15bd92191e1d5e3041a38f158291bc97.ppt
- Количество слайдов: 31
 Update on the Replacement of the HCV RNA International Standard Sally Baylis & Alan Heath, NIBSC So. GAT XX, Warsaw 12 -13 June 2007
Update on the Replacement of the HCV RNA International Standard Sally Baylis & Alan Heath, NIBSC So. GAT XX, Warsaw 12 -13 June 2007
 Replacement of the 2 nd International Standard for HCV RNA (96/798) n n Proposal made at So. GAT 2005 to replace the HCV RNA International Standard as requested by WHO Agreement that HCV 1 a genotype would be sourced & would be anti-HCV negative and diluted in plasma rather than cryosupernatant
Replacement of the 2 nd International Standard for HCV RNA (96/798) n n Proposal made at So. GAT 2005 to replace the HCV RNA International Standard as requested by WHO Agreement that HCV 1 a genotype would be sourced & would be anti-HCV negative and diluted in plasma rather than cryosupernatant
 Candidate Standards for Evaluation n n 3 anti-HCV negative window period genotype 1 a donations have been obtained The genotype of each has been confirmed by Li. PA & DNA sequencing Absence of other viral markers confirmed in these stocks Material has been freeze-dried in two batches n n Batch 1 (NIBSC Code 06/100), 2085 vials, fill CV = 0. 62% Batch 2 (NIBSC code 06/102), 2100 vials, fill CV = 1. 82%
Candidate Standards for Evaluation n n 3 anti-HCV negative window period genotype 1 a donations have been obtained The genotype of each has been confirmed by Li. PA & DNA sequencing Absence of other viral markers confirmed in these stocks Material has been freeze-dried in two batches n n Batch 1 (NIBSC Code 06/100), 2085 vials, fill CV = 0. 62% Batch 2 (NIBSC code 06/102), 2100 vials, fill CV = 1. 82%
