ada88f4fa25f6698500d2b458364a6ec.ppt
- Количество слайдов: 30

University of Abertay (P 2) Partner Report Jason Johnston, Erica Benson, Keith Harding, Isobel Pimbley, Jayanthi Nadarajan, Samantha Gale, Jon Green, Brian Grout

Presentation Outline • Part 1 - Project Management/Outputs • Visits, exchanges & conferences • Problems • Publications • Part 2 - Work Pack Highlights • DSC (WP 1 & 8) • Oxidative stress (WP 7 & 8) • Ethylene (WP 7 & 8) • DNA methylation (WP 7)

Exchanges, Visits & Conferences in 2005 • CRYMCEPT Visits • Paul Lynch (P 3) to UAD in February • Jason Johnston to Writtle College in March • Jon Green to UAD in June • Conferences (September 2005) • Society For Low Temperature Biology, York 2 -oral invited/contributed presentations by Jason Johnston/Jon Green/Brian Grout • International Association For Plant Tissue Culture, Perth, Australia “Biotechnology & Sustainable Development” Erica Benson (Plenary) Keith Harding (Contributed/Session Chair)

Problems Challenges • Potential access problems to DSC & HPLC resolved • Nuclease P 1 back in production ☺ • Growth room facilities marginal • WP 7 leader & PI (Erica Benson) returned after absence of sick leave • Erica extends a big thank you to all CRYMCEPT colleagues for their support and particularly to Jason, Keith, Paul and Brian.

Publications – books & chapters • Life In The Frozen State (2004), Fuller, B; Lane, N & Benson, EE 3 chapters authored by E. Benson WP 7/WP 8/WP 9 • Benson, E. , Johnston, J. , Muthusamy, J. , Harding, K. (2005) Physical and Engineering Perspectives of In Vitro Plant Cryopreservation, in Plant Tissue Culture Engineering, Dutta Gupta, S. , Ibaraki, Y. , Eds. , Springer, in press. WP 1 • Benson, E. , Harding, K. , Johnston, J. (2005) Cryopreservation of shoot-tips and meristems, in Cryopreservation and freeze-drying protocols, Day, J. , Stacey, G. , Eds. , Methods in Molecular Biology Humana Press, in press. WP 7/WP 8/WP 9 • Day, JG, Benson, EE, Harding K (2006) Cryopreservation of plants and algae. Molecular Biomethods Handbook 2 nd Edition, Humana Press, Eds. Walker, JM and Rapley, R. In prep. WP 8/WP 7

Publications – Journals • 3 refereed journal reviews WP 7, 8, 9 • 1 analytical methods paper (DNA Methylation) WP 7 • 1 cryopreservation international technology transfer paper WP 8 • 2 cryopreservation original papers (Thermal Analysis) WP 1 • Many more to come! • Total antioxidants paper submitted August, WP 7 • In process of preparing data for joint CRYMCEPT partners publications. WP 7/WP 8

P 2 Compliance with Deliverables & Milestones • Delivery of Deliverables/Milestones either achieved or on target for completion by the end of the project. • Outputs in progress to support D/M targets • Project exit strategy to include preparation of outputs and data for delivery in 2006 at conferences and for invited chapter contributions and original papers.

P 2 Work Pack Highlights Oxidative Stress and Thermal Analysis WP 8 and WP 9 Methods/Delivery To Wider Cryo-conservation Community

Thermal Analysis: DSC (WP 1 & 8)

WP 1 Highlights • Ribes meristems study 1 (Sherlock et al. , 2005) – Use of silica-gel as an alternative to air-flow desiccation – DSC identified optimum time for achieving Tg

WP 1 Highlights • Ribes meristems study 2 (Benson et al. , 2005) • Should 0. 75 M sucrose be included in alginate during encapsulation? • Inclusion of sucrose – Increased osmotically inactive water during desiccation – Induced a Tg earlier during desiccation and at a higher water content (30% FW) – No effect on survival for species with ca 40% shoot recovery

WP 8 DSC Highlights • Neem somatic embryos (Benson et al. , 2005) • Optimisation of encapsulation protocol • Tg after 4 hours of air-flow desiccation • Tg occurred at residual moisture 13%, 0. 02 g. H 2 O/g. DW (Ribes 25 -30%, 0. 4 g. H 2 O/g. DW) • sucrose concentration during pretreatment osmotically inactive water

