435cbc496d2145e9163a8acc05123cd7.ppt
- Количество слайдов: 151

(Unübliches zur) Menopause Bruno Müller www. Der. Endokrinologe. ch Bern

Absturz / Pause oder Abflug? Machen Sie sich selber ein Bild …

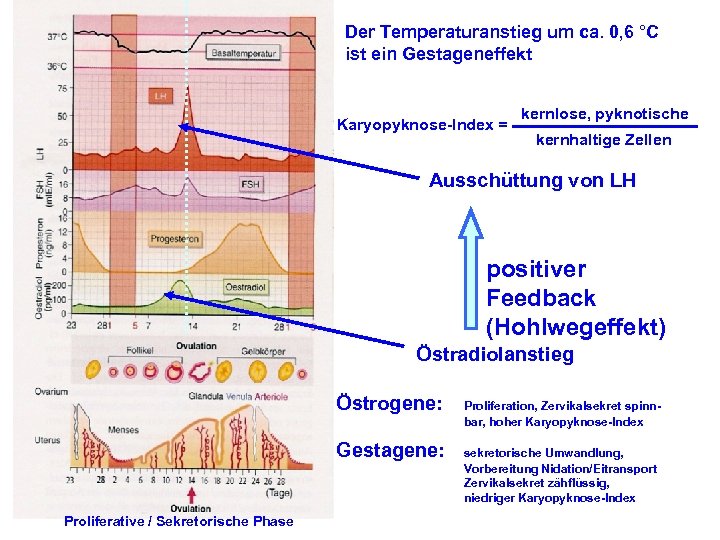
Der Temperaturanstieg um ca. 0, 6 °C ist ein Gestageneffekt Karyopyknose-Index = kernlose, pyknotische kernhaltige Zellen Ausschüttung von LH positiver Feedback (Hohlwegeffekt) Östradiolanstieg Östrogene: Gestagene: Proliferative / Sekretorische Phase Proliferation, Zervikalsekret spinnbar, hoher Karyopyknose-Index sekretorische Umwandlung, Vorbereitung Nidation/Eitransport Zervikalsekret zähflüssig, niedriger Karyopyknose-Index


Pause / Abbruch oder Abflug? • Endokrinologisch gesehen ist die Frau überaus komplex gesteuert …. • Leider sind Störfälle • Oder gar definitive Funktionsausfälle vorprogrammiert Absturz

Inhalte – key points • • Menopause allgemein: - Altern - Epidemiologie - Reparaturmechanismen

Alle versuchen, die Zeit totzuschlagen, und keiner will sterben Franz. Sprichwort


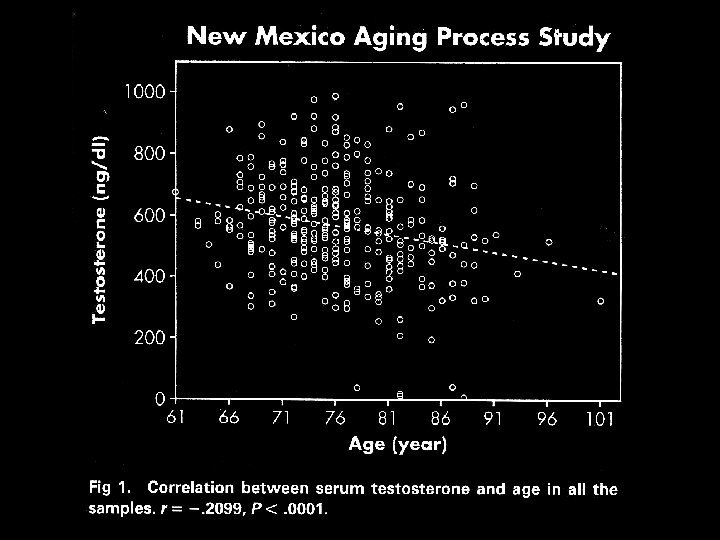
Altern wir, weil die Menge wichtiger Hormone abnimmt? Oder: nimmt die Hormonkonzentration ab, weil wir altern?

„Nun sag, wie hast du’s mit der Religion? “ Die berühmte Gretchenfrage!

Altern, am Bsp. der Oocyten 5 th SS-Monat — 7 million Geburt — 1 -2 Millionen Pubertät — 400, 000 40+ -- Rascher Oocytenverlust
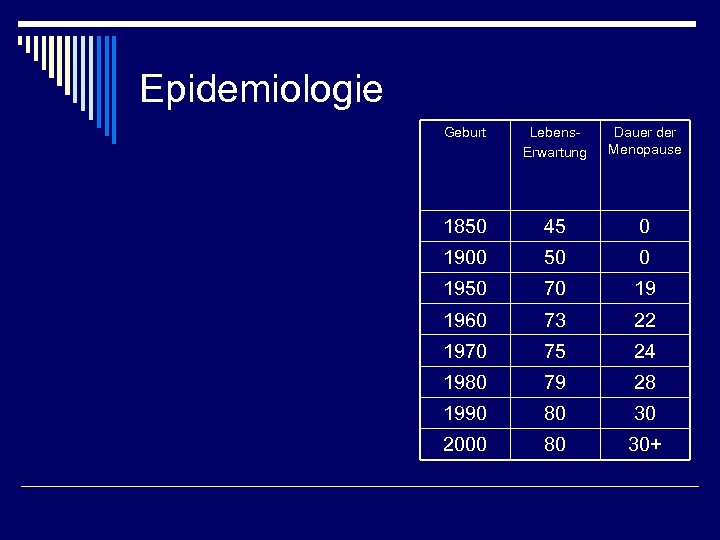
Epidemiologie Geburt Lebens. Erwartung Dauer der Menopause 1850 45 0 1900 50 0 1950 70 19 1960 73 22 1970 75 24 1980 79 28 1990 80 30 2000 80 30+
Home > Die BZg. A > Forschung > Fachdatenbanken/Marktanalysen > www. frauengesundheitsportal. de Demografische Angaben Quelle: Statistisches Bundesamt, Statistisches Jahrbuch 2007 15

Theories of Aging • Genetic – Aging is “programmed” into the genes – Certain genes are “timekeepers” for the aging process • Wear and Tear – Cumulative damage to cells from • Metabolic processes • Environmental factors – Mechanisms to resist and repair damage are critical

Mechanisms to resist and repair damage • Zelluläre • Endokrinologische • Lifestyle, Therapeutische (Hormonersatz, HRT, Alternatives)
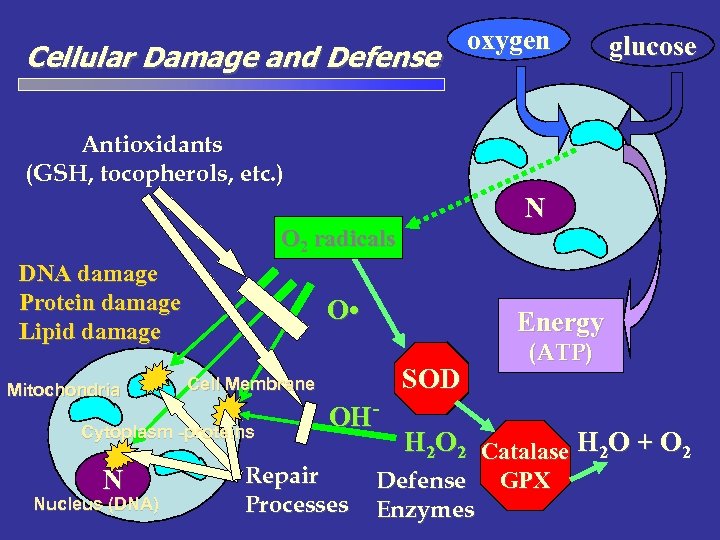
Cellular Damage and Defense oxygen glucose Antioxidants (GSH, tocopherols, etc. ) N O 2 radicals DNA damage Protein damage Lipid damage Mitochondria O • Cell Membrane Cytoplasm -proteins N Nucleus (DNA) Energy OH- Repair Processes SOD (ATP) H 2 O 2 Catalase H 2 O + O 2 Defense Enzymes GPX

Mechanisms to resist and repair damage • Zelluläre • Endokrinologische • Lifestyle, Therapeutische (Hormonersatz, HRT, Alternatives)

Menopause Sympome infolge Östrogen. Ausfall und -Entzug • • Klimakterische Beschwerden Schweißausbrüche Müdigkeit Schlaflosigkeit Nervosität Herzrasen Depressive Verstimmungen • Urogenitale Atrophie • Atrophische Veränderungen des Harntrakts und ihre Folgen (z. B. vaginale Trockenheit, Dyspareunie, häufiges Wasserlassen und Harndrang)

Wenn uns Verzweiflung überkommt, liegt das gewöhnlich daran, dass wir zu viel an die Vergangenheit und an die Zukunft denken Hl Therese von Lisieux

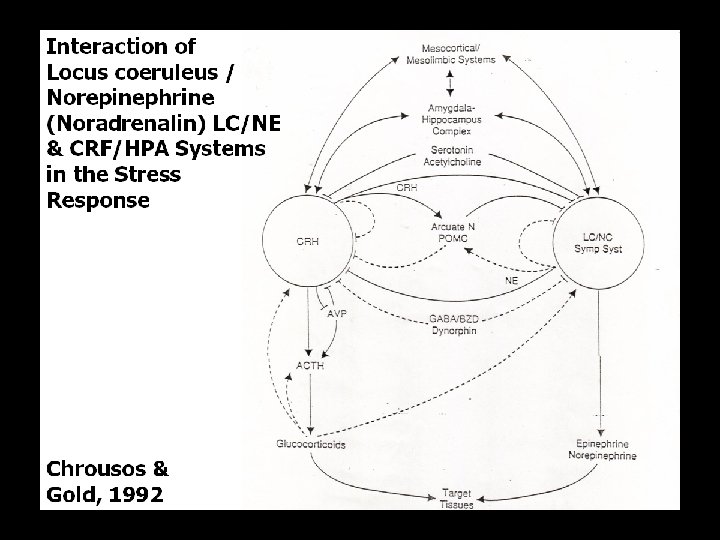
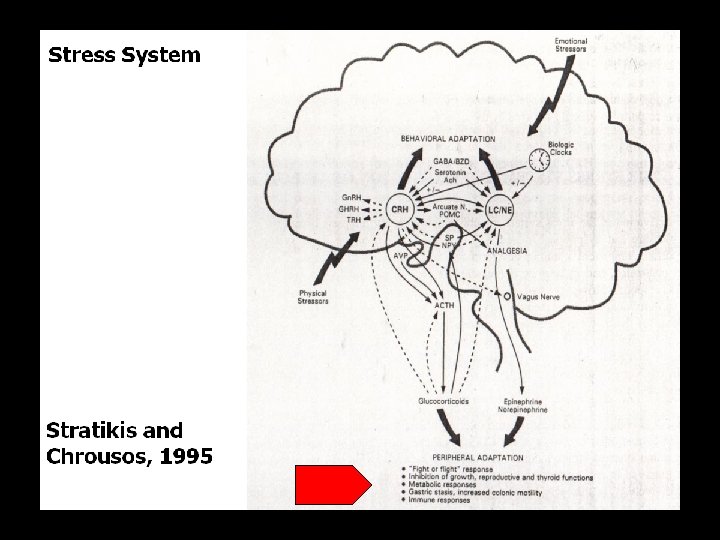
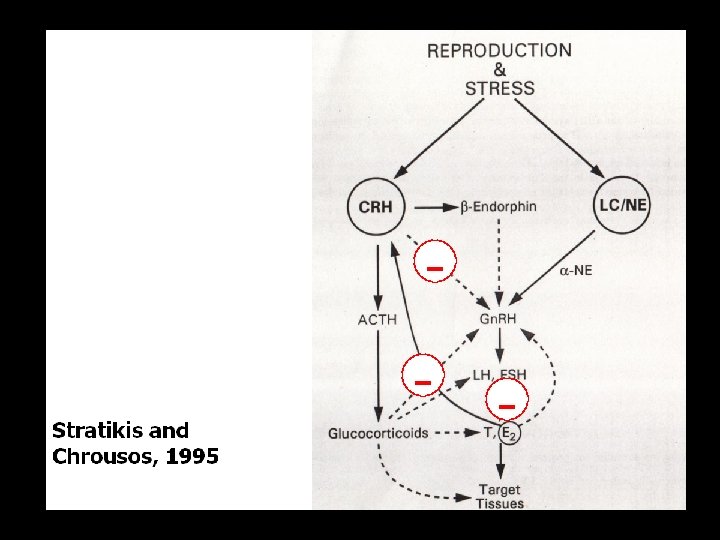
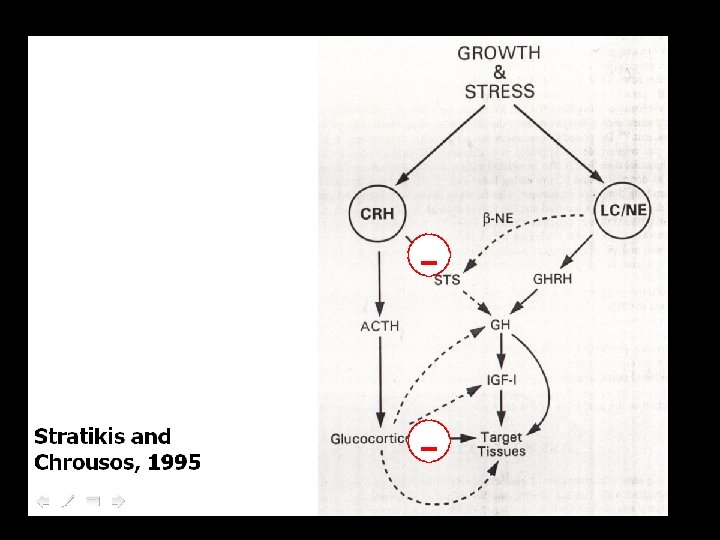
Östrogen. Ausfall und/oder –Entzug = Stress Wie reagiert das Hormonsystem?
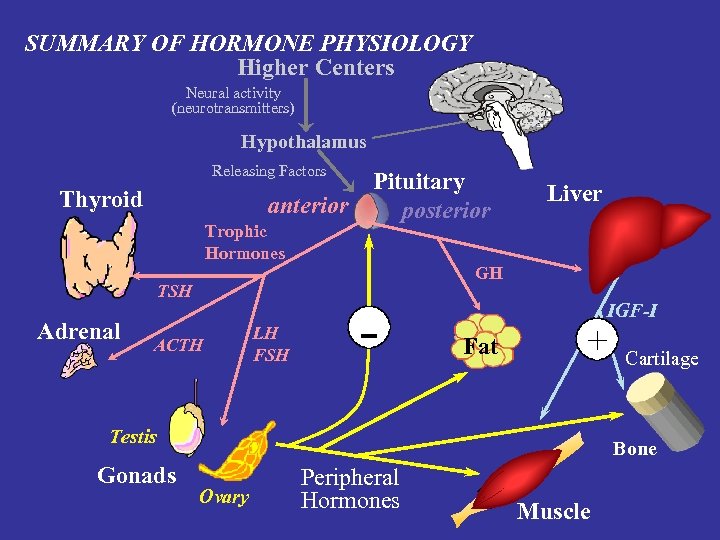
SUMMARY OF HORMONE PHYSIOLOGY Higher Centers Neural activity (neurotransmitters) Hypothalamus Releasing Factors Thyroid anterior Trophic Hormones Liver GH TSH Adrenal Pituitary posterior ACTH LH FSH - Fat + Testis Gonads IGF-I Cartilage Bone Ovary Peripheral Hormones Muscle
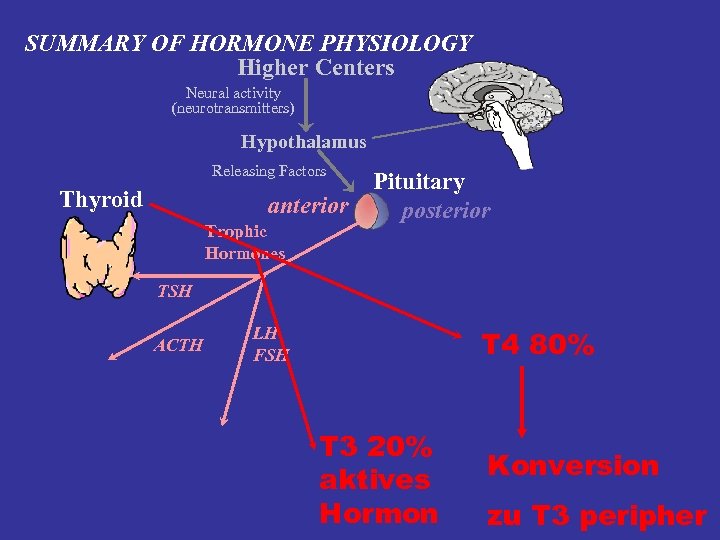
SUMMARY OF HORMONE PHYSIOLOGY Higher Centers Neural activity (neurotransmitters) Hypothalamus Releasing Factors Thyroid anterior Trophic Hormones Pituitary posterior TSH ACTH LH FSH T 4 80% T 3 20% aktives Hormon Konversion zu T 3 peripher