 Panel for Collaborative study n Sample 1 – 2 nd International Standard for HCV RNA (96/798) n Sample 2 - new freeze-dried preparation 06/100 n Sample 3 - new freeze-dried preparation 06/102 n Sample 4 – liquid/frozen bulk material (NIBSC code 06/118) that was used to prepare Samples 2 & 3
Panel for Collaborative study n Sample 1 – 2 nd International Standard for HCV RNA (96/798) n Sample 2 - new freeze-dried preparation 06/100 n Sample 3 - new freeze-dried preparation 06/102 n Sample 4 – liquid/frozen bulk material (NIBSC code 06/118) that was used to prepare Samples 2 & 3
 Collaborative Study n Collaborative study commenced in November 2006 n Participants requested to test samples in four independent assays n 33 laboratories from 14 countries participated in the study n 40 data sets received; 25 from quantitative assays & 15 from qualitative assays n NIBSC collated & analysed data
Collaborative Study n Collaborative study commenced in November 2006 n Participants requested to test samples in four independent assays n 33 laboratories from 14 countries participated in the study n 40 data sets received; 25 from quantitative assays & 15 from qualitative assays n NIBSC collated & analysed data
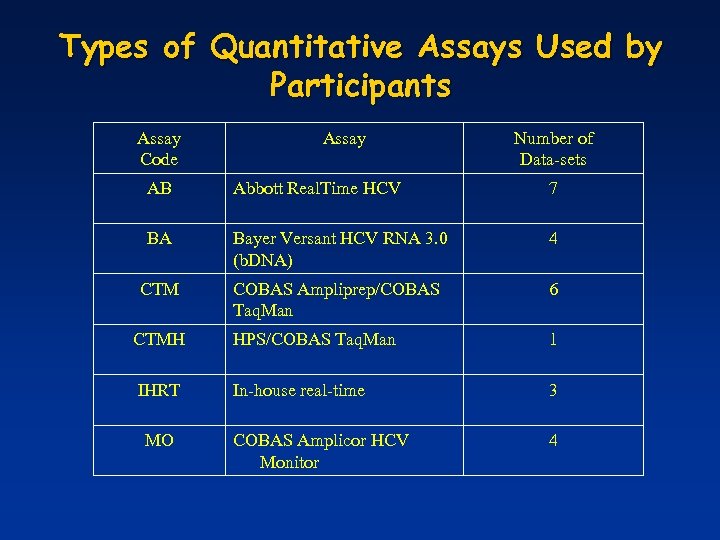 Types of Quantitative Assays Used by Participants Assay Code Assay Number of Data-sets AB Abbott Real. Time HCV 7 BA Bayer Versant HCV RNA 3. 0 (b. DNA) 4 CTM COBAS Ampliprep/COBAS Taq. Man 6 CTMH HPS/COBAS Taq. Man 1 IHRT In-house real-time 3 COBAS Amplicor HCV Monitor 4 MO
Types of Quantitative Assays Used by Participants Assay Code Assay Number of Data-sets AB Abbott Real. Time HCV 7 BA Bayer Versant HCV RNA 3. 0 (b. DNA) 4 CTM COBAS Ampliprep/COBAS Taq. Man 6 CTMH HPS/COBAS Taq. Man 1 IHRT In-house real-time 3 COBAS Amplicor HCV Monitor 4 MO
