708d337b4c5f86163f0a1f09f1a08f7c.ppt
- Количество слайдов: 42

Venous thromboembolism in Gynaecology Dr Edward Sang, Fellow, Gynaecologic Oncology University of Pretoria

Outline Introduction The coagulation cascade Contraception HRT Pathogenesis of VTE Risk factors Diagnosis of VTE Prevention of VTE Treatment of VTE

Introduction Venous thrombosis and its major complication PE are major causes of morbidity and mortality in hospitalized patients The diagnosis of acute VTE in outpatients is now being made more frequently because of increased diagnostic suspicion and the availabilty of reliable, noninvasive diagnostic tests

Introduction VTE is associated with significant morbidity Largely preventable Although venous thrombosis can occur in any vein, it most commonly occurs in the legs Superficial venous thrombosis occurs most frequently in varicosities and is usually benign and self-limiting but if extensive can be associated with DVT which is more serious

Introduction Thrombi localized in the calf veins are often smaller and therefore less commonly associated with longterm disability or clinically important PE By contrast proximal DVT involving the popliteal, femoral or iliac venous system can be complicated by PE Also, extensive damage to venous valves by the clot often leads to Postphlebitic syndrome

In Gynaecology 40% of all deaths following gynaecologic surgery can be directly attributed to PE is also the 2 nd leading cause of death in women who undergo induced abortion Most frequent cause of postop death in patients with uterine or cervical carcinoma

Contraception (RCOG) Relative risk of VTE increases in the first few months after initiating combined hormonal contraception. The risk reduces with increasing duration of use but it remains above the background risk until the combined hormonal contraceptive is stopped Not advised in women >35 years who smoke, have BMI >35 kg/m 2 or > 45 years with family h/o VTE in first degree relative Should be stopped 4 weeks before major surgery where immobilization is expected Progesterone-only methods of contraception do not appear to be associated with an increased incidence of VTE Green-top guideline no. 40, July 2010

HRT and VTE The WHI study in the USA confirmed an increase in risk of PE in women using HRT The mechanism is unclear Produces reduction in Fibrinogen and FVII activation, enhances fibrinolysis and is associated with increased resistance to activated protein C Although many of these effects are opposing, the net effect appears to be an increase in thrombin generation. Should be avoided in women with previous VTE and should be stopped should a woman develop VTE while on HRT The Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA 2002; 288: 321– 33.

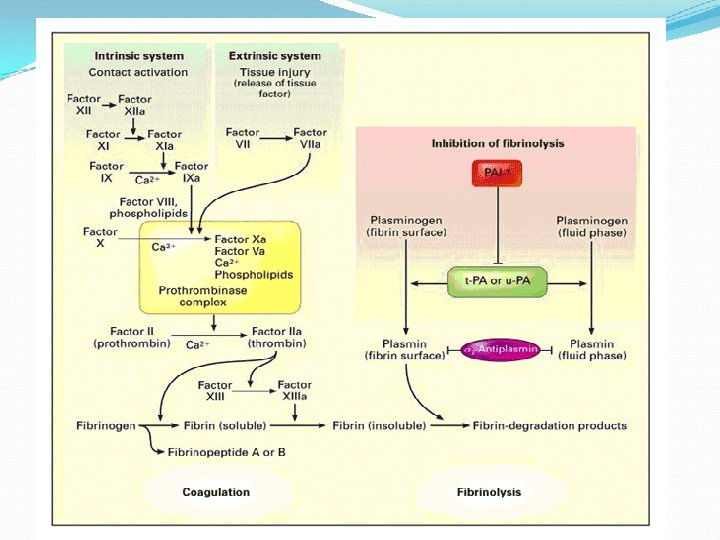
Pathogenesis of VTE Venous thrombi are composed predominantly of fibrin and red cells Pathologic or physiologic venous thrombosis occurs when activation of blood coagulation exceeds the ability of natural anticoagulant mechanisms and the fibrinolytic system to prevent clot formation

