a910c37813f31990c952105451938cdd.ppt
- Количество слайдов: 22
 Update on status of West Nile virus test, lot release and validation panel development Indira Hewlett, Ph. D CBER/FDA Blood Products Advisory Committee Meeting June 19, 2003 C B E R
Update on status of West Nile virus test, lot release and validation panel development Indira Hewlett, Ph. D CBER/FDA Blood Products Advisory Committee Meeting June 19, 2003 C B E R
 Previous FDA Actions • March BPAC discussion of FDA proposal for : - clinical study design - unit and donor management - FDA efforts in panel development C B E R
Previous FDA Actions • March BPAC discussion of FDA proposal for : - clinical study design - unit and donor management - FDA efforts in panel development C B E R
 FDA Actions – con’t Study design for test sensitivity • Repository specimens, including transfusion and community acquired WNV illness • Positive cases from prospective studies • Seroconversion panels C B E R
FDA Actions – con’t Study design for test sensitivity • Repository specimens, including transfusion and community acquired WNV illness • Positive cases from prospective studies • Seroconversion panels C B E R
 Clinical sensitivity • Testing a common set of pedigreed specimens by all candidate investigational tests to determine whether assays have equivalent sensitivity • Testing all reactive specimens identified during IND studies by all manufacturer’s assays C B E R
Clinical sensitivity • Testing a common set of pedigreed specimens by all candidate investigational tests to determine whether assays have equivalent sensitivity • Testing all reactive specimens identified during IND studies by all manufacturer’s assays C B E R
 Analytical sensitivity • FDA’s current standard for WNV NAT assays is 100 copies/ml for the individual donation • Standard may be revised as assay sensitivity improves and additional data on viremia and infectivity become available in future studies C B E R
Analytical sensitivity • FDA’s current standard for WNV NAT assays is 100 copies/ml for the individual donation • Standard may be revised as assay sensitivity improves and additional data on viremia and infectivity become available in future studies C B E R
 Unit and donor management FDA proposed foll: scheme for donor and unit management - Reactive invest. NAT results on the individual donation could be confirmed by F/U testing with invest. NAT, alternate NAT and Ig. M - If F/U sample is positive by invest. NAT or alt. NAT, donor remains deferred for an additional 28 days - Donor may be eligible for reinstatement if F/U sample prior to 28 days is NAT-ve, and Ig. M +ve C B E R
Unit and donor management FDA proposed foll: scheme for donor and unit management - Reactive invest. NAT results on the individual donation could be confirmed by F/U testing with invest. NAT, alternate NAT and Ig. M - If F/U sample is positive by invest. NAT or alt. NAT, donor remains deferred for an additional 28 days - Donor may be eligible for reinstatement if F/U sample prior to 28 days is NAT-ve, and Ig. M +ve C B E R
 Progress on test development • Multiple IND studies are in progress • Two manufacturers have publicly acknowledged existing INDs: Gen-Probe Inc. and Roche Molecular Systems Inc. • IND tests are based on NAT using pooled or individual samples • Intended use for whole blood, blood components, source plasma, bone marrow, cord blood, hematopoietic progenitor cells, tissue and organ donors C B E R
