396be5df846f89190b7642144ae2055c.ppt
- Количество слайдов: 18

Use of PMCA for Biochemical Diagnosis of Prion Diseases Claudio Soto, Ph. D Dept of Neurology, University of Texas Medical Branch and Amprion Inc.
Importance of Prion Diagnosis Food industry Blood banks Disease diagnosis Brain surgery Soto (2005) Nature Rev Microbiol. 2: 809 -819 Plasma products Clinical trials Drugs from human origin Organ transplant

The problem of Prion Diagnosis Pr. Psc Is the most specific marker for the disease. However, its levels in body fluids and tissues other than nervous system are too low to be detected Design a more sensitive test for Pr. Psc detection or Amplify the level of the marker (Pr. PSc)
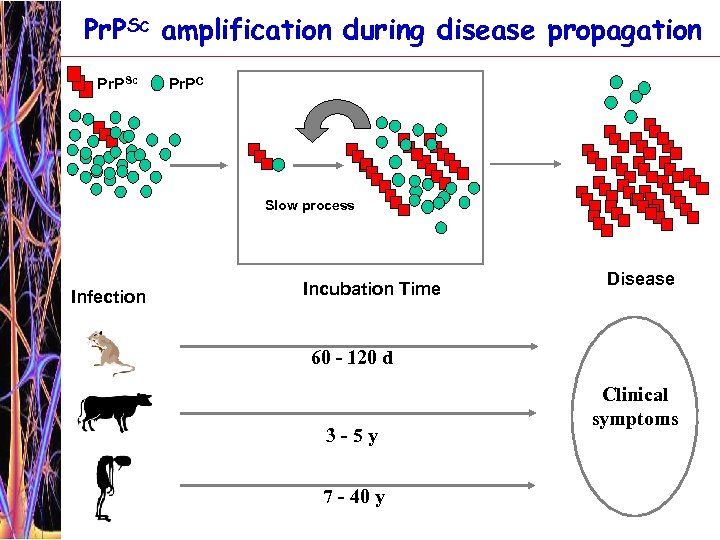
Pr. PSc amplification during disease propagation Pr. PSc Pr. PC Slow process Infection Incubation Time Disease 60 - 120 d 3 -5 y 7 - 40 y Clinical symptoms
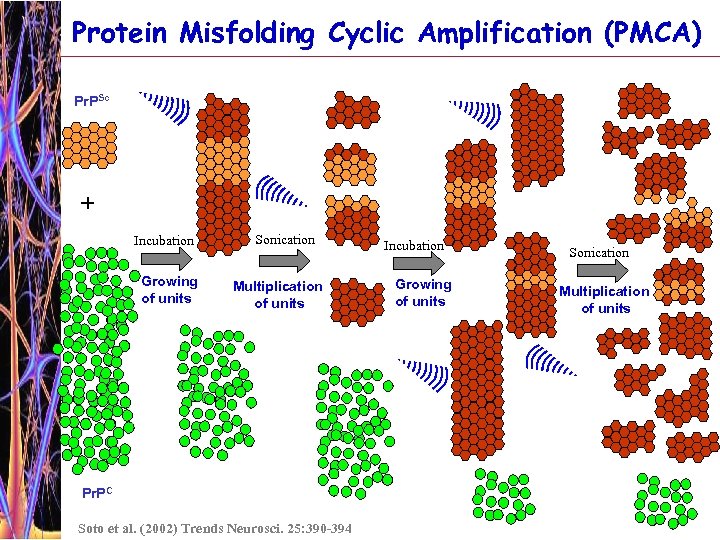
Protein Misfolding Cyclic Amplification (PMCA) Pr. PSc + Incubation Growing of units Sonication Multiplication of units Pr. PC Soto et al. (2002) Trends Neurosci. 25: 390 -394 Incubation Growing of units Sonication Multiplication of units
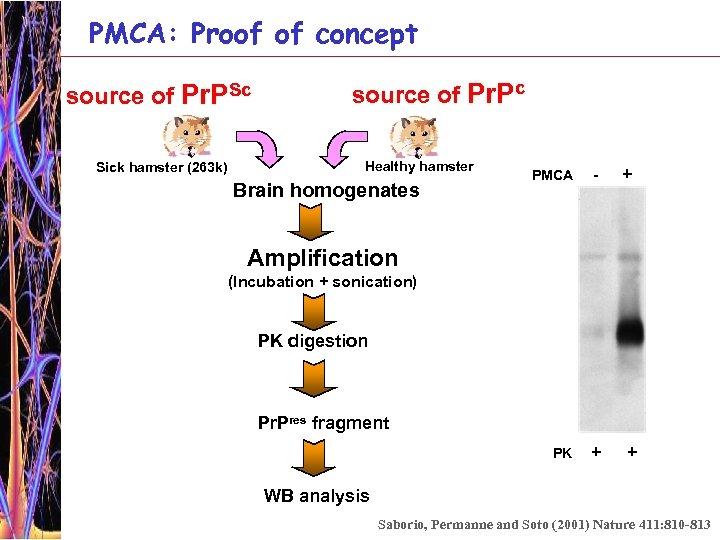
