176877725a959aec54ac6a21df29de32.ppt
- Количество слайдов: 65
 Varicella Zoster Vaccine in Preventing Zoster in Elderly People Dr David Dai Prince of Wales Hospital 21 st July, 2007
Varicella Zoster Vaccine in Preventing Zoster in Elderly People Dr David Dai Prince of Wales Hospital 21 st July, 2007
 Today’s Talk • Clinical features of Herpes Zoster • Post Herpetic Neuralgia (PHN) • Treatment • Prevention
Today’s Talk • Clinical features of Herpes Zoster • Post Herpetic Neuralgia (PHN) • Treatment • Prevention
 Herpes Zoster (Shingles): Etymology 1 = derived from Greek “herpein” meaning “to creep” zoster = a Greek word meaning “girdle” shingles = derived from Latin “cingere” herpes 1. Dorland’s Illustrated Medical Dictionary. 28 th ed. Philadelphia: WB Saunders Company, 1994: 759.
Herpes Zoster (Shingles): Etymology 1 = derived from Greek “herpein” meaning “to creep” zoster = a Greek word meaning “girdle” shingles = derived from Latin “cingere” herpes 1. Dorland’s Illustrated Medical Dictionary. 28 th ed. Philadelphia: WB Saunders Company, 1994: 759.
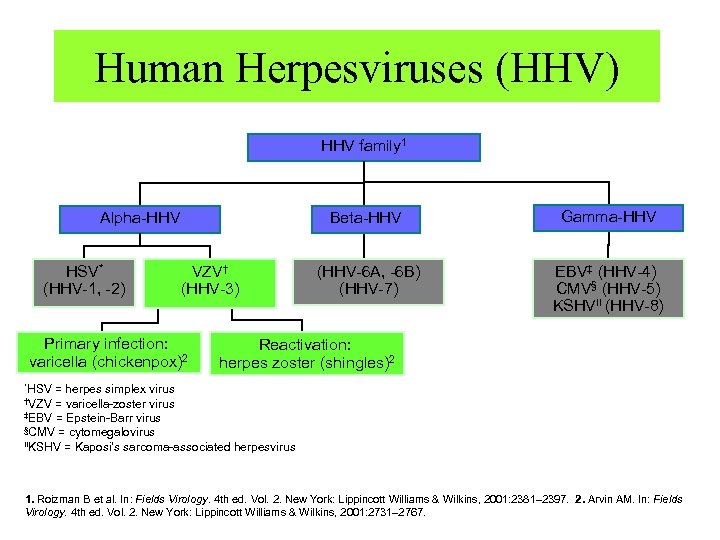 Human Herpesviruses (HHV) HHV family 1 Beta-HHV Alpha-HHV HSV* (HHV-1, -2) VZV† (HHV-3) Primary infection: varicella (chickenpox)2 Gamma-HHV (HHV-6 A, -6 B) (HHV-7) EBV‡ (HHV-4) CMV§ (HHV-5) KSHV ( ׀׀ HHV-8) Reactivation: herpes zoster (shingles)2 *HSV = herpes simplex virus †VZV = varicella-zoster virus ‡EBV = Epstein-Barr virus §CMV = cytomegalovirus ׀׀ KSHV = Kaposi’s sarcoma-associated herpesvirus 1. Roizman B et al. In: Fields Virology. 4 th ed. Vol. 2. New York: Lippincott Williams & Wilkins, 2001: 2381– 2397. 2. Arvin AM. In: Fields Virology. 4 th ed. Vol. 2. New York: Lippincott Williams & Wilkins, 2001: 2731– 2767.
Human Herpesviruses (HHV) HHV family 1 Beta-HHV Alpha-HHV HSV* (HHV-1, -2) VZV† (HHV-3) Primary infection: varicella (chickenpox)2 Gamma-HHV (HHV-6 A, -6 B) (HHV-7) EBV‡ (HHV-4) CMV§ (HHV-5) KSHV ( ׀׀ HHV-8) Reactivation: herpes zoster (shingles)2 *HSV = herpes simplex virus †VZV = varicella-zoster virus ‡EBV = Epstein-Barr virus §CMV = cytomegalovirus ׀׀ KSHV = Kaposi’s sarcoma-associated herpesvirus 1. Roizman B et al. In: Fields Virology. 4 th ed. Vol. 2. New York: Lippincott Williams & Wilkins, 2001: 2381– 2397. 2. Arvin AM. In: Fields Virology. 4 th ed. Vol. 2. New York: Lippincott Williams & Wilkins, 2001: 2731– 2767.
