966b18d19e2730684a27c3c4438dd680.ppt
- Количество слайдов: 86

Unresolved Issues in the Globalization of Clinical Trials Robert M Califf MD Vice Chancellor for Clinical Research

Globalization of Clinical Trials Ù Current State Money Ethics Cultural environment Genetic variation Ù Envisioning a positive future
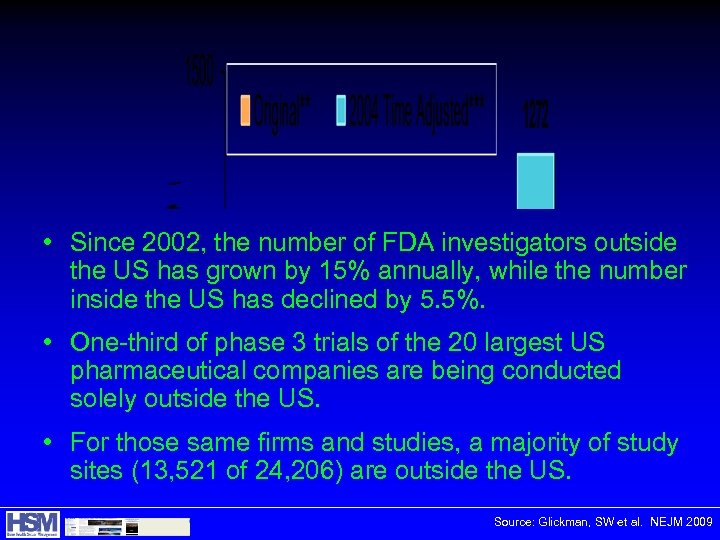
• Since 2002, the number of FDA investigators outside the US has grown by 15% annually, while the number inside the US has declined by 5. 5%. • One-third of phase 3 trials of the 20 largest US pharmaceutical companies are being conducted solely outside the US. • For those same firms and studies, a majority of study sites (13, 521 of 24, 206) are outside the US. Source: Glickman, SW et al. NEJM 2009

The Globalization of Clinical Investigators Percent of Total 1572 s Filed Sources: Tufts CSDD

Growth in Numbers of Active FDA-Regulated Investigators 2001 2003 2006 5 -yr Growth Rate Most Recent 3 -yr Growth Rate Russia 176 317 623 28. 8% 25. 3% Poland 215 314 322 8. 4% . 8% Brazil 139 296 292 16% -. 5% Costa Rica 12 20 22 12. 9% 3. 2% Argentina 174 305 462 21. 6% 14. 8% China 80 120 307 30. 9% 36. 8% India 64 145 464 48. 6% 47. 4% Sources: Tufts CSDD

The test of a first-rate intelligence is the ability to hold two opposed ideas in the mind at the same time, and still retain the ability to function. F. Scott Fitzgerald, "The Crack-Up" (1936) US novelist (1896 - 1940)

Creative Tension Ù Globalization of clinical research is a very good thing for the US and for all of us in a flat world We need more evidence to guide effective practice in the US So does every other country in the world Ù Driving globalization because of poor efficiency in the conduct of research in the US is a bad thing for the US By the way, the same thinking could be applied to any economically advantaged country that begins to think it is “above it all”

Dollars and Sense: Factors Pushing Clinical Research Out of the U. S. American Federation for Medical Research (AFMR) April 14, 2009 Kevin Schulman, MD Director, Health Sector Management Program The Fuqua School of Business Director, Center for Clinical and Genetic Economics Duke University Medical Center

Globalization of Clinical Trials—The Good Ù Larger sample sizes are needed Modest treatment effects predominate Subgroups must be validated Will become even more important in “personalized” or “stratified” medicine Ù Competition spurs improvement Ù More research will be done Ù Collaboration leads to shared learning Practice patterns Ethics Will ensure transparency on results reporting Ù Adequate sample sizes/study designs to understand genetic heterogeneity

Globalization: A Necessity Ù Proof of concept trials—there are pharmacogenomic differences Ù Efficacy trials—the context of clinical practice matters Ù Effectiveness trials—the relative costs and balance of risk and benefits are context dependent The question is not whether we globalize, it is why we do it, how we handle cultural differences and how we lump or split findings
Economic Proposition to Industry From Globalization $ duration* $1 – $10 billion* Pre-Launch No requirement to match study populations to target markets Return speed* Potential local market opportunity fall-off* ROI maximize area* $800 – $880 mil (2001) 0 Patent Launch Decrease costs or time Patent Expiration

The Good– the US Needs to Wake Up! Ù Rapid movement to moving clinical trials away from US Ù US is inferior in: Study cost index Population indicators (tx naïve patients) Business environment Ù US is better, but losing ground quickly in: Clinical research personnel qualification Study site organization Thought leader quantity
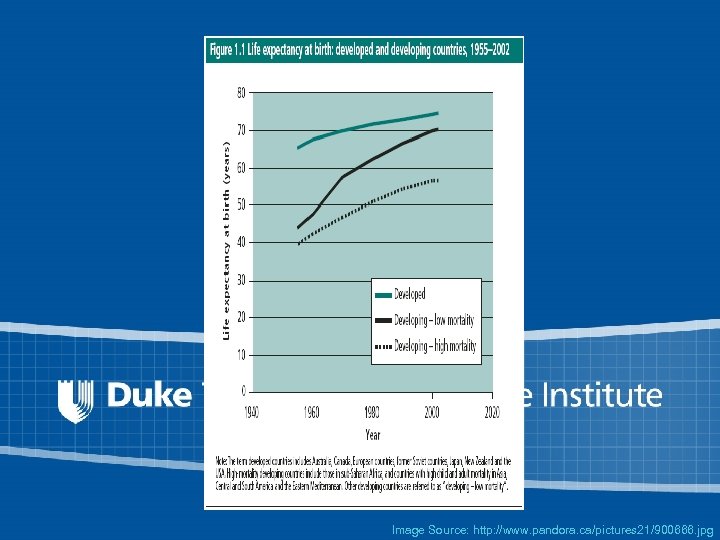
Image Source: http: //www. pandora. ca/pictures 21/900666. jpg

The Demise of Empires Ù Dominance at a point in time Ù Arrogance about superiority Ù Failure to pay attention to quality of work Ù Leaders content to “ride the wave” Ù Entrenched interests can buy stability through controlling laws and regulations Ù Inability to create or respond to innovation Ù Cost of transactions exceeds cost of actually doing the work!