PMCA: Proof of concept source of Pr. PSc Sick hamster (263 k) source of Pr. Pc Healthy hamster Brain homogenates PMCA - + Amplification (Incubation + sonication) PK digestion Pr. Pres fragment PK + + WB analysis Saborio, Permanne and Soto (2001) Nature 411: 810 -813

Automated PMCA Non Amplified l ro nt co at rm fo 96 On e- by -o ne Amplified
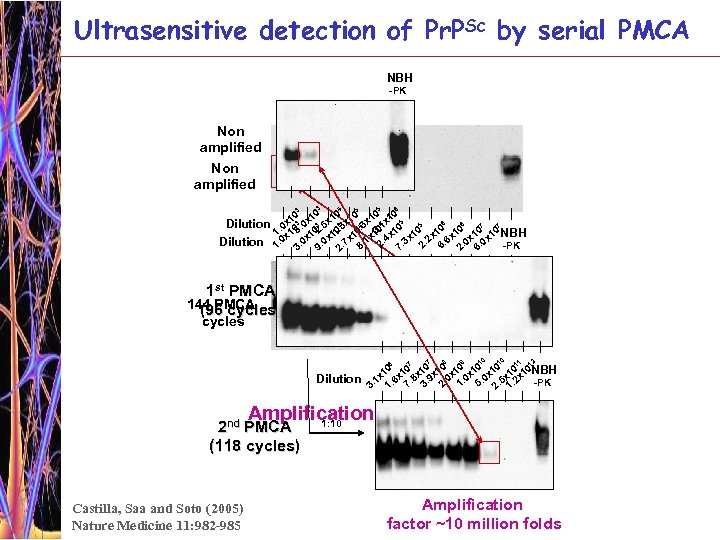
Ultrasensitive detection of Pr. PSc by serial PMCA NBH -PK Non amplified 3 Dilution 4 5 6 3 5 0 10 x 10 4 x 10 5 6 6 5 3 7 7 4 0 x 3. 0 3 5 0 3 3. 1 1. x 105 102. 11. 16. x 10 10 10 NBH 0. 0 x. 7 x. 1 2. 4. 3 x. 2. 6. 0 x 2 6 1. 3 -PK 8 7 9 2 1 st PMCA 144 PMCA ) (96 cycles 6 Dilution 7 8 9 10 10 1 2 7 1 1 0 10 10 10 0 0 NBH x 6 x 1. 8 x. 9 x. 0 x. 0 x 5 x 1 2 x 1 1 -PK 3. 1. 7 3 2 1 5 2. 1. 10 Amplification factor ~ 6600 fold 1: 10 2 nd PMCA (118 cycles) Castilla, Saa and Soto (2005) Nature Medicine 11: 982 -985 Amplification factor ~10 million folds

What is the minimum quantity detected? Scrapie LD 50 spiked 100000 1000 10 1 0 Non amplified 4 th PMCA (144 cycles) 1 st PMCA (144 cycles) 01 00 1 0 01 1 0. 0 2 nd PMCA (144 cycles) 0. 0 0. 1 Scrapie LD 50 spiked 7 th PMCA 3 rd PMCA (144 cycles) Our mathematical estimation is that 0. 001 LD 50 contains 4 th PMCA (144 cycles) around 20 -50 molecules of Pr. P monomer Saa, Castilla, and Soto (2006) J. Biol. Chem (In press) NBH -PK