 A typical case (Clin Inf Disease 1999; 28: 736 -9) • 70 year old man • Pain in RUQ, diagnosed as cholescystitis • 4 days later, rash noted by nurse; diagnosis reviewed as “Shingles” • Rash persisted in next 3 weeks • No relief of pain • 5 yrs afterwards, still severe pain
A typical case (Clin Inf Disease 1999; 28: 736 -9) • 70 year old man • Pain in RUQ, diagnosed as cholescystitis • 4 days later, rash noted by nurse; diagnosis reviewed as “Shingles” • Rash persisted in next 3 weeks • No relief of pain • 5 yrs afterwards, still severe pain
 • • • Constant deep ache Sharp pains Involuntary muscle contractions Extremely sensitive skin Unable to wear clothes over the area
• • • Constant deep ache Sharp pains Involuntary muscle contractions Extremely sensitive skin Unable to wear clothes over the area
 Medications • • Codeine, oxycodone, morphine Amitriptyline, topical lidocaine Capsaicin, mexilitine, phenytoin Carbamazepine, NSAID TENS Epidural anaesthetic/steroid injection Intercostal nerve blocks All without relief
Medications • • Codeine, oxycodone, morphine Amitriptyline, topical lidocaine Capsaicin, mexilitine, phenytoin Carbamazepine, NSAID TENS Epidural anaesthetic/steroid injection Intercostal nerve blocks All without relief
 • Falls 2 years after onset and hip fracture • Forced retirement
• Falls 2 years after onset and hip fracture • Forced retirement
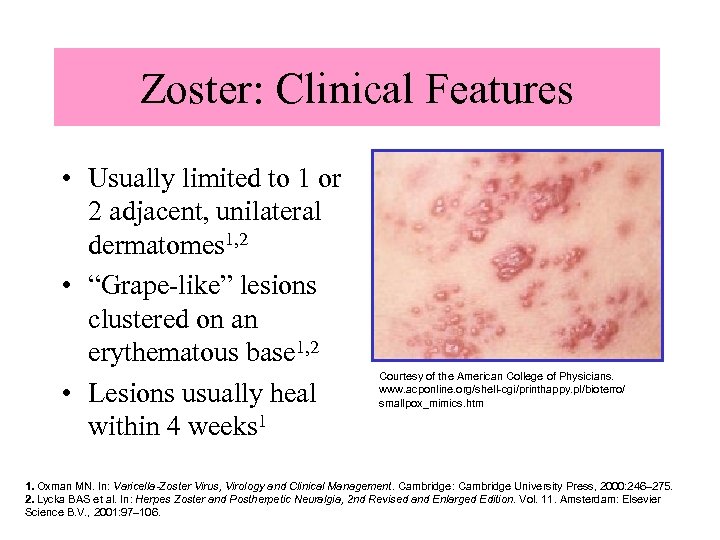 Zoster: Clinical Features • Usually limited to 1 or 2 adjacent, unilateral dermatomes 1, 2 • “Grape-like” lesions clustered on an erythematous base 1, 2 • Lesions usually heal within 4 weeks 1 Courtesy of the American College of Physicians. www. acponline. org/shell-cgi/printhappy. pl/bioterro/ smallpox_mimics. htm 1. Oxman MN. In: Varicella-Zoster Virus, Virology and Clinical Management. Cambridge: Cambridge University Press, 2000: 246– 275. 2. Lycka BAS et al. In: Herpes Zoster and Postherpetic Neuralgia, 2 nd Revised and Enlarged Edition. Vol. 11. Amsterdam: Elsevier Science B. V. , 2001: 97– 106.
Zoster: Clinical Features • Usually limited to 1 or 2 adjacent, unilateral dermatomes 1, 2 • “Grape-like” lesions clustered on an erythematous base 1, 2 • Lesions usually heal within 4 weeks 1 Courtesy of the American College of Physicians. www. acponline. org/shell-cgi/printhappy. pl/bioterro/ smallpox_mimics. htm 1. Oxman MN. In: Varicella-Zoster Virus, Virology and Clinical Management. Cambridge: Cambridge University Press, 2000: 246– 275. 2. Lycka BAS et al. In: Herpes Zoster and Postherpetic Neuralgia, 2 nd Revised and Enlarged Edition. Vol. 11. Amsterdam: Elsevier Science B. V. , 2001: 97– 106.
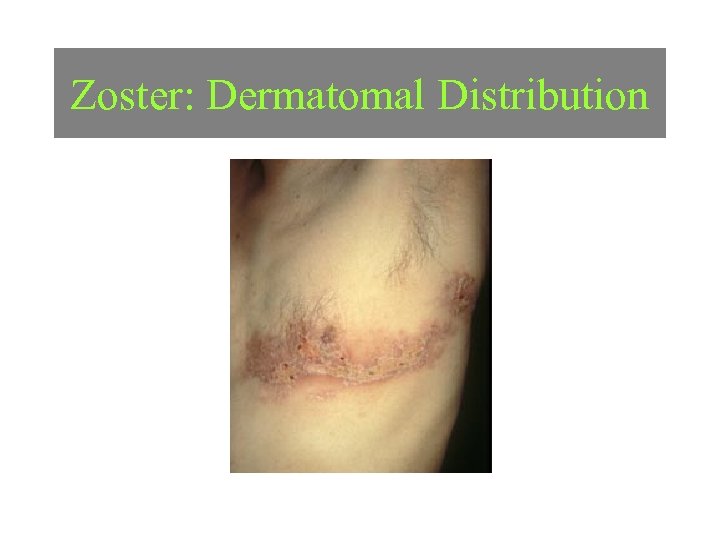 Zoster: Dermatomal Distribution Image courtesy of Charles E. Crutchfield III, MD.
Zoster: Dermatomal Distribution Image courtesy of Charles E. Crutchfield III, MD.
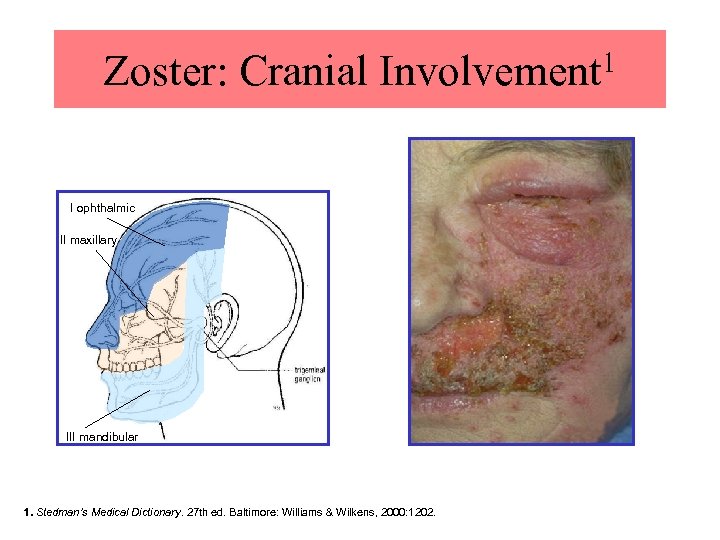 1 Zoster: Cranial Involvement I ophthalmic II maxillary III mandibular Reproduced with permission. 1 ©Diepgen TL, Yihune G et al. Dermatology Online Atlas (www. dermis. net) Reprinted with permission. 1. Stedman’s Medical Dictionary. 27 th ed. Baltimore: Williams & Wilkens, 2000: 1202.
1 Zoster: Cranial Involvement I ophthalmic II maxillary III mandibular Reproduced with permission. 1 ©Diepgen TL, Yihune G et al. Dermatology Online Atlas (www. dermis. net) Reprinted with permission. 1. Stedman’s Medical Dictionary. 27 th ed. Baltimore: Williams & Wilkens, 2000: 1202.