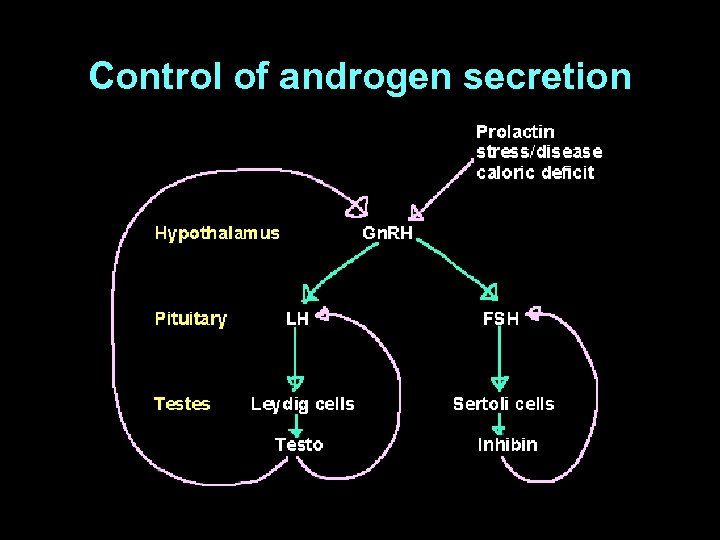
Control of androgen secretion
- -
-
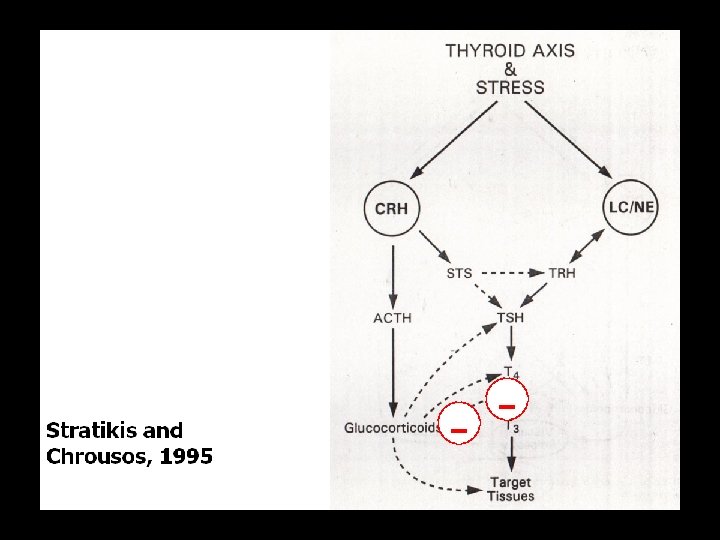
-

(physischer) Stress und endokrines System, Zusammenfassung • • (physischer) Stress führt zu Arousal der CRF-HPA-Achse vermehrter Cortisol-Produktion sekundärem Abfall von Testosteron (und Östradiol, Anstieg von Prolaktin, nicht gezeigt) • und Wachstumshormon • Verminderter Produktion (d. Konversion) von aktivem Schilddrüsen-Hormon T 3

Östrogen. Ausfall und/oder –Entzug Wie reagiert der Körper (body composition)?
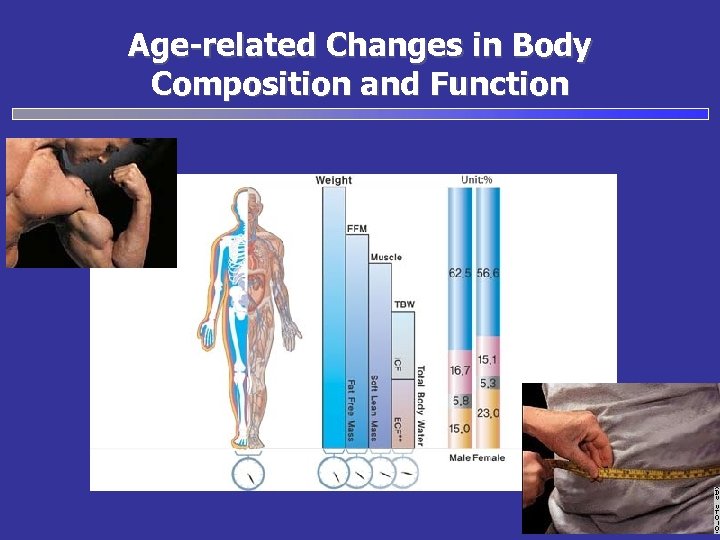
Age-related Changes in Body Composition and Function

Age-related Changes in Body Composition and Function • Body Composition – Loss of lean body (muscle) mass • Decreased strength • Decreased fitness and loss of functional capacity – Increase in total fat mass (percent body fat) • Insulin resistance (type 2 diabetes) • Increased LDL cholesterol, triglycerides, and fatty acids – Decreased bone density (negative calcium balance) • Metabolic/Physiologic Function – – Decreased protein synthesis Slower healing Reduced immune system function Altered hormone balance
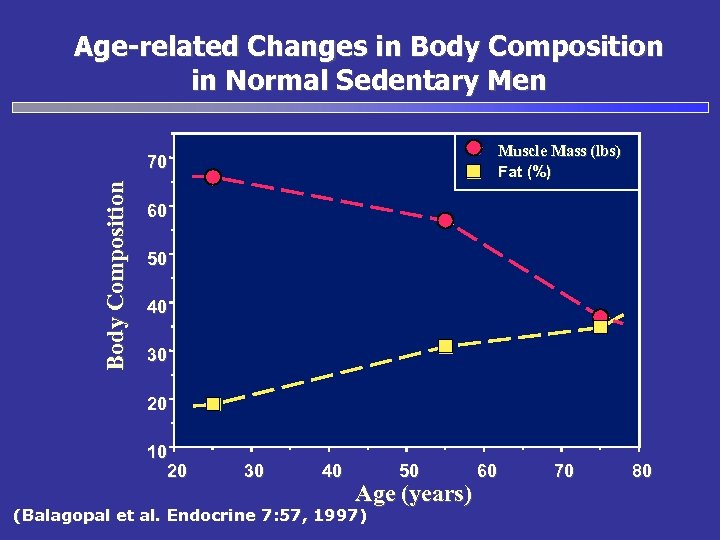
Age-related Changes in Body Composition in Normal Sedentary Men Muscle Mass (lbs) Fat (%) Body Composition 70 60 50 40 30 20 10 20 30 40 50 Age (years) (Balagopal et al. Endocrine 7: 57, 1997) 60 70 80
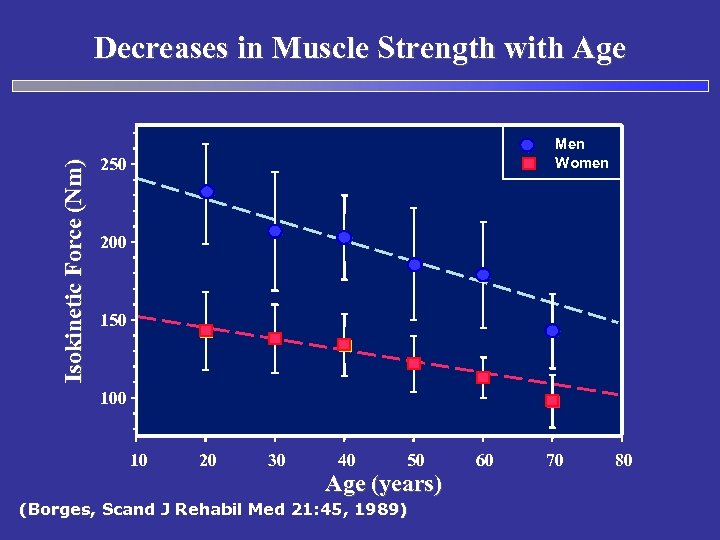
Isokinetic Force (Nm) Decreases in Muscle Strength with Age Men Women 250 200 150 10 20 30 40 50 Age (years) (Borges, Scand J Rehabil Med 21: 45, 1989) 60 70 80

Altern wir, weil die Menge wichtiger Hormone abnimmt? Oder: nimmt die Hormonkonzentration ab, weil wir altern?

