0e964ce4b09b69e20d3bf7c65e6295ad.ppt
- Количество слайдов: 22
 Universidad Politécnica de Valencia INSTITUTO DE TECNOLOGÍA QUIMICA, UPV-CSIC Drug-Biomolecule Interactions in the Excited States Miguel A. Miranda Israel, April 2010
Universidad Politécnica de Valencia INSTITUTO DE TECNOLOGÍA QUIMICA, UPV-CSIC Drug-Biomolecule Interactions in the Excited States Miguel A. Miranda Israel, April 2010
 Outline - Introduction - Photodamage to DNA - Photorepair - Conclusions
Outline - Introduction - Photodamage to DNA - Photorepair - Conclusions
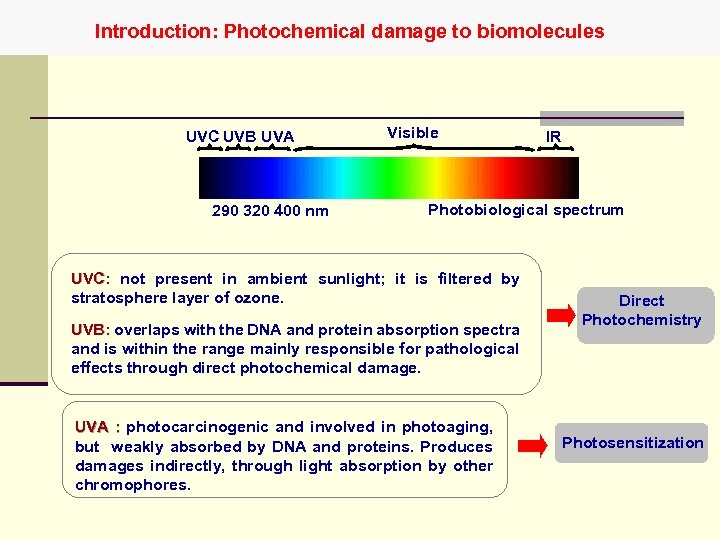 Introduction: Photochemical damage to biomolecules UVC UVB UVA 290 320 400 nm Visible IR Photobiological spectrum UVC: not present in ambient sunlight; it is filtered by stratosphere layer of ozone. UVB: overlaps with the DNA and protein absorption spectra and is within the range mainly responsible for pathological effects through direct photochemical damage. UVA : photocarcinogenic and involved in photoaging, but weakly absorbed by DNA and proteins. Produces damages indirectly, through light absorption by other chromophores. Direct Photochemistry Photosensitization
Introduction: Photochemical damage to biomolecules UVC UVB UVA 290 320 400 nm Visible IR Photobiological spectrum UVC: not present in ambient sunlight; it is filtered by stratosphere layer of ozone. UVB: overlaps with the DNA and protein absorption spectra and is within the range mainly responsible for pathological effects through direct photochemical damage. UVA : photocarcinogenic and involved in photoaging, but weakly absorbed by DNA and proteins. Produces damages indirectly, through light absorption by other chromophores. Direct Photochemistry Photosensitization
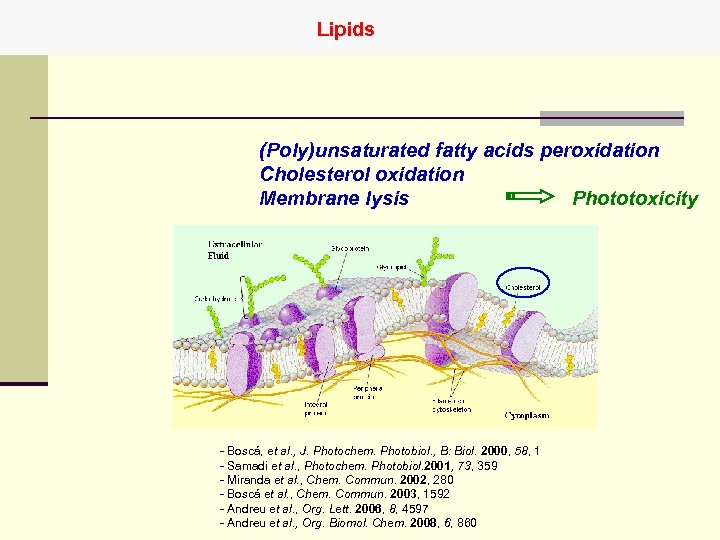 Lipids (Poly)unsaturated fatty acids peroxidation Cholesterol oxidation Phototoxicity Membrane lysis - Boscá, et al. , J. Photochem. Photobiol. , B: Biol. 2000, 58, 1 - Samadi et al. , Photochem. Photobiol. 2001, 73, 359 - Miranda et al. , Chem. Commun. 2002, 280 - Boscá et al. , Chem. Commun. 2003, 1592 - Andreu et al. , Org. Lett. 2006, 8, 4597 - Andreu et al. , Org. Biomol. Chem. 2008, 6, 860