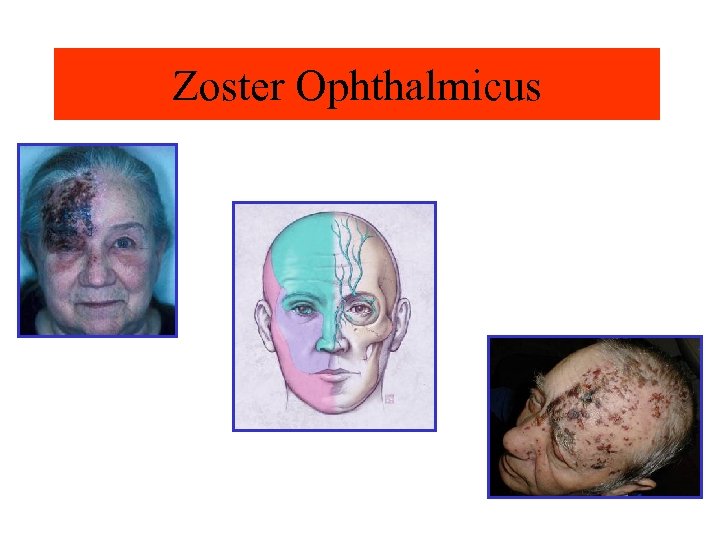 Zoster Ophthalmicus Image courtesy of Dr. Dubin’s collection (www. skinatlas. com) Image courtesy of Charles E. Crutchfield III, MD. KO Studios, Pacifica, CA. © 2002. Reprinted with permission.
Zoster Ophthalmicus Image courtesy of Dr. Dubin’s collection (www. skinatlas. com) Image courtesy of Charles E. Crutchfield III, MD. KO Studios, Pacifica, CA. © 2002. Reprinted with permission.
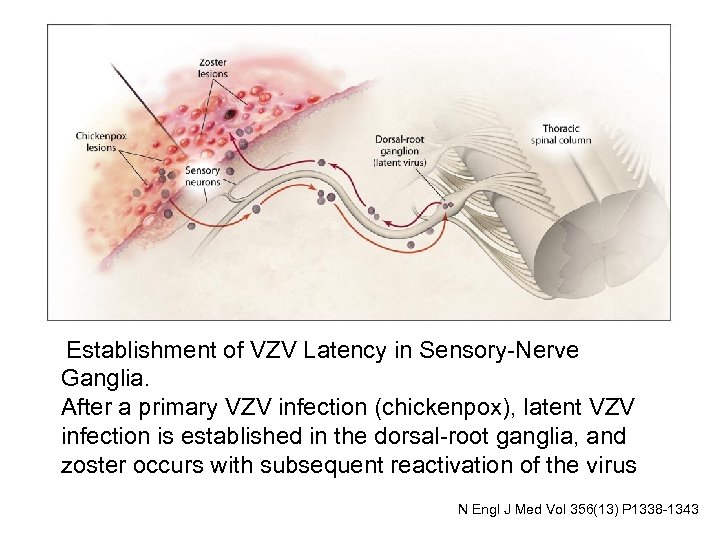 Establishment of VZV Latency in Sensory-Nerve Ganglia. After a primary VZV infection (chickenpox), latent VZV infection is established in the dorsal-root ganglia, and zoster occurs with subsequent reactivation of the virus N Engl J Med Vol 356(13) P 1338 -1343
Establishment of VZV Latency in Sensory-Nerve Ganglia. After a primary VZV infection (chickenpox), latent VZV infection is established in the dorsal-root ganglia, and zoster occurs with subsequent reactivation of the virus N Engl J Med Vol 356(13) P 1338 -1343
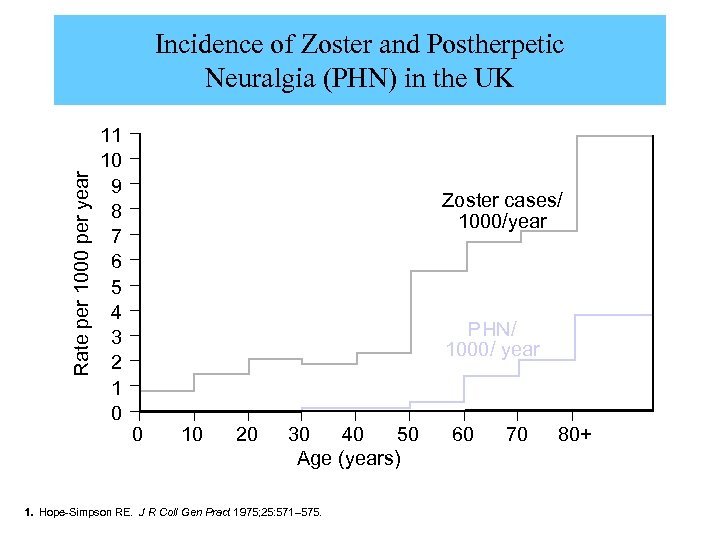 Rate per 1000 per year Incidence of Zoster and Postherpetic Neuralgia (PHN) in the UK 11 10 9 8 7 6 5 4 3 2 1 0 Zoster cases/ 1000/year PHN/ 1000/ year 0 10 20 30 40 50 Age (years) 1. Hope-Simpson RE. J R Coll Gen Pract 1975; 25: 571– 575. 60 70 80+
Rate per 1000 per year Incidence of Zoster and Postherpetic Neuralgia (PHN) in the UK 11 10 9 8 7 6 5 4 3 2 1 0 Zoster cases/ 1000/year PHN/ 1000/ year 0 10 20 30 40 50 Age (years) 1. Hope-Simpson RE. J R Coll Gen Pract 1975; 25: 571– 575. 60 70 80+
 Post Herpetic Neuralgia ( J Infect Dis 2002; 186(Suppl): 983 -90) Herpes Zoster pain 3 Phases: 1) Acute 2) Subacute: 4 -5 weeks 28. 4% 3) Chronic: > 3 months 15% > 12 months: 5 -10% Spontaneous remission: limited
Post Herpetic Neuralgia ( J Infect Dis 2002; 186(Suppl): 983 -90) Herpes Zoster pain 3 Phases: 1) Acute 2) Subacute: 4 -5 weeks 28. 4% 3) Chronic: > 3 months 15% > 12 months: 5 -10% Spontaneous remission: limited
 Effects of PHN on Patients Pain • • • Tender Burning Throbbing Stabbing Shooting/stabbing
Effects of PHN on Patients Pain • • • Tender Burning Throbbing Stabbing Shooting/stabbing
 • Allodynia • Disturbance in physical and psychological functioning • Drug dependency • Depression • Suicide
• Allodynia • Disturbance in physical and psychological functioning • Drug dependency • Depression • Suicide
