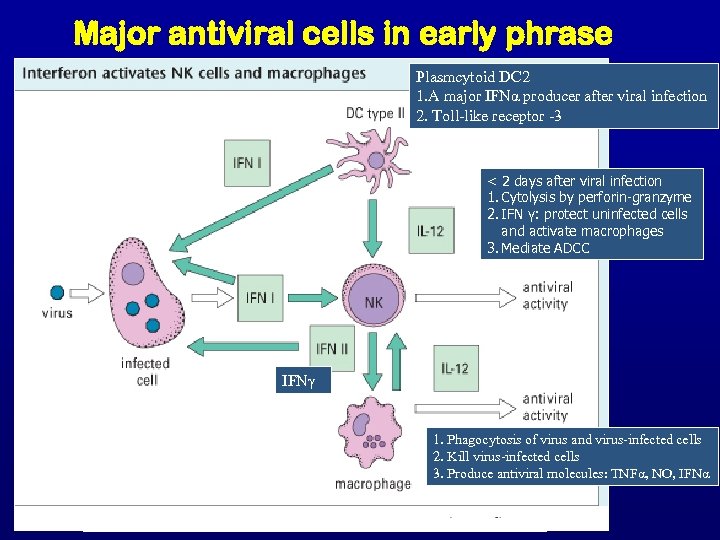 Major antiviral cells in early phrase Plasmcytoid DC 2 1. A major IFNα producer after viral infection 2. Toll-like receptor -3 < 2 days after viral infection 1. Cytolysis by perforin-granzyme 2. IFN γ: protect uninfected cells and activate macrophages 3. Mediate ADCC IFNγ 1. Phagocytosis of virus and virus-infected cells 2. Kill virus-infected cells 3. Produce antiviral molecules: TNFα, NO, IFNα
Major antiviral cells in early phrase Plasmcytoid DC 2 1. A major IFNα producer after viral infection 2. Toll-like receptor -3 < 2 days after viral infection 1. Cytolysis by perforin-granzyme 2. IFN γ: protect uninfected cells and activate macrophages 3. Mediate ADCC IFNγ 1. Phagocytosis of virus and virus-infected cells 2. Kill virus-infected cells 3. Produce antiviral molecules: TNFα, NO, IFNα
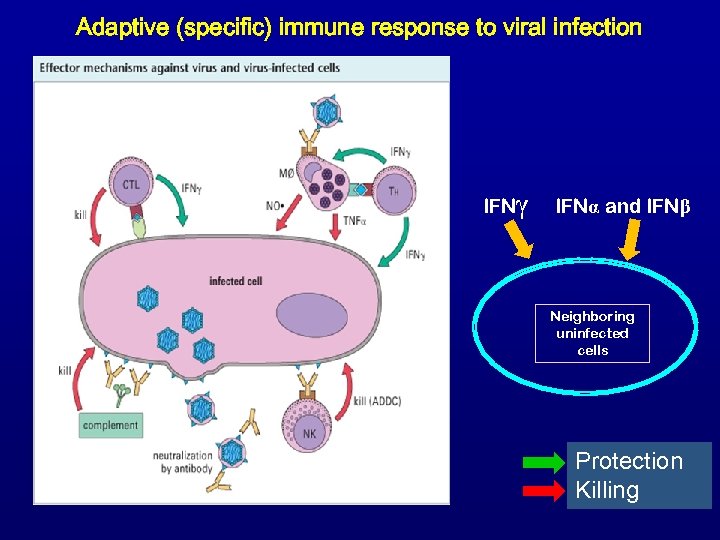 Adaptive (specific) immune response to viral infection 9 5 7 6 8 IFNγ IFNα and IFNβ Neighboring uninfected cells 1 3 4 2 Protection Killing
Adaptive (specific) immune response to viral infection 9 5 7 6 8 IFNγ IFNα and IFNβ Neighboring uninfected cells 1 3 4 2 Protection Killing
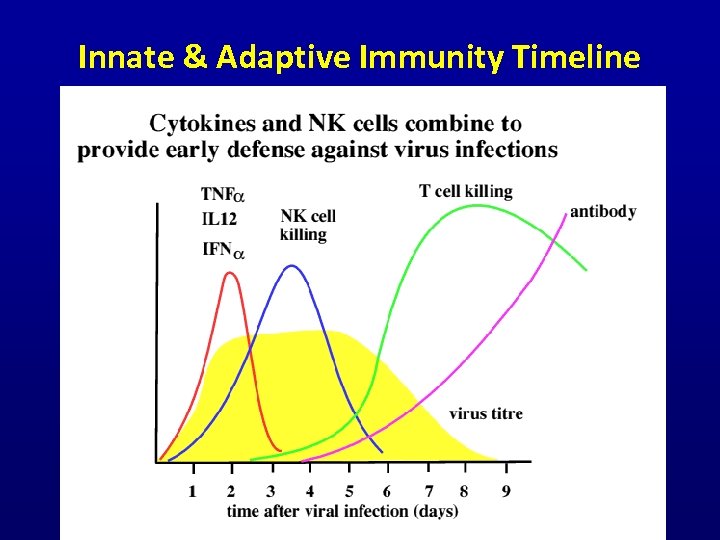 Innate & Adaptive Immunity Timeline Cambridge University Immunology Lectures (www)
Innate & Adaptive Immunity Timeline Cambridge University Immunology Lectures (www)
 Lab studies n n n n Complete Blood Count n Leucopenia, leucocytosis, thrombocytopenia, hemoconcentration, DIC Liver enzymes Alb Proteinuria universal Serological tests – Ab not detected acute phase; Direct examination blood/tissues for viral Ag enzyme immunoassay. Immunohistochemical staining liver tissue Virus isolation in cell culture RT-PCR sequencing of virus Electron microscopy specific and sensitive
Lab studies n n n n Complete Blood Count n Leucopenia, leucocytosis, thrombocytopenia, hemoconcentration, DIC Liver enzymes Alb Proteinuria universal Serological tests – Ab not detected acute phase; Direct examination blood/tissues for viral Ag enzyme immunoassay. Immunohistochemical staining liver tissue Virus isolation in cell culture RT-PCR sequencing of virus Electron microscopy specific and sensitive
 Treatment § Supportive care: • Fluid and electrolyte management • Hemodynamic monitoring • Ventilation and/or dialysis support • Steroids for adrenal crisis • Anticoagulants, IM injections, • Treat secondary bacterial infections
Treatment § Supportive care: • Fluid and electrolyte management • Hemodynamic monitoring • Ventilation and/or dialysis support • Steroids for adrenal crisis • Anticoagulants, IM injections, • Treat secondary bacterial infections
 Treatment § Manage severe bleeding complications • Cryoprecipitate (concentrated clotting factors) • Platelets • Fresh Frozen Plasma • Heparin for DIC § Ribavirin in vitro activity vs. • Lassa fever • New World Hemorrhagic fevers • Rift Valley Fever • No evidence to support use in Filovirus or Flavivirus infections
Treatment § Manage severe bleeding complications • Cryoprecipitate (concentrated clotting factors) • Platelets • Fresh Frozen Plasma • Heparin for DIC § Ribavirin in vitro activity vs. • Lassa fever • New World Hemorrhagic fevers • Rift Valley Fever • No evidence to support use in Filovirus or Flavivirus infections
 Prevention and Control
Prevention and Control
