b28aee24fb3844f96195c069c832c109.ppt
- Количество слайдов: 77
 Vaccines and Vaccine Delivery Systems of the Future Bruce G. Weniger, MD, MPH Assistant Chief for Vaccine Development Immunization Safety Branch Epidemiology and Surveillance Division National Immunization Program, CDC Course: Vaccines and Immunization - 2004 (IH 591 B / EPI 591 V) Rollins School of Public Health, Emory University, Atlanta, 19 November 2004
Vaccines and Vaccine Delivery Systems of the Future Bruce G. Weniger, MD, MPH Assistant Chief for Vaccine Development Immunization Safety Branch Epidemiology and Surveillance Division National Immunization Program, CDC Course: Vaccines and Immunization - 2004 (IH 591 B / EPI 591 V) Rollins School of Public Health, Emory University, Atlanta, 19 November 2004
 Vaccines and Vaccine Delivery Systems of the Future 1. Rapidly expanding basic science knowledge 2. Fruits of biotechnology: new and improved vaccines 3. Future vaccine constructs and technologies 4. Future (and re-emerging) vaccine delivery technologies
Vaccines and Vaccine Delivery Systems of the Future 1. Rapidly expanding basic science knowledge 2. Fruits of biotechnology: new and improved vaccines 3. Future vaccine constructs and technologies 4. Future (and re-emerging) vaccine delivery technologies
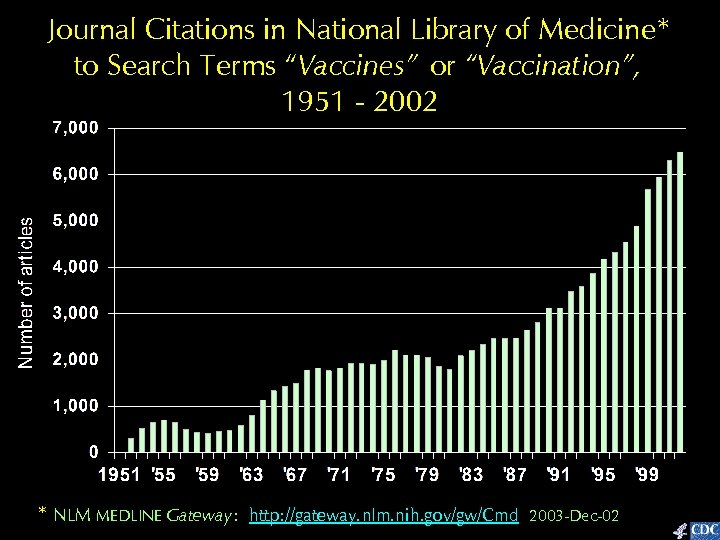 Journal Citations in National Library of Medicine* to Search Terms “Vaccines” or “Vaccination”, 1951 - 2002 * NLM MEDLINE Gateway: http: //gateway. nlm. nih. gov/gw/Cmd 2003 -Dec-02
Journal Citations in National Library of Medicine* to Search Terms “Vaccines” or “Vaccination”, 1951 - 2002 * NLM MEDLINE Gateway: http: //gateway. nlm. nih. gov/gw/Cmd 2003 -Dec-02
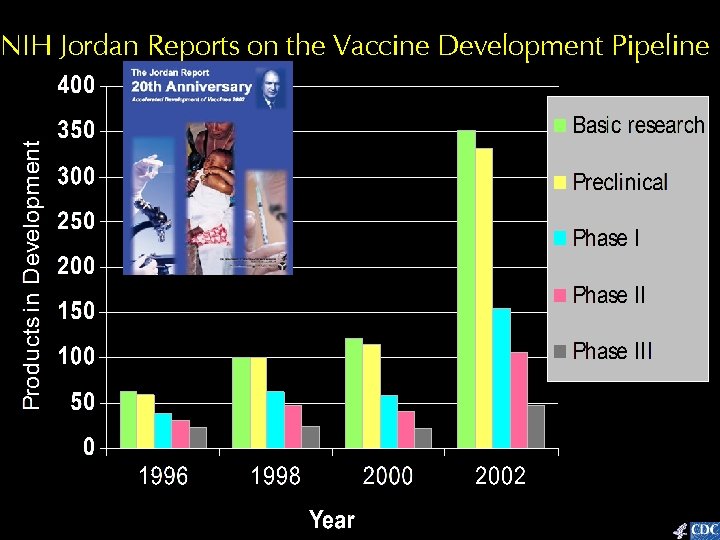 NIH Jordan Reports on the Vaccine Development Pipeline
NIH Jordan Reports on the Vaccine Development Pipeline
 Maurice Hilleman ! ! ! Walter Reed Army Institute of Research Merck and Company Shepherded many cell culture products from the laboratory to commercial availability, including:
Maurice Hilleman ! ! ! Walter Reed Army Institute of Research Merck and Company Shepherded many cell culture products from the laboratory to commercial availability, including:
 2. The Fruits of Biotechnology: New and Improved Vaccines ! ! Newly Vaccine-Preventable Diseases Recent/Expected Improvements on Existing Vaccines
2. The Fruits of Biotechnology: New and Improved Vaccines ! ! Newly Vaccine-Preventable Diseases Recent/Expected Improvements on Existing Vaccines
 Newly Vaccine-Preventable Diseases
Newly Vaccine-Preventable Diseases
 Recent/Expected Improvements on Existing Vaccines U. S. Licensed ! Hepatitis B, recombinant 1986 ! Haemophilus influenzae B, conjugated 1990 for use in infants ! Polio, inactivated, enhanced potency 1990 ! Pertussis, acellular 1992 ! Streptococcus pneumoniae , conjugated 2000 for use in infants
Recent/Expected Improvements on Existing Vaccines U. S. Licensed ! Hepatitis B, recombinant 1986 ! Haemophilus influenzae B, conjugated 1990 for use in infants ! Polio, inactivated, enhanced potency 1990 ! Pertussis, acellular 1992 ! Streptococcus pneumoniae , conjugated 2000 for use in infants
 3. Future Vaccine Constructs and Technologies ! Genetically-engineered live vectors ! Nucleic acid vaccines ! Recombinant plants and plant viruses ! Synthetic immune-stimulating complexes ! Microencapsulation of antigen ! New adjuvants
3. Future Vaccine Constructs and Technologies ! Genetically-engineered live vectors ! Nucleic acid vaccines ! Recombinant plants and plant viruses ! Synthetic immune-stimulating complexes ! Microencapsulation of antigen ! New adjuvants
 Traditional Vaccine Type Classifications - I
Traditional Vaccine Type Classifications - I
 Traditional Vaccine Type Classifications - II
Traditional Vaccine Type Classifications - II
 Newer Vaccine Type Classification(s)
Newer Vaccine Type Classification(s)
 Future Vaccine Constructs and Technologies – Recombinant Live Vectors
Future Vaccine Constructs and Technologies – Recombinant Live Vectors