Data gathered by the Tufts Center for the Study of Drug Development (Tufts CSDD) >90% of all clinical trials delayed due to over-ambitious timelines and difficulty with patient enrollment Top reasons for delays in trials: w. Protracted budget negotiations w. Slow IRB review and approval w. Poor patient recruitment and retention Estimated 20% of PIs fail to enroll a single patient and 30% under-enroll in a given trial Between 2000 and 2005 38% of PIs who participated in clinical trials in a given year did not return in a subsequent year through 2008 w. Up from 26% in previous 5 years
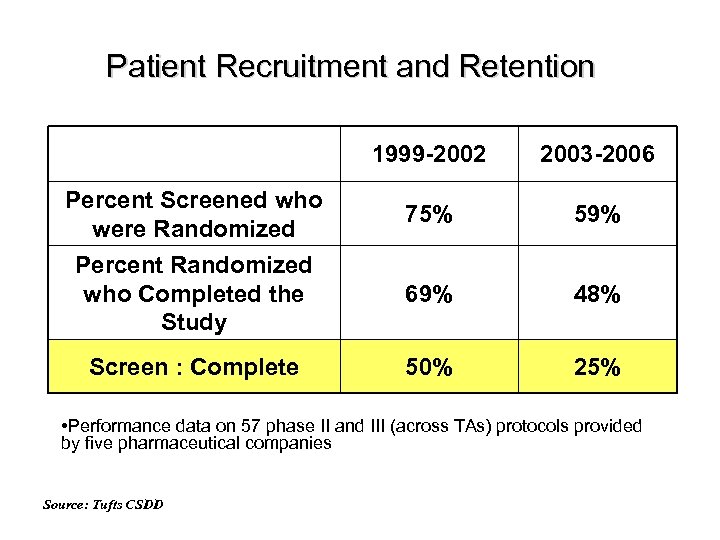
Patient Recruitment and Retention 1999 -2002 2003 -2006 Percent Screened who were Randomized 75% 59% Percent Randomized who Completed the Study 69% 48% Screen : Complete 50% 25% • Performance data on 57 phase II and III (across TAs) protocols provided by five pharmaceutical companies Source: Tufts CSDD
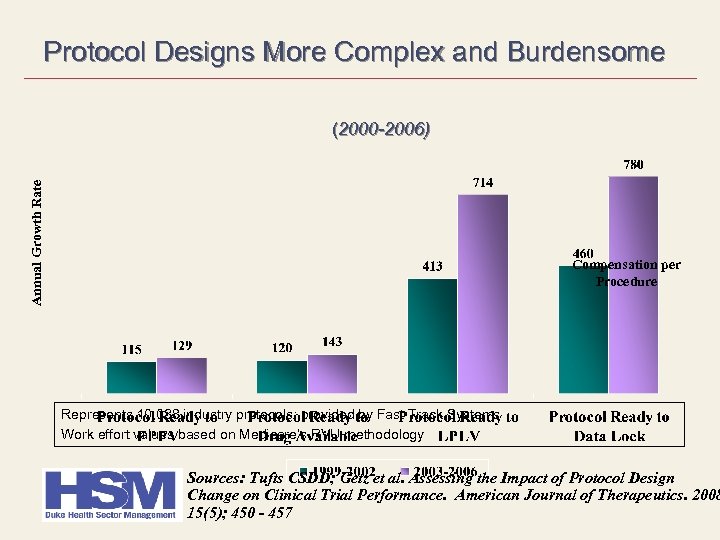
Protocol Designs More Complex and Burdensome Annual Growth Rate (2000 -2006) Compensation per Procedure Represents 10, 038 industry protocols; provided by Fast Track Systems Work effort values based on Medicare’s RVU methodology Sources: Tufts CSDD; Getz et al. Assessing the Impact of Protocol Design Change on Clinical Trial Performance. American Journal of Therapeutics. 2008 15(5); 450 - 457
Performance ‘Impact’ of protocol complexity Compared US-based pivotal trial protocols executed 1999 -2002 (lower complexity) and 2003 -2006 (higher complexity): Number of CRF pages rose to an average of 180 pages vs. 55 Controlling for treatment duration, cycle times increased substantially across all measures Enrollment rates worsened Source: Tufts CSDD

Cycle Time Metrics Median Days Elapsed 69% - 75% Increase 12% – 20% Increase Source: Tufts CSDD