Progress on test development • Multiple IND studies are in progress • Two manufacturers have publicly acknowledged existing INDs: Gen-Probe Inc. and Roche Molecular Systems Inc. • IND tests are based on NAT using pooled or individual samples • Intended use for whole blood, blood components, source plasma, bone marrow, cord blood, hematopoietic progenitor cells, tissue and organ donors C B E R
 Progress on test development • Expected start date for testing is early July, 2003 • All samples will be collected under approved IRBs with necessary informed consent • Analytical sensitivity of IND tests is comparable and between 5 -15 copies/ml C B E R
Progress on test development • Expected start date for testing is early July, 2003 • All samples will be collected under approved IRBs with necessary informed consent • Analytical sensitivity of IND tests is comparable and between 5 -15 copies/ml C B E R
 Procleix® WNV Assay • TMA-based assay for screening blood donations for West Nile virus RNA • Uses existing instrument platform as Gen-Probe’s licensed NAT blood screening assay – Procleix Semi-automated System (e. SAS) currently used with Procleix HIV-1/HCV Assay • Uses existing formulations as much as possible C B E R
Procleix® WNV Assay • TMA-based assay for screening blood donations for West Nile virus RNA • Uses existing instrument platform as Gen-Probe’s licensed NAT blood screening assay – Procleix Semi-automated System (e. SAS) currently used with Procleix HIV-1/HCV Assay • Uses existing formulations as much as possible C B E R
 Procleix® WNV Assay • Analytical sensitivity: 95% detection rate between 7 -15 copies/ml • Specificity in pre-clinical studies evaluated by testing 1180 blood donations • No cross reactivity to other blood borne viruses – HTLV, HIV-1/-2, HCV, HBV, HGV, Rubella, HAV, CMV, EBV, HCV, Parvo B 19 • No cross reactivity to other flaviviruses: Dengue (1 -4), Yellow Fever Virus, and St. Louis Encephalitis virus C B E R
Procleix® WNV Assay • Analytical sensitivity: 95% detection rate between 7 -15 copies/ml • Specificity in pre-clinical studies evaluated by testing 1180 blood donations • No cross reactivity to other blood borne viruses – HTLV, HIV-1/-2, HCV, HBV, HGV, Rubella, HAV, CMV, EBV, HCV, Parvo B 19 • No cross reactivity to other flaviviruses: Dengue (1 -4), Yellow Fever Virus, and St. Louis Encephalitis virus C B E R
 WNV IND Two Phased Clinical Protocols • Phase I: Retrospective prevalence study – 89, 000 archived American Red Cross samples from 6 high incidence areas during the 2002 season • Phase II: Prospective donor screening – Voluntary donations of whole blood and source plasma at 25 testing sites : IDT or pools (site dependent) – Nation-wide testing expected to begin by July 1, 2003 C B E R
WNV IND Two Phased Clinical Protocols • Phase I: Retrospective prevalence study – 89, 000 archived American Red Cross samples from 6 high incidence areas during the 2002 season • Phase II: Prospective donor screening – Voluntary donations of whole blood and source plasma at 25 testing sites : IDT or pools (site dependent) – Nation-wide testing expected to begin by July 1, 2003 C B E R