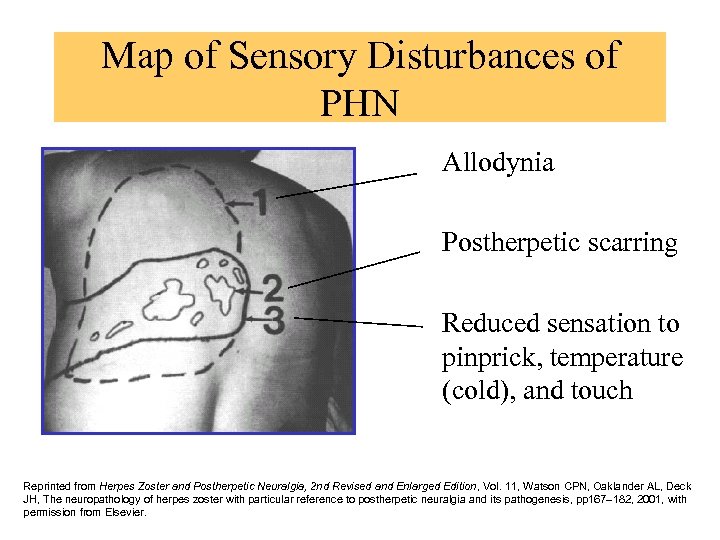 Map of Sensory Disturbances of PHN • Allodynia • Postherpetic scarring • Reduced sensation to pinprick, temperature (cold), and touch Reprinted from Herpes Zoster and Postherpetic Neuralgia, 2 nd Revised and Enlarged Edition, Vol. 11, Watson CPN, Oaklander AL, Deck JH, The neuropathology of herpes zoster with particular reference to postherpetic neuralgia and its pathogenesis, pp 167– 182, 2001, with permission from Elsevier.
Map of Sensory Disturbances of PHN • Allodynia • Postherpetic scarring • Reduced sensation to pinprick, temperature (cold), and touch Reprinted from Herpes Zoster and Postherpetic Neuralgia, 2 nd Revised and Enlarged Edition, Vol. 11, Watson CPN, Oaklander AL, Deck JH, The neuropathology of herpes zoster with particular reference to postherpetic neuralgia and its pathogenesis, pp 167– 182, 2001, with permission from Elsevier.
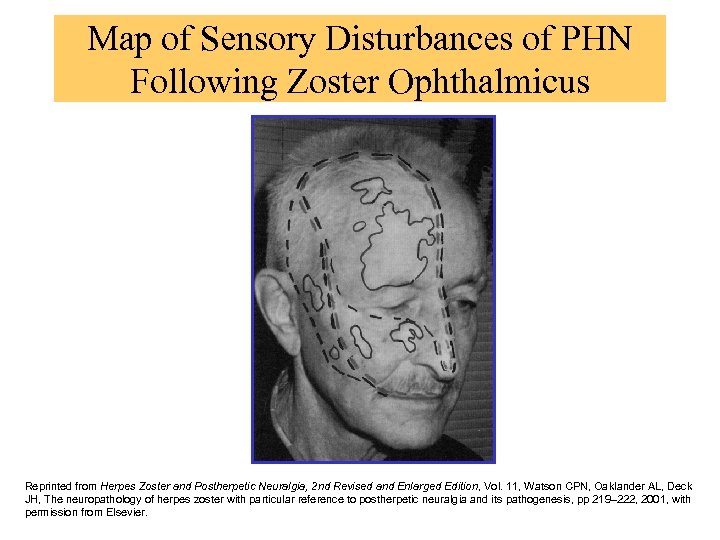 Map of Sensory Disturbances of PHN Following Zoster Ophthalmicus Reprinted from Herpes Zoster and Postherpetic Neuralgia, 2 nd Revised and Enlarged Edition, Vol. 11, Watson CPN, Oaklander AL, Deck JH, The neuropathology of herpes zoster with particular reference to postherpetic neuralgia and its pathogenesis, pp 219– 222, 2001, with permission from Elsevier.
Map of Sensory Disturbances of PHN Following Zoster Ophthalmicus Reprinted from Herpes Zoster and Postherpetic Neuralgia, 2 nd Revised and Enlarged Edition, Vol. 11, Watson CPN, Oaklander AL, Deck JH, The neuropathology of herpes zoster with particular reference to postherpetic neuralgia and its pathogenesis, pp 219– 222, 2001, with permission from Elsevier.
 Comparison of Pain Scores for Various Conditions More Pain Acute Pain Conditions 50 Chronic Pain Conditions 40 Abdominal hysterectomy 30 Acute headache Fibromyalgia Postherpetic neuralgia Zoster 20 Labor pain Postsurgical pain Mucositis 10 Atypical facial pain Musculoskeletal pain Arthritis Osteoarthritis Rheumatoid arthritis 0 Chronic cancer pain Angioplasty sheath removal Less Pain Adapted from Surgical Clinics of North America, Vol 79, Katz J, Melzack R, Measurement of pain, pp 231– 252. Copyright® 1999, with permission from Elsevier.
Comparison of Pain Scores for Various Conditions More Pain Acute Pain Conditions 50 Chronic Pain Conditions 40 Abdominal hysterectomy 30 Acute headache Fibromyalgia Postherpetic neuralgia Zoster 20 Labor pain Postsurgical pain Mucositis 10 Atypical facial pain Musculoskeletal pain Arthritis Osteoarthritis Rheumatoid arthritis 0 Chronic cancer pain Angioplasty sheath removal Less Pain Adapted from Surgical Clinics of North America, Vol 79, Katz J, Melzack R, Measurement of pain, pp 231– 252. Copyright® 1999, with permission from Elsevier.
