7fb2a221a8937d377061429d4c5f97ba.ppt
- Количество слайдов: 85

VENOUS THROMBOEMBOLISM PROPHYLAXIS for the Hospitalized Medical Patients Madel Sadili, MD, FCCP, FPCCP

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

Venous Thromboembolism (VTE) Ø DVT : Deep-vein thrombosis Ø PE : Pulmonary Embolism

Arterial Thrombosis Ø Most common cause of MI, stroke, & limb gangrene Ø Usually is initiated by the spontaneous or mechanical rupture of atherosclerotic plaque Ø Consists of platelet aggregates held together by small amounts of fibrin Ø Strategies to inhibit arterial thrombogenesis focus mainly on drugs that block platelet function but often include anticoagulant agents to prevent fibrin deposition

Venous thrombosis Ø Leads to PE (can be fatal) & to postphlebitic syndrome Ø Occurs when procoagulant stimuli overwhelm natural protective mechanisms, ie, excessive activation of coagulation with thrombophilic abnormalities, vessel wall damage or stasis; inflammatory cytokines generated after trauma, surgery, or medical illness activate endothelial cells that express adhesion molecules that attract leukocytes which elaborate tissue factor & express receptors for factor X & fibrinogen, that promotes coagulation on their surfaces; neutrophils generate O 2 free radicals & release hydrolytic enzymes, enhancing local clot formation Ø Venous thrombus is composed mainly of fibrin & RBCs Ø Anticoagulants are the drugs of choice for their prevention & treatment

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

IMPACT OF VENOUS THROMBOEMBOLISM

Hospitalization for an acute medical illness Ø Independently associated with ~8 fold increase in relative risk for VTE (Heit, et al. Arch Int Med 2000; 160) Ø 10 -30% of general medical patients may develop VTE (Cohen, et al. Thromb Haemost 2005; 94) Ø 50 -70% of symptomatic thromboembolic events, and 70 -80% of fatal PEs occur in non-surgical patients (Goldhaber, et al. Chest 2000; 118) Ø ¾ of VTE in hospitalized patients occur in acutely ill nonsurgical patients (Leizorovicz, et al. J Thrombosis & Hemostasis 2003)

Ø PE (postmortem studies) is associated with up to 10% of deaths in hospitalized patients, and only ¼ of these occur following surgery thus… ¾ of hospitalized patients who suffer a fatal PE are in fact medical patients Cohen et al. Thromb Haemost 2005; 94
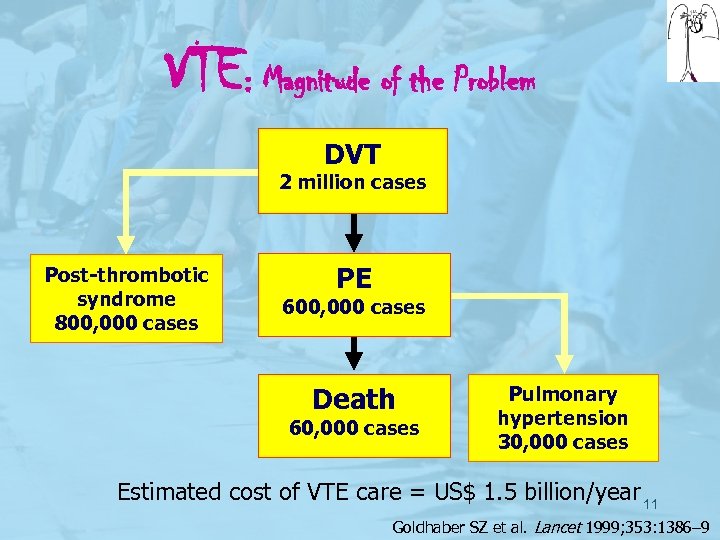
VTE: Magnitude of the Problem DVT 2 million cases Post-thrombotic syndrome 800, 000 cases PE 600, 000 cases Death 60, 000 cases Pulmonary hypertension 30, 000 cases Estimated cost of VTE care = US$ 1. 5 billion/year 11 Goldhaber SZ et al. Lancet 1999; 353: 1386– 9

Ø Therefore. . . the appropriate prophylaxis of medical inpatients offers an important opportunity to significantly reduce the burden of disease due to VTE

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

Rationale for Thromboprophylaxis in Hospitalized Patients

Rationale High Prevalence of VTE Adverse Consequences of unprevented VTE Efficacy & Effectiveness of thromboprophylaxis Description Most hospitalized px have risk factors for VTE DVT is common in many hospitalized px Hosp-acquired DVT & PE are usually clinically silent Difficult to predict which at-risk patients will develop symptomatic thromboembolic complications Screening at-risk px using PE or noninvasive testing is neither effective nor cost-effective Symptomatic DVT & PE Fatal PE Costs of investigating symptomatic patients Risks & costs of treating unprevented VTE, esp bleeding Increased future risk of recurrent VTE Chronic post-thrombotic syndrome Thromboprophylaxis is highly efficacious at preventing DVT, proximal DVT, symptomatic VTE, & fatal PE Prevention of DVT also prevents PE Cost-effectiveness of prophylaxis has repeatedly been demonstrated Geerts, et al. Chest 2001; 119: 132 S-175 S

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

Who are at risk for VTE?

Ø Ø Ø Ø Ø Ø Surgery Trauma (major or lower extremity) Immobility, paresis Malignancy Cancer therapy (hormonal, chemotx, radiotx) Previous VTE Increasing age Pregnancy & the postpartum period Estrogen-containing oral contraception or HRT Selective estrogen receptor modulators Acute medical illness Heart or respiratory failure Inflammatory bowel disease Nephrotic syndrome Myeloproliferative disease Paroxysmal nocturnal hemoglobinuria Obesity Smoking Varicose veins Central venous catheterization Inherited or acquired thrombophilia Heit, et al. Arch Int Med 2002; 162: 1245 -1248

Absolute risk of DVT in Hospitalized Patients Ø Medical px Ø Ø Ø Ø Ø Geerts, et al. Chest 2001; 119: 132 S-175 S 10 -20 % General surgery 15 -40 Major gyne surgery 15 -40 Major urologic surgery 15 -40 Neurosurgery 15 -40 Stroke 20 -50 Hip or knee arthroplasty; hip fracture surgery 40 -60 Major trauma 40 -80 Spinal cord injury 60 -80 Critical care patients 10 -80

Ø Despite consensus-group recommendations that at-risk medical patients should receive thromboprophylaxis, there is NO CONSENSUS as to which patients are at risk, thus, many patients may not receive appropriate thromboprophylaxis Cohen, et al. Thromb Haemost 2005: 94

Cohen, p 9. Fig. 2

Thromboprophylaxis in Acutely Ill Patients MEDENOX (1999) Prophylaxis of VTE in MEDical Patients with ENOXaparin Ø 40 mg, 20 mg enoxaparin vs placebo OD x 6 -14 days Ø 866 patients with heart failure, respiratory, & infectious disease Ø Primary outcome – VTE between days 1 -14 – DVT detected by bilateral venography (or duplex utz) between days 6 -14 (or earlier if clinically indicated) or documented PE Ø Duration of ff-up – 3 months Samama MM et al. N Engl J Med 1999; 341: 793– 800
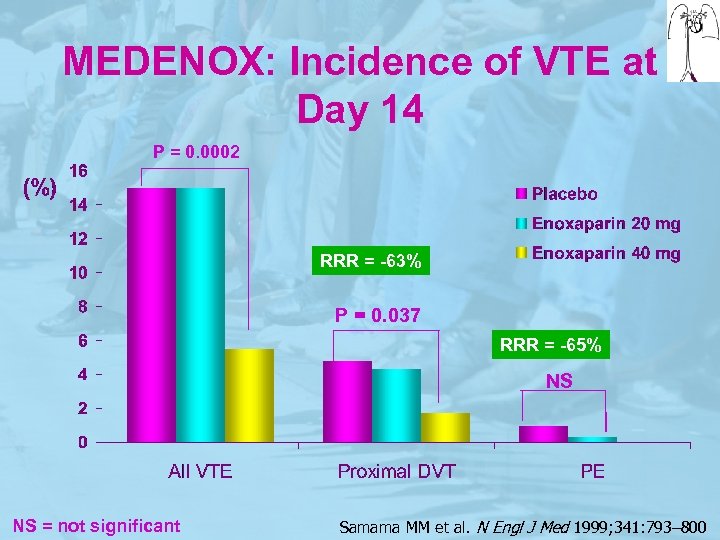
MEDENOX: Incidence of VTE at Day 14 P = 0. 0002 RRR = -63% P = 0. 037 RRR = -65% NS NS = not significant Samama MM et al. N Engl J Med 1999; 341: 793– 800