Virchow’s triad Pathogenesis of VTE first outlined by Prof Rudolph Virchow in 1858 He proposed that thrombotic disorders were associated with the triad of: Stasis Vascular injury Hypercoagulability

Pathogenesis Vessel wall injury activates coagulation and tissue damage can impair fibrinolysis by reducing synthesis of tissue plasminogen activator (t-PA) and increasing endothelial cell production of plasminogen activator inhibitor-1 (PAI-1), the major inhibitor of the fibrinolytic pathway

Pathogenesis Normally, activated coagulation factors are diluted in the flowing blood and are neutralized by inhibitors on the surface of endothelial cells or by circulating antiproteinase Activated clotting factors that escape regulation as a result of reduced levels of inhibitors or sudden generation of overwhelming amounts of these factors, trigger the coagulation system, leading to fibrin formation

Pathogenesis Homeostatic mechanisms are immediately invoked to reduce the likelihood of pathologic thrombus formation; thus, when thrombus forms, the fibrinolytic system is immediately activated as a result of the release of t-PA and urokinase from monocytes and leukocytes which are attracted to the thrombus by released fibrinopeptides and platelet products

Activation of blood coagulation Coagulation proteins circulate as inactive precursors or zymogens (except small amounts of FVII) Each zymogen is converted into an active enzyme that then activates the next zymogen in the coagulation pathway In vivo, coagulation is initiated exclusively by the TF pathway

TF pathway A proportion of circulating activated FVII (FVIIa) binds to TF at sites of vascular injury The TF-FVIIa complex then activates both FIX and FX

Common pathway FXa completes the coagulation cascade by converting prothrombin to thrombin in the presence of activated FV, phospholipid and calcium Thrombin then converts fibrinogen to fibrin, activates platelets and activates FXIII which in the presence of calcium cross-links the fibrin stabilizing the clot To ensure continuous generation of thrombin, thrombin and FXa activate FVIII and FV, markedly accelerating the coagulation reactions involving these 2 cofactors and thrombin activates FXI which in turn activates additional FIX establishing a positive feedback loop

Coagulation: intrinsic pathway Coagulation may be activated by contact of FXII with collagen on exposed subendothelium of damaged vessels or by contact with prosthetic surfaces

Coagulation FX can be activated directly by extracts of malignant cells that contain a cysteine protease, which may be one of the mechanisms by which thrombosis is induced in patients with malignant disease A factor elaborated by hypoxic endothelial cells can also directly activate FX, potentially leading to thrombosis in patients with severe venous stasis

Clinical risk factors Malignancy Extensive surgery Trauma Burns MI Local hypoxia produced by venous stasis

Venous stasis Venous return from the legs is enhanced by venous valves which prevent blood from pooling in the lower legs and by contraction of the calf muscles which propel blood up from the extremities Venous stasis may contribute to thrombogenesis by allowing stagnation of the blood with associated local hypoxia which stimulates endothelial cell release of an activator of FX Venous thrombosis is produced by immobility, venous obstruction, increased venous pressure, venous dilatation and increased blood viscosity

Inhibitors of blood coagulation Activated coagulation factors are serine proteases and their activity is modulated by several naturally occuring plasma inhibitors Important inhibitors of blood coagulation are: Antithrombin Protein C Protein S Others Factor V Leiden Prothrombin G 20210 A mutations Hyperhomocysteinaemia Antiphospholipid antibodies

DVT Pain Oedema Erythema Prominent superficial veins Non-specific: 50 -80% of patients with these signs and symptoms will not have DVT 80% of patients with symptomatic PE will not have signs and symptoms of DVT

Pulmonary embolism Has few definite symptoms But onset of respiratory distress with hypotension, chest pain and cardiac arrhythmias may be harbingers of impending death Can convert a successful operation into a postoperative fatality

Prevention of VTE Small doses of SC heparin (most widely used and studied) Unfractionated heparin LMWH Graduated compression stockings (2 nd in use after heparin) Simple, absence of major side effects Avoid tourniquet May not fit perfectly due to variation in anatomy Intermittent pneumatic compression stockings Should be used for at least 5 days postop Pneumatically inflated sleeves placed around calf/leg Various designs some as effective as heparin