 Financial Impact • UK(1994) • Life unit cost for managing PHN • ₤ 770 per patient
Financial Impact • UK(1994) • Life unit cost for managing PHN • ₤ 770 per patient
 Treatment Antiviral therapy within 72 hours of rash: • ↓duration (pain, rash healing) • ↓eye complications • ? Incidence of PHN
Treatment Antiviral therapy within 72 hours of rash: • ↓duration (pain, rash healing) • ↓eye complications • ? Incidence of PHN
 Table 1. Proportions (%) of patients (aged 50 years) with persisting pain in controlled trials of antiviral therapies for herpes zoster [4 6, 36, 40]. Drugs compared PHN pain Acyclovir Valacyclovir (800 mg 5×/day, 7 (1000 mg 10 days) 3×/day, 7 days) vs. placebo vs. acyclovir Valacyclovir Famciclovir (1000 mg 3×/day, 7 days) (500 mg vs. famciclovir 3×/day, 7 days) (500 mg 3×/day, 7 days) vs. placebo At 3 months 25 vs. 54 a 31 vs. 38 b 32 vs. 34 c 34. 9 vs. 49. 2 At 6 months 15 vs. 35 a 19. 9 vs. 25. 7 d 19 vs. 19 c 19. 5 vs. 40. 3 a NOTE. PHN, postherpetic neuralgia. a P <. 05 from 95% confidence interval (CI) for the relative risk (RR) for the difference between treatments. b Glaxo. Smith. Kine data on file. c P =. 84 from 95% CI for the RR for the difference between treatments. d P =. 08 from 95% CI for the RR for the difference between treatments J Infectious Dis 2002; 186(Suppl 1): S 83 -90
Table 1. Proportions (%) of patients (aged 50 years) with persisting pain in controlled trials of antiviral therapies for herpes zoster [4 6, 36, 40]. Drugs compared PHN pain Acyclovir Valacyclovir (800 mg 5×/day, 7 (1000 mg 10 days) 3×/day, 7 days) vs. placebo vs. acyclovir Valacyclovir Famciclovir (1000 mg 3×/day, 7 days) (500 mg vs. famciclovir 3×/day, 7 days) (500 mg 3×/day, 7 days) vs. placebo At 3 months 25 vs. 54 a 31 vs. 38 b 32 vs. 34 c 34. 9 vs. 49. 2 At 6 months 15 vs. 35 a 19. 9 vs. 25. 7 d 19 vs. 19 c 19. 5 vs. 40. 3 a NOTE. PHN, postherpetic neuralgia. a P <. 05 from 95% confidence interval (CI) for the relative risk (RR) for the difference between treatments. b Glaxo. Smith. Kine data on file. c P =. 84 from 95% CI for the RR for the difference between treatments. d P =. 08 from 95% CI for the RR for the difference between treatments J Infectious Dis 2002; 186(Suppl 1): S 83 -90
 The problem • Diagnosis in prodromal period extremely difficult • Majority of elderly patients with localised, unilateral pain does not develop herpes
The problem • Diagnosis in prodromal period extremely difficult • Majority of elderly patients with localised, unilateral pain does not develop herpes
 Later therapy still has benefit ↓duration of illness • • Immunocompromised Older Ophthalmic New lesions after 72 hours
Later therapy still has benefit ↓duration of illness • • Immunocompromised Older Ophthalmic New lesions after 72 hours
 At Risk of PHN • • Old age Prodromal pain Severe acute pain Rash ? Female Ophthalmic zoster Preexisting neurological disorder
At Risk of PHN • • Old age Prodromal pain Severe acute pain Rash ? Female Ophthalmic zoster Preexisting neurological disorder
 Figure 1. Annual Incidence of Herpes Zoster and Proportion of Patients with Postherpetic Neuralgia. Panel A shows the annual incidence of herpes zoster per 1000 persons in a general medical practice. 1 Panel B shows the percentage of patients with pain persisting after the onset of the zoster-associated rash. Data are from the placebo group in one large, double-blind treatment study. 7 Panel C shows the proportion of patients with postherpetic neuralgia according to age. 16 N Eng J Med 335(1); 32 -42, 1996
Figure 1. Annual Incidence of Herpes Zoster and Proportion of Patients with Postherpetic Neuralgia. Panel A shows the annual incidence of herpes zoster per 1000 persons in a general medical practice. 1 Panel B shows the percentage of patients with pain persisting after the onset of the zoster-associated rash. Data are from the placebo group in one large, double-blind treatment study. 7 Panel C shows the proportion of patients with postherpetic neuralgia according to age. 16 N Eng J Med 335(1); 32 -42, 1996
 Duration of Zoster-Associated Pain According to Age 1, 2 (n)2 (24) (53) (69) (136) (204) (270) Adapted with permission from Kost RG et al. 1 © 1996 Massachusetts Medical Society. 1. Kost RG et al. N Engl J Med 1996; 335: 33– 42. 2. de Moragas JM et al. AMA Arch Derm 1957; 75: 193– 196. (160)
Duration of Zoster-Associated Pain According to Age 1, 2 (n)2 (24) (53) (69) (136) (204) (270) Adapted with permission from Kost RG et al. 1 © 1996 Massachusetts Medical Society. 1. Kost RG et al. N Engl J Med 1996; 335: 33– 42. 2. de Moragas JM et al. AMA Arch Derm 1957; 75: 193– 196. (160)
 Treatment of established illness • • Tricyclics Anti-convulsants Opiate analgesics Surgery: sympathectomy, dorsal root entry zone lesion, cordotomy, thalamotomy, cingulumotomy, spinal cord and deep brain stimulation
Treatment of established illness • • Tricyclics Anti-convulsants Opiate analgesics Surgery: sympathectomy, dorsal root entry zone lesion, cordotomy, thalamotomy, cingulumotomy, spinal cord and deep brain stimulation
 The Patient Do anything to reduce the pain
The Patient Do anything to reduce the pain
 Strategy • Prevent or attenuate herpes zoster • Pain programme ( drug and non-drug)
Strategy • Prevent or attenuate herpes zoster • Pain programme ( drug and non-drug)
 Prevention of PHN • ? Antiviral therapy • ? Oral steroids ( excessive side effects) • Intervention to reduce acute inflammation and tissue damage (attenuate peripheral nocioceptive sensitisation and central hyperexcitability)
Prevention of PHN • ? Antiviral therapy • ? Oral steroids ( excessive side effects) • Intervention to reduce acute inflammation and tissue damage (attenuate peripheral nocioceptive sensitisation and central hyperexcitability)
 Shingles Prevention Study SPS A vaccine to prevent Herpes Zoster and Post Herpetic Neuralgia in Older Patients MN Oxman etal NEJM 2005(352): 2271 -84
Shingles Prevention Study SPS A vaccine to prevent Herpes Zoster and Post Herpetic Neuralgia in Older Patients MN Oxman etal NEJM 2005(352): 2271 -84
 The Question In persons > 60 yrs, does a live attenuated zoster virus( VZV) vaccine decrease the burden of illness caused by herpes zoster and incidence of PHN?