How Do Hormones Change with Normal Aging? • Estrogens- decrease to very low levels over a 1 -3 year period at menopause (between ages 4555) • Testosterone (T)- Gradual decline from age 30 onward reaching low (hypogonadal) levels in >20% of men by age 65 • Growth Hormone (GH)- Gradual decrease in secretion (and circulating IGF-I levels) from age 45 -90

How Do Hormones Change with Normal Aging? • Adrenal Steroids– Active adrenal hormones (cortisol and aldosterone) change little – DHEA, steady decrease with age to very low levels in both sexes • Thyroid- not much change in healthy men and women, but increased prevalence of hypothyroid disease in older persons. • Insulin- loss of sensitivity to insulin action with aging and obesity

Mechanisms to resist and repair damage • Zelluläre • Endokrinologische • Lifestyle, Therapeutische (Hormonersatz, HRT, Alternatives)

Wichtigste Lifestyle. Massnahme Wer die Nacht nicht ehrt, ist des Tages nicht wert Italienisches Sprichwort
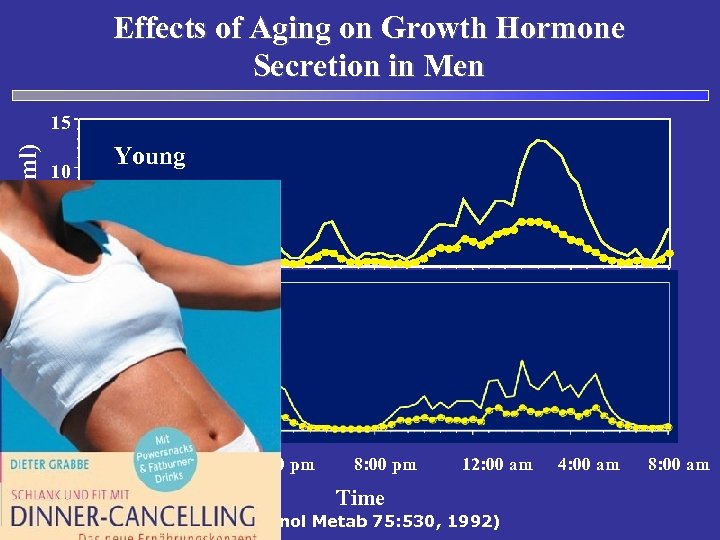
Effects of Aging on Growth Hormone Secretion in Men Growth Hormone (ng/ml) 15 10 Young 5 0 8: 00 am 12: 00 pm 4: 00 pm 8: 00 pm 12: 00 am Time (Corpas, et al. , J Clin Endocrinol Metab 75: 530, 1992) 4: 00 am 8: 00 am

Gibt es sonst was zu tun, ausser viel schlafen? Müller’sche Frage

The International Menopause Society The IMS Updated Recommendations on postmenopausal hormone therapy February 27, 2007 Climacteric 2007; 10: 181– 94

Exercise in the menopause • Any physical activity is better than being sedentary • Regular exercise reduces total and cardiovascular • • mortality Better metabolic profile, balance, muscle strength, cognition and quality of life are observed in physically active persons. Heart events, stroke, fractures and breast cancer are significantly less frequent Benefits far outweigh possible adverse consequences: the more – the better, but too much may cause harm

Exercise in the menopause: optimal exercise prescription • At least 30 minutes of moderate intensity • exercise, at least three times weekly Two additional weekly training sessions of resistance exercise may provide further benefit

AHA 2006 Diet and Lifestyle Recommendations 1 • Balance calorie intake and physical activity to achieve or maintain a healthy body weight • Consume a diet rich in vegetables and fruits • Choose whole-grain, high-fiber foods • Consume fish, especially oily fish, at least twice a week Circulation 2006; 114: 82

AHA 2006 Diet and Lifestyle Recommendations 2 • Limit intake of saturated fat to < 7% of energy, trans fat to < 1% and cholesterol to < 300 mg/day by choosing lean meats and vegetable alternatives, selecting fat-free, 1% fat and low-fat products • Choose and prepare foods with little or no salt • Increase fiber intake (beans, whole grain, other fruits and vegetables) • If you consume alcohol, do so in moderation • Quit smoking Circulation 2006; 114: 82

Mechanisms to resist and repair damage • Zelluläre • Endokrinologische • Lifestyle, Therapeutische (Hormonersatz, HRT, Alternatives)

Indikationen für eine HRT • • Klimakterische Beschwerden Schweißausbrüche Müdigkeit Schlaflosigkeit Nervosität Herzrasen Depressive Verstimmungen • Urogenitale Atrophie • • Atrophische Veränderungen des Harntrakts und ihre Folgen (z. B. vaginale Trockenheit, Dyspareunie, häufiges Wasserlassen und Harndrang) Topische niedrig dosierte Präparate sind die Behandlung der Wahl, wenn lediglich lokale Beschwerden auftreten. • Die Therapie klimakterischer Beschwerden erhält die Lebensqualität
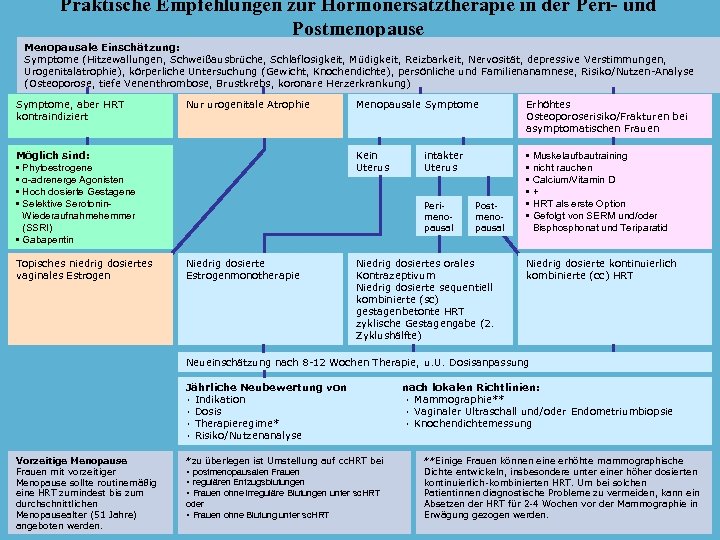
Praktische Empfehlungen zur Hormonersatztherapie in der Peri- und Postmenopause Menopausale Einschätzung: Symptome (Hitzewallungen, Schweißausbrüche, Schlaflosigkeit, Müdigkeit, Reizbarkeit, Nervosität, depressive Verstimmungen, Urogenitalatrophie), körperliche Untersuchung (Gewicht, Knochendichte), persönliche und Familienanamnese, Risiko/Nutzen-Analyse (Osteoporose, tiefe Venenthrombose, Brustkrebs, koronare Herzerkrankung) Symptome, aber HRT kontraindiziert Nur urogenitale Atrophie Topisches niedrig dosiertes vaginales Estrogen Erhöhtes Osteoporoserisiko/Frakturen bei asymptomatischen Frauen Kein Uterus Möglich sind: • Phytoestrogene • α-adrenerge Agonisten • Hoch dosierte Gestagene • Selektive Serotonin. Wiederaufnahmehemmer (SSRI) • Gabapentin Menopausale Symptome • Muskelaufbautraining • nicht rauchen • Calcium/Vitamin D • + • HRT als erste Option • Gefolgt von SERM und/oder Bisphonat und Teriparatid intakter Uterus Perimenopausal Niedrig dosierte Estrogenmonotherapie Postmenopausal Niedrig dosiertes orales Kontrazeptivum Niedrig dosierte sequentiell kombinierte (sc) gestagenbetonte HRT zyklische Gestagengabe (2. Zyklushälfte) Niedrig dosierte kontinuierlich kombinierte (cc) HRT Neueinschätzung nach 8 -12 Wochen Therapie, u. U. Dosisanpassung Jährliche Neubewertung von ۰ Indikation ۰ Dosis ۰ Therapieregime* ۰ Risiko/Nutzenanalyse Vorzeitige Menopause Frauen mit vorzeitiger Menopause sollte routinemäßig eine HRT zumindest bis zum durchschnittlichen Menopausealter (51 Jahre) angeboten werden. *zu überlegen ist Umstellung auf cc. HRT bei • postmenopausalen Frauen • regulären Entzugsblutungen • Frauen ohne irreguläre Blutungen unter sc. HRT oder • Frauen ohne Blutung unter sc. HRT nach lokalen Richtlinien: ۰ Mammographie** ۰ Vaginaler Ultraschall und/oder Endometriumbiopsie ۰ Knochendichtemessung **Einige Frauen können eine erhöhte mammographische Dichte entwickeln, insbesondere unter einer höher dosierten kontinuierlich-kombinierten HRT. Um bei solchen Patientinnen diagnostische Probleme zu vermeiden, kann ein Absetzen der HRT für 2 -4 Wochen vor der Mammographie in Erwägung gezogen werden.

Zusammenfassung HRT • Die Hormonersatztherapie sollte nur verordnet werden, wenn eine klare Indikation besteht (primär zur Behandlung klimakterischer Beschwerden). • Es gibt keine wirksamen Alternativen zur Behandlung • vasomotorischer Symptome. Die Hormonsubstitution kann bei Frauen mit erhöhtem Frakturrisiko eine Anfangsoption zur Senkung des Frakturrisikos darstellen. • Die langfristige Hormonsubstitution ist mit einigen zusätzlichen Risiken verbunden. • • • Venöse thromboembolische Erkrankungen Schlaganfall Brustkrebs (nur bei Normalgewicht) • Die Indikation zur Fortsetzung der Hormonbehandlung sollte jährlich überprüft werden.

Zu den Risiken einer HRT • Die Indikation zur Fortsetzung der Hormonbehandlung sollte jährlich überprüft werden • Unter besonderer Beachtung der Risiken einer HRT

HRT Credo • HRT bedeutet Substitution des fehlenden körpereigenen Hormons in subphysiologischer Dosis. • Die Evolution hat nicht eingeplant, dass die Lebenserwartung so ansteigt • Hätten Männer einen so starken klimakterischen Hormonabfall, gäbe es für den Mann schon lange eine HRT. • Männer haben höhere Östrogenspiegel als nicht behandelte postmenopausale Frauen.

HRT Credo • Östrogene sind nicht mutagen oder cancerogen. • Östrogene wirken evtl. als Promotor auf vorhandene noch okkulte Mammacarcinome.
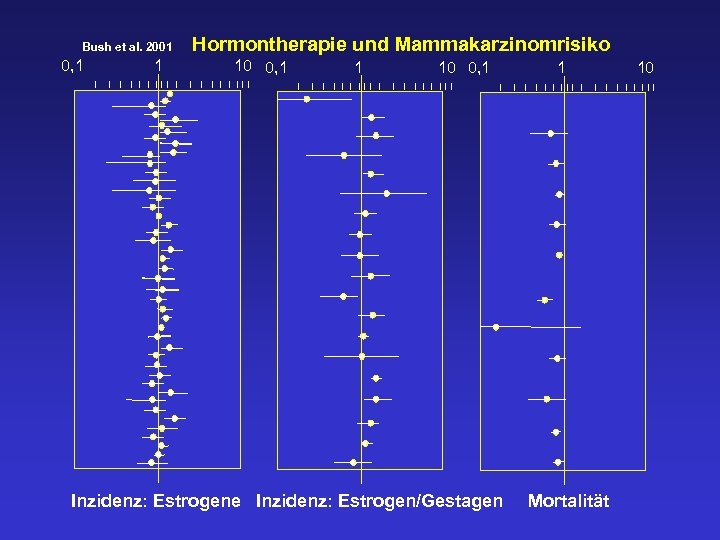
Bush et al. 2001 0, 1 1 I I I III I Hormontherapie und Mammakarzinomrisiko 10 0, 1 I I III 1 I I I III I 10 0, 1 I I III 1 I Inzidenz: Estrogene Inzidenz: Estrogen/Gestagen I I III I 10 I I III Mortalität

HRT Credo • Östrogene erhöhen bei genetisch praedisponierten Patientinnen das Thromboembolierisiko.
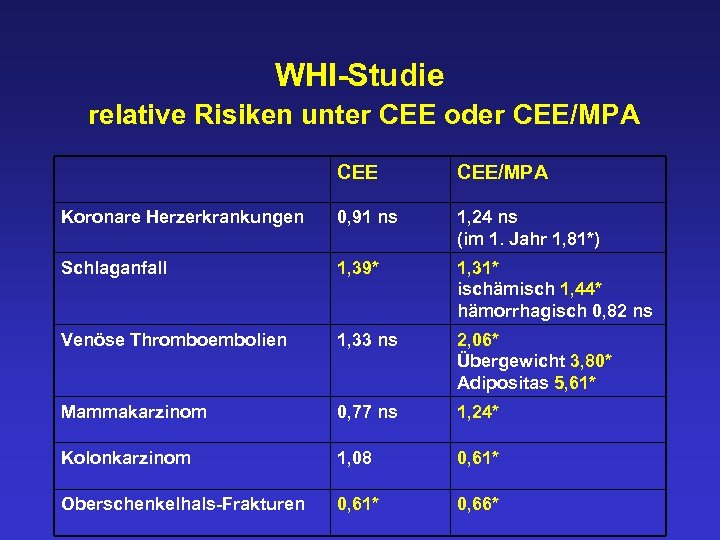
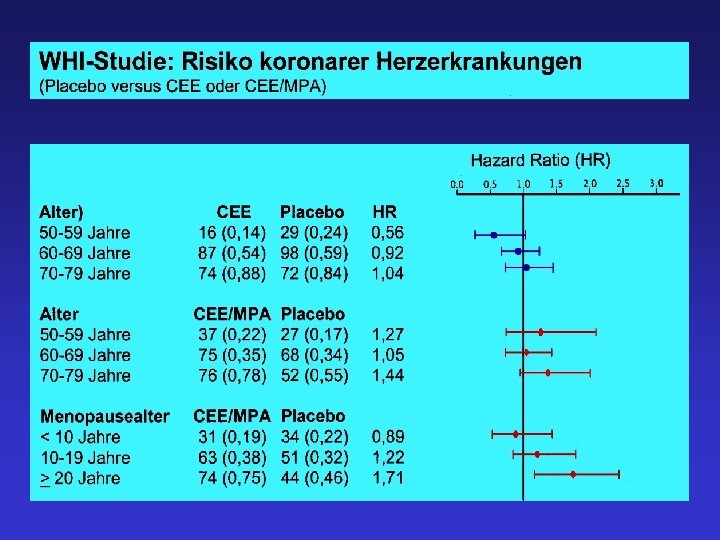
WHI-Studie relative Risiken unter CEE oder CEE/MPA Koronare Herzerkrankungen 0, 91 ns 1, 24 ns (im 1. Jahr 1, 81*) Schlaganfall 1, 39* 1, 31* ischämisch 1, 44* hämorrhagisch 0, 82 ns Venöse Thromboembolien 1, 33 ns 2, 06* Übergewicht 3, 80* Adipositas 5, 61* Mammakarzinom 0, 77 ns 1, 24* Kolonkarzinom 1, 08 0, 61* Oberschenkelhals-Frakturen 0, 61* 0, 66*
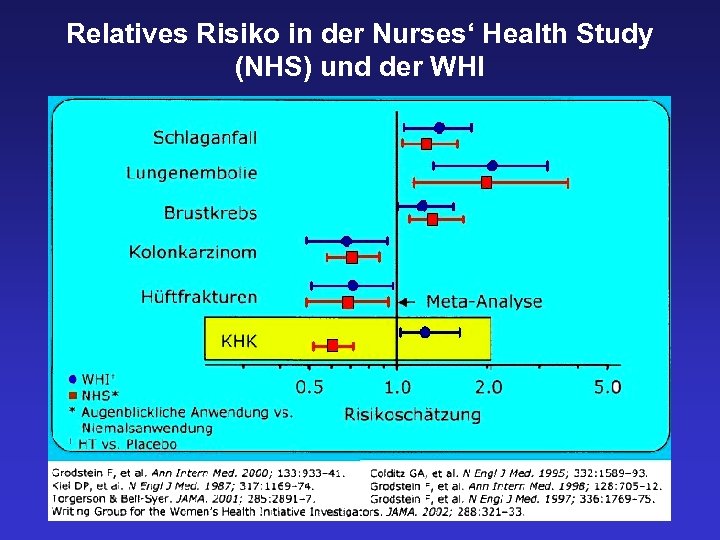
Relatives Risiko in der Nurses‘ Health Study (NHS) und der WHI

HRT Credo – HRT stellt die beste und günstigste Therapie der Wechseljahresbeschwerden dar. – Östrogene wirken osteoprotektiv. – Östrogene reduzieren das Risiko kolorektaler Carcinome.