 Future Vaccine Constructs and Technologies – Recombinant Live Vectors
Future Vaccine Constructs and Technologies – Recombinant Live Vectors
 Future Vaccine Constructs and Technologies – Nucleic Acid Vaccines
Future Vaccine Constructs and Technologies – Nucleic Acid Vaccines
 Bioengineered Plant Vaccines - 1
Bioengineered Plant Vaccines - 1
 Bioengineered Plant Vaccines - 2 Source: Charles Arntzen, Arizona State Univ. , www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. AM
Bioengineered Plant Vaccines - 2 Source: Charles Arntzen, Arizona State Univ. , www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. AM
 Bioengineered Plant Vaccines - 3 Source: Charles Arntzen, Arizona State University
Bioengineered Plant Vaccines - 3 Source: Charles Arntzen, Arizona State University
 Bioengineered Plant Vaccines - 4
Bioengineered Plant Vaccines - 4
 Synthetic Virus-like Particles as Vaccine Constructs ! ! “Virosomes™” ! “Phage T 4 Display” Cath U. Am. ) “ISCOMS®” (Berna Biotech) (CSL, Pty) “Proteosomes™” (ID Biomedical) “Alphavirus-like particles / VEE replicons” (Alpha. Vax) (Walter Reed AIR,
Synthetic Virus-like Particles as Vaccine Constructs ! ! “Virosomes™” ! “Phage T 4 Display” Cath U. Am. ) “ISCOMS®” (Berna Biotech) (CSL, Pty) “Proteosomes™” (ID Biomedical) “Alphavirus-like particles / VEE replicons” (Alpha. Vax) (Walter Reed AIR,
 Synthetic Virus-like Particles - 1
Synthetic Virus-like Particles - 1
 Synthetic Virus-like Particles - 2
Synthetic Virus-like Particles - 2
 Future Vaccine Constructs and Technologies – Microencapsulation
Future Vaccine Constructs and Technologies – Microencapsulation
 “Cochleate” Encapsulation
“Cochleate” Encapsulation
 Future Vaccine Constructs and Technologies – Adjuvants
Future Vaccine Constructs and Technologies – Adjuvants
 4. Future (and Re-emerging) Vaccine Delivery Technologies
4. Future (and Re-emerging) Vaccine Delivery Technologies
 Why Needle-syringe Alternatives Are Needed - 1
Why Needle-syringe Alternatives Are Needed - 1
 Why Needle-syringe Alternatives Are Needed - 2