 Prevention n n Nosocomial: Complete equipment sterilization & protective clothing House to house rodent trapping Better food storage & hygiene Cautious handling of rodent if used as food source If human case occurs Decrease person-to-person transmission n Isolation of infected individuals n
Prevention n n Nosocomial: Complete equipment sterilization & protective clothing House to house rodent trapping Better food storage & hygiene Cautious handling of rodent if used as food source If human case occurs Decrease person-to-person transmission n Isolation of infected individuals n
 Prevention and Control n Avoid contact with host species n Rodents Control rodent populations n Discourage rodents from entering or living in human populations n Safe clean up of rodent nests and droppings n n Insects Use insect repellents n Proper clothing and bed nets n Window screens and other barriers to insects n
Prevention and Control n Avoid contact with host species n Rodents Control rodent populations n Discourage rodents from entering or living in human populations n Safe clean up of rodent nests and droppings n n Insects Use insect repellents n Proper clothing and bed nets n Window screens and other barriers to insects n
 Vaccination § Argentine and Bolivian HF • PASSIVE IMMUNIZATION üTreat with convalescent serum containing neutralizing antibody or immune globulin § Yellow Fever • ACTIVE IMMUNIZATION üTravelers to Africa and South America n Experimental vaccines under study n Argentine HF, Rift Valley Fever, Hantavirus and Dengue HF
Vaccination § Argentine and Bolivian HF • PASSIVE IMMUNIZATION üTreat with convalescent serum containing neutralizing antibody or immune globulin § Yellow Fever • ACTIVE IMMUNIZATION üTravelers to Africa and South America n Experimental vaccines under study n Argentine HF, Rift Valley Fever, Hantavirus and Dengue HF
 VHF Personal Protective Equipment n Airborne and Contact isolation for patients with respiratory symptoms • N-95 or PAPR mask • Negative pressure isolation • Gloves • Gown • Fitted eye protection and shoe covers if going to be exposed to splash body fluids § Droplet and Contact isolation for patients without respiratory symptoms • Surgical mask • Gloves • Gown • Fitted eye protection and shoe covers if going to be exposed to splash body fluids § Environmental surfaces • Cleaned with hospital approved disinfectant • Linen incinerated, autoclaved, double-bagged for wash
VHF Personal Protective Equipment n Airborne and Contact isolation for patients with respiratory symptoms • N-95 or PAPR mask • Negative pressure isolation • Gloves • Gown • Fitted eye protection and shoe covers if going to be exposed to splash body fluids § Droplet and Contact isolation for patients without respiratory symptoms • Surgical mask • Gloves • Gown • Fitted eye protection and shoe covers if going to be exposed to splash body fluids § Environmental surfaces • Cleaned with hospital approved disinfectant • Linen incinerated, autoclaved, double-bagged for wash
 Why do VHFs make good Bioweapons? § § § § Disseminate through aerosols Low infectious dose High morbidity and mortality Cause fear and panic in the public No effective vaccine Available and can be produced in large quantity Research on weaponization has been conducted
Why do VHFs make good Bioweapons? § § § § Disseminate through aerosols Low infectious dose High morbidity and mortality Cause fear and panic in the public No effective vaccine Available and can be produced in large quantity Research on weaponization has been conducted