MEDENOX Ø The incidence of VTE was significantly lower in the 40 mg enoxaparin group (5. 5%) than in the placebo (14. 9%) Ø The benefit was maintained at 3 months

PREVENT (2003) Ø Prospective evaluation of Dalteparin efficacy for the prevention of VTE in immobilized patients Ø Largest trial (radomized, double-blind, palacebo-controlled) comparing a LMWH with placebo – Dalteparin 5000 IU OD x 14 days Ø 3706 acutely ill medical patients – CHF, acute respiratory failure, or infectious disease Ø Primary endpoint – clinically important VTE defined as objectively verified symptomatic DVT, PE, sudden death, & objectively verified asymptomatic proximal DVT. Compression UTZ done in all patients who had not reached an endpoint by day 21 Leizorovicz, et al. J Thrombosis & Haemostasis. July 2003
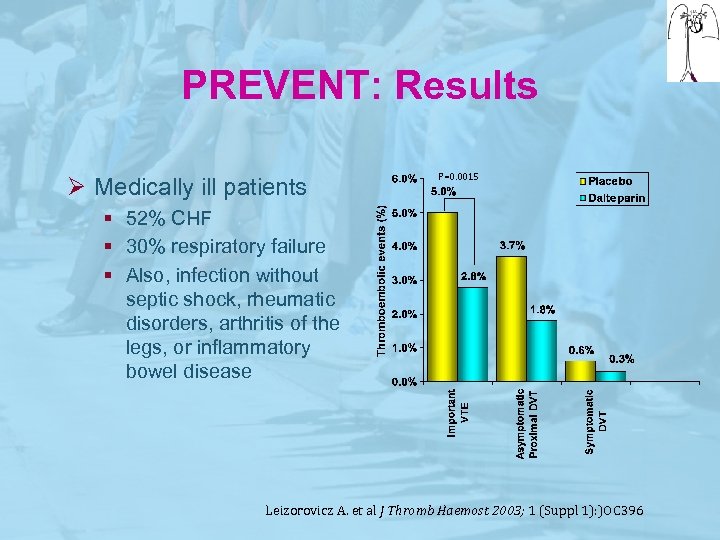
PREVENT: Results Ø Medically ill patients P=0. 0015 § 52% CHF § 30% respiratory failure § Also, infection without septic shock, rheumatic disorders, arthritis of the legs, or inflammatory bowel disease Leizorovicz A. et al J Thromb Haemost 2003; 1 (Suppl 1): )OC 396

PREVENT Ø The incidence of the composite primary outcome was 2. 77% in the dalteparin group and 4. 96% in the placebo group, a risk reduction of 45%

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

Grading of Recommendations
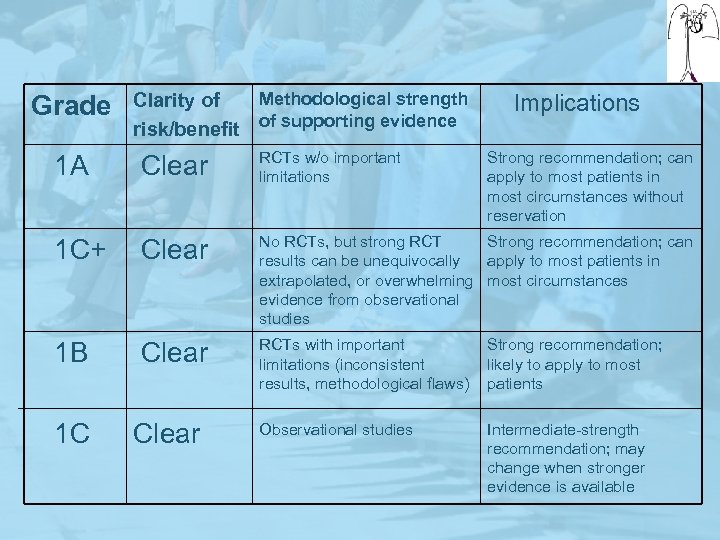
Grade Methodological strength Clarity of risk/benefit of supporting evidence Implications 1 A Clear RCTs w/o important limitations Strong recommendation; can apply to most patients in most circumstances without reservation 1 C+ Clear No RCTs, but strong RCT Strong recommendation; can results can be unequivocally apply to most patients in extrapolated, or overwhelming most circumstances evidence from observational studies 1 B Clear RCTs with important limitations (inconsistent results, methodological flaws) Strong recommendation; likely to apply to most patients 1 C Clear Observational studies Intermediate-strength recommendation; may change when stronger evidence is available
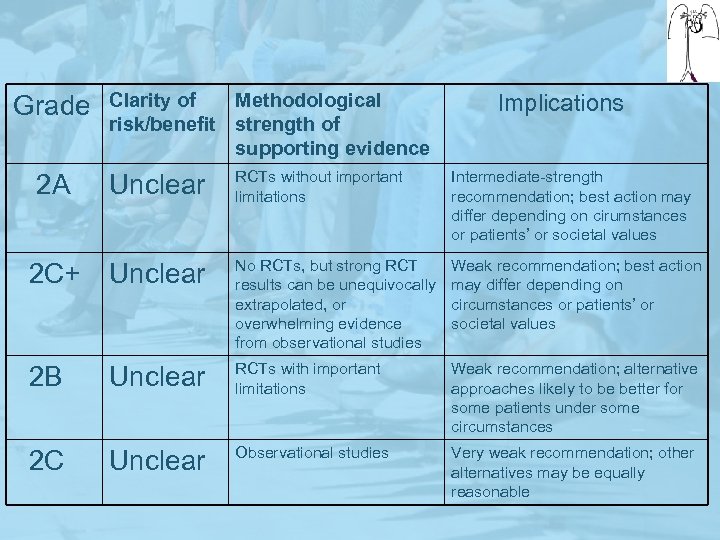
Grade Clarity of Methodological risk/benefit strength of supporting evidence Implications 2 A Unclear RCTs without important limitations Intermediate-strength recommendation; best action may differ depending on cirumstances or patients’ or societal values 2 C+ Unclear No RCTs, but strong RCT results can be unequivocally extrapolated, or overwhelming evidence from observational studies Weak recommendation; best action may differ depending on circumstances or patients’ or societal values 2 B Unclear RCTs with important limitations Weak recommendation; alternative approaches likely to be better for some patients under some circumstances 2 C Unclear Observational studies Very weak recommendation; other alternatives may be equally reasonable

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

Recommendations: Thromboprophylaxis in the Medically Ill Geerts, et al. Chest Supplement. Sept 2004; 126/3