Why Needle-syringe Alternatives Are Needed - 2
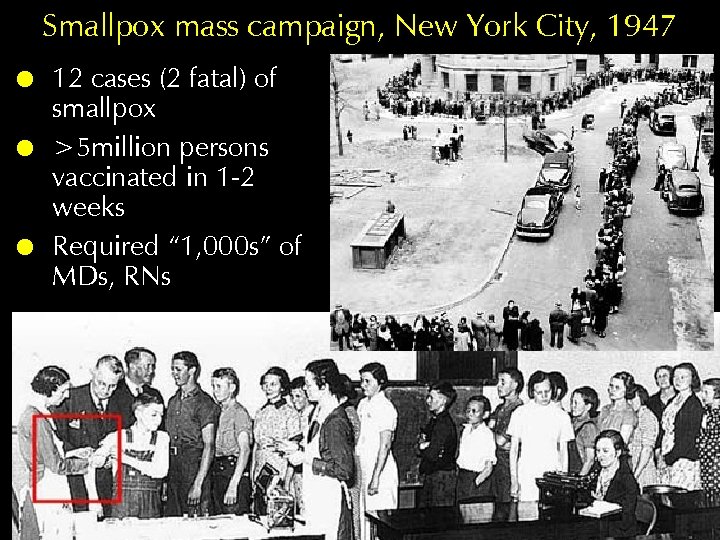 Smallpox mass campaign, New York City, 1947 ! ! ! 12 cases (2 fatal) of smallpox >5 million persons vaccinated in 1 -2 weeks Required “ 1, 000 s” of MDs, RNs
Smallpox mass campaign, New York City, 1947 ! ! ! 12 cases (2 fatal) of smallpox >5 million persons vaccinated in 1 -2 weeks Required “ 1, 000 s” of MDs, RNs
 Polio Vaccine (POL IPV) Mass Campaigns, 1950 s
Polio Vaccine (POL IPV) Mass Campaigns, 1950 s
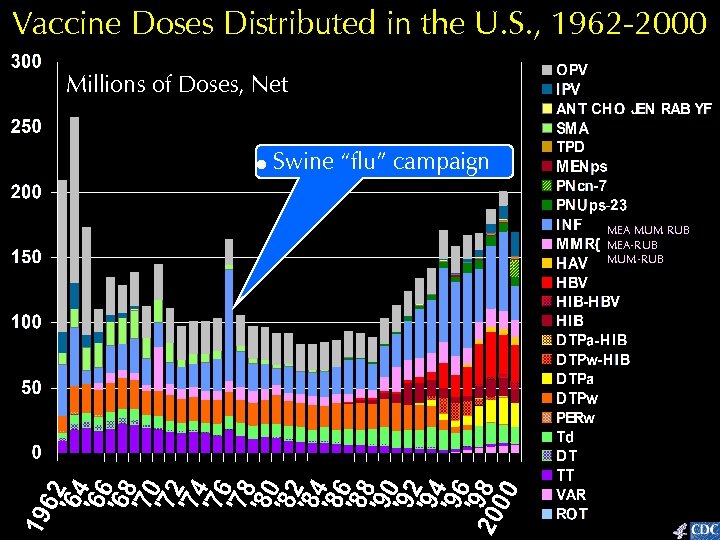 Vaccine Doses Distributed in the U. S. , 1962 -2000 Millions of Doses, Net Swine “flu” campaign MEA MUM RUB MEA-RUB MUM-RUB
Vaccine Doses Distributed in the U. S. , 1962 -2000 Millions of Doses, Net Swine “flu” campaign MEA MUM RUB MEA-RUB MUM-RUB

 Jet injectors for Mass Campaigns
Jet injectors for Mass Campaigns
 Immunogenicity of Jet-injected Vaccines www. cdc. gov/nip/dev/jetinject. htm#bibliography
Immunogenicity of Jet-injected Vaccines www. cdc. gov/nip/dev/jetinject. htm#bibliography
 Safety Concerns over Multi-use-nozzle Jet Injectors
Safety Concerns over Multi-use-nozzle Jet Injectors
 Archives of Internal Medicine 1990; 150: 1923 -1927
Archives of Internal Medicine 1990; 150: 1923 -1927
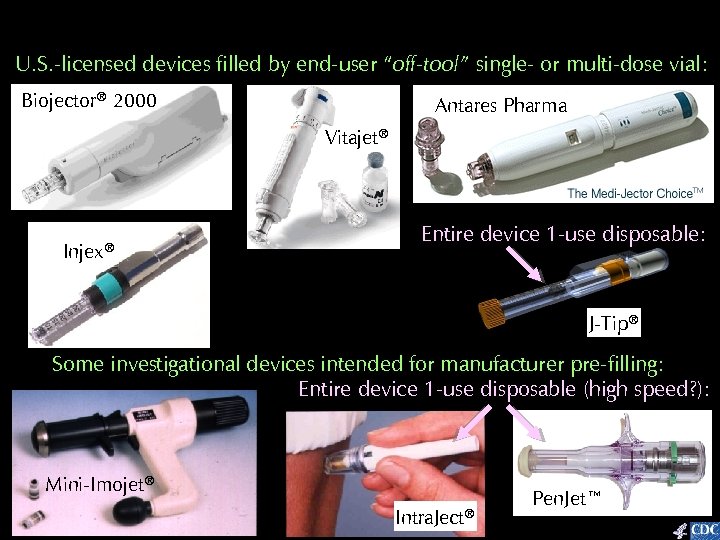 U. S. -licensed devices filled by end-user “off-tool” single- or multi-dose vial: Biojector® 2000 Antares Pharma Vitajet® Injex® Entire device 1 -use disposable: J-Tip® Some investigational devices intended for manufacturer pre-filling: Entire device 1 -use disposable (high speed? ): Mini-Imojet® Intra. Ject® Pen. Jet™
U. S. -licensed devices filled by end-user “off-tool” single- or multi-dose vial: Biojector® 2000 Antares Pharma Vitajet® Injex® Entire device 1 -use disposable: J-Tip® Some investigational devices intended for manufacturer pre-filling: Entire device 1 -use disposable (high speed? ): Mini-Imojet® Intra. Ject® Pen. Jet™