Mechanisms to resist and repair damage • Zelluläre • Endokrinologische • Lifestyle, Therapeutische (Hormonersatz, HRT, Alternatives)

Alternativen zur HRT • Kräuterextrakte können klimakterische Beschwerden lindern – ähnlich wie ein Placebo. • Phytoestrogene können die Knochenresorption verlangsamen; eine Senkung des Frakturrisikos wurde jedoch nicht gezeigt. • a-adrenerge Agonisten (z. B. Clonidin) haben eine moderate Wirkung auf Hitzewallungen. • hoch-dosierte Gestagene (5 -10 mg NETA, 20 -40 mg MPA oder Megestrolacetat / Tag) führen zu einer wirksamen Reduktion der Hitzewallungen. Die Langzeit-Auswirkungen sind bisher nicht untersucht.

Alternativen zur HRT • Tibolon bessert klimakterische Beschwerden und erhält den Knochen. • Eine Senkung des Frakturrisikos konnte nicht gezeigt werden. • Neuroaktive Medikamente (z. B. Selektive Serotonin-Wiederaufnahmehemmer: SSRIs) haben eine moderate Wirkung auf vasomotorische Beschwerden. Therapieversuch möglich, wenn eine HRT nicht geeignet ist. • Gabapentin kann Hitzewallungen reduzieren.

Conclusions • Übe Nachsicht mit Störfällen


Zusammenfassung • Menpause: subakuter Verlust der Ovarfunktion • Endokrinologisch gesehen ein Absturz • Zahlreiche Kompensations- und Reparaturmöglichkeiten:

Zusammenfassung • Kompensations- und Reparaturmöglichkeiten: • Lifestyle: Bewegung, Ernährung • HRT (beachte die Packungsbeilage) • Alternativen zur HRT

Zusammenfassung • Absturz? • Individuell gesehen Chance zu Veränderung / Persönlichkeitswachstum • Zwingt zur Auseinandersetzung mit Thema Altern

Der wahre Sinn des Lebens besteht darin, Bäume zu pflanzen, unter deren Schatten man vermutlich selber nie sitzen wird Nelson Henderson

Übliches zur Menopause Bruno Müller www. Der. Endokrinologe. ch 3010 Bern

The International Menopause Society The IMS Updated Recommendations on postmenopausal hormone therapy February 27, 2007 Climacteric 2007; 10: 181– 94

Introducing The International Menopause Society The society for the study of all aspects of the climacteric in men and women • Established in 1978 • Registered as a non-profit • organization in Geneva, Switzerland Central Office in Lancaster, UK

Introducing The International Menopause Society Officers and Board, 2005– 2008 Officers President: Amos Pines, Israel General Secretary: David Sturdee, UK Treasurer: Martin Birkhäuser, Switzerland Board members Mark Brincat, Malta Santiago Palacios, Spain Tobie De Villiers, South Africa James Pickar, USA Marco Gambacciani, Italy Regine Sitruk-Ware, USA Kobchitt Limpaphayom, Thailand Sven Skouby, Denmark Frederick Naftolin, USA Executive Director: Jean Wright, UK

Introducing The International Menopause Society The Society’s Journal, Climacteric • Editors-in-Chief: David W. Sturdee, UK • • and Alastair H. Mac. Lennan, Australia Published in six issues per year plus Supplements Indexed in Index Medicus, Medline, Current Contents Impact factor: 2. 299 11 th of 52 journals in Obstetrics & Gynecology section

Introduction • The following Recommendations express the • • views of the IMS on the principles of hormone therapy (HT) in the peri- and postmenopause periods Throughout the Recommendations, the term HT will be used to cover all therapies including estrogens, progestogens, combined therapies and tibolone The 2004 IMS Statement is still valid and serves as a basis for the current updated Recommendations Climacteric 2007; 10: 181– 94

Introduction • The IMS is aware of possible geographical variations related to different priorities of medical care, different prevalence of diseases, and country-specific attitudes of the public, the medical community and the health authorities toward menopause management, which may all impact on hormone therapy Climacteric 2007; 10: 181– 94

Introduction • The following recommendations, therefore, give • • a global and simple overview that serves as a common platform on issues related to the various aspects of hormone treatment These Recommendations were reviewed and discussed by representatives of more than 60 national and regional menopause societies from all continents These Recommendations can be easily adapted and modified according to local needs Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Hormone therapy should be part of an overall strategy including lifestyle recommendations regarding diet, exercise, smoking and alcohol for maintaining the health of postmenopausal women Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • HT must be individualized and tailored according to • • • symptoms and the need for prevention, as well as personal and family history, results of relevant investigations, the woman’s preferences and expectations The risks and benefits of HT differ for women around the time of menopause compared to those for older women HT includes a wide range of hormonal products and routes of administration, with potentially different risks and benefits The term ‘class effect’, when associated with HT, is confusing and inappropriate Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Women experiencing spontaneous or • iatrogenic menopause before the age of 45 and particularly before 40 are at higher risk for cardiovascular disease and osteoporosis They will benefit from hormone replacement, which should be given at least until the normal age of menopause Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Counseling should convey the benefits and • risks of HT in simple terms, e. g. absolute numbers rather than as percentage changes from baseline expressed as a relative risk This allows a woman and her physician to make a well-informed decision about HT Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • HT should not be recommended without a clear indication for its use Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Women taking HT should have at least an annual • • consultation to include a physical examination, update of medical history, relevant laboratory and imaging investigations and a discussion on lifestyle There are no reasons to place mandatory limitations on the length of treatment Whether or not to continue therapy should be decided at the discretion of the well-informed hormone user and her health professional, dependent upon the specific goals and an objective estimation of benefits and risks Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Dosage should be titrated to the lowest • • effective dose Lower doses of HT than have been used routinely can maintain quality of life in a large proportion of users Long-term data on lower doses regarding fracture risk and cardiovascular implications are still lacking Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Progestogen should be added to systemic • estrogen for all women with a uterus to prevent endometrial hyperplasia and cancer Natural progesterone and some progestogens have specific beneficial effects that could justify their use besides the expected actions on the endometrium Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Low-dose vaginal estrogens administered for • the relief of urogenital atrophy do not require progestogen co-medication Direct delivery of progestogen to the endometrial cavity from the vagina or by an intrauterine system is logical and may minimize systemic effects Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part I. Governing principles • Androgen replacement should be reserved for • women with clinical signs and symptoms of androgen insufficiency In women with bilateral oophorectomy or adrenal failure, androgen replacement has significant beneficial effects, in particular on health-related quality of life and sexual function Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: General • HT remains the most effective therapy for • vasomotor and estrogen-deficient urogenital symptoms Other menopause-related complaints, such as joint and muscle pains, mood swings, sleep disturbances and sexual dysfunction (including reduced libido) may improve during HT Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: General • Quality of life and sexuality are key factors • to be considered in the management of the aging individual The administration of individualized HT (including androgenic preparations when appropriate) improves both sexuality and overall quality of life Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: Postmenopausal osteoporosis • HT is effective in preventing the bone loss associated • with the menopause and decreases the incidence of all osteoporosis-related fractures, including vertebral and hip, even in patients at low risk Although the magnitude of decline in bone turnover correlates with estrogen dosage, even lower than standard-dose preparations maintain a positive influence on bone indices in most women Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: Postmenopausal osteoporosis • HT is an appropriate first-line therapy in • postmenopausal women presenting with an increased risk for fracture, particularly under the age of 60 years and for the prevention of bone loss in women with premature menopause The protective effect of HT on bone mineral density declines after cessation of therapy at an unpredictable rate, although some degree of fracture protection may remain after cessation of HT Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: Postmenopausal osteoporosis • The initiation of standard-dose HT is not • recommended for the sole purpose of the prevention of fractures after the age of 60 years The continuation of HT after the age of 60 for the sole purpose of the prevention of fractures should take into account the possible long-term effects of the specific dose and method of administration of HT, compared to other proven therapies Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: Cardiovascular disease • Cardiovascular disease is the principal cause of • • morbidity and mortality in postmenopausal women Major primary prevention measures (besides smoking cessation, and diet control) are weight loss, blood pressure reduction, and diabetes and lipid control There is evidence that HT may be cardioprotective if started around the time of menopause and continued long-term (often referred to as the ‘window of opportunity’ concept) Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: Cardiovascular disease • HT markedly reduces the risk of diabetes and, through • • improved insulin resistance, it has positive effects on other related risk factors for cardiovascular disease such as the lipid profile and metabolic syndrome In women less than 60 years old, recently menopausal, without prevalent cardiovascular disease, the initiation of HT does not cause early harm, and may reduce cardiovascular morbidity and mortality Continuation of HT beyond the age of 60 should be decided as a part of the overall risk-benefit analysis Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part II. Benefits of hormone therapy: Other • HT may reduce the risk of colon cancer • HT initiated around the time of menopause • or by younger postmenopausal women is associated with a reduced risk of Alzheimer’s disease HT has benefits for connective tissue, skin, joints and intervertebral disks Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT • Studies on the risks of postmenopausal hormone use have mainly focused on breast and endometrial cancer, venous thromboembolism (pulmonary embolism or deep vein thrombosis), stroke and coronary events Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Breast cancer • The incidence of breast cancer varies in different countries. Therefore, currently available data cannot necessarily be generalized Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Breast cancer • The degree of association between breast cancer and postmenopausal HT remains controversial. Women should be reassured that the possible risk of breast cancer associated with HT is small (less than 0. 1% per annum) Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Breast cancer • For combined HT, observational data from the Million • Women Study suggested that breast cancer risk was increased as early as the first year, raising serious reservations on possible methodologic flaws On the contrary, randomized controlled data from the Women‘s Health Initiative (WHI) Study indicate that no increased risk is observed in women initiating HT, for up to 7 years. It should be noted that the majority of subjects in the WHI Study were overweight or obese Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Breast cancer • Data from the WHI and Nurses’ Health Study suggest that • long-term estrogen-only administration for 7 and 15 years, respectively, does not increase the risk of breast cancer in American women. Recent European observational studies suggest that risk may increase after 5 years There are insufficient data to evaluate the possible differences in the incidence of breast cancer using different types and routes of estrogen, natural progesterone and progestogens, androgen administration Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Breast cancer • Baseline mammographic density correlates • with breast cancer risk. This does not necessarily apply to the increase in mammographic density induced by HT The combined estrogen–progestogen therapy-related increase in mammographic density may impede the diagnostic interpretation of mammograms Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Endometrial cancer • Unopposed estrogen administration induces a dose-related • • stimulation of the endometrium Women with a uterus should have progestogen supplementation Continuous combined estrogen–progestogen regimens are associated with a lower incidence of endometrial hyperplasia and cancer than occurs in the normal population Direct intrauterine delivery systems may have advantages Regimens containing low-/ultra-low-dose estrogen and progestogen cause less endometrial stimulation and less bleeding Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Thromboembolism and cardiovascular events • The HT-related risk for serious venous thromboembolic • • events increases with age (although minimal until age 60) and is also positively associated with obesity and thrombophilia By avoiding first-pass hepatic metabolism, transdermal estrogen may avert the risk associated with oral HT The impact on the risk of a thromboembolic event may also be affected by progestogen, depending on the type Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part III. Potential serious adverse effects of HT: Thromboembolism and cardiovascular events • Late starters of standard-dose HT may have a • • transient slightly increased risk for coronary events The risk of stroke is correlated with age. HT may increase the risk of stroke after the age of 60 Safety data from studies of low-dose and ultralow-dose regimens of estrogen and progestogen are encouraging Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part IV: Alternative treatments • The efficacy and safety of complementary • alternative medicines have not been demonstrated and further studies are required Selective serotonin reuptake inhibitors, selective noradrenaline reuptake inhibitors and gabapentin are effective in reducing vasomotor symptoms in short-term studies. Their long-term safety needs further evaluation Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part IV: Alternative treatments • There are no medical or scientific reasons • • to recommend unregistered ‘bioidentical hormones’ The measurement of hormone levels in the saliva is not clinically useful These ‘customized’ hormonal preparations have not been tested in studies, and their purity and risks are unknown Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part V: Conclusions • There is urgent need for further research, especially into the relative merits of lower doses, regimens and routes of administration Climacteric 2007; 10: 181– 94