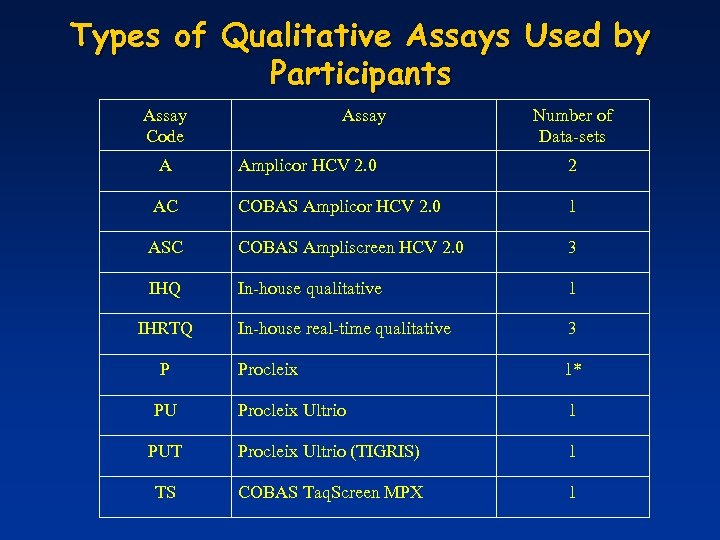 Types of Qualitative Assays Used by Participants Assay Code A Assay Number of Data-sets Amplicor HCV 2. 0 2 AC COBAS Amplicor HCV 2. 0 1 ASC COBAS Ampliscreen HCV 2. 0 3 IHQ In-house qualitative 1 In-house real-time qualitative 3 Procleix 1* Procleix Ultrio 1 PUT Procleix Ultrio (TIGRIS) 1 TS COBAS Taq. Screen MPX 1 IHRTQ P PU
Types of Qualitative Assays Used by Participants Assay Code A Assay Number of Data-sets Amplicor HCV 2. 0 2 AC COBAS Amplicor HCV 2. 0 1 ASC COBAS Ampliscreen HCV 2. 0 3 IHQ In-house qualitative 1 In-house real-time qualitative 3 Procleix 1* Procleix Ultrio 1 PUT Procleix Ultrio (TIGRIS) 1 TS COBAS Taq. Screen MPX 1 IHRTQ P PU
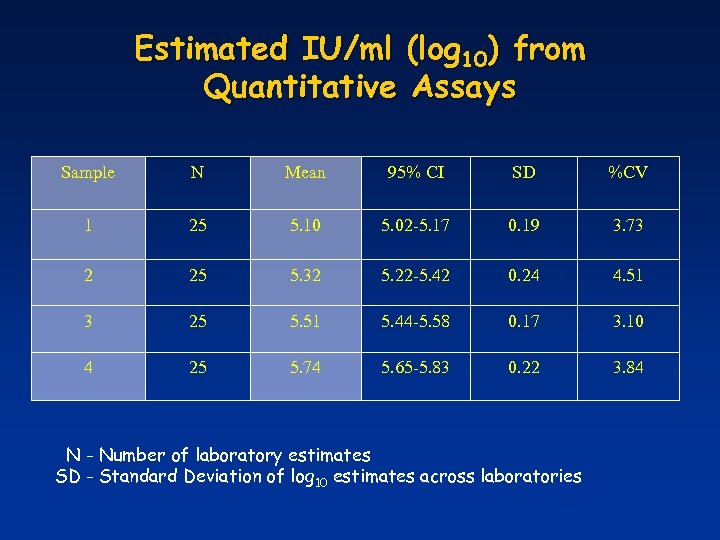 Estimated IU/ml (log 10) from Quantitative Assays Sample N Mean 95% CI SD %CV 1 25 5. 10 5. 02 -5. 17 0. 19 3. 73 2 25 5. 32 5. 22 -5. 42 0. 24 4. 51 3 25 5. 51 5. 44 -5. 58 0. 17 3. 10 4 25 5. 74 5. 65 -5. 83 0. 22 3. 84 N - Number of laboratory estimates SD - Standard Deviation of log 10 estimates across laboratories
Estimated IU/ml (log 10) from Quantitative Assays Sample N Mean 95% CI SD %CV 1 25 5. 10 5. 02 -5. 17 0. 19 3. 73 2 25 5. 32 5. 22 -5. 42 0. 24 4. 51 3 25 5. 51 5. 44 -5. 58 0. 17 3. 10 4 25 5. 74 5. 65 -5. 83 0. 22 3. 84 N - Number of laboratory estimates SD - Standard Deviation of log 10 estimates across laboratories
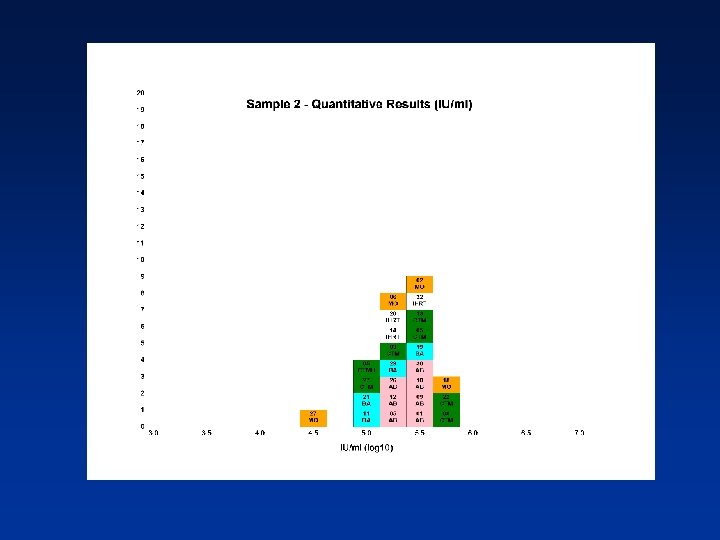
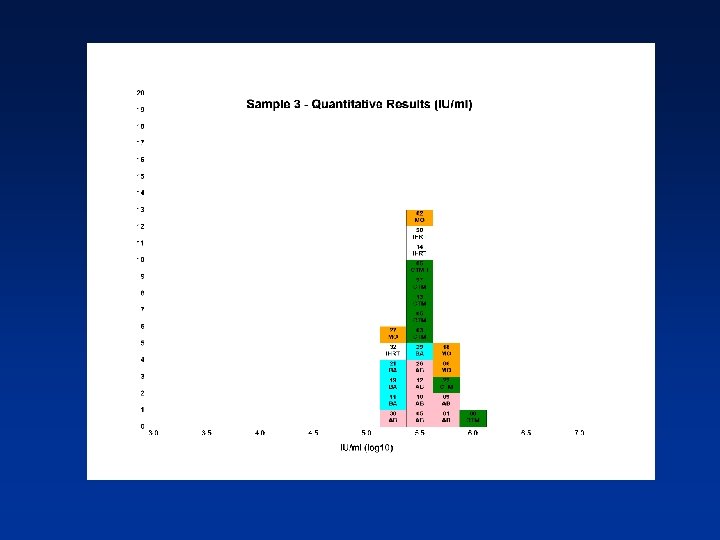
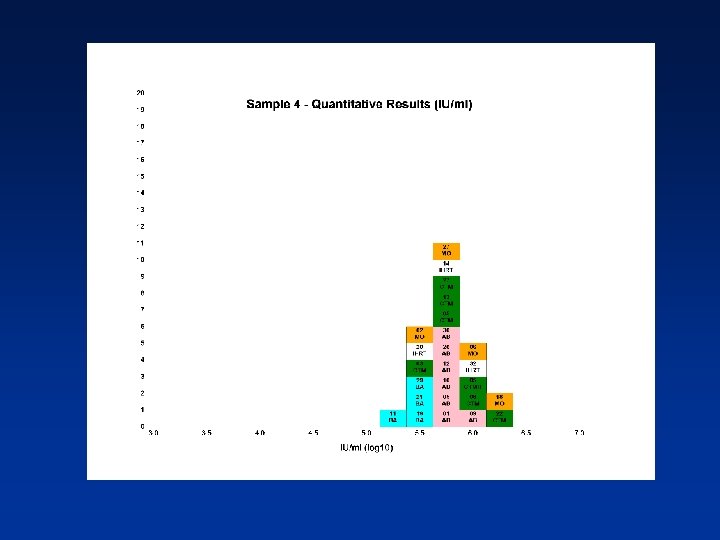
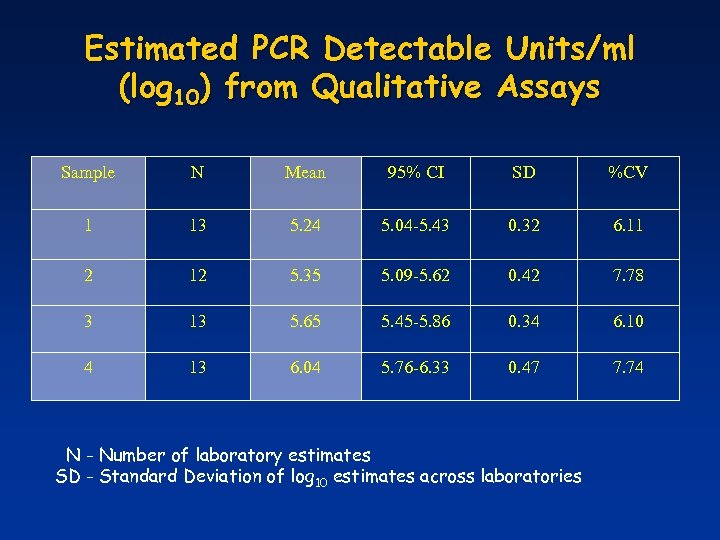 Estimated PCR Detectable Units/ml (log 10) from Qualitative Assays Sample N Mean 95% CI SD %CV 1 13 5. 24 5. 04 -5. 43 0. 32 6. 11 2 12 5. 35 5. 09 -5. 62 0. 42 7. 78 3 13 5. 65 5. 45 -5. 86 0. 34 6. 10 4 13 6. 04 5. 76 -6. 33 0. 47 7. 74 N - Number of laboratory estimates SD - Standard Deviation of log 10 estimates across laboratories