Clinical Trial Cost Estimates $ In US 2007 Millions Full Cost Industry Streamlined Industry More Streamlined
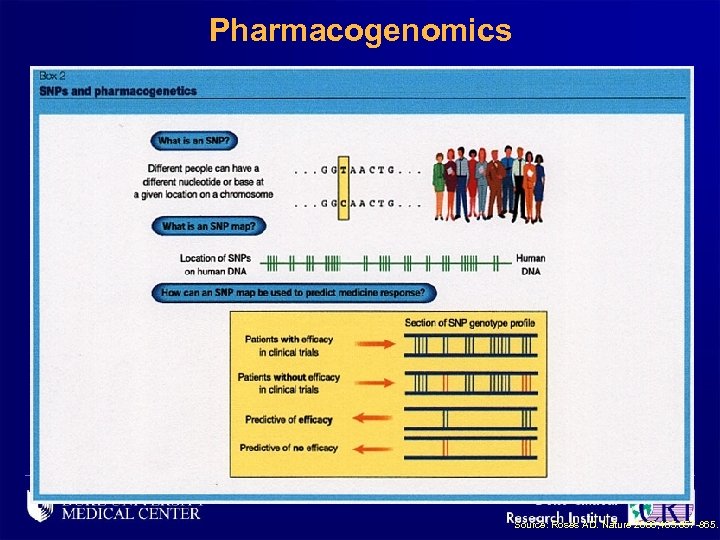
Pharmacogenomics Source: Roses AD. Nature 2000; 405: 857 -865.
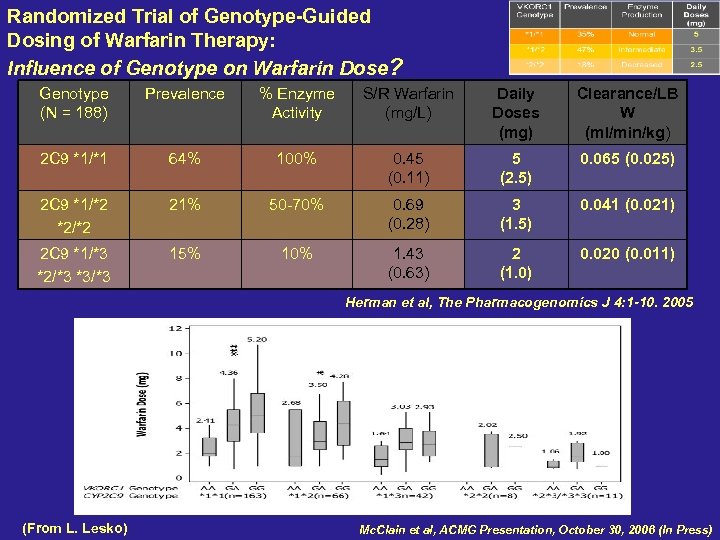
Randomized Trial of Genotype-Guided Dosing of Warfarin Therapy: Influence of Genotype on Warfarin Dose? Genotype (N = 188) Prevalence % Enzyme Activity S/R Warfarin (mg/L) Daily Doses (mg) Clearance/LB W (ml/min/kg) 2 C 9 *1/*1 64% 100% 0. 45 (0. 11) 5 (2. 5) 0. 065 (0. 025) 2 C 9 *1/*2 *2/*2 21% 50 -70% 0. 69 (0. 28) 3 (1. 5) 0. 041 (0. 021) 2 C 9 *1/*3 *2/*3 *3/*3 15% 10% 1. 43 (0. 63) 2 (1. 0) 0. 020 (0. 011) Herman et al, The Pharmacogenomics J 4: 1 -10. 2005 (From L. Lesko) Mc. Clain et al, ACMG Presentation, October 30, 2006 (In Press)

Randomized Trial of Genotype-Guided Dosing of Warfarin Therapy Boxplots of distribution of warfarin dose by CYP 2 C 9 and VKORC 1 genotype Sconce et al. Blood 2005; 106: 2329
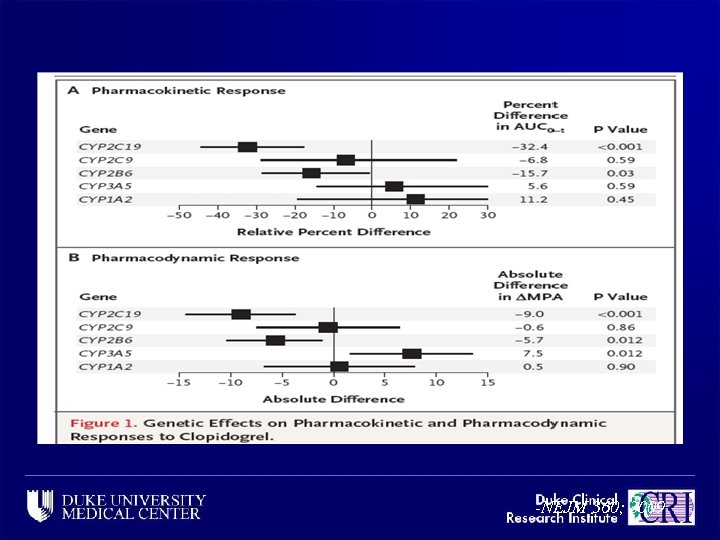
-NEJM 360; 2009
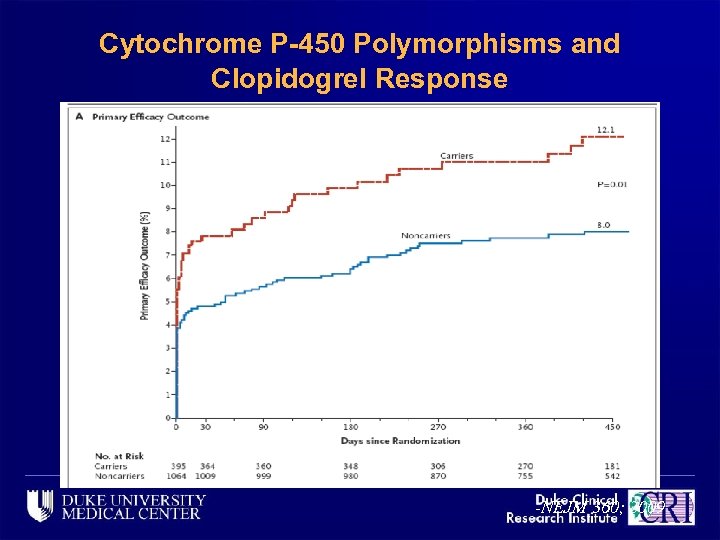
Cytochrome P-450 Polymorphisms and Clopidogrel Response -NEJM 360; 2009