General Recommendations Ø It is recommended that mechanical methods of prophylaxis be used primarily in patients who are at high risk of bleeding (Grade 1 C+) or as an adjunct to anticoagulant-based prophylaxis (Grade 2 A). Careful attention should be directed toward ensuring the proper use of, and optimal compliance with, the mechanical device (Grade 1 C+) Ø We recommend against the use of aspirin alone as prophylaxis against VTE for any patient group (Grade 1 A) Ø For each of the antithrombotic agents, it is recommended that clinicians consider the manufacturer’s suggested dosing guidelines (Grade 1 C)

General Recommendations Ø We recommend consideration of renal impairment when deciding on doses of LMWH, fondaparinux, the direct thrombin inhibitors, & other antithrombotic drugs that are cleared by the kidneys, particularly in elderly patients and those who are at high risk for bleeding (Grade 1 C+) Ø In all patients undergoing neuraxial anesthesia or analgesia, special caution when using anticoagulant prophylaxis is recommended (Grade 1 C+)

Medical Conditions Ø In acutely ill medical patients who have been admitted to the hospital with CHF or severe respiratory disease, or who are confined to bed & have 1 or more additional risk factors, including active cancer , previous VTE, sepsis, acute neurologic disease, or inflammatory bowel disease, prophylaxis with LDUH or LMWH is recommended (Grade IA) Ø In medical patients with risk factors for VTE, & in whom there is a contraindication to anticoagulant prophylaxis, the use of mechanical prophylaxis with GCS or IPC is recommended (Grade 1 C+)

Medical Condition: Acute MI Ø For all patients at high risk of systemic or venous thromboembolism (anterior MI, pump failure, previous embolus, atrial fibrillation, or LV thrombus), the administration of IV UFH while receiving streptokinase, is recommended (Grade 1 C+)

Medical Condition: Acute Ischemic Stroke Ø For acute stroke patients with restricted mobility, prophylactic low-dose subcutaneous heparin or LMWH or heparinoids is recommended (Grade 1 A). *Low-dose heparin should be restricted for 24 h after administration of thrombolytic therapy; it may be used safely in combination with aspirin. Ø For patients who have contraindications to anticoagulants, it is recommended that clinicians use intermittent pneumatic compression devices or elastic stockings (Grade 1 C)

Medical Conditions: Intracerebral Hemorrhage Ø We recommend the initial use of intermittent pnuematic compression (Grade 1 C+). Ø In stable patients, low-dose SQ heparin may be initiated as soon as the 2 nd day after the onset of the hemorrhage (Grade 2 C). Underlying values and preferences: the recommendation for SQ heparin assumes a relatively low degree of risk aversion.

Cancer Patients Ø 6 -fold increased risk of VTE compared to those without cancer Ø More specific risk estimates of VTE by cancer type, stage, and treatment approaches are still largely unknown Ø High among those with malignant brain tumors and adenocarcinoma of the ovary, pancreas, colon, stomach, lung, prostate, and kidney Ø Cancer patients undergoing surgery have at least 2 x the risk of postoperative DVT and more than 3 x the risk of fatal PE

Cancer Patients Ø Cancer patients undergoing surgical procedures receive prophylaxis that is appropriate for their current risk state (Grade 1 A). Refer to the surgical subsections. Ø Hospitalized cancer patients who are bedridden with an acute medical illness should receive prophylaxis that is appropriate for their current risk state (Grade 1 A). Refer to the medical subsection. Ø It is suggested that clinicians not routinely use prophlaxis to try to prevent thrombosis related to longterm indwelling CVCs in cancer patients (Grade 2 B).

Critical Care Ø On admission to a critical care unit, all patients should be assessed for their risk of VTE. Accordingly, most patients should receive thromboprophylaxis (Grade 1 A) Ø For patients who are at high risk for bleeding, mechanical prophylaxis with GCS &/or IPC is recommended, until the bleeding risk decreases (Grade 1 C+) Ø For ICU patients who are at moderate risk for VTE (eg, medically ill or postoperative px), LDUH or LMWH prophylaxis is recommended (Grade 1 A) Ø For patients who are at higher risk, such as that following major trauma or orthopedic surgery, LMWH prophylaxis is recommended (Grade 1 A)

BURNS Ø Burn patients with additional risk factors for VTE, including one or more of the ff: advanced age, morbid obesity, extensive or lower extremity burns, concomitant lower extremity trauma, use of a femoral venous catheter, &/or prolonged immobility (Grade 1 C+) Ø If there are no contraindications, the use of either LDUH or LMWH is recommended, starting as soon as it is considered safe to do so (Grade 1 C+)

Increased Risk of Bleeding Ø Recent surgery Ø Known bleeding disorder Ø Impaired renal function Ø Uncontrolled hypertension Ø Large ischaemic cerebral infarction Ø Active GI bleeding (peptic/bowel) Ø Use of antiplatelet drugs or NSAIDs

Lecture Outline Ø Arterial & Venous Thrombosis Ø Burden Of Disease (VTE) § Incidence Ø Rationale for Thromboprophylaxis Ø Risk Factors Ø Grading of Recommendations Ø Drugs Ø Summary

Methods of DVT Prophylaxis Ø Unfractionated heparin (UFH) Ø Low-molecular-weight heparins (LMWHs) Ø Oral anticoagulants (warfarin) Ø Pentasaccharides (fondaparinux) Ø Antiplatelet therapy Ø Mechanical compression and early ambulation

Unfractionated Heparin Ø Main anticoagulant action is mediated by the heparin/AT interaction, which inactivates thrombin factor IIa & factors Xa, IXa, & XIIa Ø Increases vessel wall permeability, suppresses proliferation of vascular smooth muscle cells, suppresses osteoblast formation, & activates osteoclasts, promoting bone loss, & HIT Ø IV infusion or SC injection (reduced bioavailabiltiy, thus, 10% higher initial dose) Ø Dose adjustment by monitoring a. PTT, or, when very high doses are given, by ACT (activated clotting time)

LMWH Ø Polysulfated glycosaminoglycans about 1/3 the molecular weight of UFH Ø Like heparin, major anticoagulant effect by activating AT Ø Administered in fixed doses, for thromboprophylaxis, or in total body weight (TBW)-adjusted doses, for therapeutic effect

LMWH Ø Reduced binding properties to proteins & cells, explaining all of the anticoagulant, phramacokinetic, & other biological differences between heparin & LMWH: -reduced ability to inactivate thrombin bec the smaller fragments cannot bind simultaneously to AT & thrombin, but, since bridging bet AT & factor Xa is less critical for factor Xa activity, the smaller fragments inactivate factor Xa almost as well as larger molecules -reduced binding to plasma proteins is responsible for the more predictable dose-response relationship of LMWHs -lower binding to macrophages & endothelial cells increases the plasma half-life of LMWHs -reduced binding to platelets & PF 4 explains lower incidence of HIT -reduced binding to osteoblasts results in lower incidence of bone loss