The Question In persons > 60 yrs, does a live attenuated zoster virus( VZV) vaccine decrease the burden of illness caused by herpes zoster and incidence of PHN?
![ZOSTAVAX® [Zoster Vaccine Live (Oka/Merck)] Product Profile • Live, attenuated varicella-zoster virus vaccine • ZOSTAVAX® [Zoster Vaccine Live (Oka/Merck)] Product Profile • Live, attenuated varicella-zoster virus vaccine •](https://present5.com/presentation/176877725a959aec54ac6a21df29de32/image-35.jpg) ZOSTAVAX® [Zoster Vaccine Live (Oka/Merck)] Product Profile • Live, attenuated varicella-zoster virus vaccine • Minimum of 19, 400 PFU* per dose • No preservative • Lyophilized product • Same excipients as VARIVAX® [Varicella Virus Vaccine Live (Oka/Merck)] • Single subcutaneous dose *PFU = plaque-forming units
ZOSTAVAX® [Zoster Vaccine Live (Oka/Merck)] Product Profile • Live, attenuated varicella-zoster virus vaccine • Minimum of 19, 400 PFU* per dose • No preservative • Lyophilized product • Same excipients as VARIVAX® [Varicella Virus Vaccine Live (Oka/Merck)] • Single subcutaneous dose *PFU = plaque-forming units
 Randomised placebo-controlled trial • • Follow up period: 3. 13 yrs (median) Setting: 22 sites in US Participants: 38, 546 persons > 60 yrs Mean age: 69 yrs, 59% men, with a history of varicella or had resided in US > 30 yrs • Immunocompromised persons excluded
Randomised placebo-controlled trial • • Follow up period: 3. 13 yrs (median) Setting: 22 sites in US Participants: 38, 546 persons > 60 yrs Mean age: 69 yrs, 59% men, with a history of varicella or had resided in US > 30 yrs • Immunocompromised persons excluded
 Randomization of Subjects Enrolled 38, 546 Adverse Event (AE) Substudy: 6616 CMI Substudy 1395 Age 60 to 69 years 20, 747 Zoster vaccine 10, 378 Placebo 10, 369 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284. Age 70 years 17, 799 Zoster vaccine 8892 Placebo 8907
Randomization of Subjects Enrolled 38, 546 Adverse Event (AE) Substudy: 6616 CMI Substudy 1395 Age 60 to 69 years 20, 747 Zoster vaccine 10, 378 Placebo 10, 369 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284. Age 70 years 17, 799 Zoster vaccine 8892 Placebo 8907
 Zoster and PHN • Zoster cases were confirmed by – Polymerase-chain-reaction (PCR) assay – Viral culture – Adjudication by a clinical evaluation committee • PHN was defined as: Zoster-associated pain rated as 3 or more on a 10 point pain scale persisting or appearing at least 90 days after rash onset
Zoster and PHN • Zoster cases were confirmed by – Polymerase-chain-reaction (PCR) assay – Viral culture – Adjudication by a clinical evaluation committee • PHN was defined as: Zoster-associated pain rated as 3 or more on a 10 point pain scale persisting or appearing at least 90 days after rash onset
 Intervention • Study group ( n= 19270): 1 subcut injection of 0. 5 m. L Oka/Merck VZV vaccine • Placebo group (n= 19276) • Vaccine has median potency of 24600 plaque-forming units
Intervention • Study group ( n= 19270): 1 subcut injection of 0. 5 m. L Oka/Merck VZV vaccine • Placebo group (n= 19276) • Vaccine has median potency of 24600 plaque-forming units
 Outcomes Vaccine efficacy: • Relative reduction in burden-of- illness sore(VEBOI): severity of and duration of herpes zoster pain • Incidence of PHN: pain rated as >3/10 Patient follow-up: 95%( modified intention to- treat- analysis)
Outcomes Vaccine efficacy: • Relative reduction in burden-of- illness sore(VEBOI): severity of and duration of herpes zoster pain • Incidence of PHN: pain rated as >3/10 Patient follow-up: 95%( modified intention to- treat- analysis)
 Definition and Measurement of the BOI Zoster BOI: Population measure Worst Pain 10 0 Days
Definition and Measurement of the BOI Zoster BOI: Population measure Worst Pain 10 0 Days
 Main Results Vaccine Efficacy
Main Results Vaccine Efficacy
 Reduction of Cumulative Incidence of Zoster Percent of Subjects With Zoster Cumulative Incidence Over Time* Placebo Zoster vaccine Logrank P-value = <0. 001 Time Since the Start of Follow-Up (in Years) *A limited number of subjects were followed beyond Year 4. 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
Reduction of Cumulative Incidence of Zoster Percent of Subjects With Zoster Cumulative Incidence Over Time* Placebo Zoster vaccine Logrank P-value = <0. 001 Time Since the Start of Follow-Up (in Years) *A limited number of subjects were followed beyond Year 4. 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
 Reduction of Cumulative Incidence of PHN Percent of Subjects With PHN Cumulative Incidence Over Time* Placebo Zoster vaccine Logrank P-value = <0. 001 Time Since the Start of Follow-Up (in Years) *A limited number of subjects were followed beyond Year 4. 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
Reduction of Cumulative Incidence of PHN Percent of Subjects With PHN Cumulative Incidence Over Time* Placebo Zoster vaccine Logrank P-value = <0. 001 Time Since the Start of Follow-Up (in Years) *A limited number of subjects were followed beyond Year 4. 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
 Overall Efficacy of the Zoster Vaccine 25%=prespecified lower bound success criterion Zoster 95% CI 51. 3% 66. 5% PHN 61. 1% BOI 0 25 50 75 Vaccine Efficacy (%) V 211. VEsummary 1 c Sept. 30, 2005 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284. 100
Overall Efficacy of the Zoster Vaccine 25%=prespecified lower bound success criterion Zoster 95% CI 51. 3% 66. 5% PHN 61. 1% BOI 0 25 50 75 Vaccine Efficacy (%) V 211. VEsummary 1 c Sept. 30, 2005 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284. 100