Cytochrome P-450 Polymorphisms and Clopidogrel Response -NEJM 360; 2009

GENEVA, SWITZERLAND—World Health Organization officials expressed disappointment Monday at the group's finding that, despite the enormous efforts of doctors, rescue workers and other medical professionals worldwide, the global death rate remains constant at 100 percent. Death, a metabolic affliction causing total shutdown of all life functions, has long been considered humanity's number one health concern. Responsible for 100 percent of all recorded fatalities worldwide, the condition has no cure. "I was really hoping, what with all those new radiology treatments, rescue helicopters, aerobics TV shows and what have you, that we might at least make a dent in it this year, " WHO Director General Dr. Gernst Bladt said. "Unfortunately, it would appear that the death rate remains constant and total, as it has inviolably since the dawn of time. "

The Good--More Research will be Done Ù Broader sources of funding Many governments now giving tax breaks for industry funded research Ù Do the research where costs are less Ù Do the research where barriers to trial initiation are less

ACC/AHA Clinical Practice Guidelines
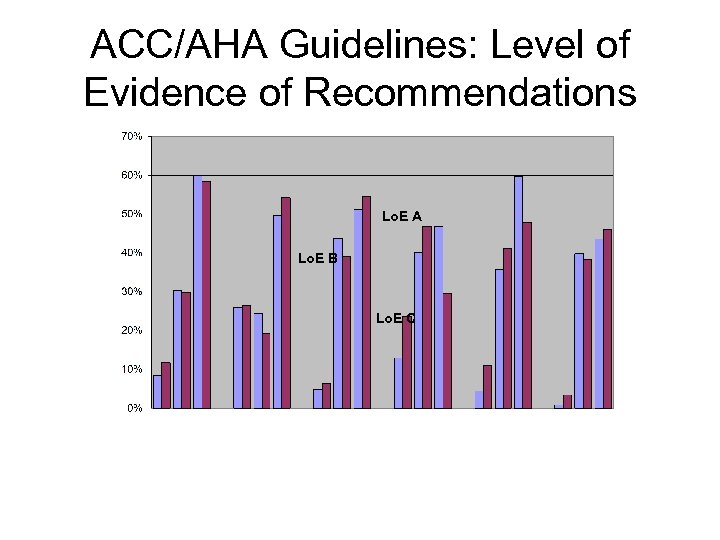
ACC/AHA Guidelines: Level of Evidence of Recommendations Lo. E A Lo. E B Lo. E C

ACC/AHA Guidelines: More Guidelines No Improvement in Proportion with High Quality! A B C A B C (2001 - Pacemaker Atrial Heart Stable Unstable PCI 2005) (1998 -2002) Fibrillation Failure (2001 Angina (1999 Angina (2000 (2001 -2006) -2002) -2007)

The Bad Ù Differences in practices can be found as a function of geography Ù Differences in outcomes can be found as a function of geography Ù Differences in treatment effect can be found as a function of geography

Current Practices in ACS Care USA vs Rest of GRACE Andrzej Budaj for the GRACE Investigators Postgraduate Medical School, Grochowski Hospital, Warsaw Poland
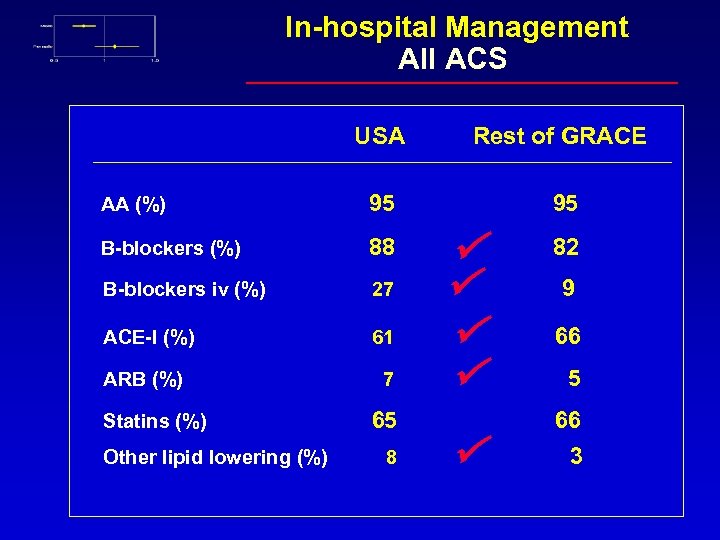
In-hospital Management All ACS USA Rest of GRACE AA (%) 95 95 B-blockers (%) 88 B-blockers iv (%) 27 ACE-I (%) 61 ARB (%) 7 Statins (%) Other lipid lowering (%) 65 8 82 9 66 5 66 3
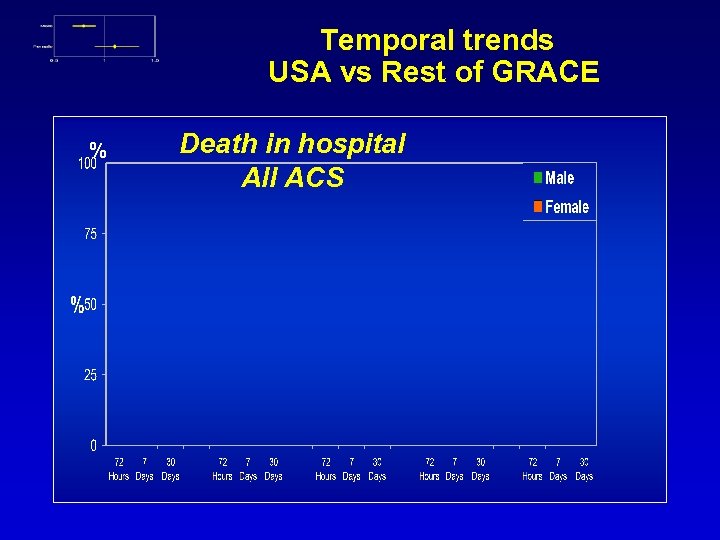
Temporal trends USA vs Rest of GRACE % Death in hospital All ACS

