14fe41ae5758e3a3548bfe3677744553.ppt
- Количество слайдов: 56

Using Covalent Structures 19/03/2018 Form of carbon Property Uses Why? Carbon – diamond Very hard Drill tips Extremely strong covalent structure Graphite Soft, conducts electricity Lubricants and making electrodes Graphite is arranged in layers that can slide over each other and it contains free electrons.
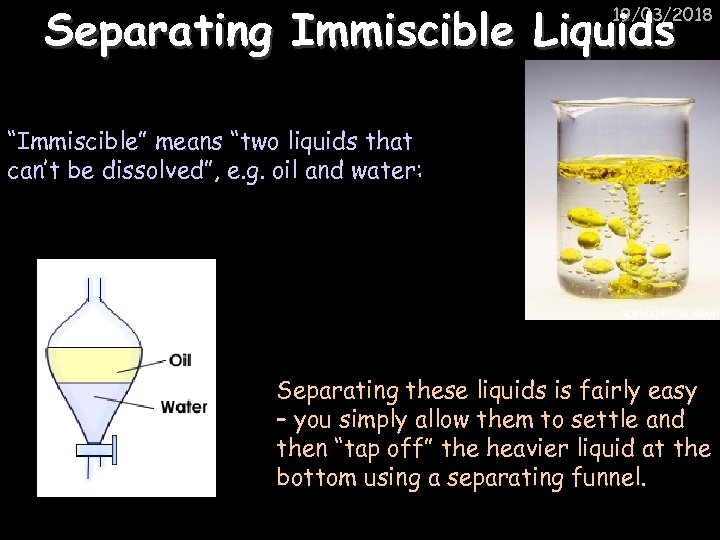
Separating Immiscible Liquids 19/03/2018 “Immiscible” means “two liquids that can’t be dissolved”, e. g. oil and water: Separating these liquids is fairly easy – you simply allow them to settle and then “tap off” the heavier liquid at the bottom using a separating funnel.
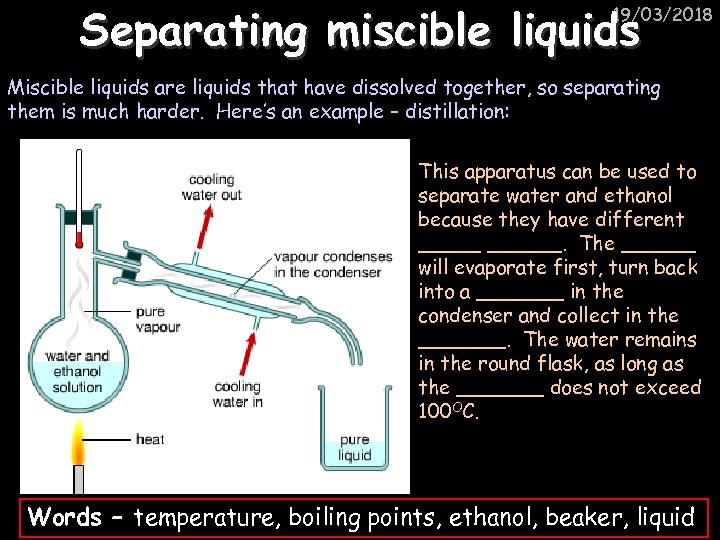
Separating miscible liquids 19/03/2018 Miscible liquids are liquids that have dissolved together, so separating them is much harder. Here’s an example – distillation: This apparatus can be used to separate water and ethanol because they have different ______. The ______ will evaporate first, turn back into a _______ in the condenser and collect in the _______. The water remains in the round flask, as long as the _______ does not exceed 100 OC. Words – temperature, boiling points, ethanol, beaker, liquid
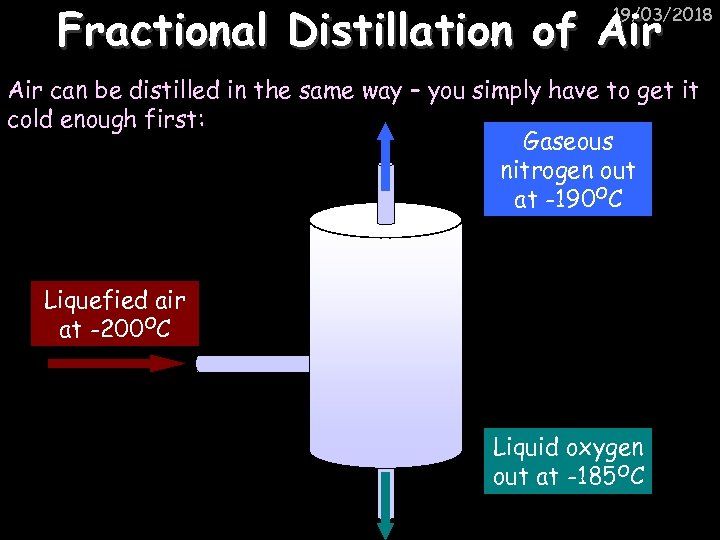
Fractional Distillation of Air 19/03/2018 Air can be distilled in the same way – you simply have to get it cold enough first: Gaseous nitrogen out at -190 OC Liquefied air at -200 OC Liquid oxygen out at -185 OC