IMS UPDATED RECOMMENDATIONS ON POSTMENOPAUSAL HORMONE THERAPY Part V: Conclusions • The safety of HT largely depends on age • Women younger than 60 years should not be • concerned about the safety profile of HT New data and re-analyses of older studies by women’s age show that, for most women, the potential benefits of HT given for a clear indication are many and the risks are few when initiated within a few years of menopause Climacteric 2007; 10: 181– 94

Adjunctive slides • The following slides may be useful for • • presentation in regard to the IMS Recommendations Some slides demonstrate data on which the statements are based. This is not, however, a full slide presentation on specific topics. The IMS is now in the process of developing an Educational Slide Kit on the main issues of adult women’s health and menopause
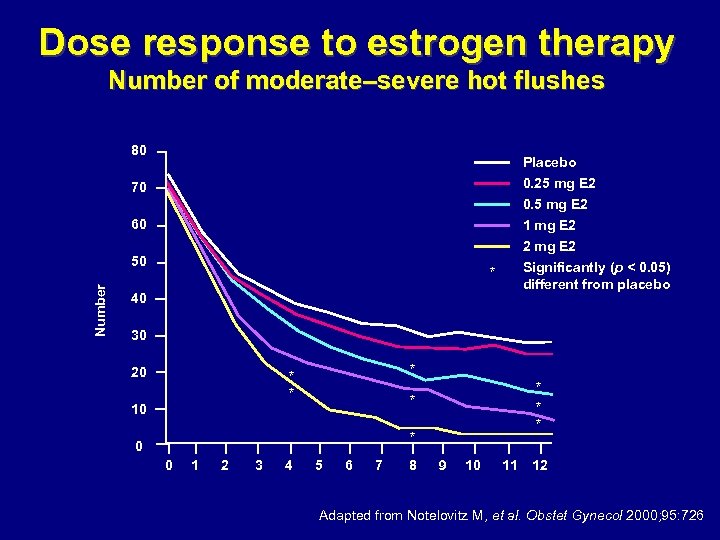
Dose response to estrogen therapy Number of moderate–severe hot flushes 80 Placebo 0. 25 mg E 2 70 0. 5 mg E 2 1 mg E 2 2 mg E 2 Significantly (p < 0. 05) different from placebo 60 Number 50 * 40 30 20 * * * * 10 * 0 0 1 2 3 4 5 6 7 8 9 10 11 12 Adapted from Notelovitz M, et al. Obstet Gynecol 2000; 95: 726
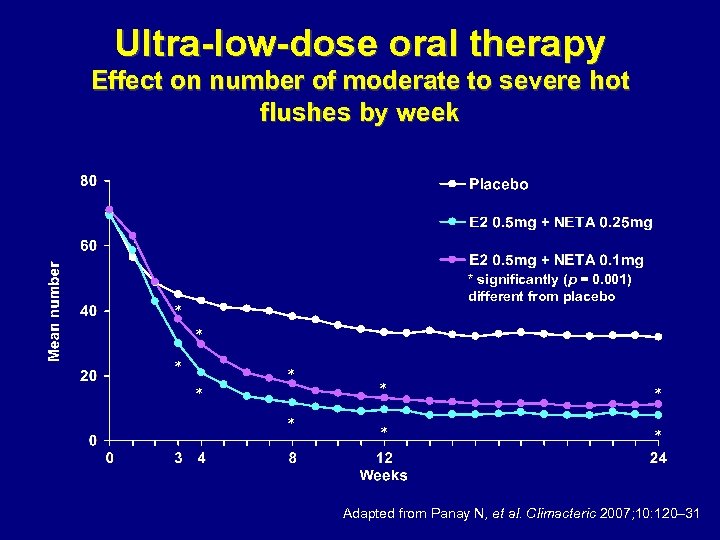
Ultra-low-dose oral therapy Effect on number of moderate to severe hot flushes by week * significantly (p = 0. 001) different from placebo * * * * * Adapted from Panay N, et al. Climacteric 2007; 10: 120– 31
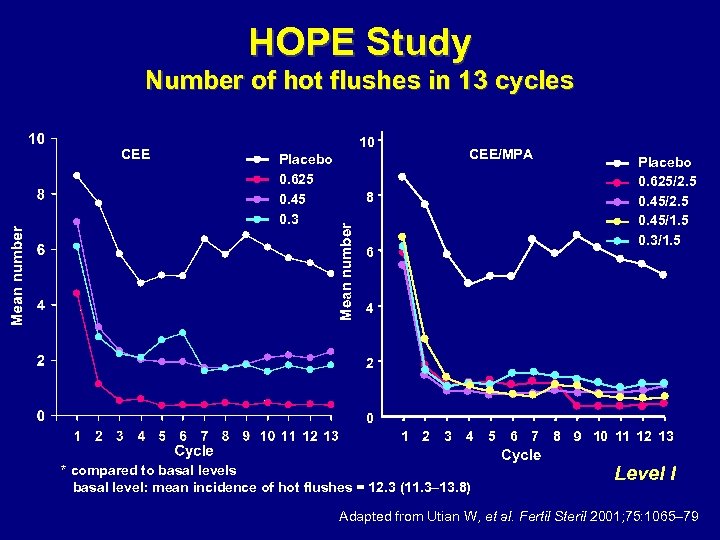
HOPE Study Number of hot flushes in 13 cycles 10 Placebo 0. 625 0. 45 0. 3 CEE/MPA 8 Mean number CEE 6 Placebo 0. 625/2. 5 0. 45/1. 5 0. 3/1. 5 4 2 0 1 2 3 4 * compared to basal levels basal level: mean incidence of hot flushes = 12. 3 (11. 3– 13. 8) 5 6 7 Cycle 8 9 10 11 12 13 Level I Adapted from Utian W, et al. Fertil Steril 2001; 75: 1065– 79
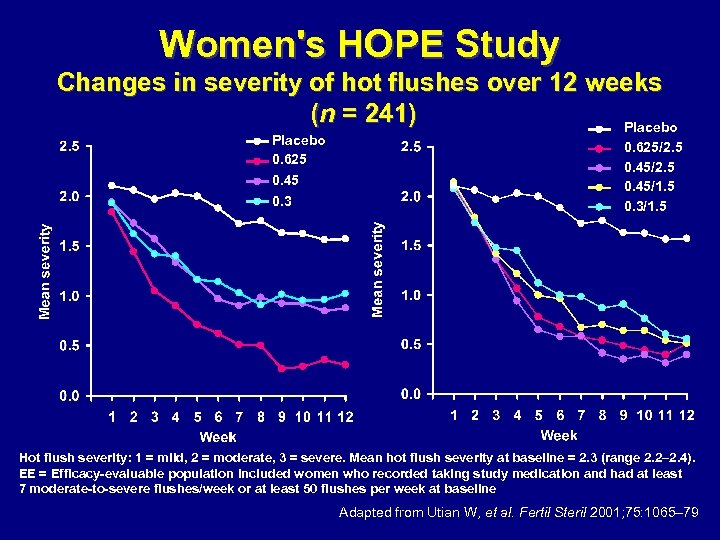
Women's HOPE Study Changes in severity of hot flushes over 12 weeks (n = 241) Placebo 0. 625 0. 45 0. 3 0. 625/2. 5 0. 45/1. 5 0. 3/1. 5 Hot flush severity: 1 = mild, 2 = moderate, 3 = severe. Mean hot flush severity at baseline = 2. 3 (range 2. 2– 2. 4). EE = Efficacy-evaluable population included women who recorded taking study medication and had at least 7 moderate-to-severe flushes/week or at least 50 flushes per week at baseline Adapted from Utian W, et al. Fertil Steril 2001; 75: 1065– 79

Unopposed ultra-low-dose transdermal estradiol • 417 postmenopausal women (60– 80 years) • • • mean 67 ± 5 years Randomly assigned to placebo or transdermal 14 µg/day for 2 years Baseline serum E 2 = 4. 8 pg/ml On treatment E 2 = 8. 6 pg/ml Johnson SR, et al. Obstet Gynecol 2005; 105: 779– 87

Unopposed ultra-low-dose transdermal estradiol Endometrial effects: • Proliferation 8. 5% vs. 1. 1% p = 0. 6 • Bleeding 12. 4% vs. 8. 6% p = 0. 3 • Atypical hyperplasia × 1 • Adenosarcoma × 1 Conclusions: ‘This therapy apparently causes little or no endometrial stimulation’ Johnson SR, et al. Obstet Gynecol 2005; 105: 779– 87
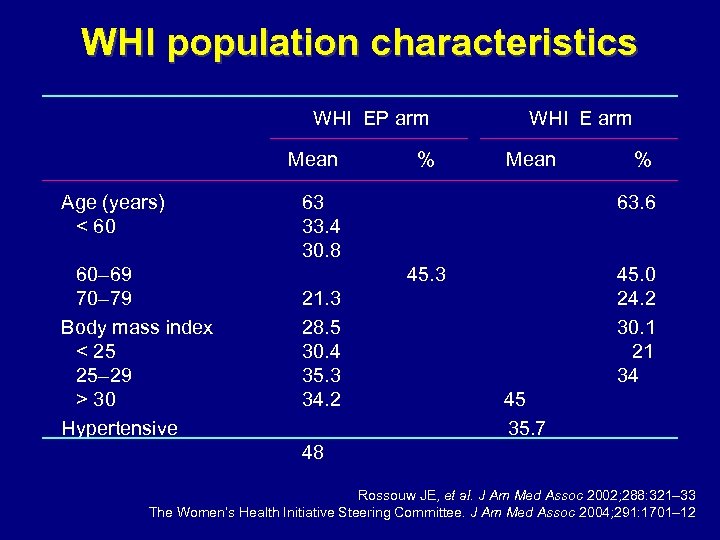
WHI population characteristics WHI EP arm Mean Age (years) < 60 60– 69 70– 79 Body mass index < 25 25– 29 > 30 Hypertensive % WHI E arm Mean 63 33. 4 30. 8 63. 6 45. 3 21. 3 28. 5 30. 4 35. 3 34. 2 % 45. 0 24. 2 30. 1 21 34 45 35. 7 48 Rossouw JE, et al. J Am Med Assoc 2002; 288: 321– 33 The Women’s Health Initiative Steering Committee. J Am Med Assoc 2004; 291: 1701– 12
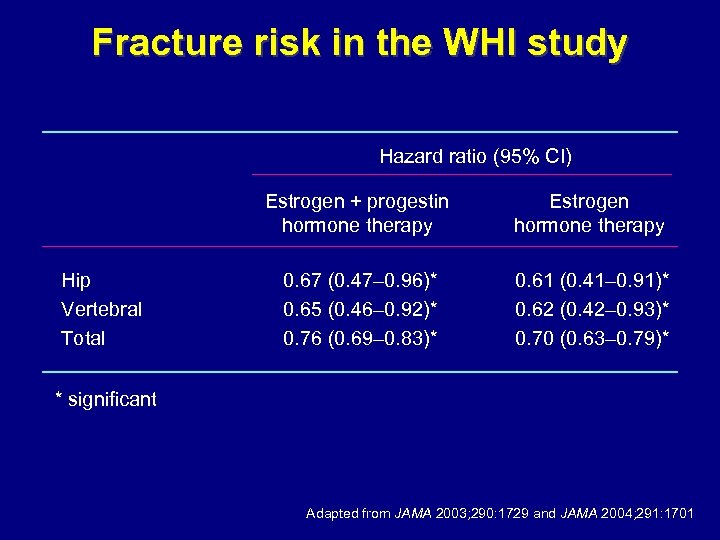
Fracture risk in the WHI study Hazard ratio (95% CI) Estrogen + progestin hormone therapy Hip Vertebral Total Estrogen hormone therapy 0. 67 (0. 47– 0. 96)* 0. 65 (0. 46– 0. 92)* 0. 76 (0. 69– 0. 83)* 0. 61 (0. 41– 0. 91)* 0. 62 (0. 42– 0. 93)* 0. 70 (0. 63– 0. 79)* * significant Adapted from JAMA 2003; 290: 1729 and JAMA 2004; 291: 1701