Death and/or MI at 30 Days Placebo Eptifibatide 16. 88 13. 89 6103 13. 68 14. 88 3357 Eptifibatide Better 9808 LB 11, 38 PT-E 2 -7 Placebo Better
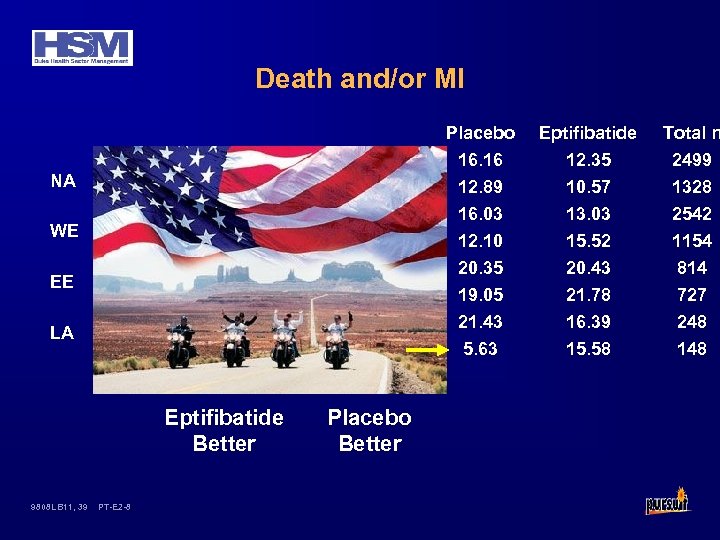
Death and/or MI Placebo LA Eptifibatide Better 9808 LB 11, 39 PT-E 2 -8 Placebo Better 2499 1328 13. 03 2542 12. 10 20. 35 19. 05 21. 43 5. 63 EE 12. 35 10. 57 16. 03 WE Total n 16. 16 12. 89 NA Eptifibatide 15. 52 20. 43 21. 78 16. 39 15. 58 1154 814 727 248 148

Timing of Angiography Overall 9808 LB 11, 40 PT-C-17 North America Western Europe Eastern Europe Latin America

Study Undertaken by FDA statisticians to evaluate possibility of systematic regional differences u Major cardiovascular outcome studies evaluated over the last 10 years u Overall study result statistically positive, ie. demonstrated overall effect u Region never pre-specified as a factor to be evaluated statistically u 16 independent studies

Estimates and confidence intervals for difference between US and Non-US treatment effects for each study In 13 of 16 , US log hazard above 0 J. Lawrence


A figure From the label
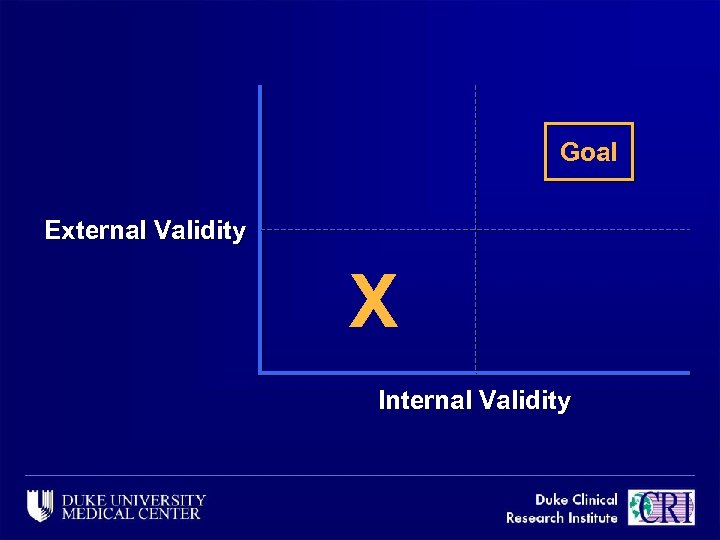
Goal External Validity X Internal Validity

u Article cites numerous examples of trials, which produce parameter estimates of questionable value to Western European decision makers and highlights differences between emerging regions and Western Europeans, which suggest the two are mutually incomparable. u Proposed that these differences may confound study outcomes, decision-making parameter estimates and data pertaining to the incidence of adverse drug interactions u Further research should be undertaken in order to explore the relationship between geographical variance and external validity, particularly where safety data derived from relatively drug naïve regions are assumed to pertain to a maximally treated populations elsewhere in the world Source: Wathal. Outsourcing Clinical Trials www. samedanltd. com

u Extrinsic factors such as medical practice, disease definition and study population may influence applicability of foreign data to an EU setting u Global drug development doesn’t necessarily support approval of unrestricted indications in an EU population u u Consider and discuss possible influence of extrinsic factors on interpretation of results and wording of indications In depth, prospective analysis of potential ethnic factors when conducting a clinical trial in a certain region. u Depending on the outcome of analyses can be decided whether certain clinical trials conducted in a specific area of the world would be relevant to EU setting or if there are reasons to perform additional clinical trials within the EU Source: European Medicines Agency, Pre-Authorization Evaluation of Medicines for Human Use. Feb. 19, 2009