Estimated PCR Detectable Units/ml (log 10) from Qualitative Assays Sample N Mean 95% CI SD %CV 1 13 5. 24 5. 04 -5. 43 0. 32 6. 11 2 12 5. 35 5. 09 -5. 62 0. 42 7. 78 3 13 5. 65 5. 45 -5. 86 0. 34 6. 10 4 13 6. 04 5. 76 -6. 33 0. 47 7. 74 N - Number of laboratory estimates SD - Standard Deviation of log 10 estimates across laboratories
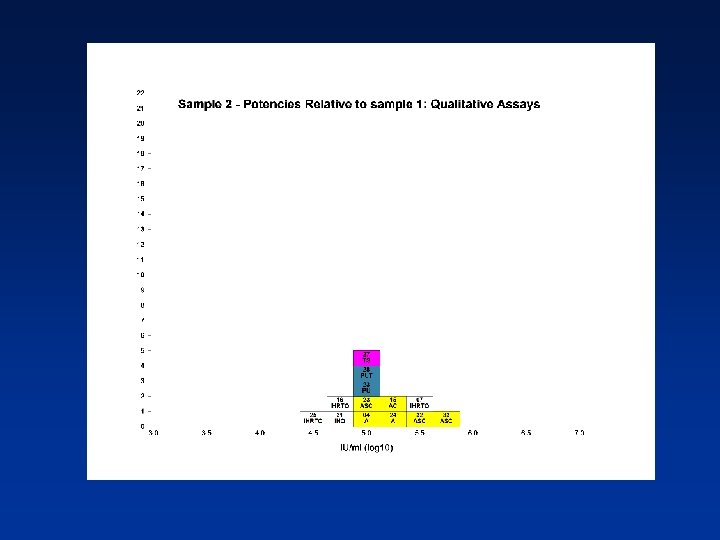
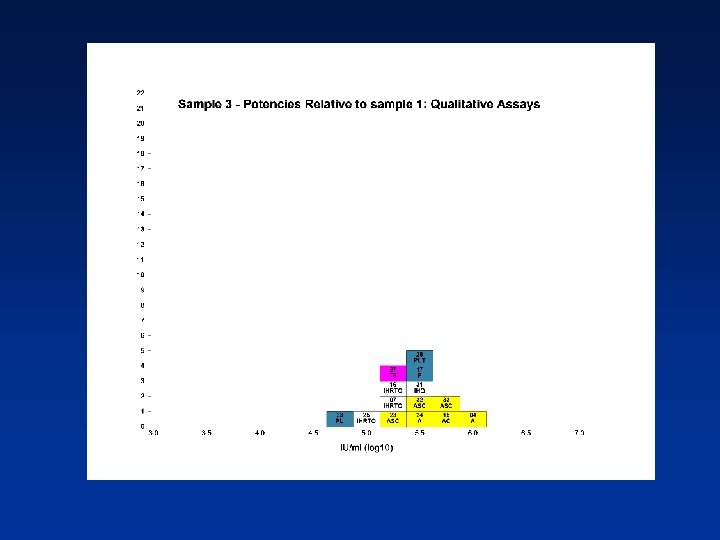
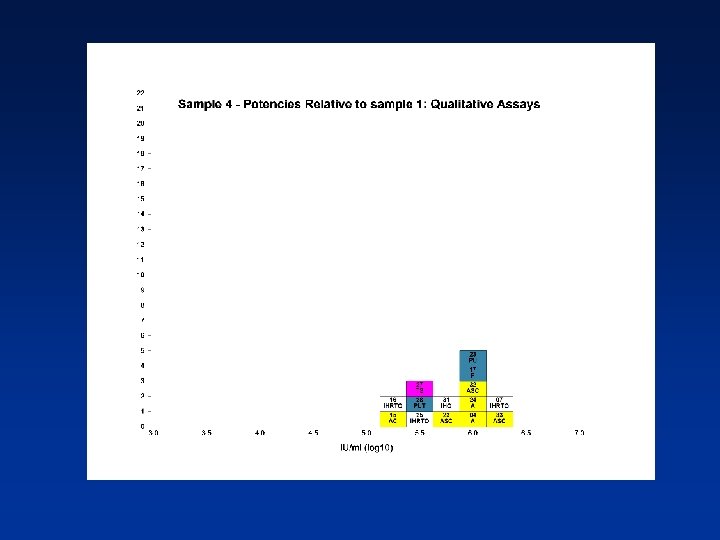
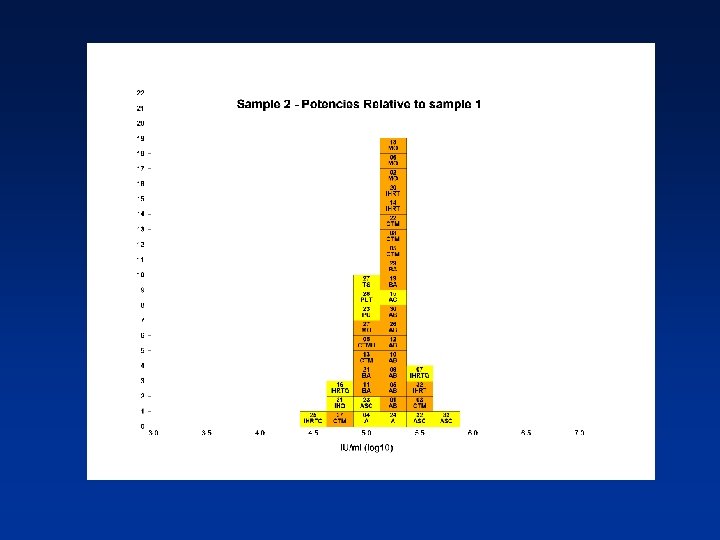
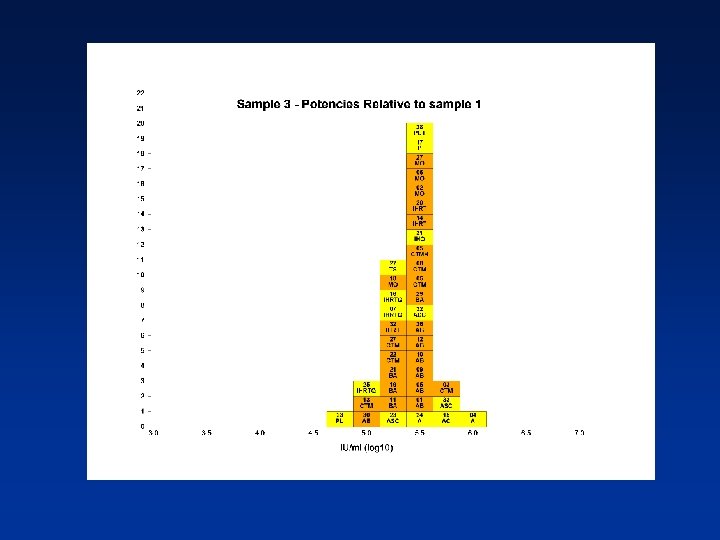
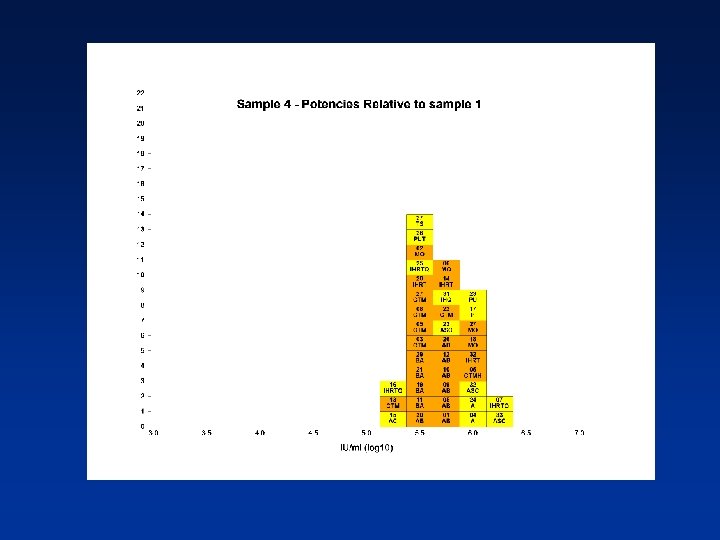
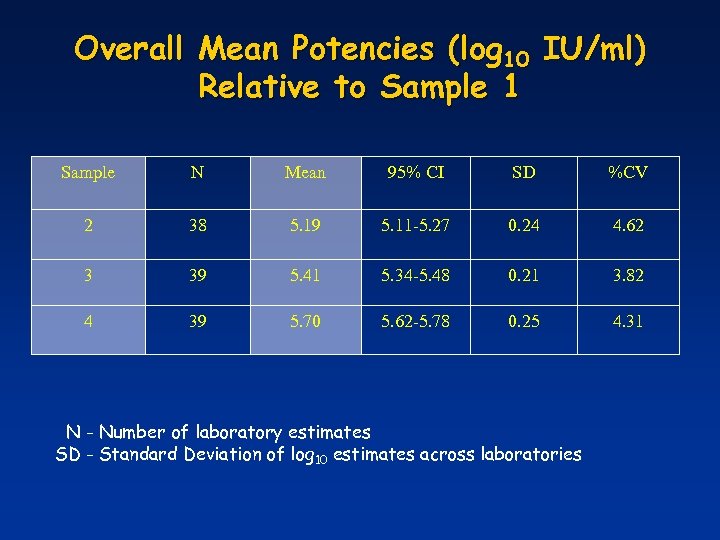 Overall Mean Potencies (log 10 IU/ml) Relative to Sample 1 Sample N Mean 95% CI SD %CV 2 38 5. 19 5. 11 -5. 27 0. 24 4. 62 3 39 5. 41 5. 34 -5. 48 0. 21 3. 82 4 39 5. 70 5. 62 -5. 78 0. 25 4. 31 N - Number of laboratory estimates SD - Standard Deviation of log 10 estimates across laboratories
Overall Mean Potencies (log 10 IU/ml) Relative to Sample 1 Sample N Mean 95% CI SD %CV 2 38 5. 19 5. 11 -5. 27 0. 24 4. 62 3 39 5. 41 5. 34 -5. 48 0. 21 3. 82 4 39 5. 70 5. 62 -5. 78 0. 25 4. 31 N - Number of laboratory estimates SD - Standard Deviation of log 10 estimates across laboratories
 Accelerated Degradation Studies Sample 2 Incubation Time n n +20°C (1 month) Sample 2 n +4°C (10. 5 months) 0. 22 0. 20 Relative potencies of accelerated degradation samples with respect to Sample 2 stored at -20ºC (log 10 drop), q. PCR analysis Stability appears similar to 96/790 and 96/798; studies on-going 96/798 from this study has not lost potency after >10 years storage at -20 ºC
Accelerated Degradation Studies Sample 2 Incubation Time n n +20°C (1 month) Sample 2 n +4°C (10. 5 months) 0. 22 0. 20 Relative potencies of accelerated degradation samples with respect to Sample 2 stored at -20ºC (log 10 drop), q. PCR analysis Stability appears similar to 96/790 and 96/798; studies on-going 96/798 from this study has not lost potency after >10 years storage at -20 ºC