Lipids (Poly)unsaturated fatty acids peroxidation Cholesterol oxidation Phototoxicity Membrane lysis - Boscá, et al. , J. Photochem. Photobiol. , B: Biol. 2000, 58, 1 - Samadi et al. , Photochem. Photobiol. 2001, 73, 359 - Miranda et al. , Chem. Commun. 2002, 280 - Boscá et al. , Chem. Commun. 2003, 1592 - Andreu et al. , Org. Lett. 2006, 8, 4597 - Andreu et al. , Org. Biomol. Chem. 2008, 6, 860
 Proteins Drug-protein photobinding Protein-protein photocrosslinking Binding site occupancy Model dyads Ø Miranda et al. J. Am. Chem. Soc. , 1999, 121, 11569. Ø Miranda et al. Chem. Commun. , 2000, 2257. Ø Perez-Prieto et al. J. Org. Chem. , 2004, 69, 374. Ø Perez-Prieto et al. J. Org. Chem. , 2004, 69, 8618. Ø Lhiaubet-Vallet et al. J. Phys. Chem. B 2007, 111, 423. Ø Vaya et al. J. Phys. Chem. B 2007, 111, 9363. Ø Vaya et al. Chem. Phys. Lett. 2010, 486, 147. Photoallergy Proteins Ø Miranda et al. Chem. Res. Toxicol. 1998, 11, 172. Ø Lahoz et al. Chem. Res. Toxicol. 2001, 1486. Ø Lhiaubet-Vallet et al. J. Am. Chem. Soc. 2004, 126, 9538. Ø Jimenez et al. J. Am. Chem Soc. 2005, 127, 10134. Ø Vaya et al. Chem. Med. Chem 2006, 1, 1015. Ø Lhiaubet-Vallet et al. J. Phys. Chem. B 2007, 111, 423. Ø Vaya et al. J. Phys. Chem. B 2008, 112, 2694. Ø Vaya et al. Chem Eur. J. 2008, 14, 11284. Ø Montanaro et al. Chem. Med. Chem 2009, 4, 1196. Ø Bueno et al J. Phys. Chem. B 2009, 113, 6861. Ø Perez-Ruiz et al, J. Phys. Chem. Lett. 2010, 829.
Proteins Drug-protein photobinding Protein-protein photocrosslinking Binding site occupancy Model dyads Ø Miranda et al. J. Am. Chem. Soc. , 1999, 121, 11569. Ø Miranda et al. Chem. Commun. , 2000, 2257. Ø Perez-Prieto et al. J. Org. Chem. , 2004, 69, 374. Ø Perez-Prieto et al. J. Org. Chem. , 2004, 69, 8618. Ø Lhiaubet-Vallet et al. J. Phys. Chem. B 2007, 111, 423. Ø Vaya et al. J. Phys. Chem. B 2007, 111, 9363. Ø Vaya et al. Chem. Phys. Lett. 2010, 486, 147. Photoallergy Proteins Ø Miranda et al. Chem. Res. Toxicol. 1998, 11, 172. Ø Lahoz et al. Chem. Res. Toxicol. 2001, 1486. Ø Lhiaubet-Vallet et al. J. Am. Chem. Soc. 2004, 126, 9538. Ø Jimenez et al. J. Am. Chem Soc. 2005, 127, 10134. Ø Vaya et al. Chem. Med. Chem 2006, 1, 1015. Ø Lhiaubet-Vallet et al. J. Phys. Chem. B 2007, 111, 423. Ø Vaya et al. J. Phys. Chem. B 2008, 112, 2694. Ø Vaya et al. Chem Eur. J. 2008, 14, 11284. Ø Montanaro et al. Chem. Med. Chem 2009, 4, 1196. Ø Bueno et al J. Phys. Chem. B 2009, 113, 6861. Ø Perez-Ruiz et al, J. Phys. Chem. Lett. 2010, 829.