 Procleix WNV Assay Early Testing Contingency Plan • Upon WNV regional outbreak, samples will be shipped and prospective testing initiated at Phase I ARC site(s) – Current testing capability limited • Archiving samples from ~June 1 st onward – Testing of samples based on regional prevalence Contingent on IRB-approval, WNV informed consent in place C B E R
Procleix WNV Assay Early Testing Contingency Plan • Upon WNV regional outbreak, samples will be shipped and prospective testing initiated at Phase I ARC site(s) – Current testing capability limited • Archiving samples from ~June 1 st onward – Testing of samples based on regional prevalence Contingent on IRB-approval, WNV informed consent in place C B E R
 Roche WNV NAT: Pre-clinical Performance Studies • PCR-based screening assay for use with pooled samples • Analytical sensitivity between 5 -7 copies/ml • No cross-reactivity seen with non-WNV microorganisms: HTLV-I/II, HIV, HCV, HBV, CMV, HSV, HAV, HPV, Varicella, Adenovirus • Clinical specificity - 400 random volunteer samples from WNV low- and high- prevalence areas C B E R
Roche WNV NAT: Pre-clinical Performance Studies • PCR-based screening assay for use with pooled samples • Analytical sensitivity between 5 -7 copies/ml • No cross-reactivity seen with non-WNV microorganisms: HTLV-I/II, HIV, HCV, HBV, CMV, HSV, HAV, HPV, Varicella, Adenovirus • Clinical specificity - 400 random volunteer samples from WNV low- and high- prevalence areas C B E R
 FDA Panel Development Efforts • Lot release panel for licensure and postmarket surveillance of NAT and Ig. M tests • Qualification panel for evaluation of relative sensitivities of investigational NAT and Ig. M assays C B E R
FDA Panel Development Efforts • Lot release panel for licensure and postmarket surveillance of NAT and Ig. M tests • Qualification panel for evaluation of relative sensitivities of investigational NAT and Ig. M assays C B E R
 FDA NAT Panels • FDA NY 99 and FDA-Hu 2002 isolates characterized by genetic sequencing • Viral infectivity determination – PFUdetermined at both FDA and NY Dept. of Health Laboratories, and by cytopathic assays at FDA • RNA concentration measurements – Fluorescence and Optical density determination – Taq. Man • Final panel specifications are being established through collaborative studies C B E R
FDA NAT Panels • FDA NY 99 and FDA-Hu 2002 isolates characterized by genetic sequencing • Viral infectivity determination – PFUdetermined at both FDA and NY Dept. of Health Laboratories, and by cytopathic assays at FDA • RNA concentration measurements – Fluorescence and Optical density determination – Taq. Man • Final panel specifications are being established through collaborative studies C B E R
 PFU Results on FDA Isolates • At FDA – NY 99 (CDC Flamingo Isolate) 108/m. L – Hu. WNV 2002 108/m. L • At NY State Dept. of Health – NY 99 (CDC Flamingo Isolate) 5. 5 x 107/m. L – Hu. WNV 2002 9. 5 x 106/m. L C B E R
PFU Results on FDA Isolates • At FDA – NY 99 (CDC Flamingo Isolate) 108/m. L – Hu. WNV 2002 108/m. L • At NY State Dept. of Health – NY 99 (CDC Flamingo Isolate) 5. 5 x 107/m. L – Hu. WNV 2002 9. 5 x 106/m. L C B E R