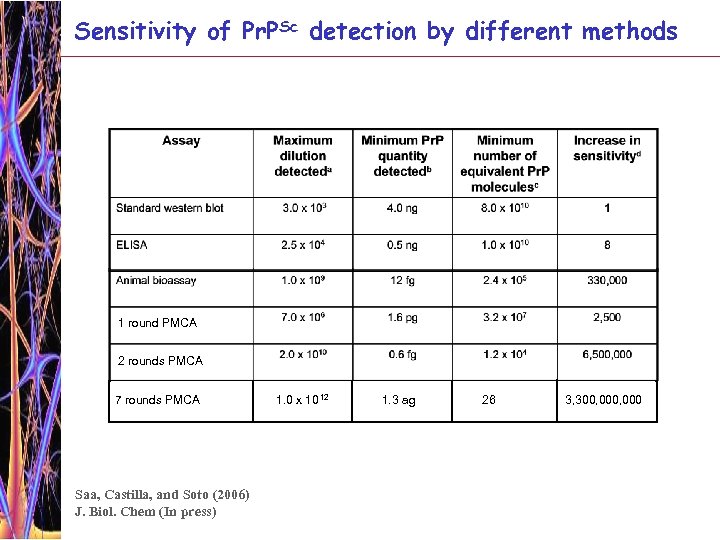
Sensitivity of Pr. PSc detection by different methods 1 round PMCA 2 rounds PMCA 7 rounds PMCA Saa, Castilla, and Soto (2006) J. Biol. Chem (In press) 1. 0 x 10 12 1. 3 ag 26 3, 300, 000

Can we detect Pr. PSc in blood? How much Pr. PSc is there in blood? Symptomatic phase Plasma Buffy coat 0. 1 pg/ml = 2 x 107 molecules 1 pg/ml = 2 x 108 molecules Incubation period (pre-symptomatic phase) Plasma 0. 005 -0. 01 pg/ml = 1 -2 x 106 molecules Buffy coat 0. 05 -0. 1 pg/ml = 1 -2 x 107 molecules Taken from Brown et al (2001) J. Lab Clin. Invest. 137: 5 -13 In other words sensitivity of Pr. PSc detection as compared with standard western blot has to be increase by 100, 000 – 1, 000 fold Our sensitivity Single PMCA: 65 pg/ml = 1. 3 x 109 molecules/ml Double PMCA: 0. 02 pg/ml = 4 x 105 molecules/ml 7 th rounds PMCA: 0. 015 fg/ml = 500 molecules/ml
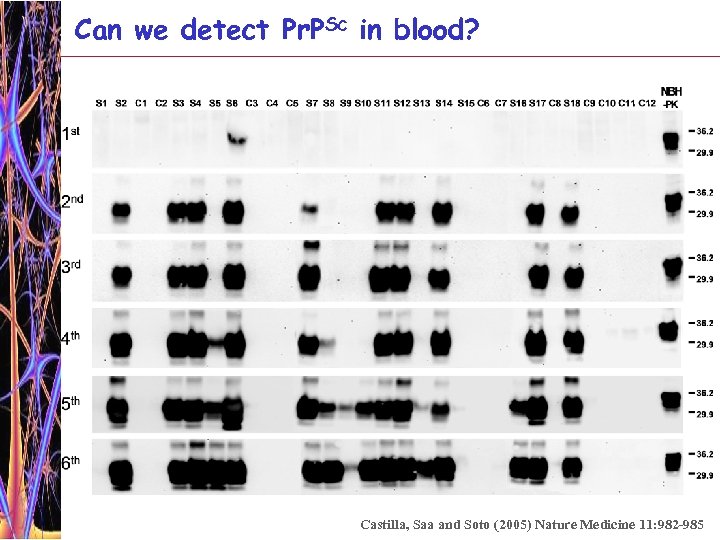
Can we detect Pr. PSc in blood? Castilla, Saa and Soto (2005) Nature Medicine 11: 982 -985
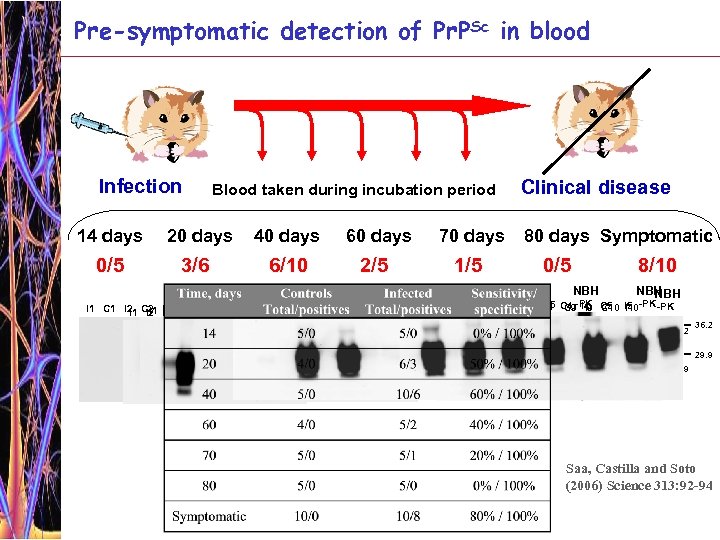