Chromatography 19/03/2018 Chromatography can be used to separate a mixture of different inks. Some example questions… R G B X 1) Ink X contains two different colours. What are they? 1 2 3 2) Which ink is ink Z made out of? Z
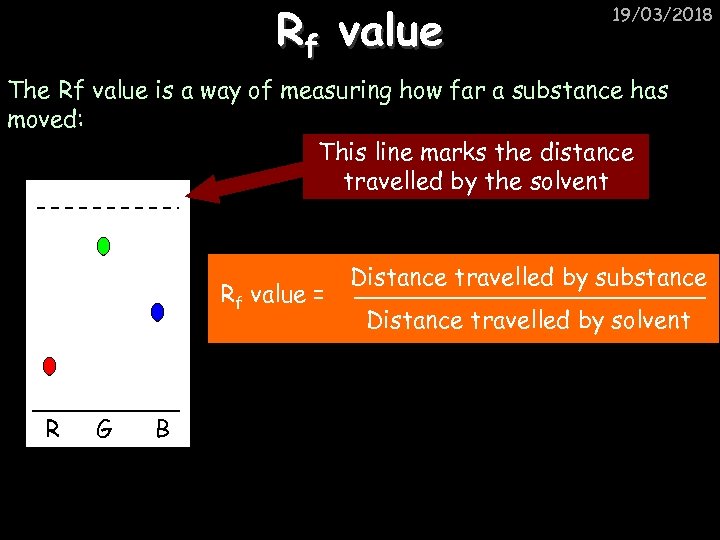
Rf value 19/03/2018 The Rf value is a way of measuring how far a substance has moved: This line marks the distance travelled by the solvent Rf value = R G B Distance travelled by substance Distance travelled by solvent
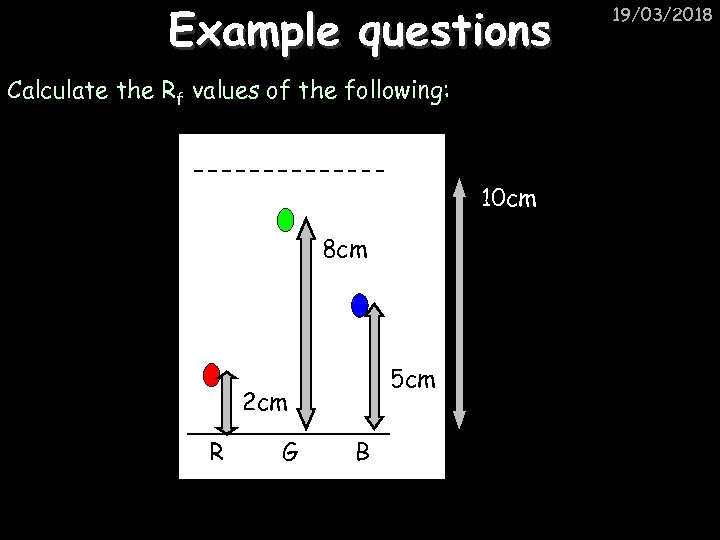
Example questions Calculate the Rf values of the following: 10 cm 8 cm 5 cm 2 cm R G B 19/03/2018
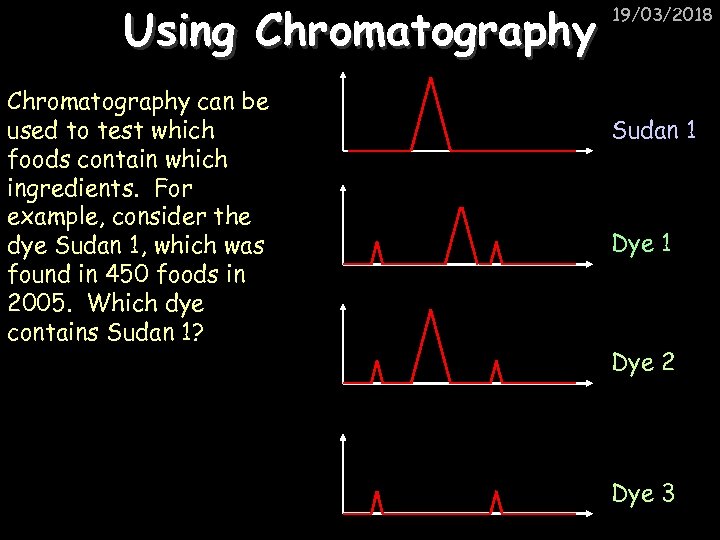
Using Chromatography can be used to test which foods contain which ingredients. For example, consider the dye Sudan 1, which was found in 450 foods in 2005. Which dye contains Sudan 1? 19/03/2018 Sudan 1 Dye 2 Dye 3

19/03/2018 Topic 4 – Groups in the Periodic Table
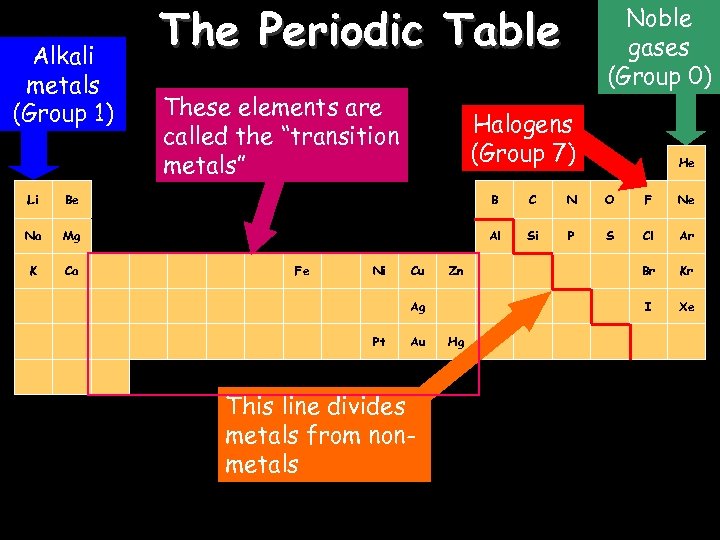
Alkali metals (Group 1) The Periodic Table These elements are called the “transition H metals” 19/03/2018 Noble gases (Group 0) Halogens (Group 7) He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Br Kr I Xe Fe Ni Cu Zn Ag Pt Au This line divides metals from nonmetals Hg
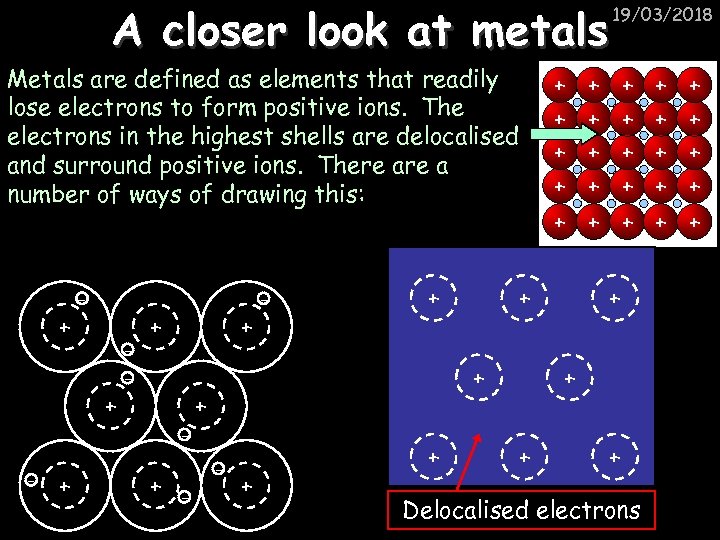
A closer look at metals Metals are defined as elements that readily lose electrons to form positive ions. The electrons in the highest shells are delocalised and surround positive ions. There a number of ways of drawing this: + - + + + + + + + + + - - + + 19/03/2018 + Delocalised electrons