 Efficacy of the Zoster Vaccine by Age Stratum Zoster Ages 60 to 69 Ages 70 95% CI PHN BOI 0 20 40 60 80 100 Vaccine Efficacy (%) v 211 ACM. Vbar 1 a Dec. 6, 2005
Efficacy of the Zoster Vaccine by Age Stratum Zoster Ages 60 to 69 Ages 70 95% CI PHN BOI 0 20 40 60 80 100 Vaccine Efficacy (%) v 211 ACM. Vbar 1 a Dec. 6, 2005
 Shingles Prevention Study: Summary of Efficacy • The zoster vaccine was highly effective in reducing the incidence and morbidity from zoster and PHN – Reduced the incidence of zoster by 51% – Reduced the incidence of PHN by 67% – Reduced the BOI associated with zoster by 61% 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
Shingles Prevention Study: Summary of Efficacy • The zoster vaccine was highly effective in reducing the incidence and morbidity from zoster and PHN – Reduced the incidence of zoster by 51% – Reduced the incidence of PHN by 67% – Reduced the BOI associated with zoster by 61% 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
 Vaccine Efficacy by Age Group • The zoster vaccine benefited the 60 - to 69 -yearolds by reducing the incidence of zoster 1: – 60 -to-69 -year-old group: 63. 9% – 70 -year-old group: 37. 6% • The zoster vaccine benefited the 70 -year-olds by decreasing disease severity 1 – Efficacy for pain BOI was similar in both age groups – Efficacy for PHN was similar in both age groups 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
Vaccine Efficacy by Age Group • The zoster vaccine benefited the 60 - to 69 -yearolds by reducing the incidence of zoster 1: – 60 -to-69 -year-old group: 63. 9% – 70 -year-old group: 37. 6% • The zoster vaccine benefited the 70 -year-olds by decreasing disease severity 1 – Efficacy for pain BOI was similar in both age groups – Efficacy for PHN was similar in both age groups 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
 The AE Monitoring Substudy AE Injection Site Erythema* Pain / tenderness* Swelling* Hematoma Pruritus Warmth Zoster Vaccine (N=3345) % Placebo (N=3271) % 35. 8 34. 5 26. 2 1. 6 7. 1 1. 7 7. 0 8. 5 4. 5 1. 4 1. 0 0. 3 *Designates a solicited adverse experience. Injection-site adverse experiences solicited only from Days 0– 4 postvaccination. 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
The AE Monitoring Substudy AE Injection Site Erythema* Pain / tenderness* Swelling* Hematoma Pruritus Warmth Zoster Vaccine (N=3345) % Placebo (N=3271) % 35. 8 34. 5 26. 2 1. 6 7. 1 1. 7 7. 0 8. 5 4. 5 1. 4 1. 0 0. 3 *Designates a solicited adverse experience. Injection-site adverse experiences solicited only from Days 0– 4 postvaccination. 1. Oxman MN et al. N Engl J Med 2005; 352: 2271– 2284.
 Serious side effects 5 subjects: 1) asthma on D 2 2) PMR on D 3 3) Anaphylactoid reaction 90 minures 4) PMR on D 15 5) Goodpasture syndrome D 52
Serious side effects 5 subjects: 1) asthma on D 2 2) PMR on D 3 3) Anaphylactoid reaction 90 minures 4) PMR on D 15 5) Goodpasture syndrome D 52
 Shingles Prevention Study: Summary of Safety • The zoster vaccine has demonstrated a favorable safety profile. • Transient injection-site reactions occurred in approximately 50% of the vaccine recipients. • There were clinically important differences between the zoster vaccine and placebo with respect to – SAEs (1. 4% in each group) – Systemic clinical AEs • Following a dose of zoster vaccine, vaccineassociated rashes were uncommon (0. 1%)
Shingles Prevention Study: Summary of Safety • The zoster vaccine has demonstrated a favorable safety profile. • Transient injection-site reactions occurred in approximately 50% of the vaccine recipients. • There were clinically important differences between the zoster vaccine and placebo with respect to – SAEs (1. 4% in each group) – Systemic clinical AEs • Following a dose of zoster vaccine, vaccineassociated rashes were uncommon (0. 1%)
 Conclusion In persons >60 yrs, a live attenuated varicella-zoster virus vaccine decreased the burden of illness caused herpes zoster and the incidence of PHN. The incidence of herpes zoster was also reduced to a greater extent in vaccine recipients
Conclusion In persons >60 yrs, a live attenuated varicella-zoster virus vaccine decreased the burden of illness caused herpes zoster and the incidence of PHN. The incidence of herpes zoster was also reduced to a greater extent in vaccine recipients
 Figure 3. Host Factors in Latent VZV Infection and Reactivation. Varicella is the primary infection caused by VZV, and its resolution is associated with the induction of VZV-specific memory T cells (blue line). Memory immunity to VZV may be boosted periodically by exposure to varicella or silent reactivation from latency (red peaks). VZV-specific memory T cells decline with age. The decline below a threshold (dashed green line) correlates with an increased risk of zoster. The occurrence of zoster, in turn, is associated with an increase in VZVspecific T cells. The administration of zoster vaccine to older persons may prevent VZV-specific T cells from dropping below the threshold for the occurrence of zoster (dashed blue line). (Reprinted from Arvin. 19) N Engl J Med Vol 356(13) P 1338 -1343
Figure 3. Host Factors in Latent VZV Infection and Reactivation. Varicella is the primary infection caused by VZV, and its resolution is associated with the induction of VZV-specific memory T cells (blue line). Memory immunity to VZV may be boosted periodically by exposure to varicella or silent reactivation from latency (red peaks). VZV-specific memory T cells decline with age. The decline below a threshold (dashed green line) correlates with an increased risk of zoster. The occurrence of zoster, in turn, is associated with an increase in VZVspecific T cells. The administration of zoster vaccine to older persons may prevent VZV-specific T cells from dropping below the threshold for the occurrence of zoster (dashed blue line). (Reprinted from Arvin. 19) N Engl J Med Vol 356(13) P 1338 -1343