Fondaparinux Ø New parenteral indirect factor Xa inhibitor, with no activity against thrombin Ø Excellent bioavailability after SQ injection with a plasma half-life of 17 h, thus given OD Ø Does not bind to platelets or PF 4 (no heparin/PF 4 complex), thus, no HIT Ø Phase III trial for thromboprophylaxis at 2. 5 mg OD
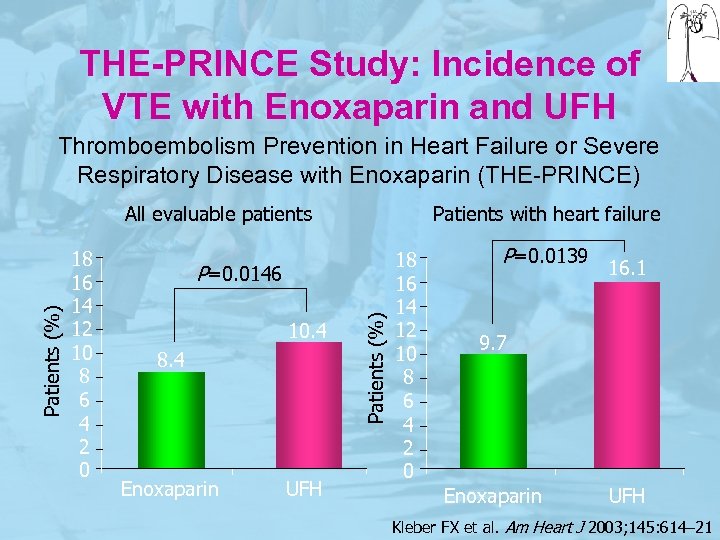
THE-PRINCE Study: Incidence of VTE with Enoxaparin and UFH Thromboembolism Prevention in Heart Failure or Severe Respiratory Disease with Enoxaparin (THE-PRINCE) 18 16 14 12 10 8 6 4 2 0 Patients with heart failure P=0. 0146 10. 4 8. 4 Enoxaparin UFH Patients (%) All evaluable patients 18 16 14 12 10 8 6 4 2 0 P=0. 0139 16. 1 9. 7 Enoxaparin UFH Kleber FX et al. Am Heart J 2003; 145: 614– 21

VTE Prevention Non-Pharmacologic Methods Ø Ambulation Ø Elastic or Graduated compression stockings (GCS) Ø Intermittent pneumatic compression (IPC) devices Ø Arteriovenous foot pumps (VFP)

Mechanical Methods of Prophylaxis Ø Increase venous outflow and/or reduce stasis within the leg veins Ø Primary attraction is the lack of bleeding potential, therefore, are considered for patients with high bleeding risks Ø Must select the correct size of the device, must properly apply them, and must ensure that they are removed for only a short time each day, and that they do not impede ambulation

Recommendation: Mechanical Methods of Prophylaxis Ø Should be used primarily in patients who are at high risk of bleeding (Grade 1 C+), or an an adjunct to anticoagulant-based prophylaxis (Grade 2 A) Ø Careful attention must be directed toward ensuring the proper use of, and optimal compliance with, the mechanical device (Grace 1 C+)