Properties of metals 19/03/2018 Metals have very high melting points (which means that they are usually _____) whereas non-metals will melt at lower ______ All metals conduct heat and _____ very well, whereas non-metals don’t (usually) Metals are strong and ______ but bendable. Non-metals are usually _____ or they will snap. Metals will _____ when freshly cut or scratched, whereas non-metals are usually dull. Metals have higher _______ than non-metals (i. e. they weigh more) Metals can be used to make ______ (a mixture of different metals) Words - alloys, electricity, solids, weak, densities, temperatures, tough, shine
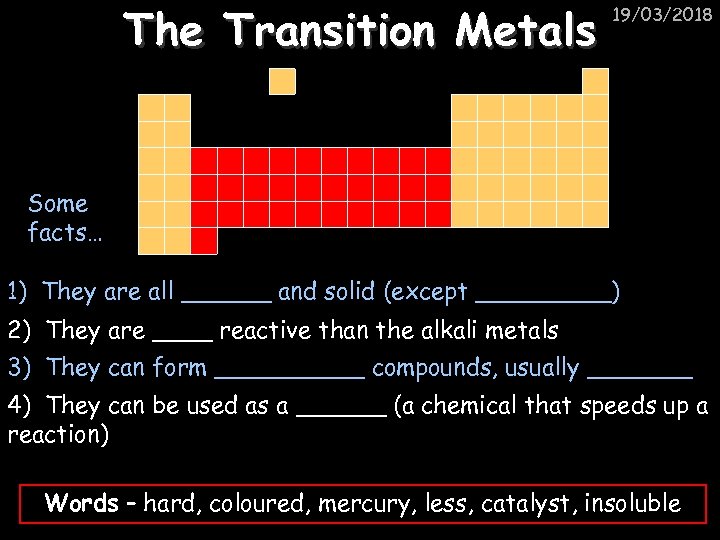
The Transition Metals 19/03/2018 Some facts… 1) They are all ______ and solid (except _____) 2) They are ____ reactive than the alkali metals 3) They can form _____ compounds, usually _______ 4) They can be used as a ______ (a chemical that speeds up a reaction) Words – hard, coloured, mercury, less, catalyst, insoluble
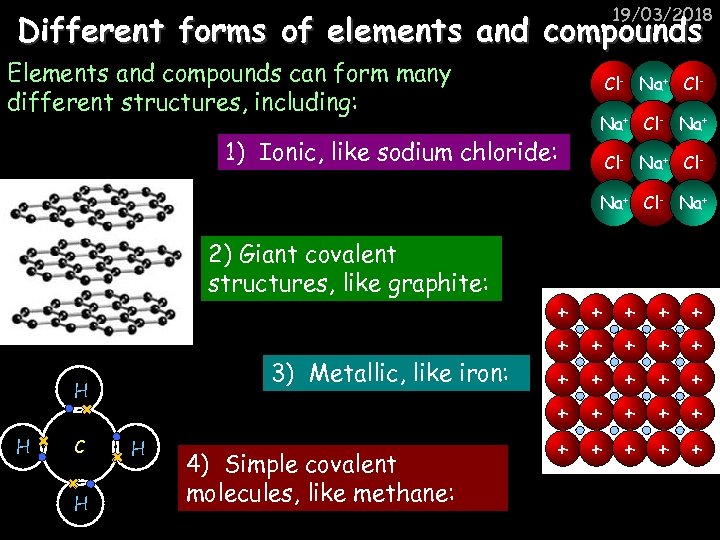
19/03/2018 Different forms of elements and compounds Elements and compounds can form many different structures, including: Cl- Na+ Cl- 1) Ionic, like sodium chloride: Na+ Cl- Na+ 2) Giant covalent structures, like graphite: 3) Metallic, like iron: H H C H H 4) Simple covalent molecules, like methane: + + + + + + +
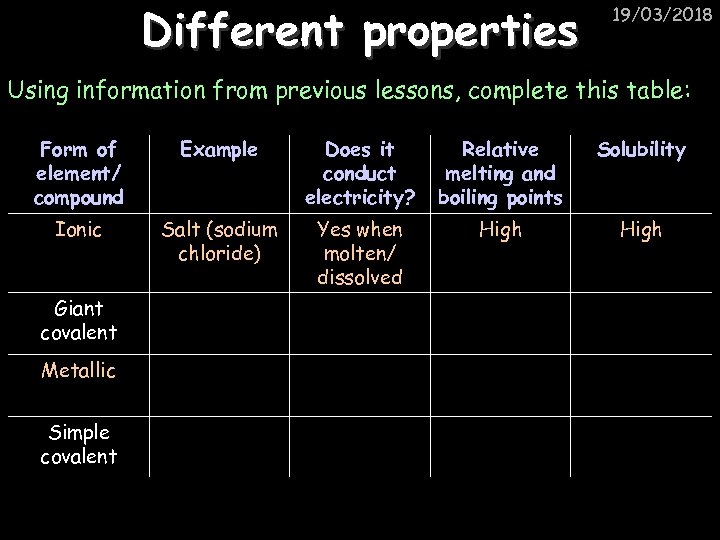
Different properties 19/03/2018 Using information from previous lessons, complete this table: Form of element/ compound Example Does it conduct electricity? Relative melting and boiling points Solubility Ionic Salt (sodium chloride) Yes when molten/ dissolved High Giant covalent Metallic Simple covalent
Group 1 – The alkali metals 19/03/2018 Li Na K Rb Cs Fr Watch video of these metals reacting with water (from Sky One’s Brainiac)