 Proposal for 3 rd International Standard for HCV RNA n n n Real-time & accelerated degradation data indicate that Sample 2 (06/100) is stable & suitable for long term use Propose that 06/100 be established as the 3 rd International Standard for HCV RNA with a unitage of 5. 19 log 10 IU/ml (4. 89 log 10 IU/vial) Report will be submitted to ECBS by July
Proposal for 3 rd International Standard for HCV RNA n n n Real-time & accelerated degradation data indicate that Sample 2 (06/100) is stable & suitable for long term use Propose that 06/100 be established as the 3 rd International Standard for HCV RNA with a unitage of 5. 19 log 10 IU/ml (4. 89 log 10 IU/vial) Report will be submitted to ECBS by July
 Acknowledgements n n n Nita Shah & David Padley NIBSC Professor Ewa Brojer, Institute of Hematology & Blood Transfusion, Warsaw Dr Richard Smith, National Genetics Institute, Los Angeles n Dr Vivienne James, HPA, Colindale n Dr Michael Chudy, WHO, Geneva n Collaborative study participants
Acknowledgements n n n Nita Shah & David Padley NIBSC Professor Ewa Brojer, Institute of Hematology & Blood Transfusion, Warsaw Dr Richard Smith, National Genetics Institute, Los Angeles n Dr Vivienne James, HPA, Colindale n Dr Michael Chudy, WHO, Geneva n Collaborative study participants
 HCV RNA Collaborative Study Participants n n n n n S Baylis, N Shah T Cuypers M Koppelman C Defer L Fanning G Gesu A Goglio P Grant P Iudicone L Jarvis, A Cleland C Jork S Kerby A Klotz, M Gessner R Madej B Miller S Mizusawa C Negru M Nübling South Mimms, UK Amsterdam, The Netherlands Lille, France Cork, Eire Milan, Italy Bergamo, Italy London, UK Rome, Italy Edinburgh, UK Springe, Germany Bethesda, USA Vienna, Austria Pleasanton, USA Karlsruhe, Germany Tokyo, Japan Timişoara , Romania Langen, Germany