Summary Ø VTE is an important clinical problem worldwide Ø Thromboprophylaxis for the medically-ill patients who are at risk for VTE is effective & safe Ø The concensus recommends against the use of aspirin alone for thromoprophylaxis Ø LDUH, LMWH, VKA, Fondaparinux, and mechanical devices are recommended for thromboprophylaxis.
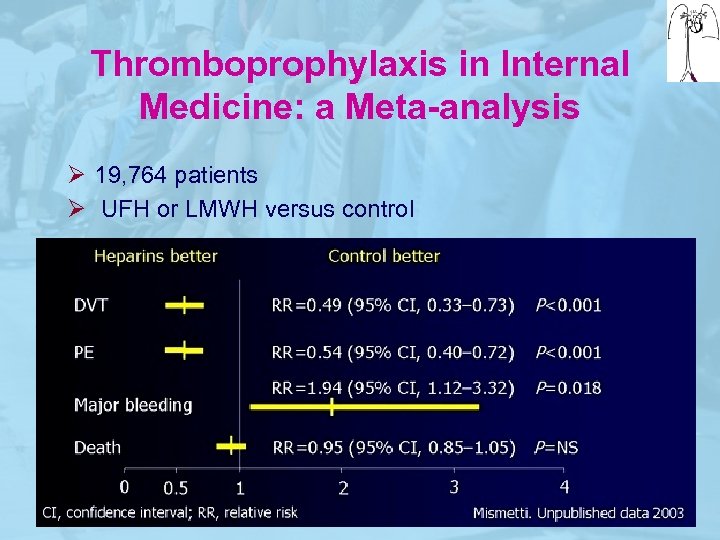
Thromboprophylaxis in Internal Medicine: a Meta-analysis Ø 19, 764 patients Ø UFH or LMWH versus control
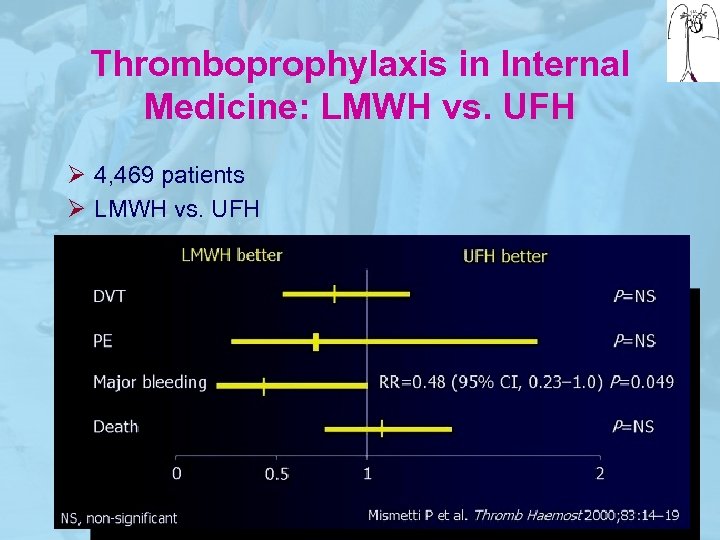
Thromboprophylaxis in Internal Medicine: LMWH vs. UFH Ø 4, 469 patients Ø LMWH vs. UFH
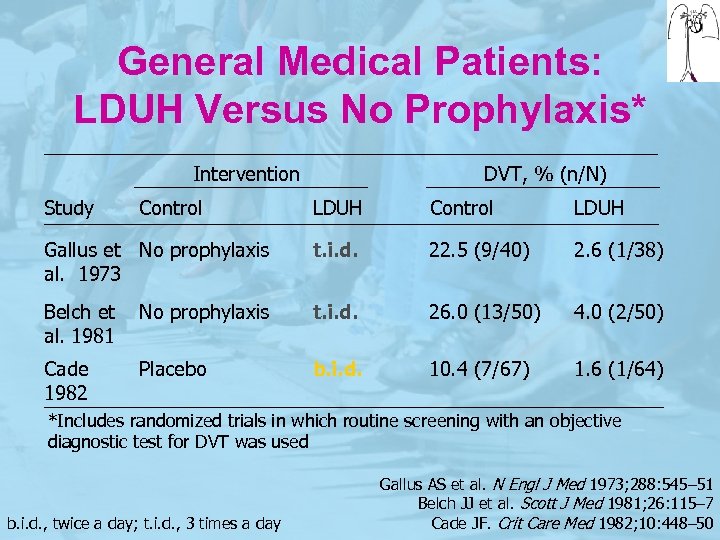
General Medical Patients: LDUH Versus No Prophylaxis* Intervention Study Control DVT, % (n/N) LDUH Control LDUH Gallus et No prophylaxis al. 1973 t. i. d. 22. 5 (9/40) 2. 6 (1/38) Belch et al. 1981 No prophylaxis t. i. d. 26. 0 (13/50) 4. 0 (2/50) Cade 1982 Placebo b. i. d. 10. 4 (7/67) 1. 6 (1/64) *Includes randomized trials in which routine screening with an objective diagnostic test for DVT was used b. i. d. , twice a day; t. i. d. , 3 times a day Gallus AS et al. N Engl J Med 1973; 288: 545– 51 Belch JJ et al. Scott J Med 1981; 26: 115– 7 Cade JF. Crit Care Med 1982; 10: 448– 50
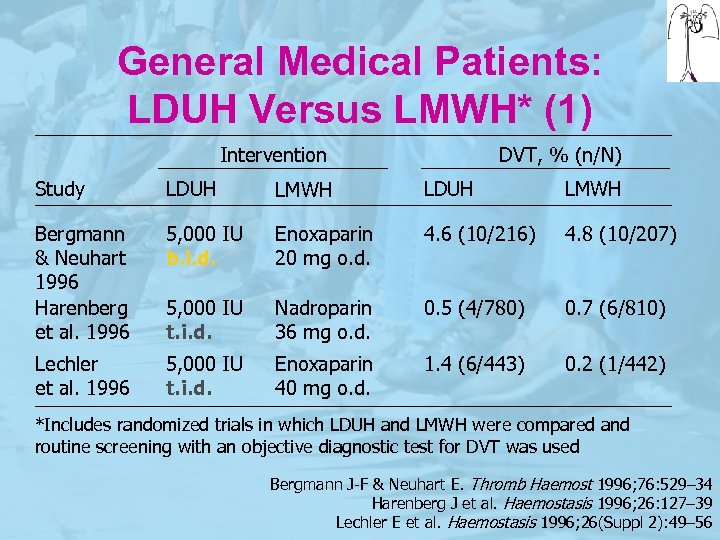
General Medical Patients: LDUH Versus LMWH* (1) Intervention DVT, % (n/N) Study LDUH LMWH Bergmann & Neuhart 1996 Harenberg et al. 1996 5, 000 IU b. i. d. Enoxaparin 20 mg o. d. 4. 6 (10/216) 4. 8 (10/207) 5, 000 IU t. i. d. Nadroparin 36 mg o. d. 0. 5 (4/780) 0. 7 (6/810) Lechler et al. 1996 5, 000 IU t. i. d. Enoxaparin 40 mg o. d. 1. 4 (6/443) 0. 2 (1/442) *Includes randomized trials in which LDUH and LMWH were compared and routine screening with an objective diagnostic test for DVT was used Bergmann J-F & Neuhart E. Thromb Haemost 1996; 76: 529– 34 Harenberg J et al. Haemostasis 1996; 26: 127– 39 Lechler E et al. Haemostasis 1996; 26(Suppl 2): 49– 56
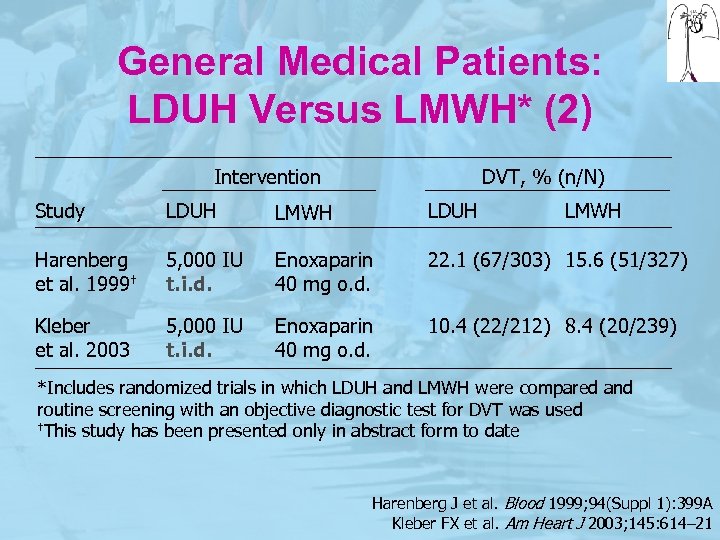
General Medical Patients: LDUH Versus LMWH* (2) Intervention DVT, % (n/N) Study LDUH LMWH Harenberg et al. 1999† 5, 000 IU t. i. d. Enoxaparin 40 mg o. d. 22. 1 (67/303) 15. 6 (51/327) Kleber et al. 2003 5, 000 IU t. i. d. Enoxaparin 40 mg o. d. 10. 4 (22/212) 8. 4 (20/239) *Includes randomized trials in which LDUH and LMWH were compared and routine screening with an objective diagnostic test for DVT was used †This study has been presented only in abstract form to date Harenberg J et al. Blood 1999; 94(Suppl 1): 399 A Kleber FX et al. Am Heart J 2003; 145: 614– 21
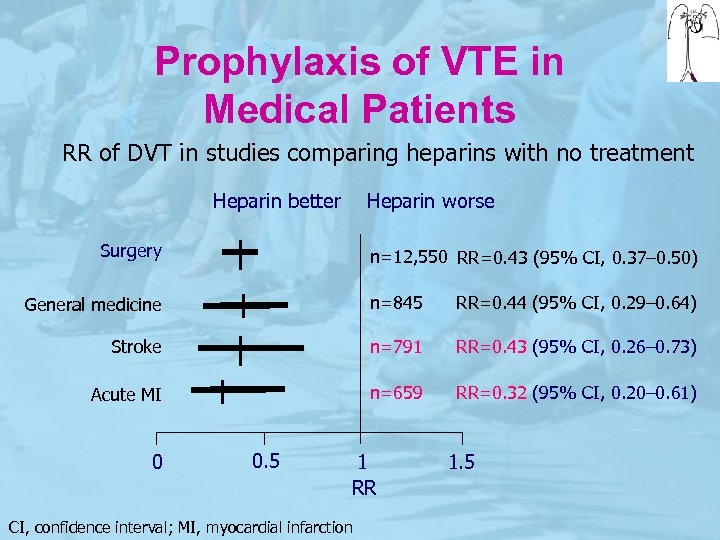
Prophylaxis of VTE in Medical Patients RR of DVT in studies comparing heparins with no treatment Heparin better Heparin worse Surgery n=12, 550 RR=0. 43 (95% CI, 0. 37– 0. 50) General medicine n=845 RR=0. 44 (95% CI, 0. 29– 0. 64) Stroke n=791 RR=0. 43 (95% CI, 0. 26– 0. 73) Acute MI n=659 RR=0. 32 (95% CI, 0. 20– 0. 61) 0 0. 5 1 RR CI, confidence interval; MI, myocardial infarction 1. 5

Thromboprophylaxis in Internal Medicine: Risk: Benefit Ratio Ø Risk: benefit ratio of heparins vs. controls § 50% reduction in risk of symptomatic PE § 2 -fold increase in major bleeding Ø Risk: benefit ratio of LMWH versus UFH § Similar effect on symptomatic PE § 50% reduction in risk of major bleeding Ø LMWHs are effective and safe
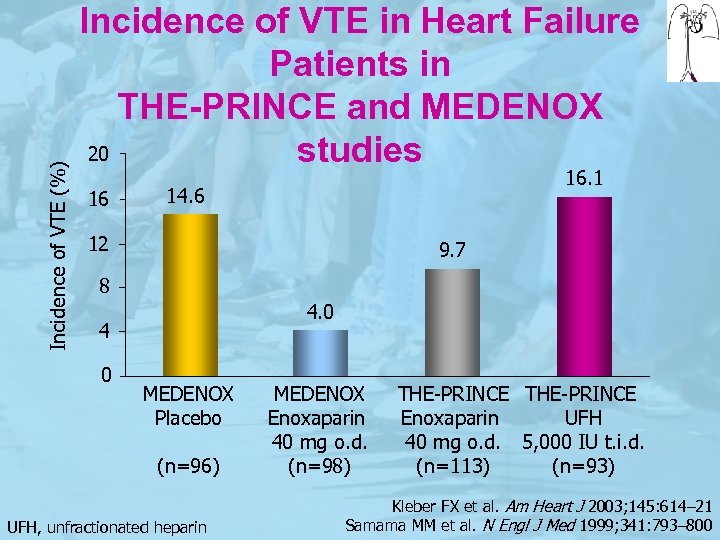
Incidence of VTE (%) Incidence of VTE in Heart Failure Patients in THE-PRINCE and MEDENOX 20 studies 16 16. 1 14. 6 12 9. 7 8 4. 0 4 0 MEDENOX Placebo (n=96) UFH, unfractionated heparin MEDENOX Enoxaparin 40 mg o. d. (n=98) THE-PRINCE Enoxaparin UFH 40 mg o. d. 5, 000 IU t. i. d. (n=113) (n=93) Kleber FX et al. Am Heart J 2003; 145: 614– 21 Samama MM et al. N Engl J Med 1999; 341: 793– 800