 Nucleic acids Base damage Photogenotoxicity Photomutagenicity Thymine dimerization Guanine oxidation Ø Lhiaubet-Vallet et al. Photochem. Photobiol. 2003, 77, 487. Ø Lhiaubet-Vallet et al. J. Phys Chem. B 2004, 108, 14148. Ø Bosca et al. J. Am. Chem. Soc. 2006, 128, 6318. Ø Lhiaubet-Vallet et al. J. Phys. Chem. B 2007 111, 7409. Ø Trzcionka et al. Chem. Bio. Chem 2007, 4, 367. Ø Lhiaubet-Vallet et al. Photochem. Photobiol. 2009, 85, 861 Ø Agapakis et al. Photochem. Photobiol. 2000, 71, 499 Ø Belvedere et al. Chem. Res. Toxicol. 2002, 15, 1142. Ø Cuquerella et al. Chem. Res. Toxicol. 2003, 16, 562. Ø Lhiaubet-Vallet et al. Toxicol. in vitro 2003, 17, 651. Ø Chatgilialoglu et al. Chem. Res. Toxicol. 2007, 20, 1820. Ø Paris et al. Org. Lett. 2008, 10, 4409. Stereodifferentiation (chiral drug/nucleosides) Ø Encinas et al. Chem. Phys. Chem, 2004, 5, 1704. Ø Lhiaubet-Vallet et al. J. Am. Chem. Soc. 2005, 12774. Ø Encinas et al. Chem. Commun. 2005, 272. Ø Belmadoui et al. Chem. Eur. J. 2006, 12, 553. Ø Jimenez et al. Org. Biomol. Chem. 2008, 6, 860.
Nucleic acids Base damage Photogenotoxicity Photomutagenicity Thymine dimerization Guanine oxidation Ø Lhiaubet-Vallet et al. Photochem. Photobiol. 2003, 77, 487. Ø Lhiaubet-Vallet et al. J. Phys Chem. B 2004, 108, 14148. Ø Bosca et al. J. Am. Chem. Soc. 2006, 128, 6318. Ø Lhiaubet-Vallet et al. J. Phys. Chem. B 2007 111, 7409. Ø Trzcionka et al. Chem. Bio. Chem 2007, 4, 367. Ø Lhiaubet-Vallet et al. Photochem. Photobiol. 2009, 85, 861 Ø Agapakis et al. Photochem. Photobiol. 2000, 71, 499 Ø Belvedere et al. Chem. Res. Toxicol. 2002, 15, 1142. Ø Cuquerella et al. Chem. Res. Toxicol. 2003, 16, 562. Ø Lhiaubet-Vallet et al. Toxicol. in vitro 2003, 17, 651. Ø Chatgilialoglu et al. Chem. Res. Toxicol. 2007, 20, 1820. Ø Paris et al. Org. Lett. 2008, 10, 4409. Stereodifferentiation (chiral drug/nucleosides) Ø Encinas et al. Chem. Phys. Chem, 2004, 5, 1704. Ø Lhiaubet-Vallet et al. J. Am. Chem. Soc. 2005, 12774. Ø Encinas et al. Chem. Commun. 2005, 272. Ø Belmadoui et al. Chem. Eur. J. 2006, 12, 553. Ø Jimenez et al. Org. Biomol. Chem. 2008, 6, 860.