Group 1 – The alkali metals 19/03/2018 Some facts… 1) These metals all have ___ electron in their outer shell. 2) Density increases as you go down the group, while melting point ____ 2) Reactivity increases as you go _______ the group. This is because the electrons are further away from the _______ every time a _____ is added, so they are given up more easily. 3) They all react with water to form an alkali (hence their name) and _____, e. g: Potassium + water 2 K(s) + 2 H 2 O(l) potassium hydroxide + hydrogen 2 KOH(aq) + H 2(g) Words – down, one, shell, hydrogen, nucleus, decreases
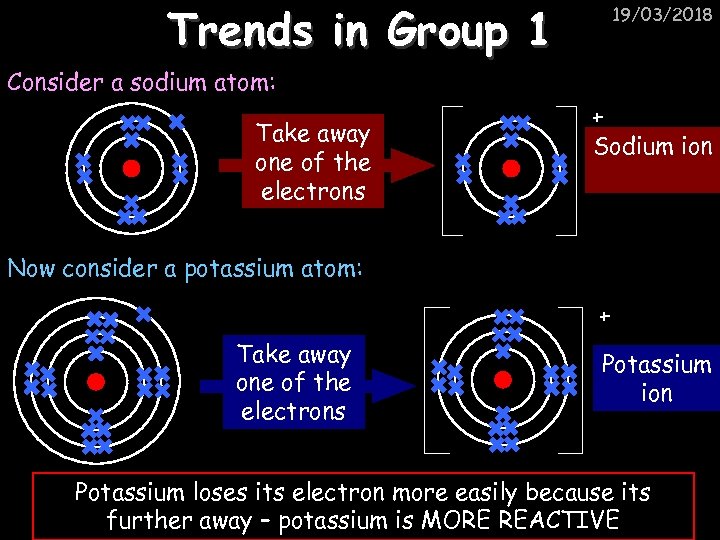
Trends in Group 1 19/03/2018 Consider a sodium atom: Take away one of the electrons + Sodium ion Now consider a potassium atom: + Take away one of the electrons Potassium ion Potassium loses its electron more easily because its further away – potassium is MORE REACTIVE
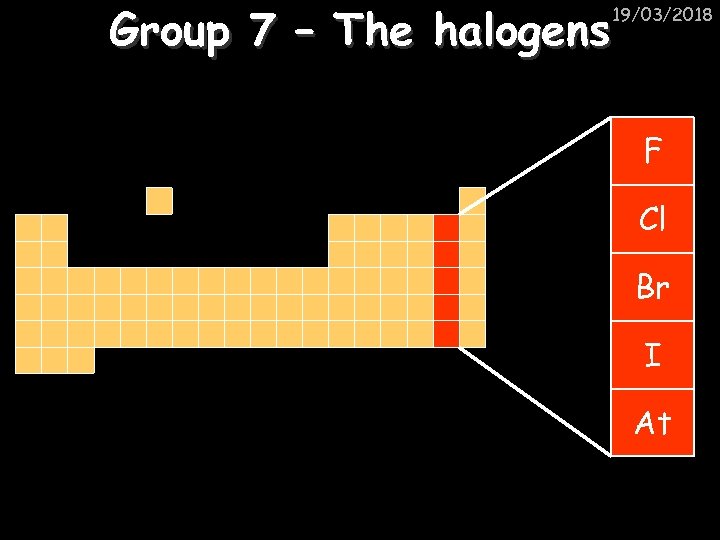
Group 7 – The halogens 19/03/2018 F Cl Br I At
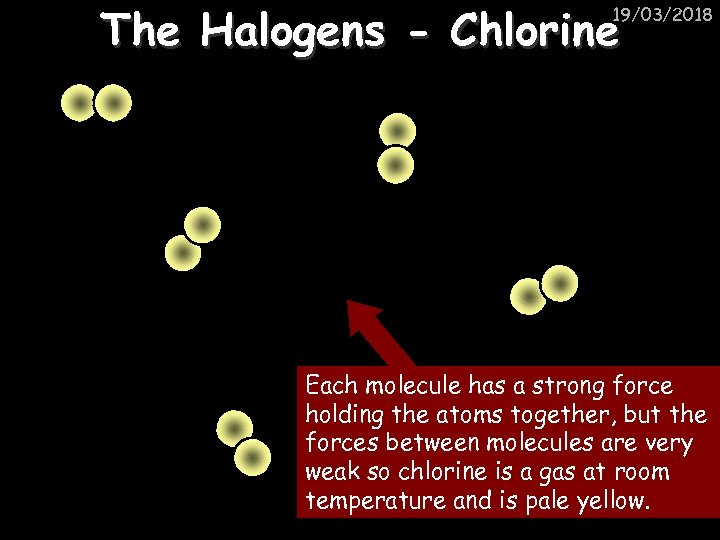
The Halogens - Chlorine 19/03/2018 Each molecule has a strong force holding the atoms together, but the forces between molecules are very weak so chlorine is a gas at room temperature and is pale yellow.
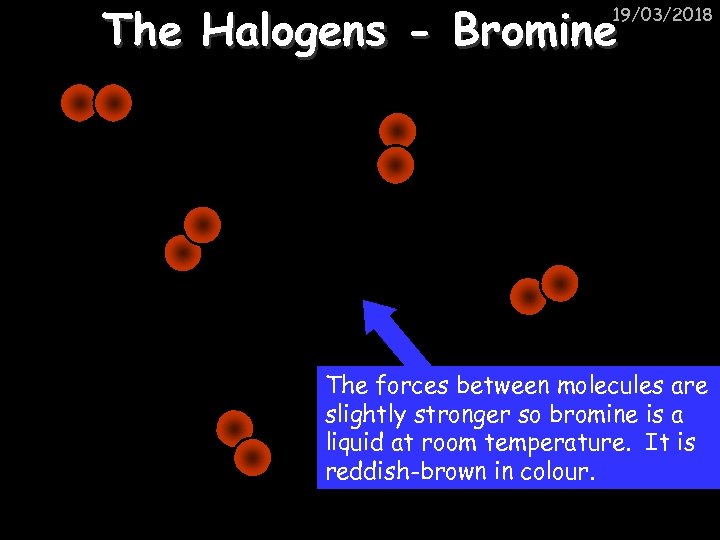
The Halogens - Bromine 19/03/2018 The forces between molecules are slightly stronger so bromine is a liquid at room temperature. It is reddish-brown in colour.