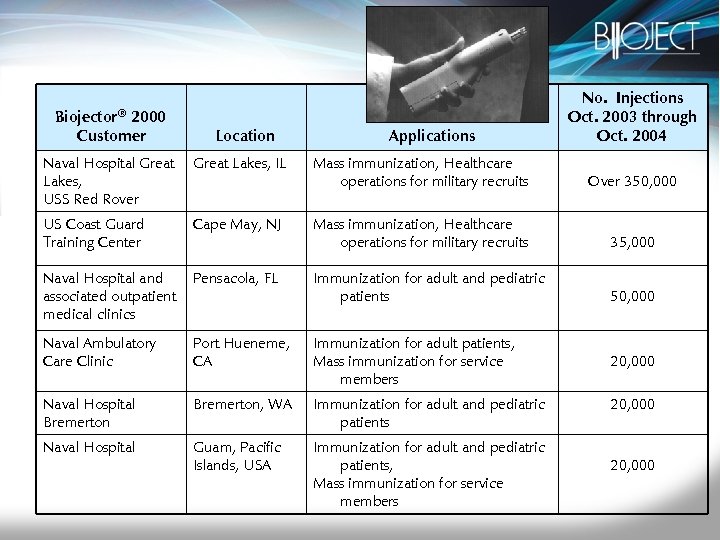 Biojector® 2000 Customer Location Applications No. Injections Oct. 2003 through Oct. 2004 Naval Hospital Great Lakes, USS Red Rover Great Lakes, IL Mass immunization, Healthcare operations for military recruits Over 350, 000 US Coast Guard Training Center Cape May, NJ Mass immunization, Healthcare operations for military recruits 35, 000 Naval Hospital and associated outpatient medical clinics Pensacola, FL Immunization for adult and pediatric patients 50, 000 Naval Ambulatory Care Clinic Port Hueneme, CA Immunization for adult patients, Mass immunization for service members Naval Hospital Bremerton, WA Immunization for adult and pediatric patients Naval Hospital Guam, Pacific Islands, USA Immunization for adult and pediatric patients, Mass immunization for service members 20, 000
Biojector® 2000 Customer Location Applications No. Injections Oct. 2003 through Oct. 2004 Naval Hospital Great Lakes, USS Red Rover Great Lakes, IL Mass immunization, Healthcare operations for military recruits Over 350, 000 US Coast Guard Training Center Cape May, NJ Mass immunization, Healthcare operations for military recruits 35, 000 Naval Hospital and associated outpatient medical clinics Pensacola, FL Immunization for adult and pediatric patients 50, 000 Naval Ambulatory Care Clinic Port Hueneme, CA Immunization for adult patients, Mass immunization for service members Naval Hospital Bremerton, WA Immunization for adult and pediatric patients Naval Hospital Guam, Pacific Islands, USA Immunization for adult and pediatric patients, Mass immunization for service members 20, 000
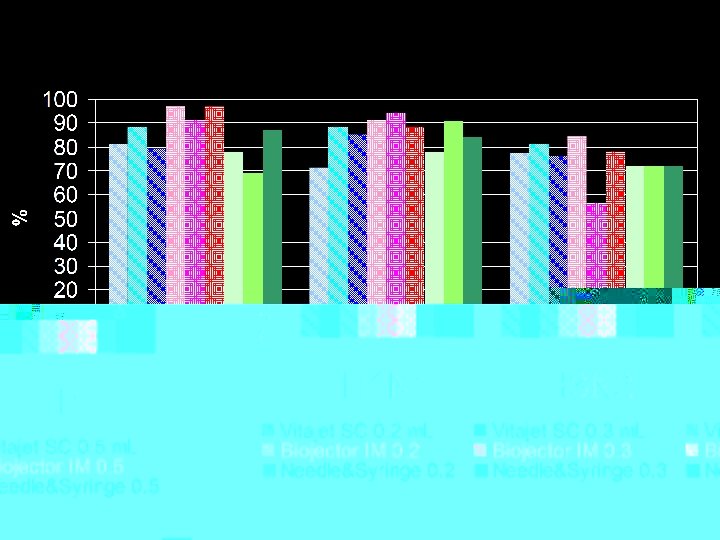 JI-SC JI-IM NS-IM JI-SC: JI-IM: NS-IM: Source: Jackson, et al. Vaccine 2001; 19: 4703 -4709
JI-SC JI-IM NS-IM JI-SC: JI-IM: NS-IM: Source: Jackson, et al. Vaccine 2001; 19: 4703 -4709
 Lectra. Jet® – Motorized Model
Lectra. Jet® – Motorized Model
 Lectra. Jet ® Cartridges
Lectra. Jet ® Cartridges
 Demonstration of Lectra. Jet® Jet Injector, High-speed Model
Demonstration of Lectra. Jet® Jet Injector, High-speed Model