 - Introduction - Photodamage to DNA - Photorepair - Conclusions
- Introduction - Photodamage to DNA - Photorepair - Conclusions
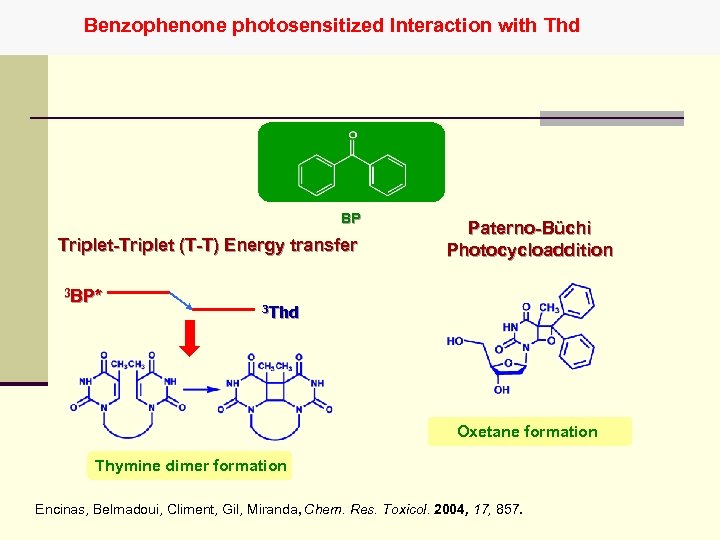 Benzophenone photosensitized Interaction with Thd BP Triplet-Triplet (T-T) Energy transfer 3 BP* Paterno-Büchi Photocycloaddition 3 Thd Oxetane formation Thymine dimer formation Encinas, Belmadoui, Climent, Gil, Miranda, Chem. Res. Toxicol. 2004, 17, 857.
Benzophenone photosensitized Interaction with Thd BP Triplet-Triplet (T-T) Energy transfer 3 BP* Paterno-Büchi Photocycloaddition 3 Thd Oxetane formation Thymine dimer formation Encinas, Belmadoui, Climent, Gil, Miranda, Chem. Res. Toxicol. 2004, 17, 857.
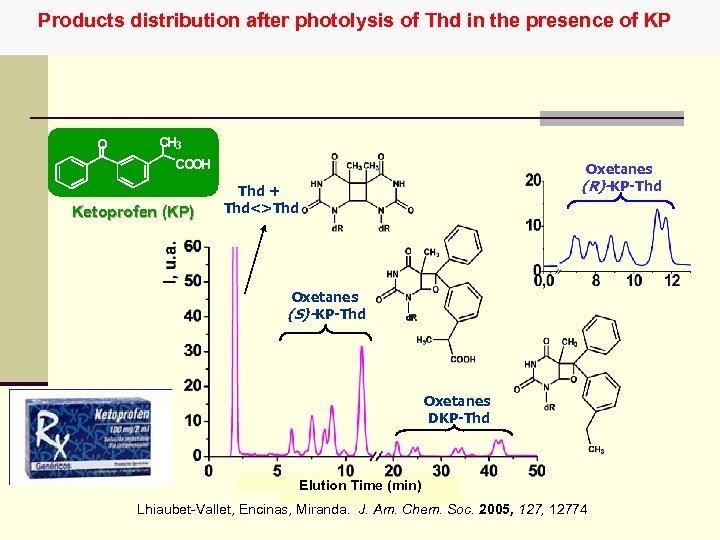 Products distribution after photolysis of Thd in the presence of KP O CH 3 COOH Ketoprofen (KP) Oxetanes (R)-KP-Thd + Thd<>Thd Oxetanes (S)-KP-Thd Oxetanes DKP-Thd Elution Time (min) Lhiaubet-Vallet, Encinas, Miranda. J. Am. Chem. Soc. 2005, 12774
Products distribution after photolysis of Thd in the presence of KP O CH 3 COOH Ketoprofen (KP) Oxetanes (R)-KP-Thd + Thd<>Thd Oxetanes (S)-KP-Thd Oxetanes DKP-Thd Elution Time (min) Lhiaubet-Vallet, Encinas, Miranda. J. Am. Chem. Soc. 2005, 12774