Oxidative stress (WP 7 & 8)
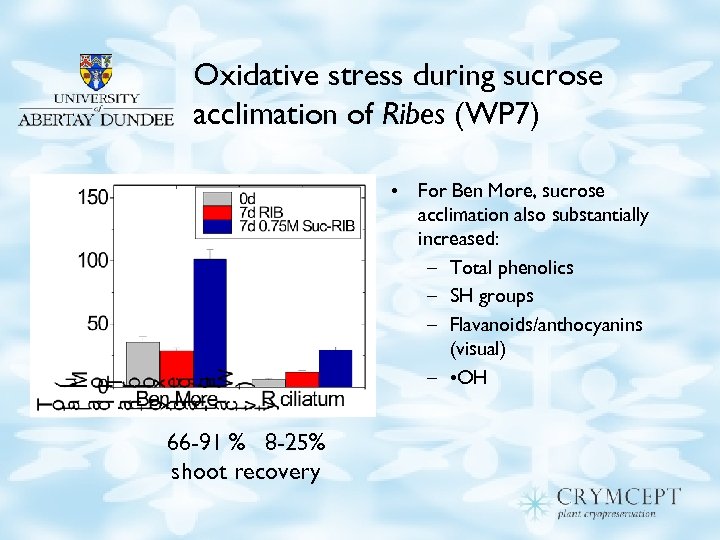
Oxidative stress during sucrose acclimation of Ribes (WP 7) • For Ben More, sucrose acclimation also substantially increased: – Total phenolics – SH groups – Flavanoids/anthocyanins (visual) – • OH 66 -91 % 8 -25% shoot recovery
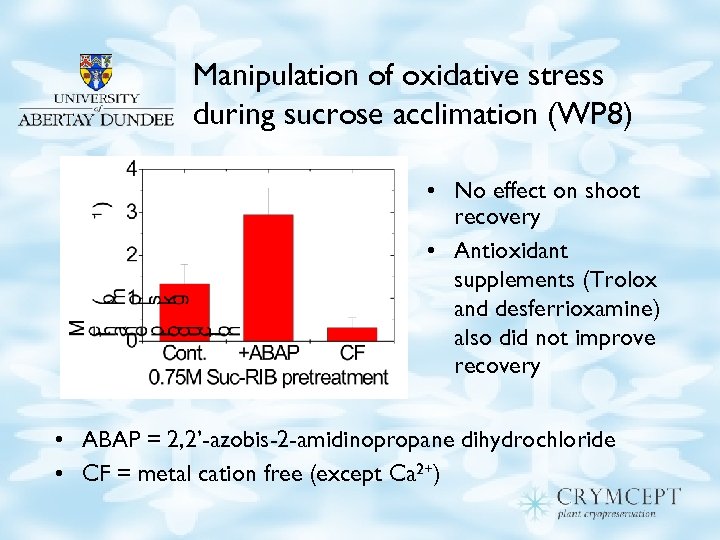
Manipulation of oxidative stress during sucrose acclimation (WP 8) • No effect on shoot recovery • Antioxidant supplements (Trolox and desferrioxamine) also did not improve recovery • ABAP = 2, 2’-azobis-2 -amidinopropane dihydrochloride • CF = metal cation free (except Ca 2+)

Oxidative stress (WP 8) • Manipulation of OS during recovery had no effect on survival: – Cation free medium ± antioxidants (desferrioxamine & Trolox) – Cation free medium for control meristems, but for stressed meristems – Cation free medium prevented polyphenol blackening, but temporarily!

Oxidative stress (WP 8) • Oxidative stress markers developed for Ribes for culture performance were successfully applied to Sitka spruce shoots.