The Halogens - Iodine 19/03/2018 Iodine is a solid at room temperature but with gentle heating it will melt. The atoms will remain in pairs. In solid form iodine is grey like metal but gaseous iodine is purple.
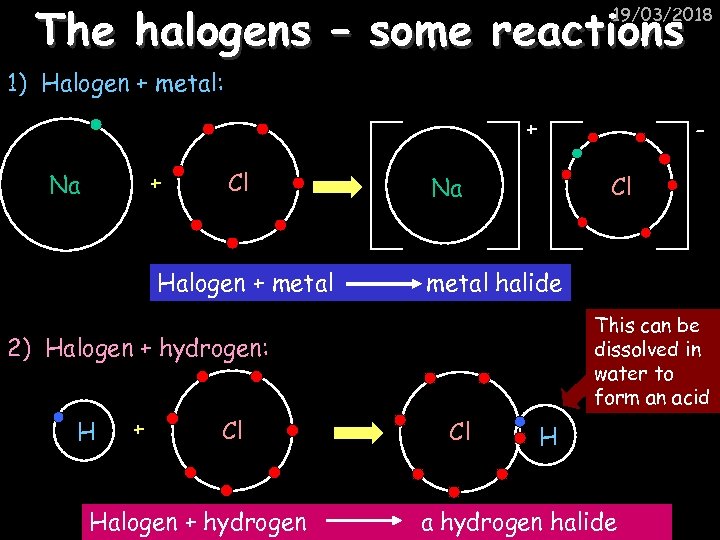
The halogens – some reactions 19/03/2018 1) Halogen + metal: + + Na Cl Halogen + metal Cl Na metal halide This can be dissolved in water to form an acid 2) Halogen + hydrogen: H + Cl Halogen + hydrogen Cl H a hydrogen halide
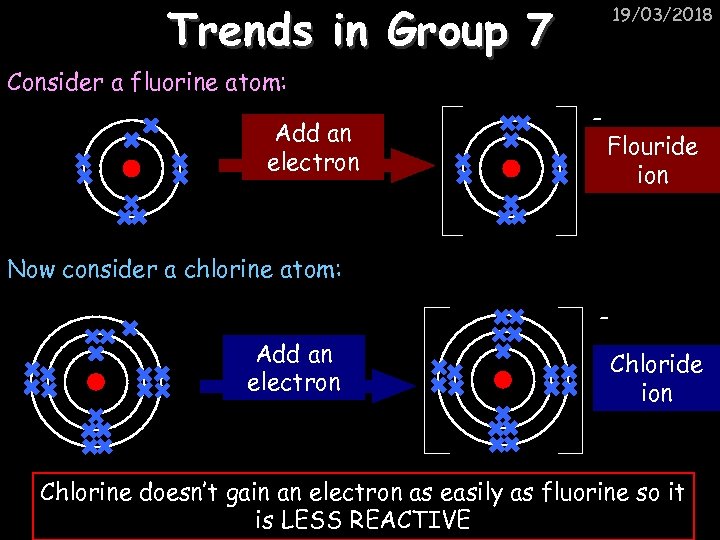
Trends in Group 7 19/03/2018 Consider a fluorine atom: Add an electron - Flouride ion Now consider a chlorine atom: Add an electron Chloride ion Chlorine doesn’t gain an electron as easily as fluorine so it is LESS REACTIVE
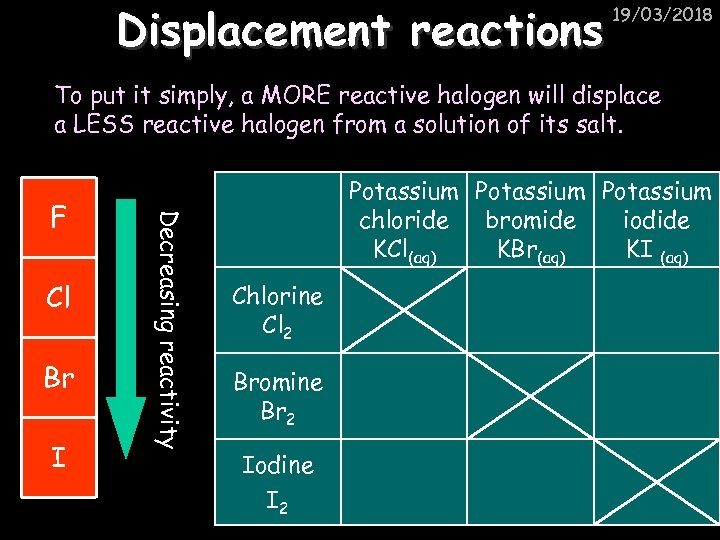
Displacement reactions 19/03/2018 To put it simply, a MORE reactive halogen will displace a LESS reactive halogen from a solution of its salt. Cl Br I Decreasing reactivity F Potassium chloride bromide iodide KCl(aq) KBr(aq) KI (aq) Chlorine Cl 2 Bromine Br 2 Iodine I 2
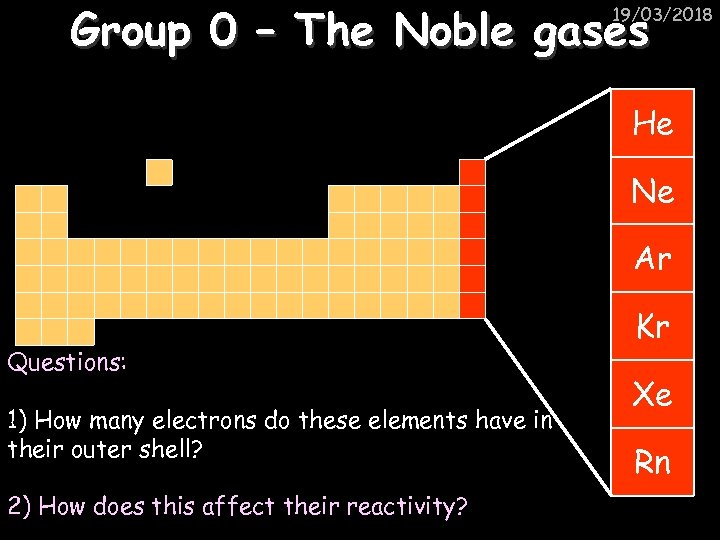
Group 0 – The Noble gases 19/03/2018 He Ne Ar Questions: 1) How many electrons do these elements have in their outer shell? 2) How does this affect their reactivity? Kr Xe Rn

19/03/2018 How the Noble Gases were discovered A while ago we discussed the idea that Mendeleev left gaps in the periodic table to account for undiscovered elements: Li Be B C N O F Na Mg Al I discovered most of the Noble Gases around the year 1900. I did this by: 1) Noticing the density of nitrogen made in certain reactions differed from the density of nitrogen in air 2) I then developed a hypothesis about the existence of undiscovered elements Sir William Ramsay, 1852 -1916 3) I then tested this hypothesis with further experiments. Ramsay’s blue plaque outside a house in Notting Hill