PREVENT: Study Design Ø Dalteparin vs. Placebo in medically ill patients (n=3, 706) Ø Primary endpoint: reduction in clinically important VTE § § Objectively verified symptomatic DVT Objectively verified asymptomatic proximal DVT Fatal and non-fatal PE Sudden death Ø Selection of Patients: Day 3 Ø Randomizations: Day 1 Ø Bilateral ultrasonography Treatment Period Ø Follow-up period PREVENT, Prospective Evaluation of Dalteparin Efficacy In Immobilized Patients Trial Vaitkus PT et al Vasc Med 2002; 7: 269 -73

PREVENT: Incidence of VTE on Day 21 Incidence of VTE (%) Dalteparin Placebo (n=1, 518) (n=1, 473) 2. 77 4. 96 Difference in incidence 2. 19 (– 3. 57 to – 0. 81) Risk ratio 0. 55 (0. 38– 0. 80) P=0. 0015 (Cochran-Mantel-Haenszel test) Leizorovicz A et al. J Thromb Haemost 2003; 1(Suppl 1): OC 396

EXCLAIM: Extended Clinical Prophylaxis in Acutely ill Medical Patients

Thromboprophylaxis in Medical Patients: Real World Data Ø DVT Free § Routine preventive efforts among inpatients are not widely practiced, especially among acutely ill medical patients Ø IMPROVE 2 § International Medical Prevention Registry on Venous Thromboembolism § Most acutely ill medical patients do not receive thromboprophylaxis during hospitalization Goldhaber SZ & Tapson VF. J Thromb Haemost 2003; 1 (Suppl 1): P 1470 Anderson FA et al. J Thromb Haemost 2003; 1 (Suppl 1): P 1438

DVT FREE Registry (1) Ø Of the 5, 451 patients, 50% (n=2, 725) were diagnosed with DVT as outpatients or in the emergency department and 50% (n=2, 726) were diagnosed as inpatients Ø Overall, 71% (n=3, 894) of patients received no prophylaxis for DVT within 30 days prior to diagnosis Ø Of the 29% (n=1, 557) of patients who did receive prophylaxis, 30% (n=410) were diagnosed with DVT as outpatients and 70% (n=1, 147) as inpatients Goldhaber SZ & Tapson VF. J Thromb Haemost 2003; 1(Suppl 1): P 1470

DVT FREE Registry (2) Ø Surgical patients (those with a history of surgery within 3 months prior to diagnosis) were far more likely to receive DVT prophylaxis than non-surgical patients Ø The vast majority of non-surgical patients (80%; n=2, 295) received no prophylaxis within 30 days before diagnosis Goldhaber SZ & Tapson VF. J Thromb Haemost 2003; 1(Suppl 1): P 1470
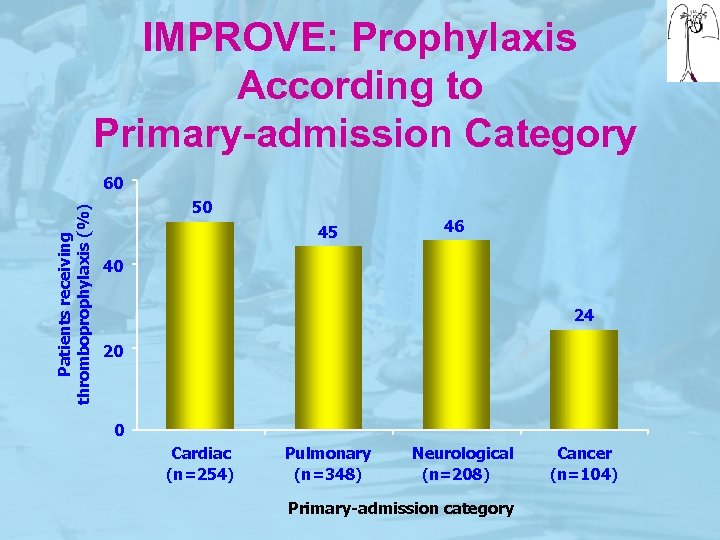
IMPROVE: Prophylaxis According to Primary-admission Category Patients receiving thromboprophylaxis (%) 60 50 45 46 40 24 20 0 Cardiac (n=254) Pulmonary (n=348) Neurological (n=208) Primary-admission category Cancer (n=104)
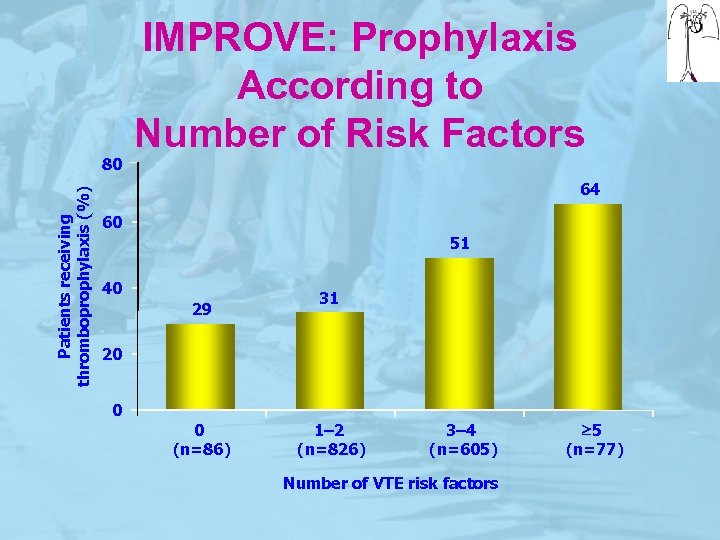
Patients receiving thromboprophylaxis (%) 80 IMPROVE: Prophylaxis According to Number of Risk Factors 64 60 51 40 29 31 20 0 0 (n=86) 1– 2 (n=826) 3– 4 (n=605) Number of VTE risk factors 5 (n=77)
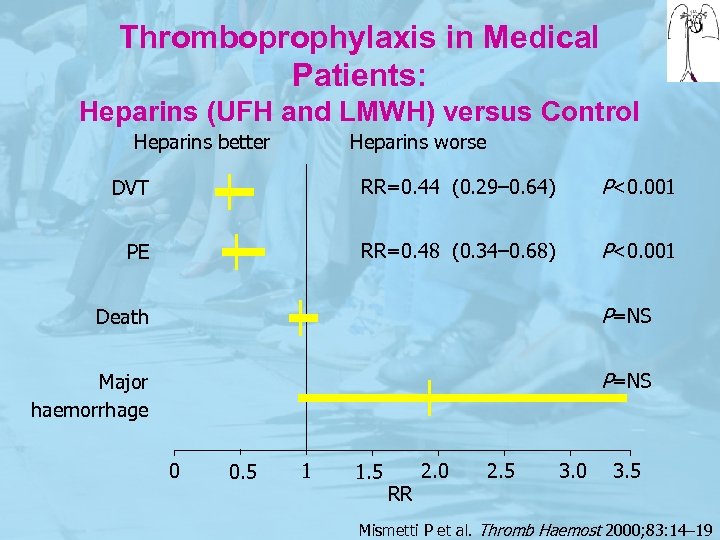
Thromboprophylaxis in Medical Patients: Heparins (UFH and LMWH) versus Control Heparins better Heparins worse DVT RR=0. 44 (0. 29– 0. 64) P<0. 001 PE RR=0. 48 (0. 34– 0. 68) P<0. 001 Death P=NS Major haemorrhage P=NS 0 0. 5 1 1. 5 RR 2. 0 2. 5 3. 0 3. 5 Mismetti P et al. Thromb Haemost 2000; 83: 14– 19
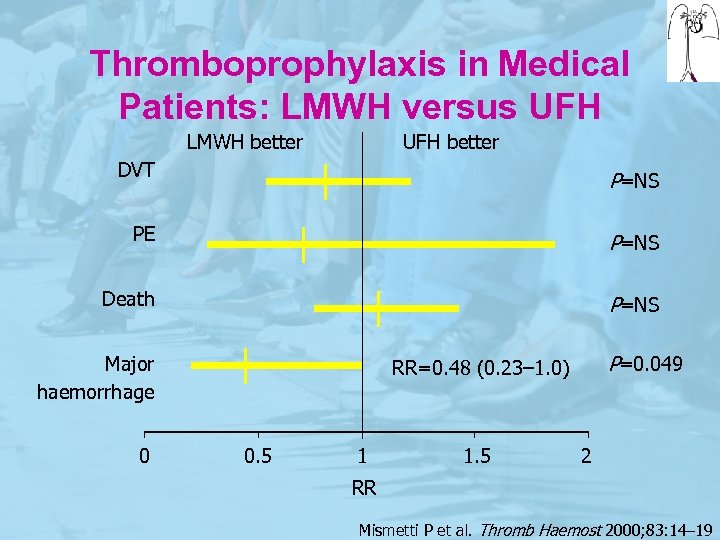
Thromboprophylaxis in Medical Patients: LMWH versus UFH LMWH better UFH better DVT P=NS PE P=NS Death P=NS Major haemorrhage 0 P=0. 049 RR=0. 48 (0. 23– 1. 0) 0. 5 1 1. 5 2 RR Mismetti P et al. Thromb Haemost 2000; 83: 14– 19