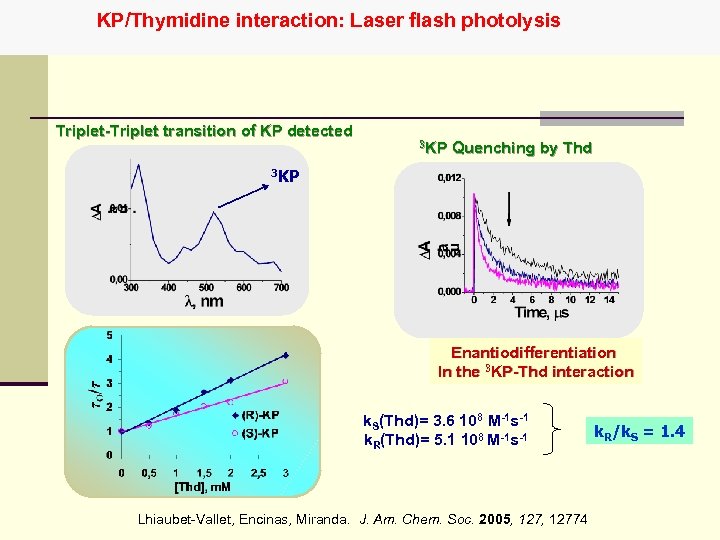 KP/Thymidine interaction: Laser flash photolysis Triplet-Triplet transition of KP detected 3 KP Quenching by Thd 3 KP Enantiodifferentiation In the 3 KP-Thd interaction k. S(Thd)= 3. 6 108 M-1 s-1 k. R(Thd)= 5. 1 108 M-1 s-1 Lhiaubet-Vallet, Encinas, Miranda. J. Am. Chem. Soc. 2005, 12774 k. R/k. S = 1. 4
KP/Thymidine interaction: Laser flash photolysis Triplet-Triplet transition of KP detected 3 KP Quenching by Thd 3 KP Enantiodifferentiation In the 3 KP-Thd interaction k. S(Thd)= 3. 6 108 M-1 s-1 k. R(Thd)= 5. 1 108 M-1 s-1 Lhiaubet-Vallet, Encinas, Miranda. J. Am. Chem. Soc. 2005, 12774 k. R/k. S = 1. 4
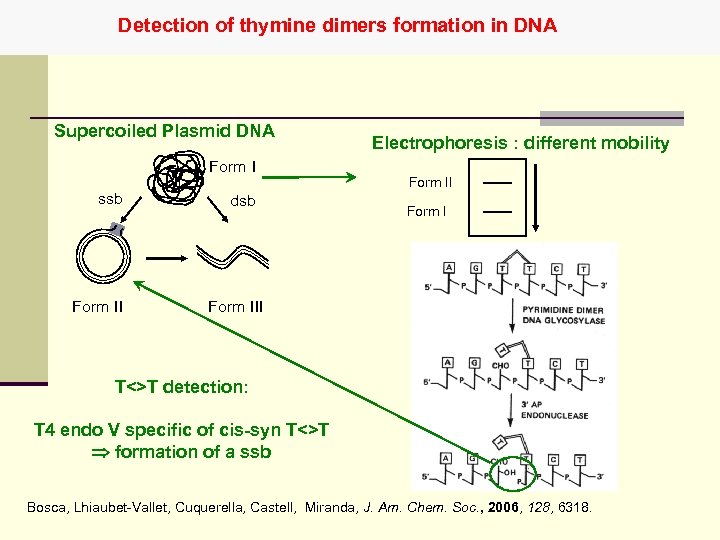 Detection of thymine dimers formation in DNA Supercoiled Plasmid DNA Form I ssb Form II dsb Electrophoresis : different mobility Form III T<>T detection: T 4 endo V specific of cis-syn T<>T formation of a ssb Bosca, Lhiaubet-Vallet, Cuquerella, Castell, Miranda, J. Am. Chem. Soc. , 2006, 128, 6318.