WHI: unopposed estrogen Estrogen (n = 5076) Placebo (n = 5196) Hazard ratio (95% CI) p Total joint replacement 119 169 0. 73 (0. 58– 0. 93) 0. 01 Hip joint replacement 28 53 0. 55 (0. 35– 0. 88) 0. 01 Knee joint replacement 93 121 0. 8 (0. 61– 1. 05) 0. 11 Compliance more than 80% Adapted from Cirillo, et al. Arthritis Rheumatism 2006
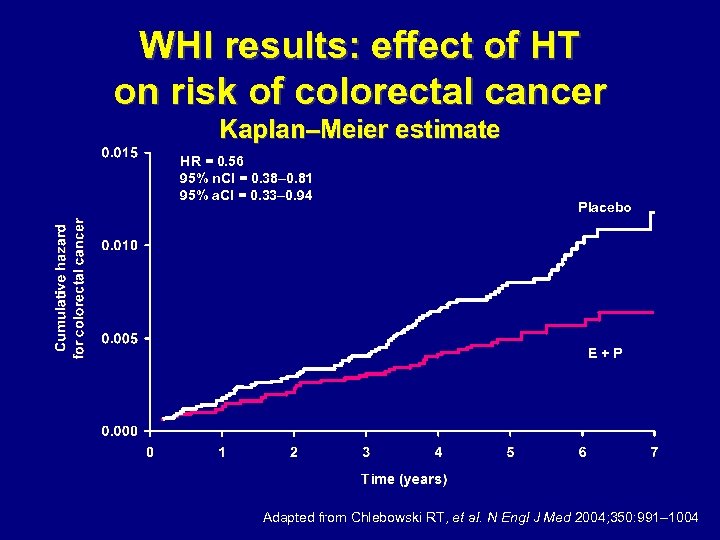
WHI results: effect of HT on risk of colorectal cancer Kaplan–Meier estimate HR = 0. 56 95% n. CI = 0. 38– 0. 81 95% a. CI = 0. 33– 0. 94 Placebo E+P Adapted from Chlebowski RT, et al. N Engl J Med 2004; 350: 991– 1004
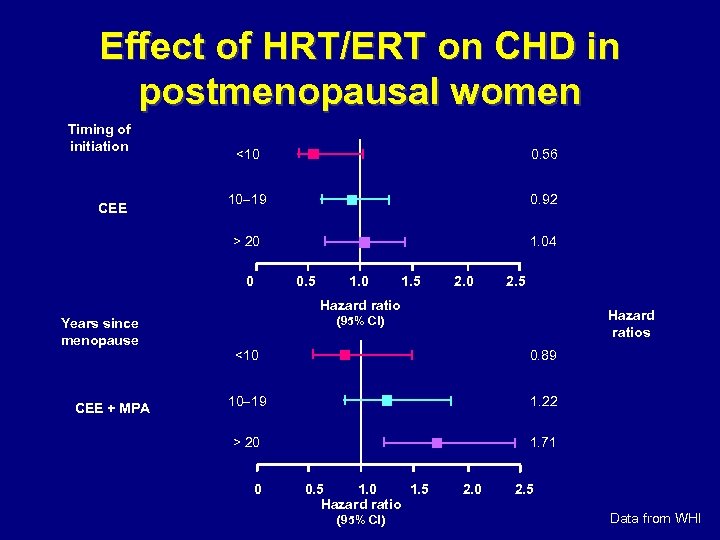
Effect of HRT/ERT on CHD in postmenopausal women Timing of initiation 0. 56 10 19 0. 92 > 20 CEE <10 1. 04 0 0. 5 1. 0 1. 5 2. 0 2. 5 Hazard ratio Years since menopause CEE + MPA Hazard ratios (95% CI) <10 0. 89 10 19 1. 22 > 20 1. 71 0 0. 5 1. 0 1. 5 Hazard ratio (95% CI) 2. 0 2. 5 Data from WHI
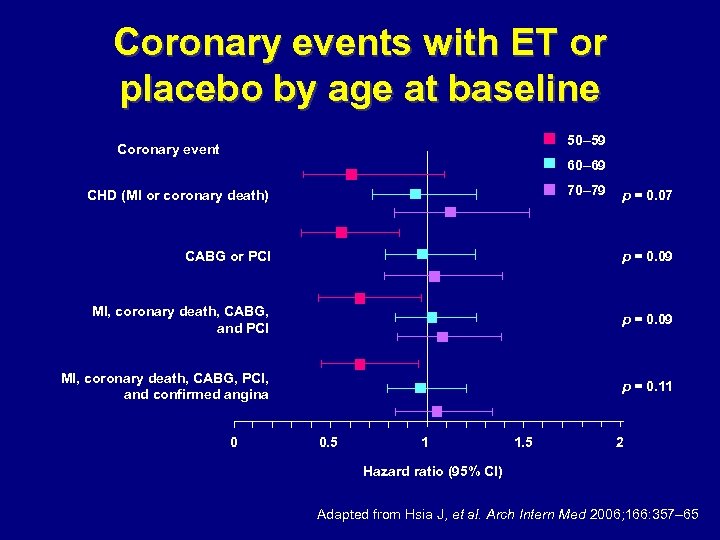
Coronary events with ET or placebo by age at baseline 50– 59 Coronary event 60– 69 70– 79 CHD (MI or coronary death) p = 0. 07 CABG or PCI p = 0. 09 MI, coronary death, CABG, and PCI p = 0. 09 MI, coronary death, CABG, PCI, and confirmed angina p = 0. 11 0 0. 5 1 1. 5 2 Hazard ratio (95% CI) Adapted from Hsia J, et al. Arch Intern Med 2006; 166: 357– 65
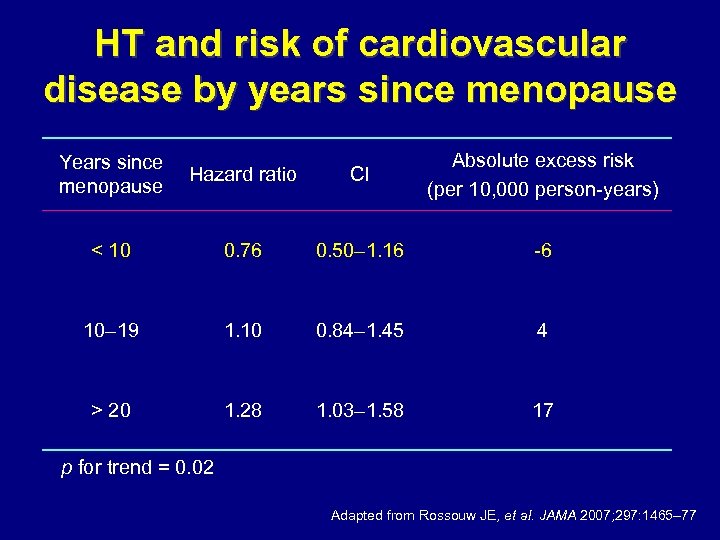
HT and risk of cardiovascular disease by years since menopause Years since menopause Hazard ratio CI Absolute excess risk (per 10, 000 person-years) < 10 0. 76 0. 50– 1. 16 -6 10– 19 1. 10 0. 84– 1. 45 4 > 20 1. 28 1. 03– 1. 58 17 p for trend = 0. 02 Adapted from Rossouw JE, et al. JAMA 2007; 297: 1465– 77
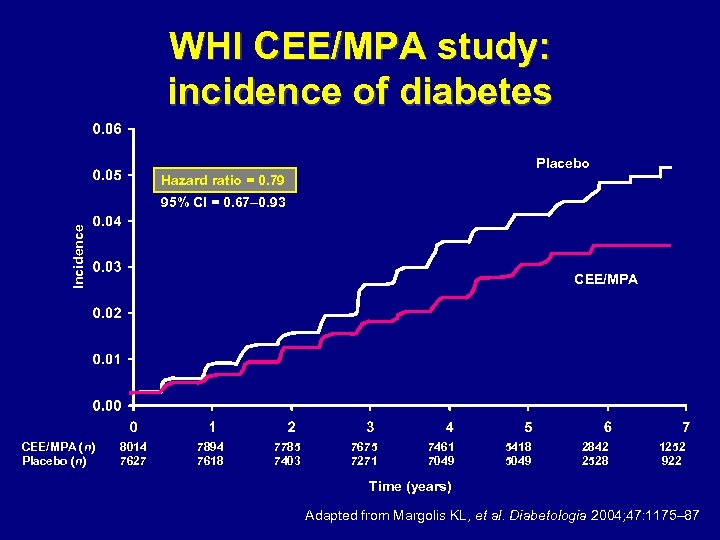
WHI CEE/MPA study: incidence of diabetes Placebo Hazard ratio = 0. 79 Incidence 95% CI = 0. 67– 0. 93 CEE/MPA (n) Placebo (n) CEE/MPA 8014 7627 7894 7618 7785 7403 7675 7271 7461 7049 5418 5049 2842 2528 1252 922 Time (years) Adapted from Margolis KL, et al. Diabetologia 2004; 47: 1175– 87
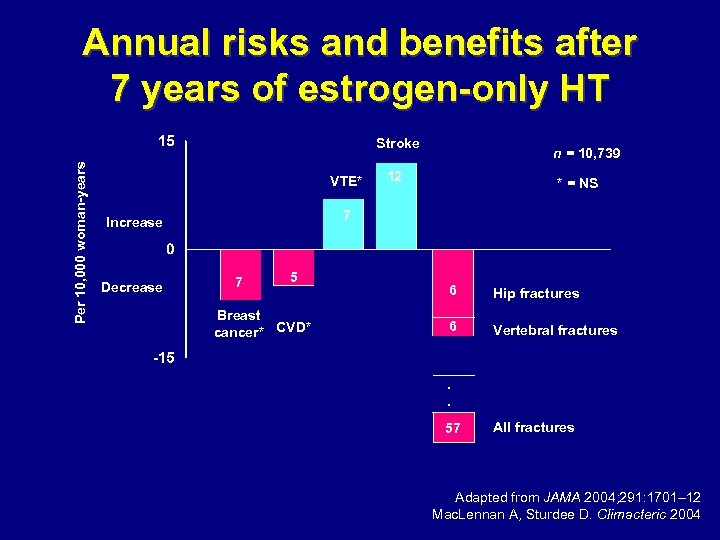
Annual risks and benefits after 7 years of estrogen-only HT Per 10, 000 woman-years Stroke VTE* 12 * = NS 7 Increase Decrease n = 10, 739 7 5 Breast cancer* CVD* 6 Hip fractures 6 Vertebral fractures . . , 57 All fractures Adapted from JAMA 2004; 291: 1701– 12 Mac. Lennan A, Sturdee D. Climacteric 2004
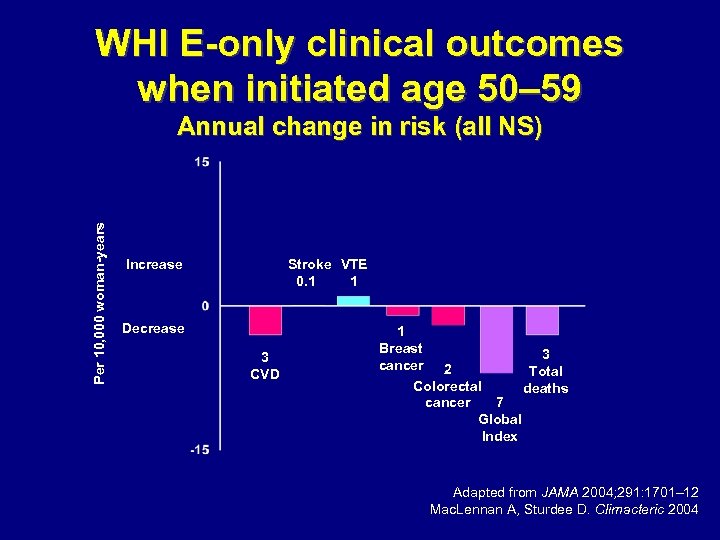
WHI E-only clinical outcomes when initiated age 50– 59 Per 10, 000 woman-years Annual change in risk (all NS) Stroke VTE 0. 1 1 Increase Decrease 3 CVD 1 Breast 3 cancer 2 Total Colorectal deaths 7 cancer Global Index Adapted from JAMA 2004; 291: 1701– 12 Mac. Lennan A, Sturdee D. Climacteric 2004
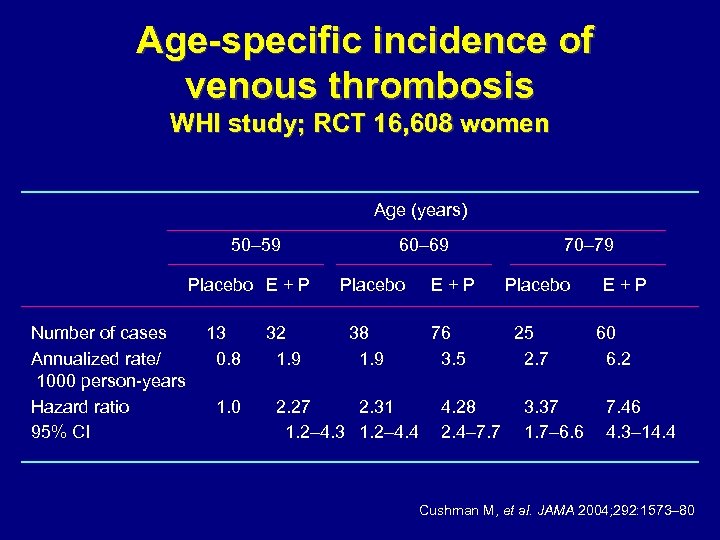
Age-specific incidence of venous thrombosis WHI study; RCT 16, 608 women Age (years) 50– 59 Placebo E + P Number of cases Annualized rate/ 1000 person-years Hazard ratio 95% CI 13 0. 8 1. 0 32 1. 9 60– 69 Placebo 38 1. 9 2. 27 2. 31 1. 2– 4. 3 1. 2– 4. 4 E+P 76 3. 5 4. 28 2. 4– 7. 7 70– 79 Placebo 25 2. 7 3. 37 1. 7– 6. 6 E+P 60 6. 2 7. 46 4. 3– 14. 4 Cushman M, et al. JAMA 2004; 292: 1573– 80
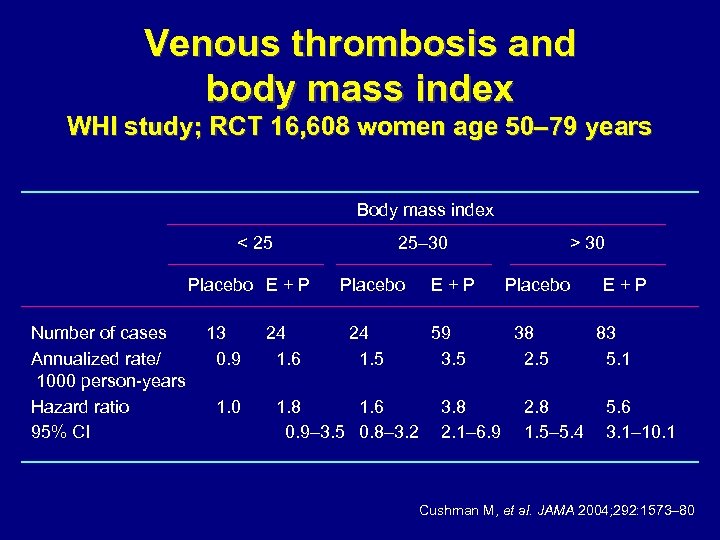
Venous thrombosis and body mass index WHI study; RCT 16, 608 women age 50– 79 years Body mass index < 25 25– 30 Placebo E + P Number of cases Annualized rate/ 1000 person-years Hazard ratio 95% CI 13 0. 9 1. 0 24 1. 6 Placebo 24 1. 5 1. 8 1. 6 0. 9– 3. 5 0. 8– 3. 2 E+P 59 3. 5 3. 8 2. 1– 6. 9 > 30 Placebo 38 2. 5 2. 8 1. 5– 5. 4 E+P 83 5. 1 5. 6 3. 1– 10. 1 Cushman M, et al. JAMA 2004; 292: 1573– 80