u Verification at time of evaluation of marketing authorization application that trials have been conducted in accordance with GCP and ethical standards u Greater transparency of this process and its outcome should be described in the European Public Assessment Report (EPAR) u Increased GCP inspection including further extension of GCP policy on increasing numbers of routine inspections as part of the need for greater supervision of the conduct and ethical standards of clinical trials performed outside the EEA Source: European Medicines Agency, EMEA strategy paper: acceptance of clinical trials conducted in the third countries, for evaluation in Marketing Authorization Applications. Feb. 5, 2008
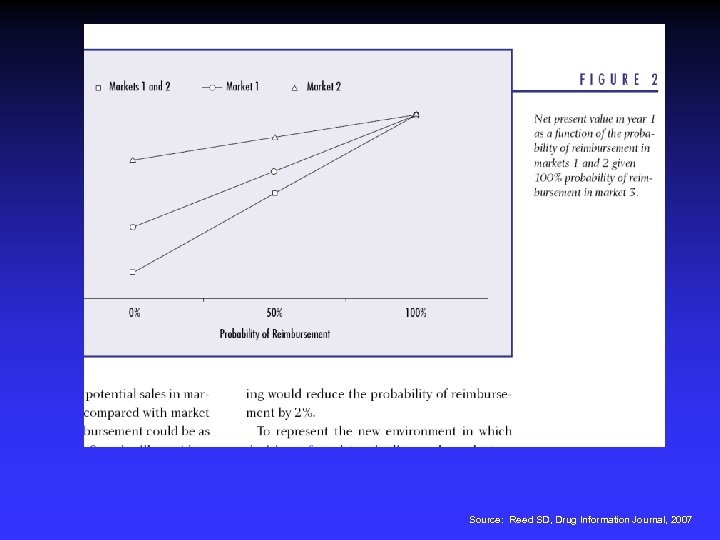
Source: Reed SD, Drug Information Journal, 2007
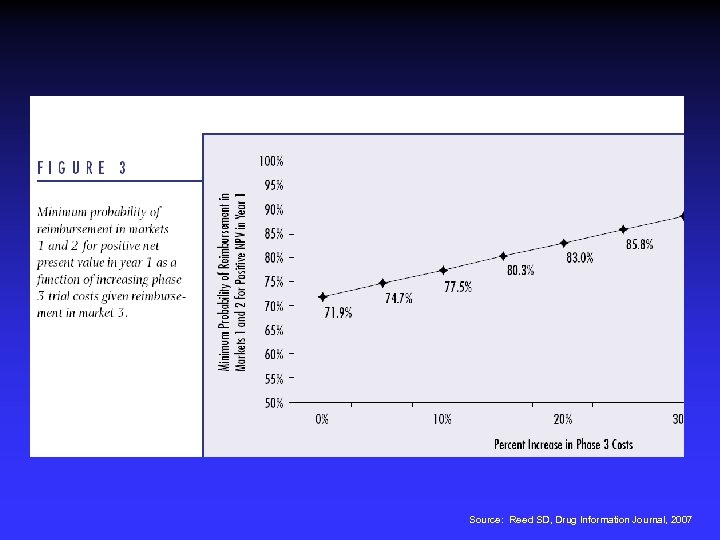
Source: Reed SD, Drug Information Journal, 2007

Source: Reed SD, Drug Information Journal, 2007

The Ugly Ù Differences in reported adverse events can be found in different countries Is there a relationship between relative financial incentive and lower rates of side effects? Ù Differences in adherence can be found in different countries Is there a relationship between lack of access to health care outside of the trial and adherence?
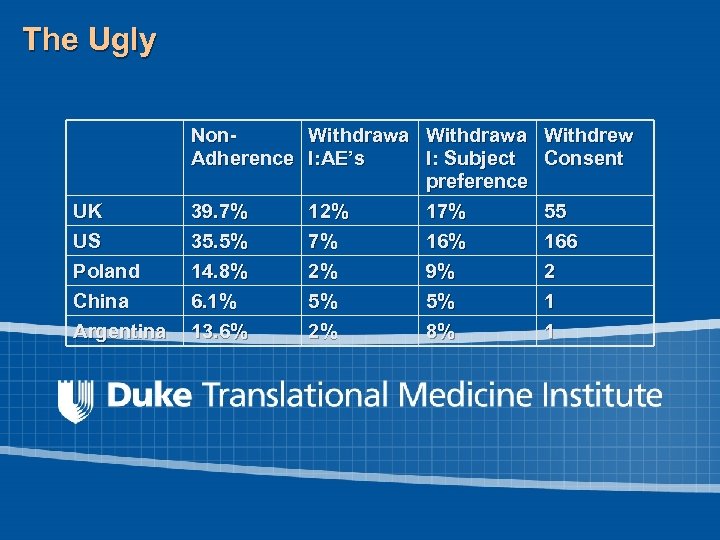
The Ugly UK US Non. Withdrawa Withdrew Adherence l: AE’s l: Subject Consent preference 39. 7% 12% 17% 55 35. 5% 7% 166 Poland China 14. 8% 6. 1% 2% 5% 9% 5% 2 1 Argentina 13. 6% 2% 8% 1

The Obscene Ù When the per patient reimbursement exceeds reasonable levels, the human experimentation entrepreneurs will be tempted Ù When the FDA cannot inspect, the cheaters will figure out how to get the data looking real good!
Source: http: //www. tampabay. com/news/business/article 934677. ece