HCV RNA Collaborative Study Participants n n n n n S Baylis, N Shah T Cuypers M Koppelman C Defer L Fanning G Gesu A Goglio P Grant P Iudicone L Jarvis, A Cleland C Jork S Kerby A Klotz, M Gessner R Madej B Miller S Mizusawa C Negru M Nübling South Mimms, UK Amsterdam, The Netherlands Lille, France Cork, Eire Milan, Italy Bergamo, Italy London, UK Rome, Italy Edinburgh, UK Springe, Germany Bethesda, USA Vienna, Austria Pleasanton, USA Karlsruhe, Germany Tokyo, Japan Timişoara , Romania Langen, Germany
 HCV RNA Collaborative Study Participants n n n n JM Pawlotsky M Pecorari G Pisani A Portela Moreira S Sauleda H Schindl G Schneider A da Silva R Smith S Tassen P Taylor A van der Zanden YC Yang S Yerly M Zaccanelli Créteil, France Modena, Italy Rome, Italy Madrid, Spain Barcelona, Spain Vienna, Austria Des Plaines, USA Lisbon, Portugal Los Angeles, USA Brentwood, UK London, UK Apeldoorn, The Netherlands Taipei, Taiwan Geneva, Switzerland Treviglio, Italy
HCV RNA Collaborative Study Participants n n n n JM Pawlotsky M Pecorari G Pisani A Portela Moreira S Sauleda H Schindl G Schneider A da Silva R Smith S Tassen P Taylor A van der Zanden YC Yang S Yerly M Zaccanelli Créteil, France Modena, Italy Rome, Italy Madrid, Spain Barcelona, Spain Vienna, Austria Des Plaines, USA Lisbon, Portugal Los Angeles, USA Brentwood, UK London, UK Apeldoorn, The Netherlands Taipei, Taiwan Geneva, Switzerland Treviglio, Italy
 Update on the Stability of the HAV RNA International Standard Sally Baylis, Nita Shah & Alan Heath, NIBSC So. GAT XX, Warsaw 12 -13 June 2007
Update on the Stability of the HAV RNA International Standard Sally Baylis, Nita Shah & Alan Heath, NIBSC So. GAT XX, Warsaw 12 -13 June 2007