VTE: route of administration and progestogens ESTHER study Route/progestogen Odds ratio 95% CI Oral 4. 2 1. 5– 11. 6 Transdermal 0. 9 0. 4– 2. 1 Micronized progesterone 0. 7 0. 3– 1. 9 Pregnanes 0. 9 0. 4– 2. 3 Norpregnanes 3. 9 1. 5– 10. 0 Canonico M, et al. Circulation 2007; 115: 820– 2
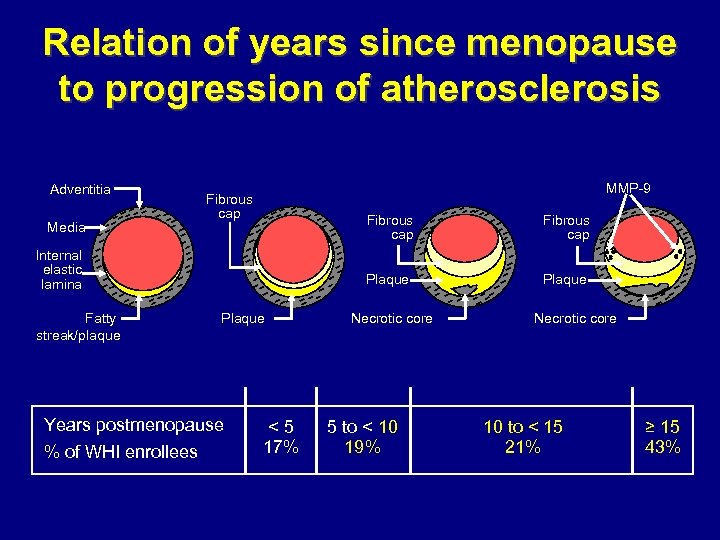
Relation of years since menopause to progression of atherosclerosis Adventitia Media MMP-9 Fibrous cap Plaque Internal elastic lamina Fatty streak/plaque Plaque Years postmenopause % of WHI enrollees <5 17% Fibrous cap Plaque Necrotic core 5 to < 10 19% Necrotic core 10 to < 15 21% ≥ 15 43%
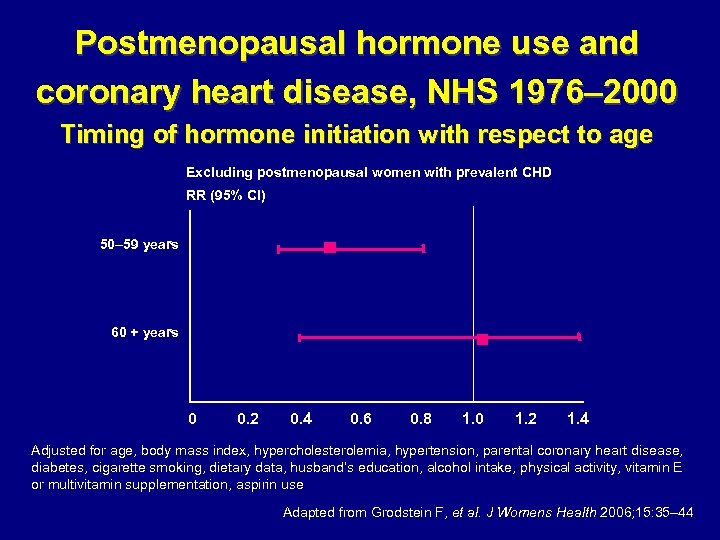
Postmenopausal hormone use and coronary heart disease, NHS 1976– 2000 Timing of hormone initiation with respect to age Excluding postmenopausal women with prevalent CHD RR (95% CI) 50– 59 years 60 + years 0 0. 2 0. 4 0. 6 0. 8 1. 0 1. 2 1. 4 Adjusted for age, body mass index, hypercholesterolemia, hypertension, parental coronary heart disease, diabetes, cigarette smoking, dietary data, husband’s education, alcohol intake, physical activity, vitamin E or multivitamin supplementation, aspirin use Adapted from Grodstein F, et al. J Womens Health 2006; 15: 35– 44
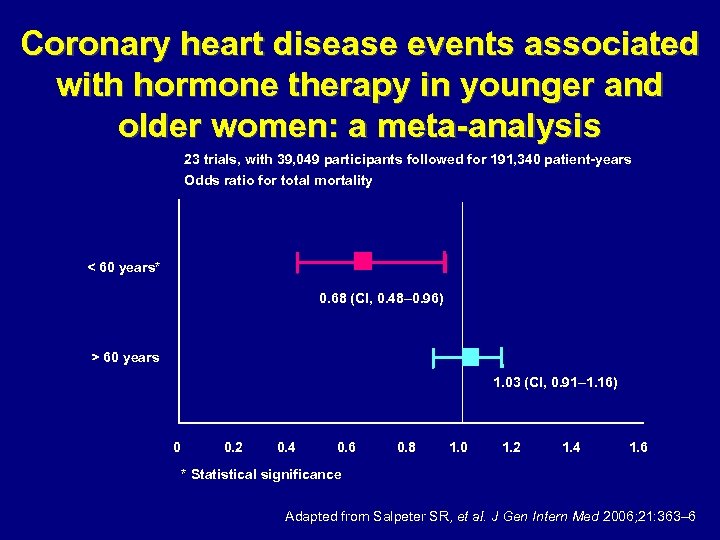
Coronary heart disease events associated with hormone therapy in younger and older women: a meta-analysis 23 trials, with 39, 049 participants followed for 191, 340 patient-years Odds ratio for total mortality < 60 years* 0. 68 (CI, 0. 48– 0. 96) > 60 years 1. 03 (CI, 0. 91– 1. 16) 0 0. 2 0. 4 0. 6 0. 8 1. 0 1. 2 1. 4 1. 6 * Statistical significance Adapted from Salpeter SR, et al. J Gen Intern Med 2006; 21: 363– 6

Summary of published results on incidence of endometrial cancer in relation to use of hormone therapy (HT) Type of HT Relative risk (95% CI) Unopposed estrogen < 1 year 1– 4 years 5– 9 years 10+ years Sequential E + P; ever vs. never CCEPT from WHI 1. 4 (1. 0– 1. 8) 2. 8 (2. 3– 3. 5) 5. 9 (4. 7– 7. 5) 9. 5 (7. 4– 12. 3) 1. 3 (1. 1– 1. 4) 0. 8 (0. 5– 1. 4) CCEPT, continuous combined estrogen and progestogen; E, estrogen; P, progestogen Adapted from Grady D, et al. Obstet Gynecol 1995; 85: 304– 13 Beral V, et al. J Epidemiol Biostat 1999; 4: 191– 215 Anderson GL, et al. JAMA 2003; 290: 1739– 44
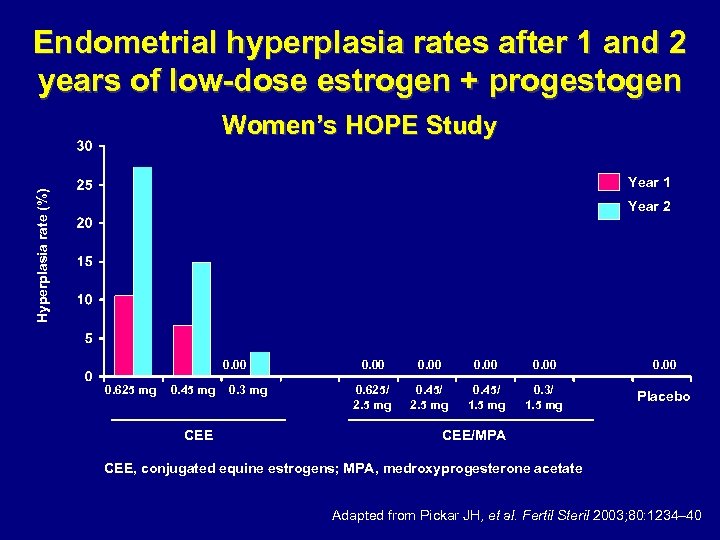
Endometrial hyperplasia rates after 1 and 2 years of low-dose estrogen + progestogen Women’s HOPE Study Hyperplasia rate (%) Year 1 Year 2 0. 00 0. 625 mg 0. 45 mg CEE 0. 3 mg 0. 00 0. 625/ 2. 5 mg 0. 45/ 1. 5 mg 0. 3/ 1. 5 mg Placebo CEE/MPA CEE, conjugated equine estrogens; MPA, medroxyprogesterone acetate Adapted from Pickar JH, et al. Fertil Steril 2003; 80: 1234– 40
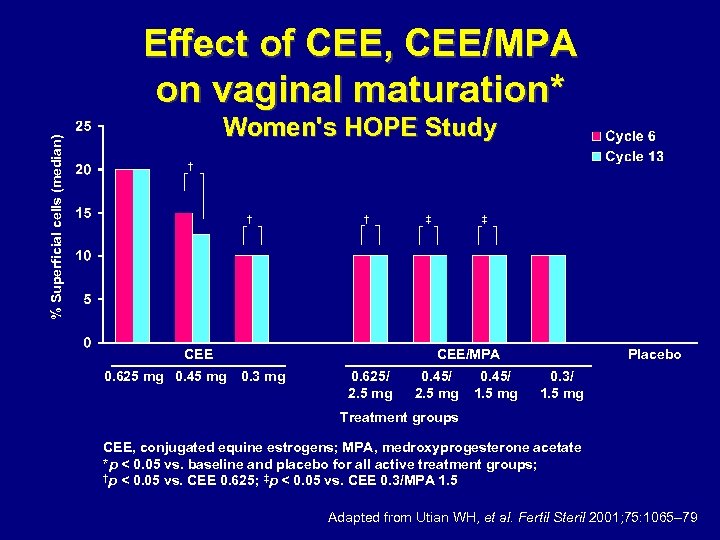
% Superficial cells (median) Effect of CEE, CEE/MPA on vaginal maturation* Women's HOPE Study † † † CEE 0. 625 mg 0. 45 mg 0. 3 mg ‡ ‡ CEE/MPA 0. 625/ 2. 5 mg 0. 45/ 1. 5 mg Placebo 0. 3/ 1. 5 mg Treatment groups CEE, conjugated equine estrogens; MPA, medroxyprogesterone acetate *p < 0. 05 vs. baseline and placebo for all active treatment groups; †p < 0. 05 vs. CEE 0. 625; ‡p < 0. 05 vs. CEE 0. 3/MPA 1. 5 Adapted from Utian WH, et al. Fertil Steril 2001; 75: 1065– 79
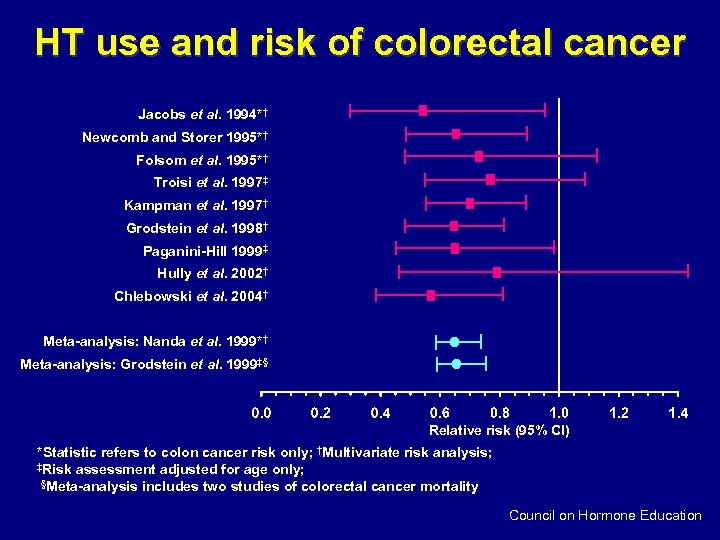
HT use and risk of colorectal cancer Jacobs et al. 1994*† Newcomb and Storer 1995*† Folsom et al. 1995*† Troisi et al. 1997‡ Kampman et al. 1997† Grodstein et al. 1998† Paganini-Hill 1999‡ Hully et al. 2002† Chlebowski et al. 2004† Meta-analysis: Nanda et al. 1999*† Meta-analysis: Grodstein et al. 1999‡§ Relative risk (95% CI) *Statistic refers to colon cancer risk only; †Multivariate risk analysis; ‡Risk assessment adjusted for age only; §Meta-analysis includes two studies of colorectal cancer mortality Council on Hormone Education
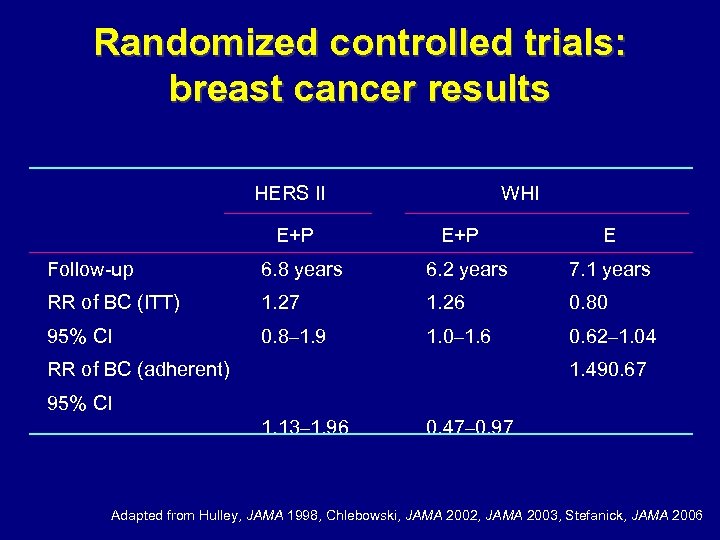
Randomized controlled trials: breast cancer results HERS II E+P WHI E+P E Follow-up 6. 8 years 6. 2 years 7. 1 years RR of BC (ITT) 1. 27 1. 26 0. 80 95% CI 0. 8– 1. 9 1. 0– 1. 6 0. 62– 1. 04 RR of BC (adherent) 1. 490. 67 95% CI 1. 13– 1. 96 0. 47– 0. 97 Adapted from Hulley, JAMA 1998, Chlebowski, JAMA 2002, JAMA 2003, Stefanick, JAMA 2006