 Convenient Intradermal Injection by Jet Injection Older MUNJI models ID nozzle IM nozzle
Convenient Intradermal Injection by Jet Injection Older MUNJI models ID nozzle IM nozzle
 Convenient Intradermal Injection by Jet Injection Newer DCJI models
Convenient Intradermal Injection by Jet Injection Newer DCJI models
 Future (and Re-emerging) Vaccine Delivery Technologies
Future (and Re-emerging) Vaccine Delivery Technologies
 Live, trivalent, attenuated influenza vaccine
Live, trivalent, attenuated influenza vaccine
 Virosome intranasal influenza vaccine
Virosome intranasal influenza vaccine
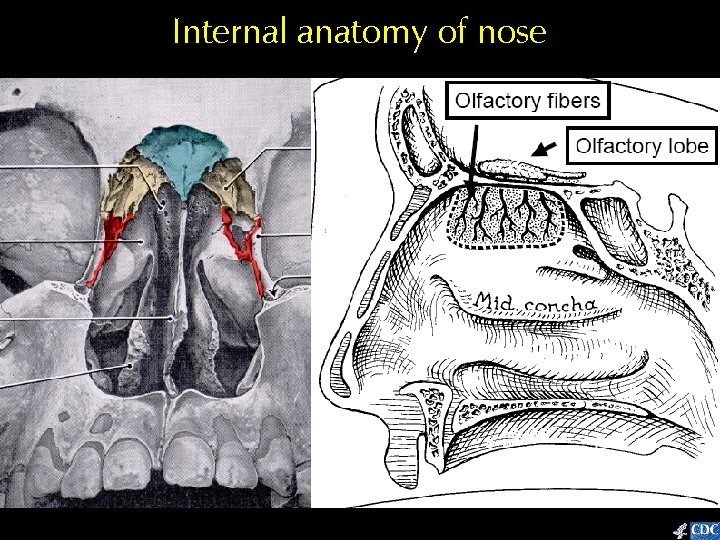 Internal anatomy of nose
Internal anatomy of nose
 Intranasal Delivery of “Proteosomes™” ! ! Nanoparticles from bacterial outer membrane protein ID Biomedical, Canada Source: Louis Fries, ID Biomedical Corp. ; www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. PM/
Intranasal Delivery of “Proteosomes™” ! ! Nanoparticles from bacterial outer membrane protein ID Biomedical, Canada Source: Louis Fries, ID Biomedical Corp. ; www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. PM/
 Source: Louis Fries, ID Biomedical Corp. ; www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. PM/
Source: Louis Fries, ID Biomedical Corp. ; www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. PM/
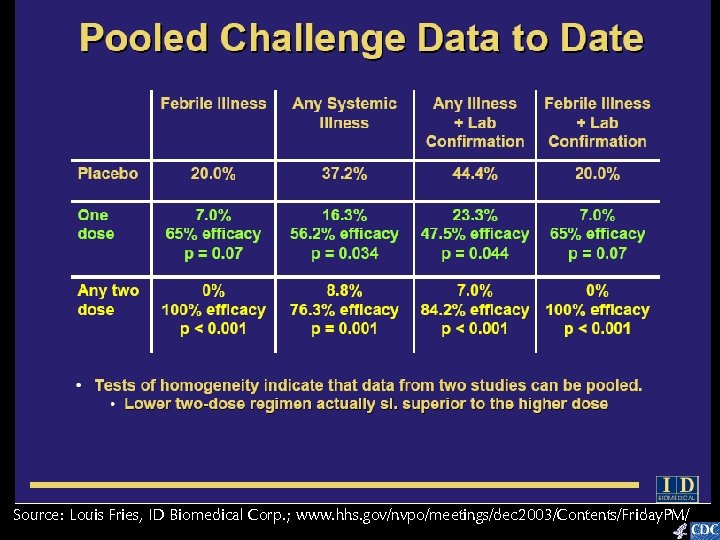 Source: Louis Fries, ID Biomedical Corp. ; www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. PM/
Source: Louis Fries, ID Biomedical Corp. ; www. hhs. gov/nvpo/meetings/dec 2003/Contents/Friday. PM/
 Optinose “Bi-directional” Nasal Delivery Device - Video http: //www. optinose. no/technology/optinose_bidirectionalvideo. asp
Optinose “Bi-directional” Nasal Delivery Device - Video http: //www. optinose. no/technology/optinose_bidirectionalvideo. asp
 Future (and Re-emerging) Vaccine Delivery Technologies
Future (and Re-emerging) Vaccine Delivery Technologies
 Aerosol measles vaccination Bulletin W HO ©October 2002 Infectious Diseases in Children ©May 2000