Pre-symptomatic detection of Pr. PSc in blood Infection Blood taken during incubation period 14 days 20 days 40 days 60 days 70 days 0/5 3/6 6/10 2/5 1/5 NBH NBH Clinical disease 80 days Symptomatic 0/5 8/10 NBH NBH -PK C 1 C 2 C 3 I 5 I 4 C 5 I 2 C 6 I 3 C 7 I 7 C 5 I 1 C 1 I 2 C 2 I 3 I 4 I 3 I 5 I 4 I 6 I 5 C 1 C 1 C 5 C 2 I 1 I 2 C 3 I 3 C 2 C 3 C 4 I 1 C 2 C 3 I 5 C 1 I 1 C 2 I 2 C 3 I 3 C 4 I 4 C 5 I 5 -PK C 4 I 6 I 5 C 2 I 1 -PK-PK C 1 I 3 I 4 C 1 C 5 -PK C 4 -PK I 6 C 4 I 4 C 8 I 8 C 9 I 9 C 10 I 1 I 2 I 7 C 3 I 9 I 2 I 8 C 4 I 10 I 1 I 2 I 3 C 3 36. 2 29. 9 36. 2 29. 9 Saa, Castilla and Soto (2006) Science 313: 92 -94
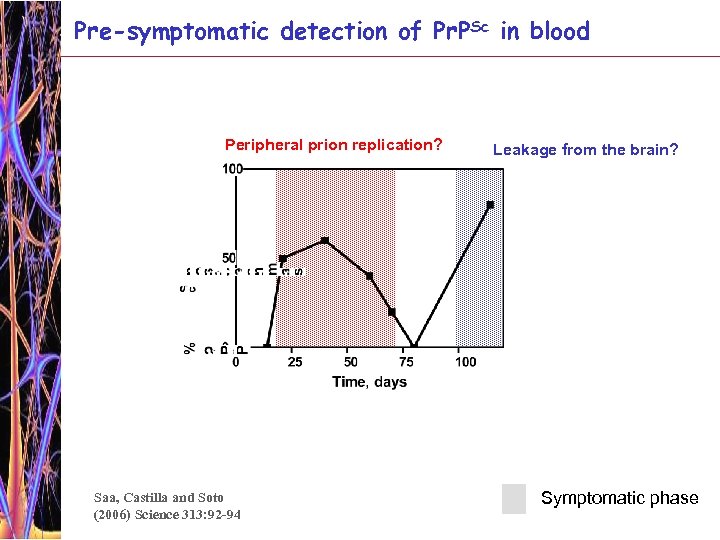
Pre-symptomatic detection of Pr. PSc in blood Peripheral prion replication? Saa, Castilla and Soto (2006) Science 313: 92 -94 Leakage from the brain? Symptomatic phase
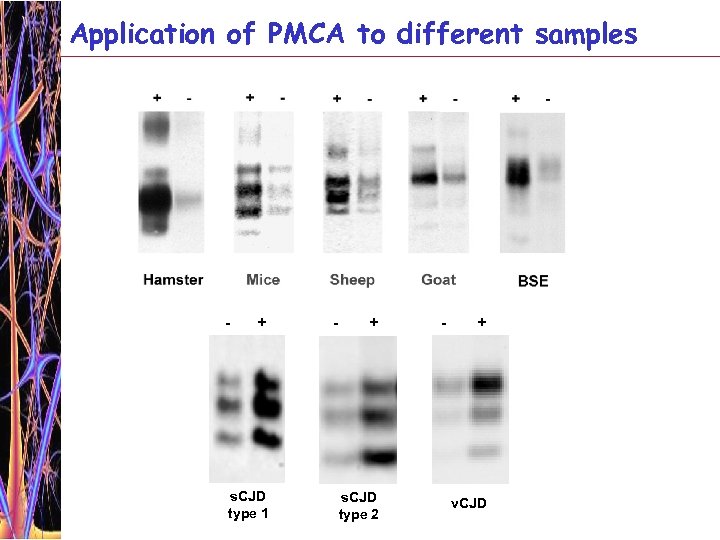
Application of PMCA to different samples - + s. CJD type 1 - + s. CJD type 2 - + v. CJD

What is next? üAdapt and optimize blood detection of Pr. PSc in relevant natural samples (cow, human, sheep, deer) üLarge scale study to evaluate the detection of Pr. PSc in blood of healthy donors in countries with high risk of v. CJD (UK, France, etc) üStudy earliest