Body mass index: the risk with hormone therapy is (more) apparent in lean women • BMI > 24. 4 kg/m 2 – no additional risk • • • Schairer C, et al. JAMA 2000; 283: 485– 91 BMI > 26 kg/m 2 – no additional risk Rosenberg L, et al. Arch Intern Med 2006; 166: 760– 5 Inverse relationship between the risk and BMI with estrogen or combined hormone therapy Million Women Study. Reeves GK, et al. Lancet Oncol 2006; 7: 910– 18 80% of users have a BMI < 25 E 3 N-EPIC. Fournier A, et al. Int J Cancer 2005; 114: 448– 54
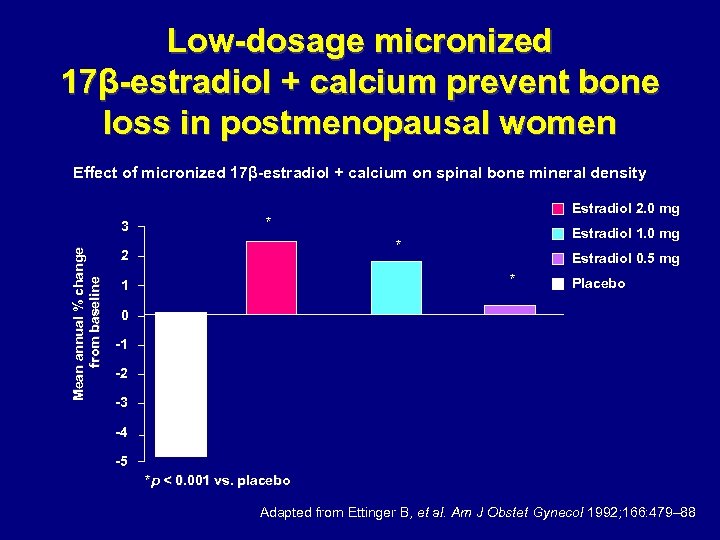
Low-dosage micronized 17β-estradiol + calcium prevent bone loss in postmenopausal women Effect of micronized 17β-estradiol + calcium on spinal bone mineral density Mean annual % change from baseline 3 Estradiol 2. 0 mg * Estradiol 1. 0 mg * 2 Estradiol 0. 5 mg * 1 Placebo 0 -1 -2 -3 -4 -5 *p < 0. 001 vs. placebo Adapted from Ettinger B, et al. Am J Obstet Gynecol 1992; 166: 479– 88

Osteoporosis and bone strength Genetics Diet Exercise Hormones Bone density Architecture Turnover rate Damage accumulation Degree of mineralization + Bone quality Bone strength Adapted from The NIH Consensus Development Panel on Osteoporosis. JAMA 2001; 285: 785– 95

Different effects of estrogen therapy on connective tissue Genital organs (improved) Decreased skin thickness (reversed) Cerebral changes (Alzheimer’s decreased) Estrogen therapy Bone loss (stopped and reversed) CVS effects (including carotids) Cartilage
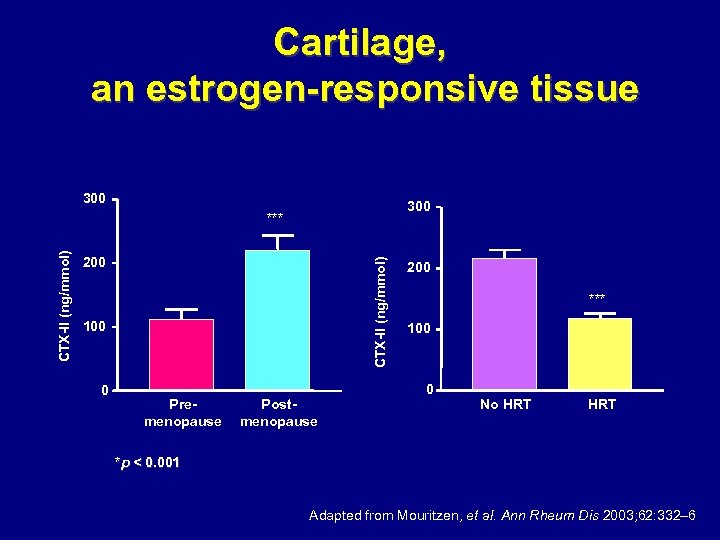
Cartilage, an estrogen-responsive tissue 300 200 CTX-II (ng/mmol) *** 100 0 Premenopause Postmenopause 200 *** 100 0 No HRT *p < 0. 001 Adapted from Mouritzen, et al. Ann Rheum Dis 2003; 62: 332– 6

Selective serotonin and/or noradrenaline reuptake inhibitors • Newer SNRI formulations: – Extended release venlafaxine • 51% reduction in hot flushes/sweats • Less nausea Evans ML, et al. Obstet Gynecol 2005; 105: 161– 6 • Desvenlafaxine succinate – in development – Selective NA & 5 HT reuptake inhibitor – Good plasma / brain ratios in animal models Deecher DC, et al. J Pharmacol Exp Ther 2006; 318: 657– 65
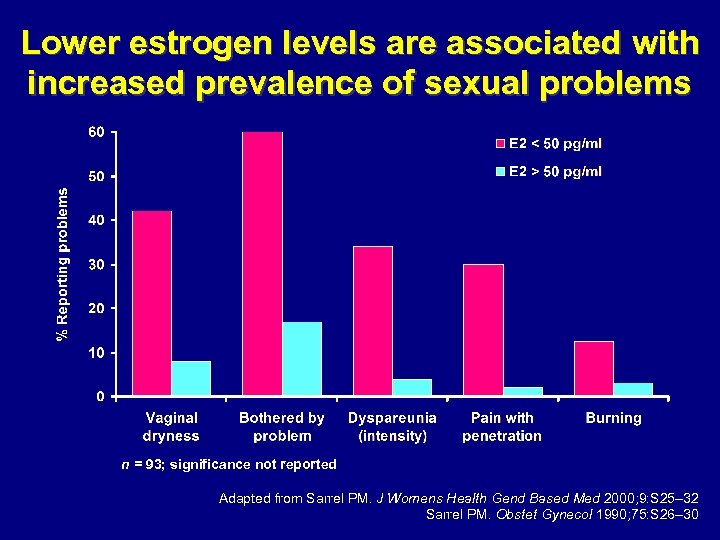
Lower estrogen levels are associated with increased prevalence of sexual problems n = 93; significance not reported Adapted from Sarrel PM. J Womens Health Gend Based Med 2000; 9: S 25– 32 Sarrel PM. Obstet Gynecol 1990; 75: S 26– 30
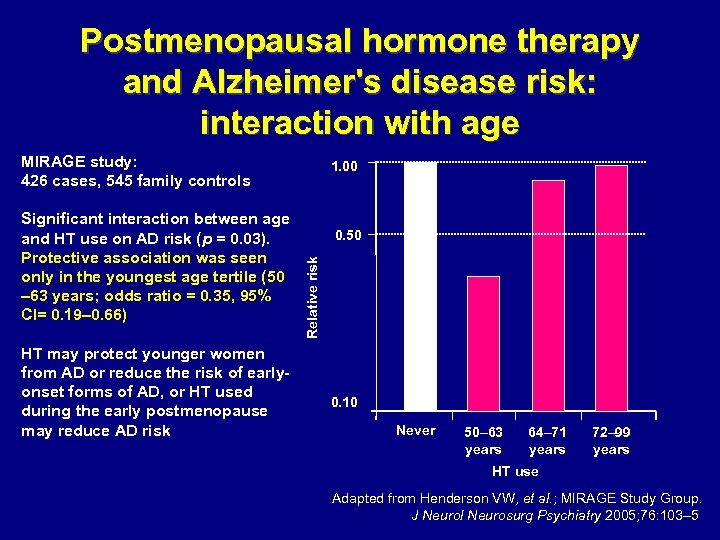
Postmenopausal hormone therapy and Alzheimer's disease risk: interaction with age MIRAGE study: 426 cases, 545 family controls HT may protect younger women from AD or reduce the risk of earlyonset forms of AD, or HT used during the early postmenopause may reduce AD risk 0. 50 Relative risk Significant interaction between age and HT use on AD risk (p = 0. 03). Protective association was seen only in the youngest age tertile (50 – 63 years; odds ratio = 0. 35, 95% CI= 0. 19– 0. 66) 1. 00 0. 10 Never 50– 63 years 64– 71 years 72– 99 years HT use Adapted from Henderson VW, et al. ; MIRAGE Study Group. J Neurol Neurosurg Psychiatry 2005; 76: 103– 5
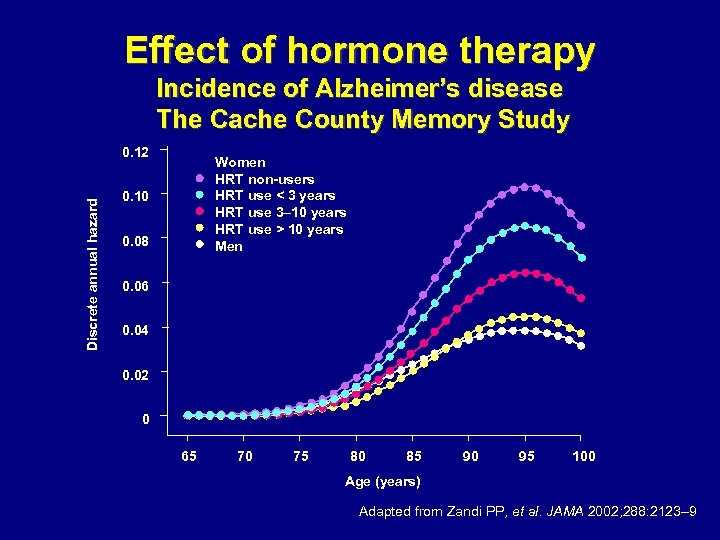
Effect of hormone therapy Incidence of Alzheimer’s disease The Cache County Memory Study Discrete annual hazard 0. 12 Women HRT non-users HRT use < 3 years HRT use 3– 10 years HRT use > 10 years Men 0. 10 0. 08 0. 06 0. 04 0. 02 0 65 70 75 80 85 90 95 100 Age (years) Adapted from Zandi PP, et al. JAMA 2002; 288: 2123– 9

Exercise in the menopause • Any physical activity is better than being sedentary • Regular exercise reduces total and cardiovascular • • mortality Better metabolic profile, balance, muscle strength, cognition and quality of life are observed in physically active persons. Heart events, stroke, fractures and breast cancer are significantly less frequent Benefits far outweigh possible adverse consequences: the more – the better, but too much may cause harm

Exercise in the menopause: optimal exercise prescription • At least 30 minutes of moderate intensity • • exercise, at least three times weekly Two additional weekly training sessions of resistance exercise may provide further benefit Injury to the musculo-articulo-skeletal system should be avoided

AHA 2006 Diet and Lifestyle Recommendations 1 • Balance calorie intake and physical activity to achieve or maintain a healthy body weight • Consume a diet rich in vegetables and fruits • Choose whole-grain, high-fiber foods • Consume fish, especially oily fish, at least twice a week Circulation 2006; 114: 82

AHA 2006 Diet and Lifestyle Recommendations 2 • Limit intake of saturated fat to < 7% of energy, trans fat to < 1% and cholesterol to < 300 mg/day by choosing lean meats and vegetable alternatives, selecting fat-free, 1% fat and low-fat products • Choose and prepare foods with little or no salt • Increase fiber intake (beans, whole grain, other fruits and vegetables) • If you consume alcohol, do so in moderation • Quit smoking Circulation 2006; 114: 82
435cbc496d2145e9163a8acc05123cd7.ppt