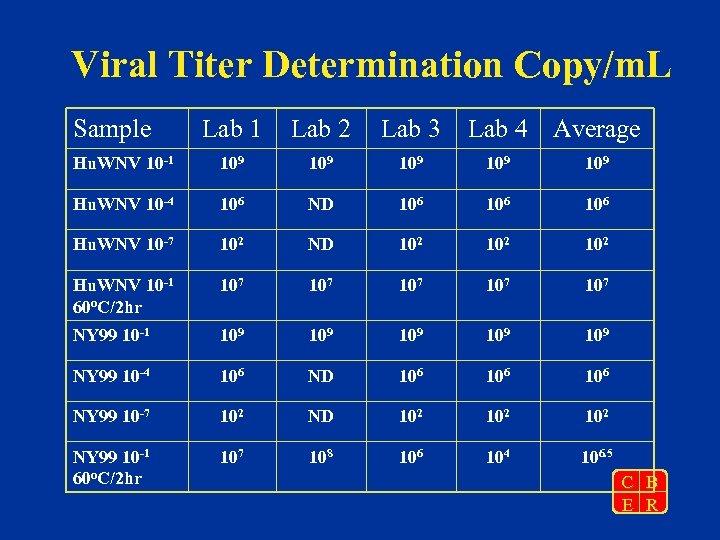 Viral Titer Determination Copy/m. L Sample Lab 1 Lab 2 Lab 3 Lab 4 Average Hu. WNV 10 -1 109 109 109 Hu. WNV 10 -4 106 ND 106 106 Hu. WNV 10 -7 102 ND 102 102 Hu. WNV 10 -1 60 o. C/2 hr 107 107 107 NY 99 10 -1 109 109 109 NY 99 10 -4 106 ND 106 106 NY 99 10 -7 102 ND 102 102 NY 99 10 -1 60 o. C/2 hr 107 108 106 104 106. 5 C B E R
Viral Titer Determination Copy/m. L Sample Lab 1 Lab 2 Lab 3 Lab 4 Average Hu. WNV 10 -1 109 109 109 Hu. WNV 10 -4 106 ND 106 106 Hu. WNV 10 -7 102 ND 102 102 Hu. WNV 10 -1 60 o. C/2 hr 107 107 107 NY 99 10 -1 109 109 109 NY 99 10 -4 106 ND 106 106 NY 99 10 -7 102 ND 102 102 NY 99 10 -1 60 o. C/2 hr 107 108 106 104 106. 5 C B E R
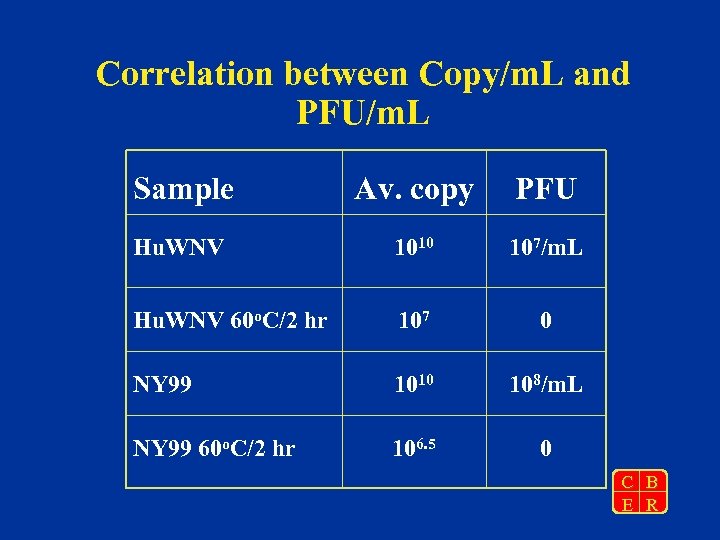 Correlation between Copy/m. L and PFU/m. L Sample Av. copy PFU Hu. WNV 1010 107/m. L Hu. WNV 60 o. C/2 hr 107 0 NY 99 1010 108/m. L NY 99 60 o. C/2 hr 106. 5 0 C B E R
Correlation between Copy/m. L and PFU/m. L Sample Av. copy PFU Hu. WNV 1010 107/m. L Hu. WNV 60 o. C/2 hr 107 0 NY 99 1010 108/m. L NY 99 60 o. C/2 hr 106. 5 0 C B E R
 FDA Plan for Qualification Panel • At least 100 pedigreed clinical specimens – RNA positive only – Ig. M positive only – Dual RNA and Ig. M positive • FDA also recommends that all reactive specimens identified in IND clinical trials be made available to all manufacturers through sharing of samples C B E R
FDA Plan for Qualification Panel • At least 100 pedigreed clinical specimens – RNA positive only – Ig. M positive only – Dual RNA and Ig. M positive • FDA also recommends that all reactive specimens identified in IND clinical trials be made available to all manufacturers through sharing of samples C B E R
 FDA Ig. M panel • Panel will consist of clinical specimens containing varying titers of antibodies to WNV and some members that are also NAT positive • Panel will be evaluated in collaborative studies using various candidate Ig. M assays • Specifications for NAT and Ig. M panels will be established based on results of collaborative studies C B E R
FDA Ig. M panel • Panel will consist of clinical specimens containing varying titers of antibodies to WNV and some members that are also NAT positive • Panel will be evaluated in collaborative studies using various candidate Ig. M assays • Specifications for NAT and Ig. M panels will be established based on results of collaborative studies C B E R
 Summary • Both NY 99 and FDA-Hu 2002 stocks have a viral titer of 1010 copies/m. L • PFU titers at both NY State Dept of Health Laboratory and at FDA were three logs lower than copy numbers • Heat treatment virus results in loss of infectivity and 2 to 3 log reduction in copy number determined by Taq. Man C B E R
Summary • Both NY 99 and FDA-Hu 2002 stocks have a viral titer of 1010 copies/m. L • PFU titers at both NY State Dept of Health Laboratory and at FDA were three logs lower than copy numbers • Heat treatment virus results in loss of infectivity and 2 to 3 log reduction in copy number determined by Taq. Man C B E R
 Acknowledgements • • • Gen-Probe, Inc Roche Molecular Systems, Inc. Maria Rios, CBER, FDA Robert Lanciotti, CDC Laura Kramer, New York Department of Health C B E R
Acknowledgements • • • Gen-Probe, Inc Roche Molecular Systems, Inc. Maria Rios, CBER, FDA Robert Lanciotti, CDC Laura Kramer, New York Department of Health C B E R