Group 0 – The Noble gases 19/03/2018 Some facts… 1) All of the noble gases have a full outer shell, so they are very ______ 2) They all have _____ melting and boiling points and are inflammable 3) They exist as single atoms rather then _____ molecules 4) Helium is ____ then air and is used in balloons and airships (as well as for talking in a silly voice) 5) Argon is used in light bulbs (because it is so unreactive) and argon , krypton and ____ are used in fancy lights Words – neon, stable, low, diatomic, lighter
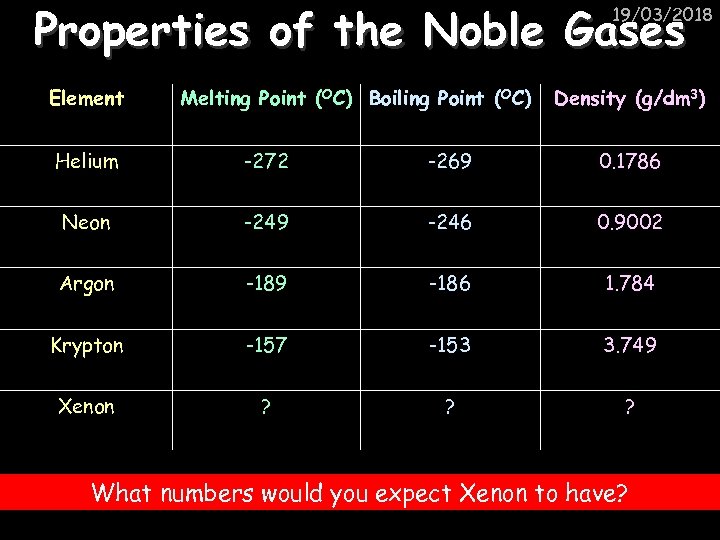
Properties of the Noble Gases 19/03/2018 Element Melting Point (OC) Boiling Point (OC) Density (g/dm 3) Helium -272 -269 0. 1786 Neon -249 -246 0. 9002 Argon -189 -186 1. 784 Krypton -157 -153 3. 749 Xenon ? ? ? What numbers would you expect Xenon to have?

Topic 5 – Chemical Reactions 19/03/2018

19/03/2018 Endothermic and exothermic reactions Step 1: Energy must be SUPPLIED to break bonds: Step 2: Energy is RELEASED when new bonds are made: Energy A reaction is EXOTHERMIC if more energy is RELEASED then SUPPLIED. If more energy is SUPPLIED then is RELEASED then the reaction is ENDOTHERMIC

19/03/2018 Common examples of these reactions Are these reactions exothermic or endothermic? Burning Photosynthesis Cooling packs Hand warmer packs
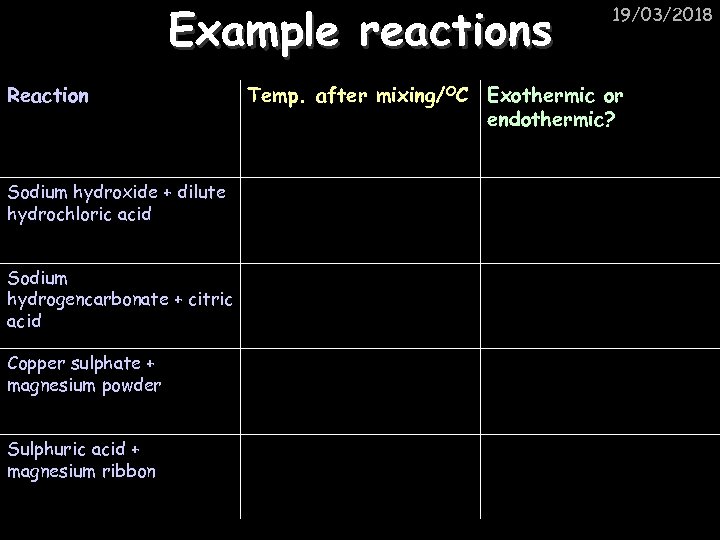
Example reactions Reaction Sodium hydroxide + dilute hydrochloric acid Sodium hydrogencarbonate + citric acid Copper sulphate + magnesium powder Sulphuric acid + magnesium ribbon 19/03/2018 Temp. after mixing/OC Exothermic or endothermic?
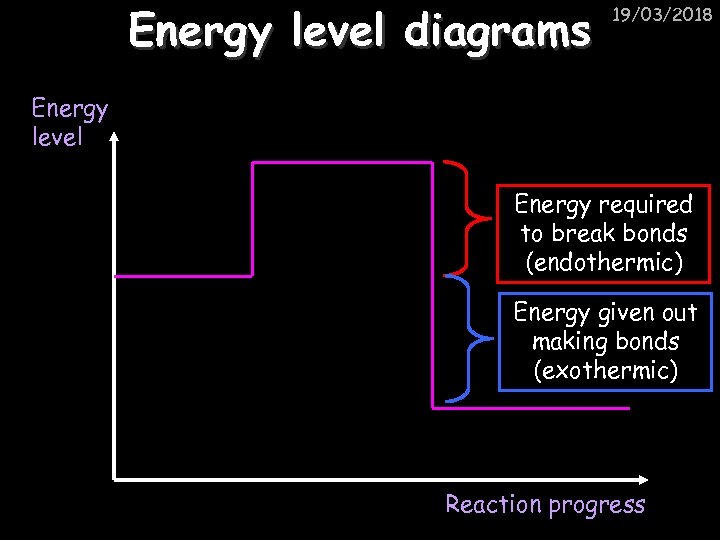
Energy level diagrams 19/03/2018 Energy level Energy required to break bonds (endothermic) Energy given out making bonds (exothermic) Reaction progress

Exothermic vs endothermic: 19/03/2018 EXOTHERMIC – more energy is given out than is taken in (e. g. burning, respiration) ENDOTHERMIC – energy is taken in but not necessarily given out (e. g. photosynthesis)

Rates of Reaction 19/03/2018 Oh no! Here comes another one and it’s got more energy… Here comes another one. Look at how slow it’s going… It missed! Here comes an acid particle… No effect! It didn’t have enough energy! Hi. I’m Mike Marble. I’m about to have some acid poured onto me. Let’s see what happens…

Measuring the Rate of Reaction 19/03/2018 Two common methods:

Rates of Reaction 19/03/2018 Chemical reactions occur when different atoms or molecules _____ with each other but they HAVE to collide with enough _______. Basically, the more collisions we get and the more energetic they are the _______ the reaction goes. The rate at which the reaction happens depends on four things: 1) The _______ of the reactants, 2) Their concentration 3) Their surface area 4) The ______ the reactants are under Words – energy, quicker, pressure, temperature, collide