DNA methylation (WP 7)
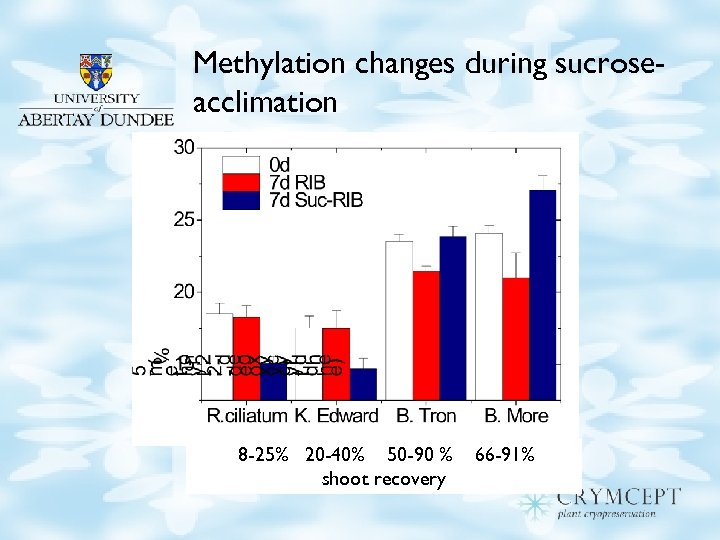
Methylation changes during sucroseacclimation 8 -25% 20 -40% 50 -90 % shoot recovery 66 -91%
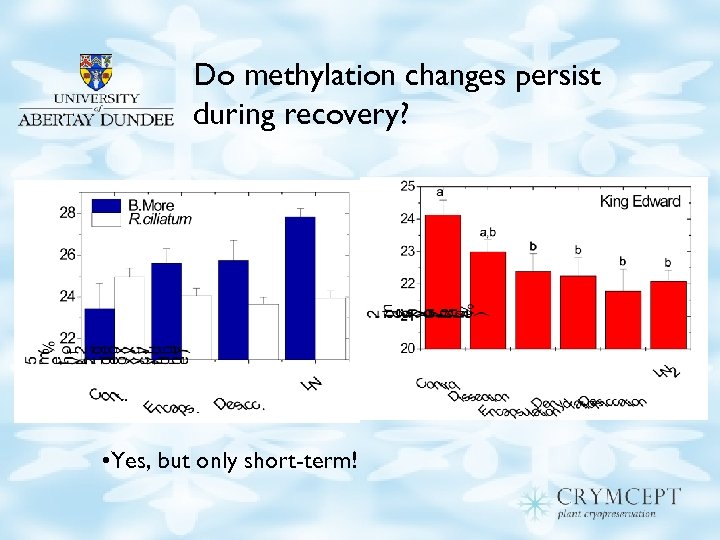
Do methylation changes persist during recovery? • Yes, but only short-term!

Ethylene (WP 7 & 8)

Genotype comparison of ethylene production during cryo (WP 7) • Ethylene an major hormone in plants – Radial cell expansion – Senescence – Mediator of stress responses • Contrasting ethylene production between Ribes genotypes with different % shoot recovery after cryo • Cause, consequence or coincidence?
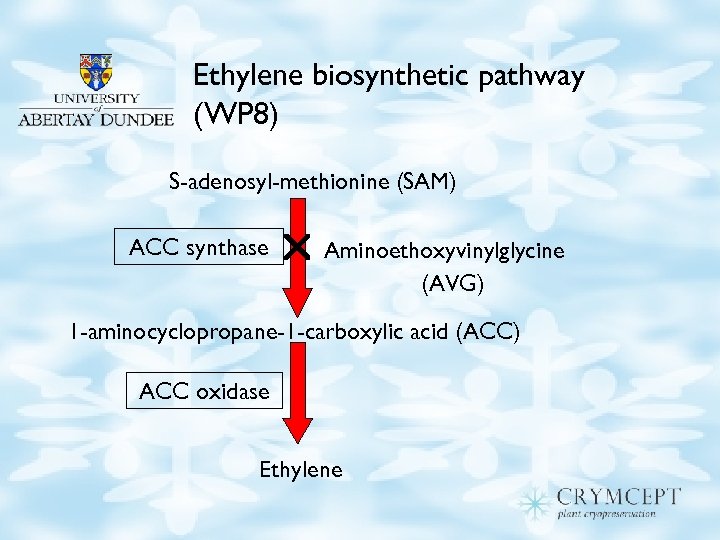
Ethylene biosynthetic pathway (WP 8) S-adenosyl-methionine (SAM) ACC synthase Aminoethoxyvinylglycine (AVG) 1 -aminocyclopropane-1 -carboxylic acid (ACC) ACC oxidase Ethylene
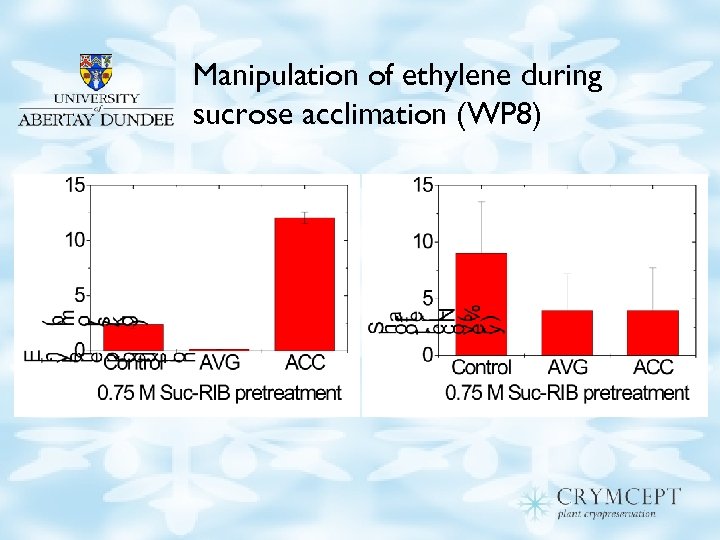
Manipulation of ethylene during sucrose acclimation (WP 8)