time in which Pr. PSc can be detected in humans (primate model, familial cases) and in cattle (experimental infection model) üOptimize the method for detection in other blood components (plasma, red cells) and other biological fluids (urine, CSF). üDevelop the technology into a high throughput and practical test

Rodrigo Morales June Yowtak Joaquín Castilla, Ph. D Lisbell Estrada Karim Abid, Ph. D Becky Daniels Paula Saá Claudio Soto, Ph. D Jorge de Castro

Former lab members Gabriela Saborio, MD Celine Adessi, Ph. D Kinsey Maundrell, Ph. D Bruno Permanne, Ph. D Youcef Fezoui, Ph. D Raphaele Buser, Ph. D Milene Russelaskis, Ph. D Claudio Hetz, Ph. D Sergio Benavent Laurence Anderes Marie-Jose Frossard Santiago Fraga Elizabeth Vial Sergio Peano, MD Thomas Ruckle, Ph. D Collaborators Case Western Reserve University Pierluigi Gambetti CJD Unit, Edinburgh, UK Robert Will James Ironside Istituto Carlo Besta, Italy Fabrizio Tagliavini US Department of Agriculture Juergen Richt Istituto Superiore di Sanita, Italy Maurizio Pocchiari University of Kentucky Glenn Telling University of Edinburgh, UK Jean Manson University of Zurich, Switzerland Adriano Aguzzi Mathias Heikenwalder
396be5df846f89190b7642144ae2055c.ppt