Catalysts Summary 19/03/2018 Catalysts are used to ____ __ a reaction to increase the rate at which a product is made or to make a process ____. They are not normally ___ __ in a reaction. Cars use catalytic converters to remove unwanted gases. They take gases like carbon _______ and react them with water to form carbon dioxide. They have a large _____ and work best at high temperatures. Carbon monoxide + oxygen carbon dioxide Words – surface area, speed up, used up, cheaper, monoxide

Topic 6 – Quantitative Chemistry 19/03/2018

Mass and atomic number revision 19/03/2018 Particle Relative Mass Relative Charge Proton 1 +1 Neutron 1 0 Electron Very small -1 MASS NUMBER = number of protons + number of neutrons SYMBOL PROTON NUMBER = number of protons (obviously)

Relative formula mass, Mr 19/03/2018 The relative formula mass of a compound is the relative atomic masses of all the elements in the compound added together. E. g. water H 2 O: Relative atomic mass of O = 16 Relative atomic mass of H = 1 Therefore Mr for water = 16 + (2 x 1) = 18 Work out Mr for the following compounds: 1) HCl H=1, Cl=35 so Mr = 36 2) Na. OH Na=23, O=16, H=1 so Mr = 40 3) Mg. Cl 2 Mg=24, Cl=35 so Mr = 24+(2 x 35) = 94 4) H 2 SO 4 H=1, S=32, O=16 so Mr = (2 x 1)+32+(4 x 16) = 98 5) K 2 CO 3 K=39, C=12, O=16 so Mr = (2 x 39)+12+(3 x 16) = 138

Empirical formulae 19/03/2018 Empirical formulae is simply a way of showing how many atoms are in a molecule (like a chemical formula). For example, Ca. O, Ca. CO 3, H 20 and KMn. O 4 are all empirical formulae. Here’s how to work them out: A classic exam question: Find the simplest formula of 2. 24 g of iron reacting with 0. 96 g of oxygen. Step 1: Divide both masses by the relative atomic mass: For iron 2. 24/56 = 0. 04 For oxygen 0. 96/16 = 0. 06 Step 2: Write this as a ratio and simplify: 0. 04: 0. 06 is equivalent to 2: 3 Step 3: Write the formula: 2 iron atoms for 3 oxygen atoms means the formula is Fe 2 O 3

Example questions 19/03/2018 1) Find the empirical formula of magnesium oxide which contains 48 g of magnesium and 32 g of oxygen. Mg. O 2) Find the empirical formula of a compound that contains 42 g of nitrogen and 9 g of hydrogen. NH 3 3) Find the empirical formula of a compound containing 20 g of calcium, 6 g of carbon and 24 g of oxygen. Ca. CO 3

Calculating percentage mass 19/03/2018 If you can work out Mr then this bit is easy… Percentage mass (%) = Mass of element Ar Relative formula mass Mr x 100% Calculate the percentage mass of magnesium in magnesium oxide, Mg. O: Ar for magnesium = 24 Ar for oxygen = 16 Mr for magnesium oxide = 24 + 16 = 40 Therefore percentage mass = 24/40 x 100% = 60% Calculate the percentage mass of the following: 1) Hydrogen in hydrochloric acid, HCl 3% 2) Potassium in potassium chloride, KCl 52% 3) Calcium in calcium chloride, Ca. Cl 2 36% 4) Oxygen in water, H 2 O 89%

Recap questions 19/03/2018 Work out the relative formula mass of: 1) Carbon dioxide CO 2 44 2) Calcium oxide Ca. O 56 3) Methane CH 4 16 Work out the percentage mass of: 1) Carbon in carbon dioxide CO 2 27% 2) Calcium in calcium oxide Ca. O 71% 3) Hydrogen in methane CH 4 25%
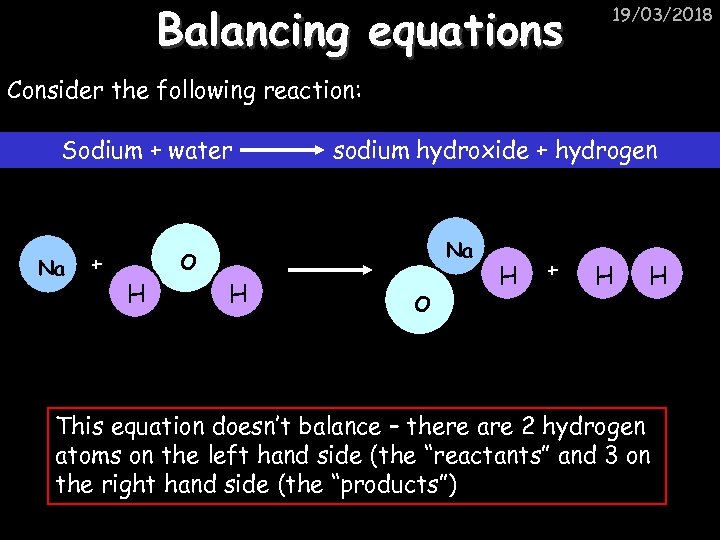
Balancing equations 19/03/2018 Consider the following reaction: Sodium + water Na + sodium hydroxide + hydrogen Na O H H O H + H H This equation doesn’t balance – there are 2 hydrogen atoms on the left hand side (the “reactants” and 3 on the right hand side (the “products”)
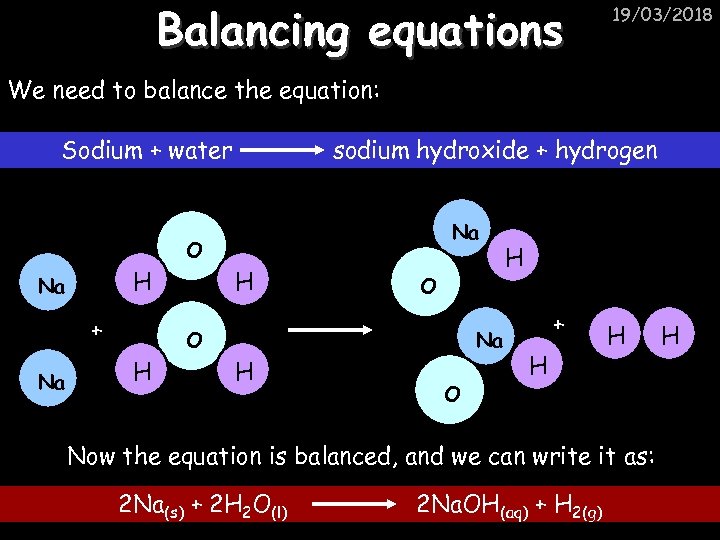
Balancing equations 19/03/2018 We need to balance the equation: Sodium + water sodium hydroxide + hydrogen Na O H Na + Na H O O H Na H O H + H H Now the equation is balanced, and we can write it as: 2 Na(s) + 2 H 2 O(l) 2 Na. OH(aq) + H 2(g) H
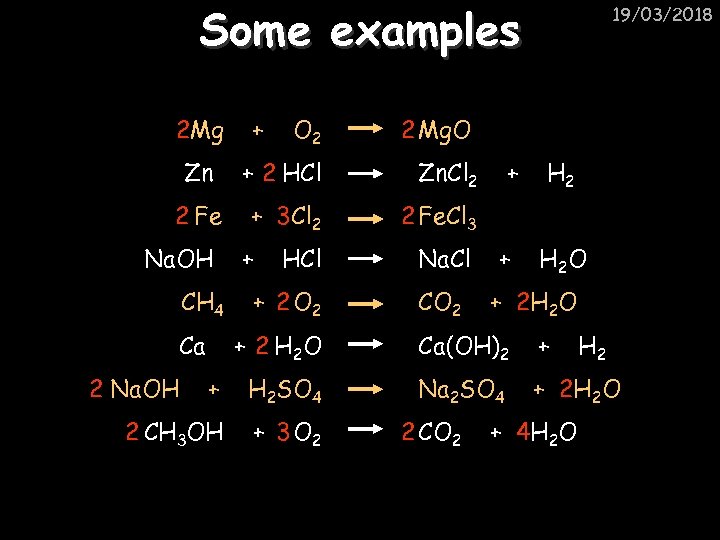
Some examples 2 Mg O 2 2 Mg. O Zn + 2 HCl Zn. Cl 2 2 Fe + 3 Cl 2 2 Fe. Cl 3 Na. OH CH 4 Ca + 19/03/2018 + HCl + 2 O 2 Na. Cl CO 2 + + H 2 O + 2 H 2 O + 2 H 2 O Ca(OH)2 + + H 2 SO 4 Na 2 SO 4 + 2 H 2 O 2 CH 3 OH + 3 O 2 2 Na. OH 2 CO 2 + 4 H 2 O H 2