Aerosol measles vaccination Bulletin W HO ©October 2002 Infectious Diseases in Children ©May 2000
 A New Device for Measles Mass Vaccination by Aerosol Low Thermal/Mechanical Stress on Virus Compatible with Nasal Prong or Mask
A New Device for Measles Mass Vaccination by Aerosol Low Thermal/Mechanical Stress on Virus Compatible with Nasal Prong or Mask
 Aerosol inhalation measles vaccination
Aerosol inhalation measles vaccination
 Future (and Re-emerging) Vaccine Delivery Technologies
Future (and Re-emerging) Vaccine Delivery Technologies
 Cutaneous Immunization - I
Cutaneous Immunization - I
 Source: Powder. Ject plc
Source: Powder. Ject plc
 Cutaneous Immunization - II
Cutaneous Immunization - II
 “Epidermal Powder Immunization” - 1
“Epidermal Powder Immunization” - 1
 “Epidermal Powder Immunization” - 2 Human trial: trivalent inactivated influenza vaccine
“Epidermal Powder Immunization” - 2 Human trial: trivalent inactivated influenza vaccine
 On. Vax™ Microenhancer Arrays, Becton-Dickinson Mikszta, et al. Nature Medicine April 2002; 8(4): 415 -19
On. Vax™ Microenhancer Arrays, Becton-Dickinson Mikszta, et al. Nature Medicine April 2002; 8(4): 415 -19
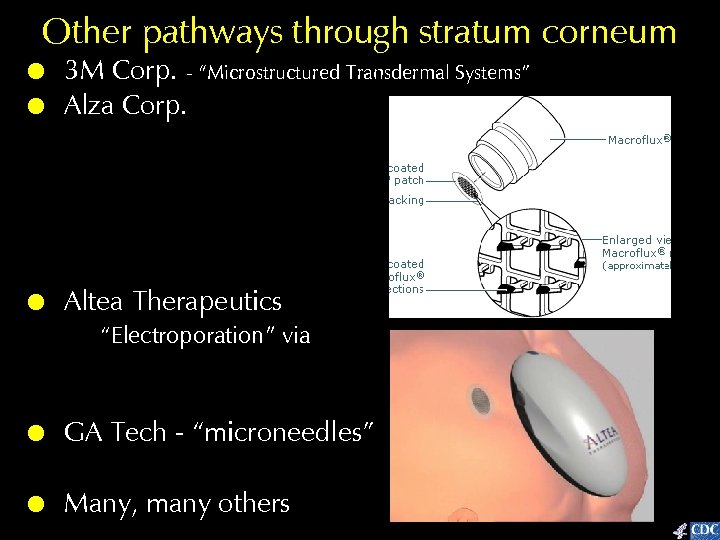 Other pathways through stratum corneum ! ! 3 M Corp. - “Microstructured Transdermal Systems” Alza Corp. ! Altea Therapeutics Macroflux® “Electroporation” via Pass. Port™ ! GA Tech - “microneedles” ! Many, many others
Other pathways through stratum corneum ! ! 3 M Corp. - “Microstructured Transdermal Systems” Alza Corp. ! Altea Therapeutics Macroflux® “Electroporation” via Pass. Port™ ! GA Tech - “microneedles” ! Many, many others
 Laser “Ablation” of Stratum Corneum Norwood Abbey, Ltd. Victoria, Australia www. norwoodabbey. com
Laser “Ablation” of Stratum Corneum Norwood Abbey, Ltd. Victoria, Australia www. norwoodabbey. com
 Cutaneous Immunization - III
Cutaneous Immunization - III
 Iomai, Inc. , Gaithersburg, MD, USA
Iomai, Inc. , Gaithersburg, MD, USA
 Source: Iomai, Inc.
Source: Iomai, Inc.