SAFETY of THROMBOPROPHYLAXIS
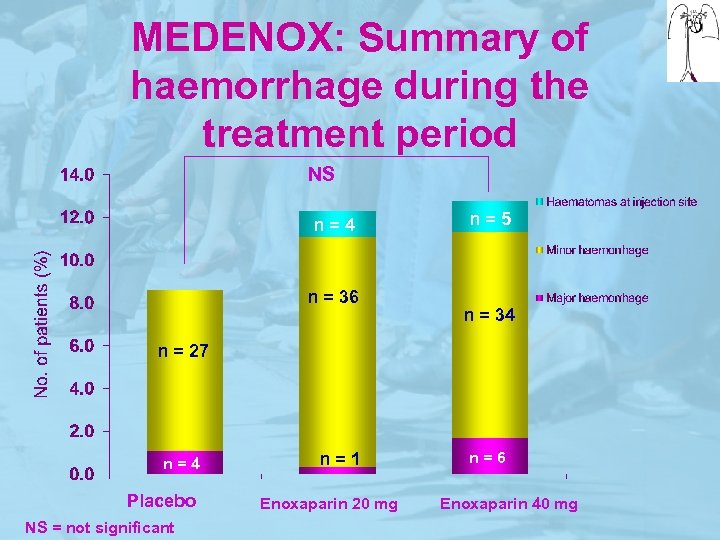
MEDENOX: Summary of haemorrhage during the treatment period NS n=4 n = 36 n=5 n = 34 n = 27 n=4 Placebo NS = not significant n=1 Enoxaparin 20 mg n=6 Enoxaparin 40 mg
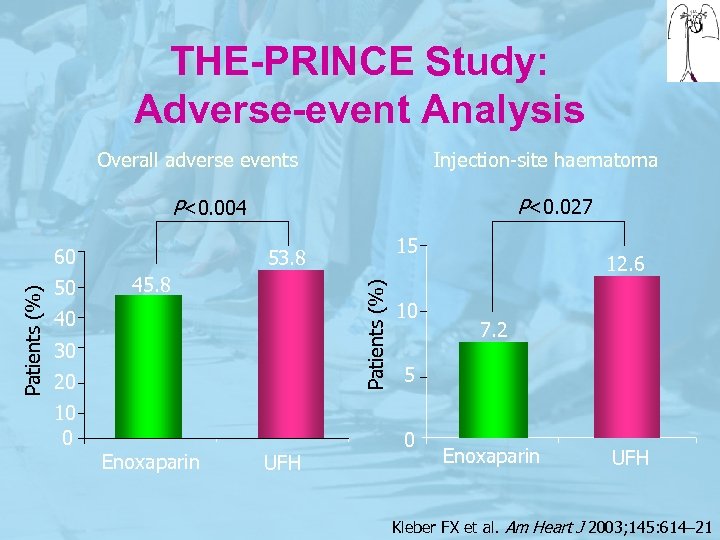
THE-PRINCE Study: Adverse-event Analysis Overall adverse events Injection-site haematoma P<0. 027 P<0. 004 50 45. 8 40 30 20 10 0 15 53. 8 Patients (%) 60 Enoxaparin UFH 10 12. 6 7. 2 5 0 Enoxaparin UFH Kleber FX et al. Am Heart J 2003; 145: 614– 21
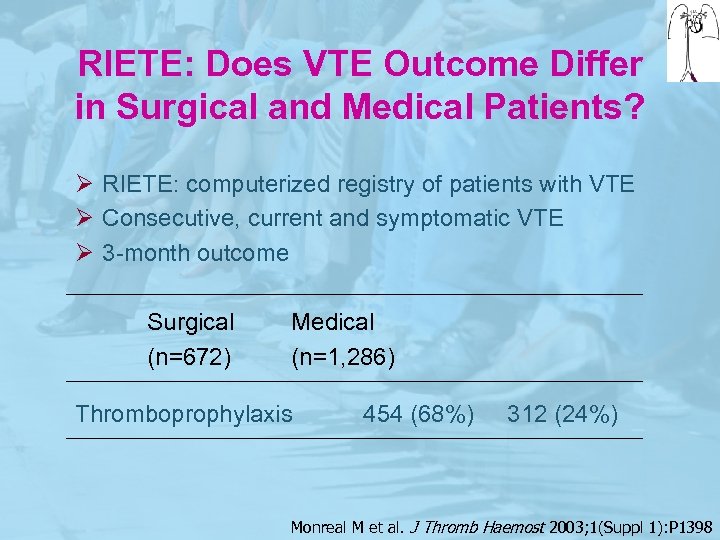
RIETE: Does VTE Outcome Differ in Surgical and Medical Patients? Ø RIETE: computerized registry of patients with VTE Ø Consecutive, current and symptomatic VTE Ø 3 -month outcome Surgical (n=672) Medical (n=1, 286) Thromboprophylaxis 454 (68%) 312 (24%) Monreal M et al. J Thromb Haemost 2003; 1(Suppl 1): P 1398