 Unresolved Issues (ACP Journal club 2005, 143(3): 61) • Cost-effectiveness • Optimal age at first administration • Duration of protection
Unresolved Issues (ACP Journal club 2005, 143(3): 61) • Cost-effectiveness • Optimal age at first administration • Duration of protection
 Additional Comments • • • ( MN Oxman , Human Vaccine 2007, 3: 2, 64 -68) Efficacy persist for at least 4 years Young subjects: prevent HZ Older subjects: attenuate HZ No use in PHN treatment or HZ Transmission of vaccine virus no risk
Additional Comments • • • ( MN Oxman , Human Vaccine 2007, 3: 2, 64 -68) Efficacy persist for at least 4 years Young subjects: prevent HZ Older subjects: attenuate HZ No use in PHN treatment or HZ Transmission of vaccine virus no risk
 • 86% of SPS subjects who develop HZ received famcyclovir and analgesia, and 2/3 received anti-viral treatment within 72 hrs; therefore, severity and duration of HZ pain reduced and understimate efficacy of VZV vaccine
• 86% of SPS subjects who develop HZ received famcyclovir and analgesia, and 2/3 received anti-viral treatment within 72 hrs; therefore, severity and duration of HZ pain reduced and understimate efficacy of VZV vaccine
 Cost -effectiveness • QOL and functional of older persons with HZ comparable to CCF, MI, DM, major depression (Neurol; 45 Suppl 8: S 52 -3) • PHN cause severe functional impairment, fatigue, anorexia, weight loss, insomnia, difficult concentration, depression, isolation, loss of independence and death
Cost -effectiveness • QOL and functional of older persons with HZ comparable to CCF, MI, DM, major depression (Neurol; 45 Suppl 8: S 52 -3) • PHN cause severe functional impairment, fatigue, anorexia, weight loss, insomnia, difficult concentration, depression, isolation, loss of independence and death
 Figure 2. Cost per quality-adjusted life-year (QALY) gained as a function of duration of vaccine efficacy (from 3 to 30 years) and total vaccination costs (from $50 to $500). Total vaccination costs include unit vaccine cost, a public awareness campaign, administration costs, patient Ann Intern Med 2006; 145: 317 -325 travel time and time receiving vaccine, and cost of treating adverse events.
Figure 2. Cost per quality-adjusted life-year (QALY) gained as a function of duration of vaccine efficacy (from 3 to 30 years) and total vaccination costs (from $50 to $500). Total vaccination costs include unit vaccine cost, a public awareness campaign, administration costs, patient Ann Intern Med 2006; 145: 317 -325 travel time and time receiving vaccine, and cost of treating adverse events.
 • Cost of vaccine US$150 per dose, assuming duration of efficacy 10 yrs: • $ 2600 per HZ prevented • $ 7500 per PHN prevented • NNV to prevent a case of HZ: 18 • NNV to prevent a clinically significant case of PHN: 1009
• Cost of vaccine US$150 per dose, assuming duration of efficacy 10 yrs: • $ 2600 per HZ prevented • $ 7500 per PHN prevented • NNV to prevent a case of HZ: 18 • NNV to prevent a clinically significant case of PHN: 1009
 Improved Zoster vaccine • Increased potency ( ↑quantity of infective VZV and amount of VZV antigens) • 5 times the current vaccine • Likely to be well tolerated • Effective inactivated zoster vaccine to protect immunodeficient population
Improved Zoster vaccine • Increased potency ( ↑quantity of infective VZV and amount of VZV antigens) • 5 times the current vaccine • Likely to be well tolerated • Effective inactivated zoster vaccine to protect immunodeficient population
 ZOSTAVAQX® Stored frozen at an average temperature of -15ºC or colder until it is reconstituted for injection.
ZOSTAVAQX® Stored frozen at an average temperature of -15ºC or colder until it is reconstituted for injection.
 Recommendations The author has already been vaccinated and has purchased the vaccine and administrated to his wife
Recommendations The author has already been vaccinated and has purchased the vaccine and administrated to his wife
 Preventive Dermatology (J Am Acad Derm 2007; 56: 675 -6) Vaccination with live attenuated VZV vaccine should be made available to all immunocompetent persons aged 60 yrs and older during routine office visits; and before immunosuppresive atherapy
Preventive Dermatology (J Am Acad Derm 2007; 56: 675 -6) Vaccination with live attenuated VZV vaccine should be made available to all immunocompetent persons aged 60 yrs and older during routine office visits; and before immunosuppresive atherapy
 ACIP Provisional Recommendations ( October 25, 2006) • A single dose of zoster vaccine is recommended for adults 60 years of age and older whether or not they report a prior episode of herpes zoster. Persons with chronic medical conditions may be vaccinated unless a contraindication or precaution exists for their condition. • Contraindications and precautions to use of zoster vaccine are available at http: //www. fda. gov/Cber/label/zosmer 052506 LB. htm
ACIP Provisional Recommendations ( October 25, 2006) • A single dose of zoster vaccine is recommended for adults 60 years of age and older whether or not they report a prior episode of herpes zoster. Persons with chronic medical conditions may be vaccinated unless a contraindication or precaution exists for their condition. • Contraindications and precautions to use of zoster vaccine are available at http: //www. fda. gov/Cber/label/zosmer 052506 LB. htm
 To give or not to give? Give Patient treatment experience Storage Cost
To give or not to give? Give Patient treatment experience Storage Cost