Diagnosis of VTE Venography Impedence plethysmography Doppler ultrasound MRI/MRI Venography

Venography Is the reference standard for the diagnosis of DVT However: is uncomfortable, invasive Requires injection of contrast which may cause an allergic reaction or renal inury 5% risk of phlebitis Therefore not routinely used

125 labeled I Fibrinogen scanning Involves IV injection of isotope-labeled fibrinogen which is expected to be incorporated into the evolving thrombus and can be imaged by a scintillation scanner High correlation to venography Not used much as is cumbersome

Impedence plethysmography Based on the principal of electrical resistance in specific areas of the body When there is resistance to blood flow due to thrombus, there is marked reduction in the electrical resistance over that vessel Most useful in proximal DVT but relatively poor in visualizing thrombi below the knee due small size and slow flow rates in soleal sinuses
Doppler ultrasound Most widely used imaging technique for diagnosis of VTE Measures flow velocity in blood vessels A reflected sound signal is converted to both an audible form and visual image on a computer screen A thrombus causes a decrease in the reflected signal that can be heard or visualized Most machines now use color enhancement to identify arteries (red) and veins (blue)

Duplex Doppler ultrasound Combines real-time and doppler ultrasound Allows a radiologist to visualize a vessel and identify any thrombus in it

Light Reflection Rheography Uses infrared light directed at the skin The backscattered rays are quantitated, which allows an estimation of blood volume Low-cost and sensitive tool for DVT detection Not accurate in the diagnosis of DVT in pregnancy

Indirect CT Venography Uses IV contrast medium injection followed by CT scanning of the limbs or chest Detects more VTE than CT pulmonary angiography

MRI/MRI Venography Use differences in signal intensities to distinguish flowing blood from stagnant blood (clot) Does not need contrast so can be used in pregnant women MRI still in its infancy in VTE diagnosis

Nonimaging methods Automated quantitative D-dimer assay 95% NPV No proven consistent correlation between a positive D -dimer assay and venous thrombosis

Diagnosis of PE: Pulmonary angiogram Reference standard in diagnosis of PE A negative result excludes PE but tertiary pulmonary arteries must be visualized Problems of allergy and patients allergic to radioopaque dye should not undergo pulmonary angio

Ventilation-perfusion lung scan The lung scan consists of a perfusion and ventilation component Perfusion: particles of isotopically labeled microaggregates of human albumin are injected IV after which they become trapped in the pulmonary capillary bed. Their distribution reflects lung blood flow and is recorded with an external photoscanner A normal perfusion scan excludes PE but an abnormal one is none-specific Ventilation: uses radioactive gases or aerosols inhaled or exhaled by the patient while a gamma camera records the distribution of radioactivity in the alveolae The ventilation imaging improves the specificity of perfusion scanning

Prevention of VTE Unfractionated heparin LMWH Graduated compression stockings Intermittent pneumatic compression stockings

Treatment of VTE UFH: bolus dose (5000 -10, 000 IU or 80 IU/kg) then IV infusion (30 -40, 000 IU/day or 18 IU/kg) Monitored using APTT LMWH e. g. Clexane (Enoxaparin) 1. 5 mg/kg daily Monotored using anti-Xa activity (target is 0. 5 -1 U/ml) Warfarin started and heparin continued until INR is 2 -3

Heparin Acts as an anticoagulant by activating antithrombin and accelerates the rate at which antithrombin inhibits clotting enzymes esp. thrombin and FXa Heparin also enhances the inhibition of FIXa, FXIa and FVIIa bound to TF by antithrombin

Warfarin A vitamin K antagonist Interferes with synthesis of Vit K-dependent clotting factors: prothrombin, Fs VII, IX and X

LMWH MW of 5000 (1/3 that f UFH) Compared to UFH More effective Less haemorrhagic risk Lower mortality
708d337b4c5f86163f0a1f09f1a08f7c.ppt