RIETE: VTE Characteristics Surgical (n=671) Distal DVT Proximal DVT PE 259 (39%) Medical (n=1, 286) P value 117 (17%) 295 (44%) 526 (41%) 115 (9%) 645 (50%) NS 0. 001 0. 009 Monreal M et al. J Thromb Haemost 2003; 1(Suppl 1): P 1398
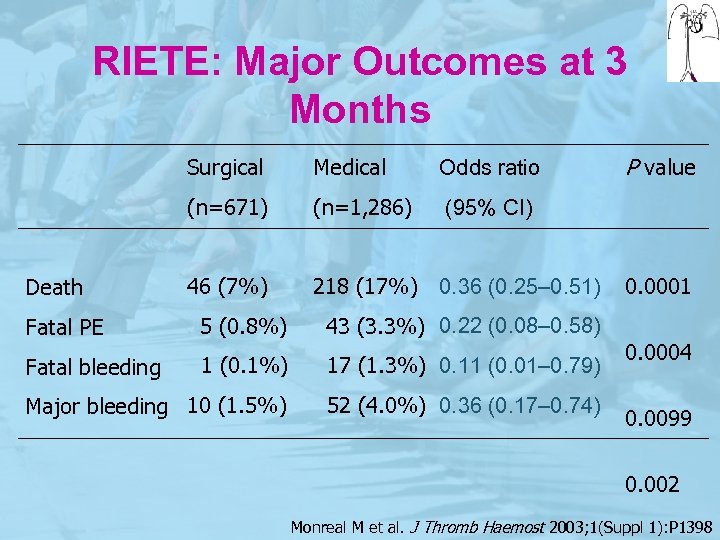
RIETE: Major Outcomes at 3 Months Surgical Odds ratio (n=671) Death Medical (n=1, 286) (95% CI) 46 (7%) 218 (17%) 0. 36 (0. 25– 0. 51) P value Fatal PE 5 (0. 8%) 43 (3. 3%) 0. 22 (0. 08– 0. 58) Fatal bleeding 1 (0. 1%) 17 (1. 3%) 0. 11 (0. 01– 0. 79) Major bleeding 10 (1. 5%) 52 (4. 0%) 0. 36 (0. 17– 0. 74) 0. 0001 0. 0004 0. 0099 0. 002 Monreal M et al. J Thromb Haemost 2003; 1(Suppl 1): P 1398

Safety of LMWHs in Medical Patients Hemorrhage Ø Anecdotal reports of ADRs induced by LMWHs Ø In 1997, two cases of fatal bleeding were reported (cancer in one case, morbid obesity and cardiac failure in the other). Patients were over 80 years with worsening renal insufficiency (Hôtel Dieu, Paris, France) Ø Increased bleeding in cardiac patients in trials with a high dose of enoxaparin ADR, adverse drug reaction
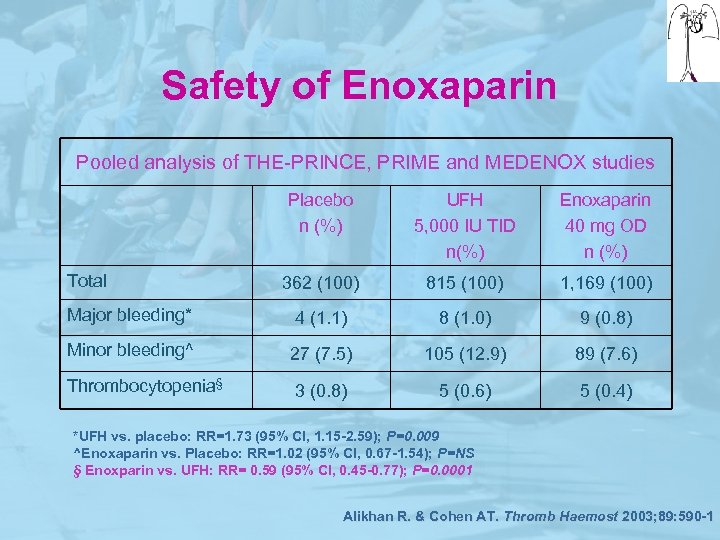
Safety of Enoxaparin Pooled analysis of THE-PRINCE, PRIME and MEDENOX studies Placebo n (%) UFH 5, 000 IU TID n(%) Enoxaparin 40 mg OD n (%) 362 (100) 815 (100) 1, 169 (100) Major bleeding* 4 (1. 1) 8 (1. 0) 9 (0. 8) Minor bleeding^ 27 (7. 5) 105 (12. 9) 89 (7. 6) Thrombocytopenia§ 3 (0. 8) 5 (0. 6) 5 (0. 4) Total *UFH vs. placebo: RR=1. 73 (95% CI, 1. 15 -2. 59); P=0. 009 ^Enoxaparin vs. Placebo: RR=1. 02 (95% CI, 0. 67 -1. 54); P=NS § Enoxparin vs. UFH: RR= 0. 59 (95% CI, 0. 45 -0. 77); P=0. 0001 Alikhan R. & Cohen AT. Thromb Haemost 2003; 89: 590 -1

Tolerability of LMWHs: Bleeding Events Ø Patients from medical departments: cardiology, geriatrics, angiology, infectious disease Ø More than 50% of patients received aspirin Ø Prolonged treatment of more than 10 days in 27% of patients Ø Bleeding events observed in 15/334 (4. 7%) of patients § 7 (7%) during curative treatment § 8 (3. 4%) during thromboprophylaxis § increased bleeding risk when creatinine clearance <20 ml/min • Digestive hemorrhage • Ecchymoses • Gingival bleeding 3 2 2 Haematoma Epistaxis 7 1 § 13 cases of thrombocytosis, 4 moderate thrombocytopenia, 1 hepatic cytosis (doubtful causal relationship) Cestac P et al. Drug Saf 2003; 26: 197– 207

LMWHs: Safe and Costeffective Ø The good safety profile of LMWHs is an important characteristic of these drugs which allows wide international use and substitution for UFH in most clinical indications Success factor Ø Socio-economic studies have concluded that LMWHs are cost-effective 1, 2 § MEDENOX trial: for patients in a tertiary-care setting, incremental cost-effectiveness of enoxaparin 40 mg versus placebo was US$ 87/VTE avoided 1 A et al. Can Respir J 2002; 9: 169– 77 2 de Lissovoy G & Subedi P. Am J Manag Care 2002; 8: 1082– 8 1 Lamy
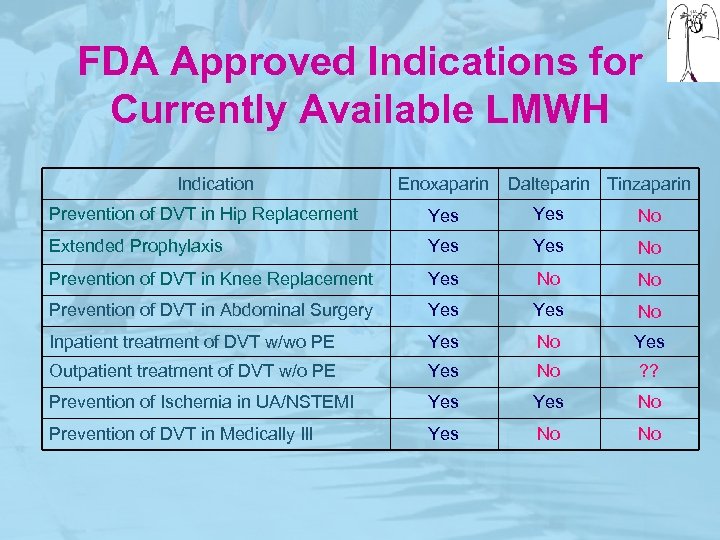
FDA Approved Indications for Currently Available LMWH Indication Enoxaparin Dalteparin Tinzaparin Prevention of DVT in Hip Replacement Yes No Extended Prophylaxis Yes No Prevention of DVT in Knee Replacement Yes No No Prevention of DVT in Abdominal Surgery Yes No Inpatient treatment of DVT w/wo PE Yes No Yes Outpatient treatment of DVT w/o PE Yes No ? ? Prevention of Ischemia in UA/NSTEMI Yes No Prevention of DVT in Medically Ill Yes No No

SUMMARY and RECOMMENDATIONS
7fb2a221a8937d377061429d4c5f97ba.ppt