Source: http: //www. tampabay. com/news/business/article 934633. ece

Source: http: //www. tampabay. com/opinion/essays/article 934654. ece

Ethical Concerns in clinical trials in India: an investigation • Lapatinib, Glaxo. Smith. Kline – The majority of breast cancer patients in India cannot afford proper treatment. This trial required seriously ill patients who had not received treatment for their condition. – Their economic vulnerability forces patients in India to take part in trials in order to get access to treatment and to disregard the potential risks that participating in clinical trials entails. By carrying out this clinical trial in India Glaxo. Smith. Kline (GSK) took advantage of the vulnerable position of breast cancer patients. Source: Srinivasan S, et al. Ethical concerns in clinical trials in India: an investigation. Center for Studies in Ethics and Rights, Mumbai, India. Feb. 2009

Ethical Concerns in clinical trials in India: an investigation • Risperidone, Johnson & Johnson – Patients in trial were suffering from an acute attack of a psychiatric condition that would have caused them much distress. – They were harmed because they were taken off all treatment before they were put on either the active drug or a placebo. – Those on the placebo were also harmed because they were deprived of an effective treatment Source: Srinivasan S, et al. Ethical concerns in clinical trials in India: an investigation. Center for Studies in Ethics and Rights, Mumbai, India. Feb. 2009

Ethical Concerns in clinical trials in India: an investigation • Quetiapine fumurate extended release, Astra. Zeneca – These two placebo-controlled trials of quetiapine were conducted on patients with schizophrenia. An immediate release formulation of the drug had already been approved and these trials were of an extended release version of the drug. – A patient in one of the quetiapine trials committed suicide after 173 days of being on placebo. Source: Srinivasan S, et al. Ethical concerns in clinical trials in India: an investigation. Center for Studies in Ethics and Rights, Mumbai, India. Feb. 2009


Factors Pulling Research Offshore • • • Cost: cost per patient may be 1/10 the cost in the US (Garnier, JP). Availability: untreated or under-treated patients may speed recruitment of patients to clinical trials reducing the time costs of research. Regulations: local processes may be more variable (and potentially less restrictive) than in the US. Source: Garnier JP. Rebuilding the R&D engine in big pharmacy. Harv Bus Rev 2008; 86: 68 -76.

Factors Pushing Research Offshore • • Costs (Clinical Care) Costs (Administrative) • • Systems that developed to govern single site studies are inefficient in oversight of multicenter, multinational studies IRB (redundant site level process) Contracting (redundant site level process) Compliance (research as a potential criminal issue)
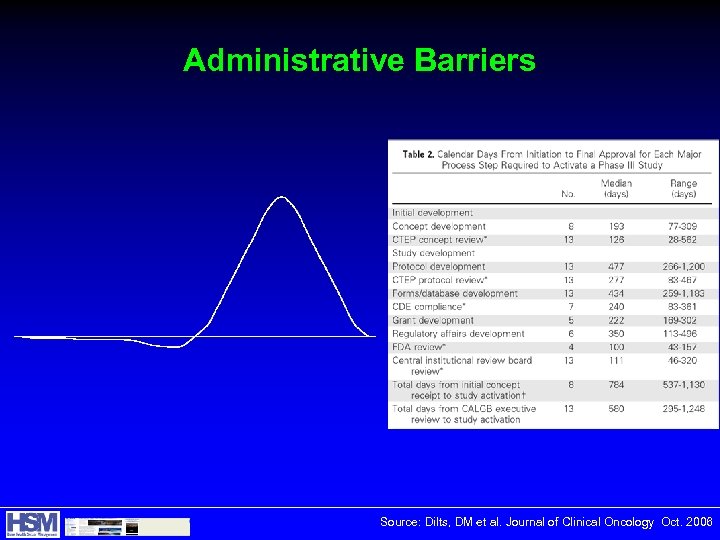
Administrative Barriers Source: Dilts, DM et al. Journal of Clinical Oncology Oct. 2006

IRB Approval Timeline for a Focus Group discussing Health Insurance with Latinos Event Time Comments Study Submitted to IRB as Exempt Aug 2007 IRB rejected exempt status Discussions with IRB about rejection of exempt status Aug-Sep 2007 Focus groups may not qualify for IRB exempt status Study rewritten and submitted as expedited Oct 2007 Initial study approved by IRB Feb 2008 Local cultural center requests major changes to study Apr 2008 Continued negotiations between local cultural center and IRB requested extensive changes Incorporate relevant cultural concerns in initial study design May-Sep 2008 Rewritten study submitted to IRB Oct 2008 Final Study Approved Jan 2009 IRB requested additional changes

Contracting for Clinical Research “…the system would be better served if there were universally accepted contractual language … Such language would help safeguard the integrity of the research process. ” -JEFFREY M. DRAZEN Editor-in-Chief, New England Journal of Medicine Source: Drazen, JM. NEJM Oct. 24, 2002

(1) I am familiar with, and will comply with, applicable federal regulations and guidance for the protection of human subjects: HHS regulations at 45 FR 46 and associated guidance; FDA regulations at 21 CFR Parts 50, 54, 56, 312, 314, 601, 812, and 814 and associated guidance; the HIPAA privacy regulations at 45 CFR Parts 160 and 164 and associated guidance; the DUHS Federal-Wide Assurance; and relevant institutional policies and procedures for the protection of human research subjects. Source: DUHS Principal Investigator Agreement form. Version 6/25/08

Source: Glickman, SW et al. NEJM 2009.