19/03/2018 Calculating the mass of a product E. g. what mass of magnesium oxide is produced when 60 g of magnesium is burned in air? Step 1: READ the equation: 2 Mg + O 2 2 Mg. O IGNORE the oxygen in step 2 – the question doesn’t ask for it Step 2: WORK OUT the relative formula masses (Mr): 2 Mg = 2 x 24 = 48 2 Mg. O = 2 x (24+16) = 80 Step 3: LEARN and APPLY the following 3 points: 1) 48 g of Mg makes 80 g of Mg. O 2) 1 g of Mg makes 80/48 = 1. 66 g of Mg. O 3) 60 g of Mg makes 1. 66 x 60 = 100 g of Mg. O

1) When water is electrolysed it breaks down into hydrogen and 19/03/2018 oxygen: 2 H 2 O 2 H 2 + O 2 What mass of hydrogen is produced by the electrolysis of 6 g of water? Work out Mr: 2 H 2 O = 2 x ((2 x 1)+16) = 36 2 H 2 = 2 x 2 = 4 1. 36 g of water produces 4 g of hydrogen 2. So 1 g of water produces 4/36 = 0. 11 g of hydrogen 3. 6 g of water will produce (4/36) x 6 = 0. 66 g of hydrogen 2) What mass of calcium oxide is produced when 10 g of calcium burns? 2 Ca + O 2 Mr: 2 Ca = 2 x 40 = 80 2 Ca. O = 2 x (40+16) = 112 80 g produces 112 g so 10 g produces (112/80) x 10 = 14 g of Ca. O 3) What mass of aluminium is produced from 100 g of aluminium oxide? 2 Al 2 O 3 4 Al + 3 O 2 Mr: 2 Al 2 O 3 = 2 x((2 x 27)+(3 x 16)) = 204 4 Al = 4 x 27 = 108 204 g produces 108 g so 100 g produces (108/204) x 100 = 52. 9 g of Al 2 O 3

Another method 19/03/2018 Try using this equation: Mass of product IN GRAMMES Mass of reactant IN GRAMMES Mr of product Mr of reactant Q. When water is electrolysed it breaks down into hydrogen and oxygen: 2 H 2 O 2 H 2 + O 2 What mass of hydrogen is produced by the electrolysis of 6 g of water? Mass of product IN GRAMMES 6 g 4 36 So mass of product = (4/36) x 6 g = 0. 66 g of hydrogen

Problems with this technique 19/03/2018 Calculating the amount of a product may not always give you a reliable answer. . . 1) The reaction may not have completely _______ 2) The reaction may have been _______ 3) Some of the product may have been ____ 4) Some of the reactants may have produced other _______ The amount of product that is made is called the “____”. This number can be compared to the maximum theoretical amount as a percentage, called the “percentage yield”. Words – lost, yield, finished, reversible, products

Percentage Yield 19/03/2018 Theoretical yield = the amount of product that should be made as calculated from the masses of atoms Actual yield = what was actually produced in a reaction Percentage yield = actual yield (in g) theoretical yield Example question: 65 g of zinc reacts with 73 g of hydrochloric acid and produces 102 g of zinc chloride. What is the percentage yield? Zn + 2 HCl Zn. Cl 2 + H 2 The theoretical yield is 136 g (using Cl = 35. 5) so the % yield is 75%

Percentage yield = Actual yield Predicted yield 19/03/2018 X 100% Some example questions: 1) The predicted yield of an experiment to make salt was 10 g. If 7 g was made what is the percentage yield? 70% 2) Dave is trying to make water. If he predicts to make 15 g but only makes 2 g what is the percentage yield? 13% 3) Sarah performs an experiment and has a percentage yield of 33%. If she made 50 g what was she predicted to make? 150 g

Chemical Economics 19/03/2018 Hi. We’re industrial scientists and we want to make lots of chemicals and sell them to make money. What problems would we face? Possible problems with making chemicals: Therefore we need reactions and processes that give us a high percentage yield where all of the products are useful and the reactions happen 1) Reactions often produce chemicals that aren’t commercially useful or that can’t be sold 2) Reactions can also produce chemicals that present environmental and social problems.
14fe41ae5758e3a3548bfe3677744553.ppt