 Background – Hepatitis A Virus RNA IS n The 1 st International Standard for HAV RNA (00/560) and a second batch (00/562) were prepared in Q 1, 2001 n 00/560 was evaluated in an international collaborative study, with data returned from 16 laboratories n The mean log 10 equivalents/ml were 5. 29 (00/560) and 5. 07 (00/562) n 1 st International Standard (00/560) was established in 2003 with 50, 000 IU/vial n ECBS noted that 00/562 may be a replacement for 00/560
Background – Hepatitis A Virus RNA IS n The 1 st International Standard for HAV RNA (00/560) and a second batch (00/562) were prepared in Q 1, 2001 n 00/560 was evaluated in an international collaborative study, with data returned from 16 laboratories n The mean log 10 equivalents/ml were 5. 29 (00/560) and 5. 07 (00/562) n 1 st International Standard (00/560) was established in 2003 with 50, 000 IU/vial n ECBS noted that 00/562 may be a replacement for 00/560
 Stability Data 00/560 & 00/562 n Accelerated thermal degradation samples for 00/560 and 00/562, stored at +4ºC for ~6 years were tested in parallel with samples of 00/560 stored at -20 ºC n 00/560 – a drop on 0. 1 log 10 n 00/562 – a drop on 1. 0 log 10 n Conclude that 00/560 is highly stable
Stability Data 00/560 & 00/562 n Accelerated thermal degradation samples for 00/560 and 00/562, stored at +4ºC for ~6 years were tested in parallel with samples of 00/560 stored at -20 ºC n 00/560 – a drop on 0. 1 log 10 n 00/562 – a drop on 1. 0 log 10 n Conclude that 00/560 is highly stable
 Proposal to Replace the Parvovirus B 19 DNA International Standard Sally Baylis, NIBSC So. GAT XX, Warsaw 12 -13 June 2007
Proposal to Replace the Parvovirus B 19 DNA International Standard Sally Baylis, NIBSC So. GAT XX, Warsaw 12 -13 June 2007