Selection of Patients in Multinational Trials • Problem: Research in communities that are not intended to be major markets for the products under testing can be ethically problematic • Solutions – Sponsors should describe how trial populations match intended markets – Create target enrollment according to region on the basis of intended use of product, similar to target enrollment of women and minorities Source: Glickman, SW et al. NEJM 2009.

Transparency of Clinical Trial Results in Developing Countries • Problem: Protection of publication rights and access to trial data for investigators is necessary to preserve the integrity of research • Solutions – Publish all data regardless of research location, and reinforce requirements according to FDA Amendments Act of 2007 – Preserve publication rights globally through legal agreements at onset of trial – Create trial leadership that incorporates representatives of countries involved in study Source: Glickman, SW et al. NEJM 2009.

Regulatory Oversight of International Clinical Research • Problem: Regulatory agencies have little information on trials conducted outside their countries • Solutions – Mechanism for sharing regulatory oversight among government agencies worldwide – Public registry of IRBs and inventory of country-specific provisions for ethical oversight – Comprehensive study of the globalization of clinical research by IOM or WHO – Central statistical monitoring system to find unusual data patterns suspicious for fraud Source: Glickman, SW et al. NEJM 2009.

Training and Experience of Clinical Investigators • Problem: Investigators in developing countries are typically less experienced than investigators in developed countries • Solutions – Formal training programs for clinical research for investigators in developing countries to expand global clinical research leadership capacity and improve collaboration worldwide – Mechanism for tracking investigators who are trained to conduct clinical trials and those who have been prohibited from conducting trials Source: Glickman, SW et al. NEJM 2009.

Genomic Information in Drug Development • Problem: Lack of pharmacogenomic data for subjects limits confidence in generalizability of results • Solutions – Expand FDA Voluntary Genomic Data Submissions program to international regulatory agencies – Develop global data warehousing and data analysis capabilities Source: Glickman, SW et al. NEJM 2009.

IRB Quality and Inefficiency • Problem: Redundancy in review process may harm patient safety by requiring diversion of effort to unnecessary procedures and practices • Solutions – Greater use of centralized IRBs (eg, Central IRB Initiative, European Union Clinical Trials Directive) – Mutual acceptance of proposal review in consortia (eg, Biomedical Research Alliance of New York) – Streamlined best practices to reduce unnecessary work for investigators (eg, Clinical Trials Transformation Initiative) Source: Glickman, SW et al. NEJM 2009.

Payment Compliance • Problem: Increased costs and delays in payment for research subjects divert financial support from research to administration and make research less attractive to investigators because of risk of criminal penalties • Solutions – Establish nonpunitive mechanism for reconciliation of payment – Expand mechanisms to pay for usual care services (eg, within Medicare and Medicaid) Source: Glickman, SW et al. NEJM 2009.

Commercial Contracts • Problem: Variety of contracting practices brings complexity and delays to research • Solutions – Adopt standard contract language for clinical research agreements Source: Glickman, SW et al. NEJM 2009.

Confidentiality Agreements in Commercial Contracts • Problem: Confidentiality agreements reduce the transparency and efficiency of clinical research • Solutions – Adopt standard confidentiality language for clinical research agreements Source: Glickman, SW et al. NEJM 2009.

A collaborative effort to find solutions In light of these issues the U. S. FDA’s Office of Critical Path Programs and Duke University joined together as founding members of a publicprivate partnership: The Clinical Trials Transformation Initiative (CTTI) All stakeholders are involved in this initiative including government, industry, academia, patient advocates, clinical investigators, professional societies, and others

Mission To identify practices that through broad adoption will increase the quality and efficiency of clinical trials

Scope CTTI will conduct projects in support of its mission to identify practices that will increase the quality and efficiency of clinical trials w“Quality” - the ability to effectively answer the intended question about the benefits and risks of a medical product (therapeutic or diagnostic) or procedure, while assuring protection of human subjects

Scope (continued) CTTI will generate evidence about how to improve the design and execution of clinical trials w. Projects about design will address principles generally applicable to clinical trials to assure that they are fit to accomplish their intended purpose.

Scope (continued) While CTTI focuses on clinical trials, it may study other types of clinical research (e. g. , registries) that can provide data to regulatory agencies. Although CTTI will concentrate initially on the design and conduct of clinical trials in the United States, it seeks to identify practice improvements that can be applied internationally.

Executive Committee FDA: (Rachel Behrman, OC, Co-chair; Bob Temple, CDER, Bram Zuckerman, CDRH) Duke: (Rob Califf, Co-chair) NIH liaison: (Amy Patterson) Industry: (Glenn Gormley, Jay Siegel, Susan Alpert, Alberto Grignolo) Academia: (David De. Mets) Patient representative: (Nancy Roach) At-large representative: (Ken Getz) Non-US regulatory liaison: (Hans-Georg Eichler, EMEA) Steering Committee co-chairs, ex officio (James Ferguson, Briggs Morrison)

Member Organizations Category # organizations Pharmaceutical companies 7 Clinical research organizations 7 Academic institutions 7 Professional societies 7 Device companies 6 Biotechnology companies 5 Government 4 (FDA, CMS, NIH, OHRP) Clinical investigator groups 3 Trade organizations 3 Patient representatives 2 (TBD) Private equity firm 1 Regulatory law firm 1 *Began recruiting members May 2008

Projects Priority areas defined by Executive Committee: w. Design principles w. Data quality and quantity (including monitoring) w. Study start-up w. Adverse event reporting Information about the process for submission, review, and approval of projects available at CTTI Web site: www. trialstransformation. org/projects

Life Expectancy at Birth: Developed and Developing Countries, 1955 -2002 Source: World Health Report 2003
966b18d19e2730684a27c3c4438dd680.ppt