 Cutaneous Immunization - IV
Cutaneous Immunization - IV
 Future (and Re-emerging) Vaccine Delivery Technologies
Future (and Re-emerging) Vaccine Delivery Technologies
 Aerosol Anthrax and Plague Vaccines Source: Jim Eyles, Dstl, MOD, www. hhs. gov/nvpo/meetings/dec 2003/Contents/
Aerosol Anthrax and Plague Vaccines Source: Jim Eyles, Dstl, MOD, www. hhs. gov/nvpo/meetings/dec 2003/Contents/
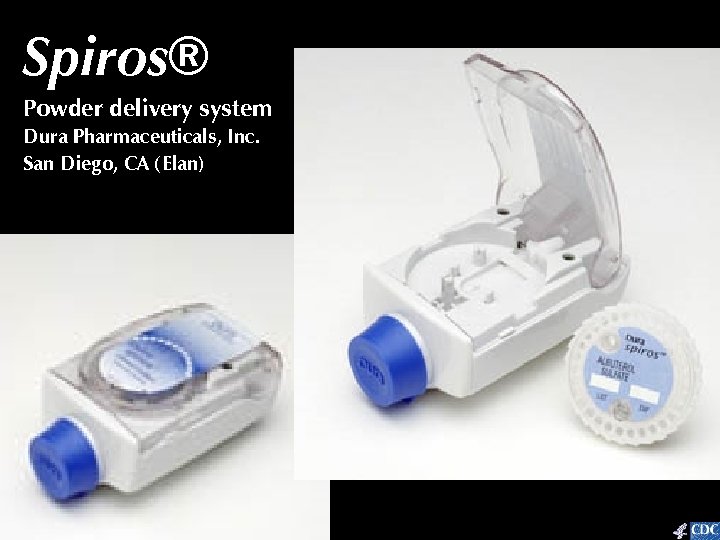 Spiros® Powder delivery system Dura Pharmaceuticals, Inc. San Diego, CA (Elan)
Spiros® Powder delivery system Dura Pharmaceuticals, Inc. San Diego, CA (Elan)
 Future (and Re-emerging) Vaccine Delivery Technologies
Future (and Re-emerging) Vaccine Delivery Technologies
 Oral vaccines
Oral vaccines
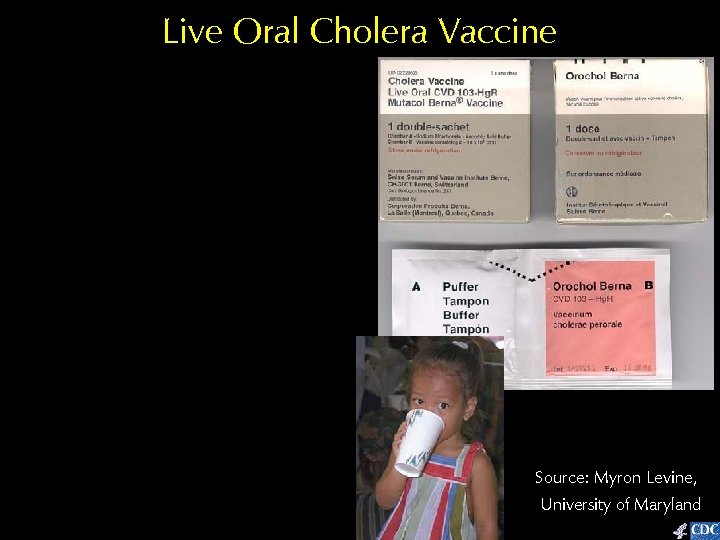 Live Oral Cholera Vaccine Source: Myron Levine, University of Maryland
Live Oral Cholera Vaccine Source: Myron Levine, University of Maryland
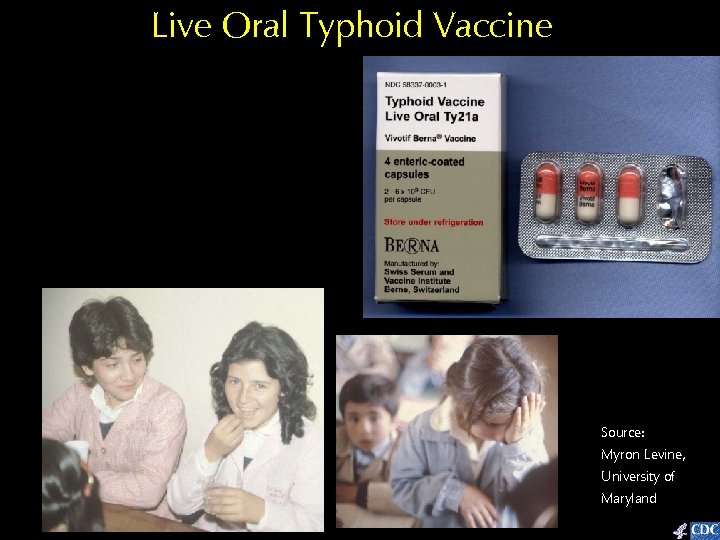 Live Oral Typhoid Vaccine Source: Myron Levine, University of Maryland
Live Oral Typhoid Vaccine Source: Myron Levine, University of Maryland
 Future (and Re-emerging) Vaccine Delivery Technologies Thank you.
Future (and Re-emerging) Vaccine Delivery Technologies Thank you.