 Replacement of the Parvovirus B 19 DNA IS 99/800 n The 1 st International Standard for B 19 V DNA was established by the WHO ECBS in October 2000 n Estimated date of exhaustion of the IS will be 2008/9 at current rate of usage n n Materials coded AA (99/800) & BB (99/802) showed no significant difference in potency in the collaborative study ECBS noted that BB (made from the same stock as AA) could be reserved for potential future use as a replacement standard
Replacement of the Parvovirus B 19 DNA IS 99/800 n The 1 st International Standard for B 19 V DNA was established by the WHO ECBS in October 2000 n Estimated date of exhaustion of the IS will be 2008/9 at current rate of usage n n Materials coded AA (99/800) & BB (99/802) showed no significant difference in potency in the collaborative study ECBS noted that BB (made from the same stock as AA) could be reserved for potential future use as a replacement standard
 Candidate 2 nd International Standard n n n Materials coded AA (99/800) & BB (99/802) showed no significant difference in potency in the original collaborative study ECBS noted that BB (prepared from the same stock as AA) could be reserved for potential future use as a replacement standard Current study designed to demonstrate the equivalence of the candidate replacement (BB) to AA n n Real-time data on samples AA & BB Accelerated degradation data for samples AA & BB
Candidate 2 nd International Standard n n n Materials coded AA (99/800) & BB (99/802) showed no significant difference in potency in the original collaborative study ECBS noted that BB (prepared from the same stock as AA) could be reserved for potential future use as a replacement standard Current study designed to demonstrate the equivalence of the candidate replacement (BB) to AA n n Real-time data on samples AA & BB Accelerated degradation data for samples AA & BB