Manipulation of ethylene during recovery (WP 8) • Stimulation of ethylene production during recovery had no effect on shoot recovery • Stimulation of ethylene production by ACC only occurred in meristems that survived cryo – May indicate membrane damage in non-surviving meristems • AVG inhibited meristem growth – Ethylene maybe essential for meristem growth

Model for cryo-injury
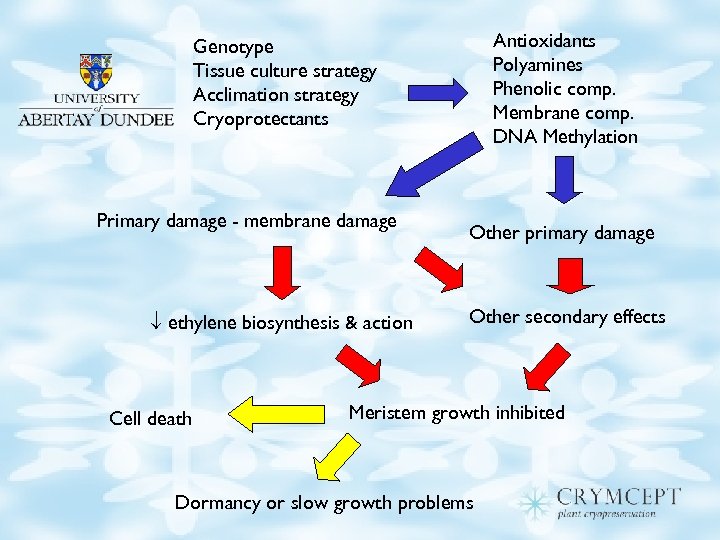
Antioxidants Polyamines Phenolic comp. Membrane comp. DNA Methylation Genotype Tissue culture strategy Acclimation strategy Cryoprotectants Primary damage - membrane damage ethylene biosynthesis & action Cell death Other primary damage Other secondary effects Meristem growth inhibited Dormancy or slow growth problems

Conclusions • DSC effective at optimising cryo-protocols for tropical & temperates species, but cannot explain genotype survival differences for Ribes meristems – Physiological problem • Accumulation of antioxidants during pretreatment important for recovery • Tissue culture and pretreatment regimes required that enhance endogenous antioxidants

Conclusions • DNA methylation changes during sucrose pretreatment – May have some genetic stability implications – May be required for survival! • Ethylene not involved in sucrose acclimation responses • Ethylene maybe essential for meristem growth

Acknowledgements • Thanks to all partners • Paul Lynch for assisting with P 2 and WP 7 PI activities
ada88f4fa25f6698500d2b458364a6ec.ppt