Detection of thymine dimers formation in DNA Supercoiled Plasmid DNA Form I ssb Form II dsb Electrophoresis : different mobility Form III T<>T detection: T 4 endo V specific of cis-syn T<>T formation of a ssb Bosca, Lhiaubet-Vallet, Cuquerella, Castell, Miranda, J. Am. Chem. Soc. , 2006, 128, 6318.
 DNA-photosensitization Agarose gel electrophoresis T<>T • p. BR + FQ (20 m. M) + UVA (355 nm) • enzymatic treatment with T 4 endonuclease V No T<>T ENX, NFX and PFX sensitize T<>T but ANFX does not!! Form II Form I T<>T formation native DNA Bosca, Lhiaubet-Vallet, Cuquerella, Castell, Miranda, J. Am. Chem. Soc. , 2006, 128, 6318. Lhiaubet-Vallet, Cuquerella, Castell, Bosca, Miranda, J. Phys. Chem. B, 2007, 111, 7409.
DNA-photosensitization Agarose gel electrophoresis T<>T • p. BR + FQ (20 m. M) + UVA (355 nm) • enzymatic treatment with T 4 endonuclease V No T<>T ENX, NFX and PFX sensitize T<>T but ANFX does not!! Form II Form I T<>T formation native DNA Bosca, Lhiaubet-Vallet, Cuquerella, Castell, Miranda, J. Am. Chem. Soc. , 2006, 128, 6318. Lhiaubet-Vallet, Cuquerella, Castell, Bosca, Miranda, J. Phys. Chem. B, 2007, 111, 7409.
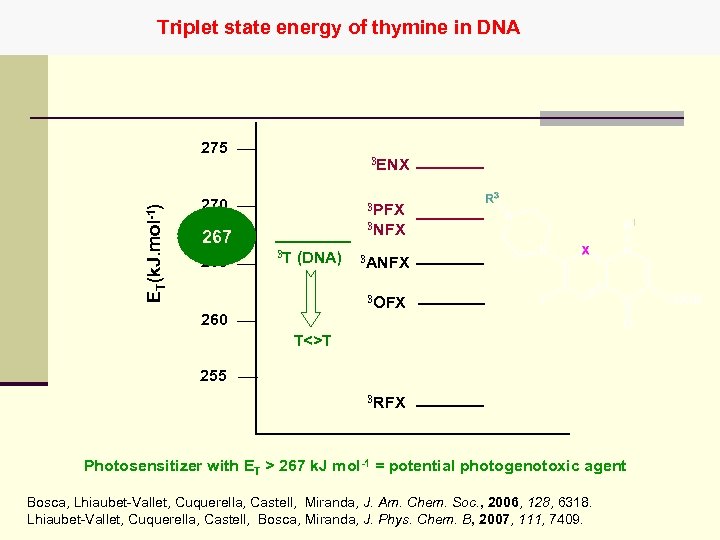 Triplet state energy of thymine in DNA ET(k. J. mol-1) 275 3 ENX 270 3 PFX 3 NFX 267 265 3 T (DNA) 3 ANFX 3 OFX 260 T<>T 255 3 RFX Photosensitizer with ET > 267 k. J mol-1 = potential photogenotoxic agent Bosca, Lhiaubet-Vallet, Cuquerella, Castell, Miranda, J. Am. Chem. Soc. , 2006, 128, 6318. Lhiaubet-Vallet, Cuquerella, Castell, Bosca, Miranda, J. Phys. Chem. B, 2007, 111, 7409.
Triplet state energy of thymine in DNA ET(k. J. mol-1) 275 3 ENX 270 3 PFX 3 NFX 267 265 3 T (DNA) 3 ANFX 3 OFX 260 T<>T 255 3 RFX Photosensitizer with ET > 267 k. J mol-1 = potential photogenotoxic agent Bosca, Lhiaubet-Vallet, Cuquerella, Castell, Miranda, J. Am. Chem. Soc. , 2006, 128, 6318. Lhiaubet-Vallet, Cuquerella, Castell, Bosca, Miranda, J. Phys. Chem. B, 2007, 111, 7409.
 - Introduction - Photodamage to biomolecules - Photorepair - Conclusions
- Introduction - Photodamage to biomolecules - Photorepair - Conclusions
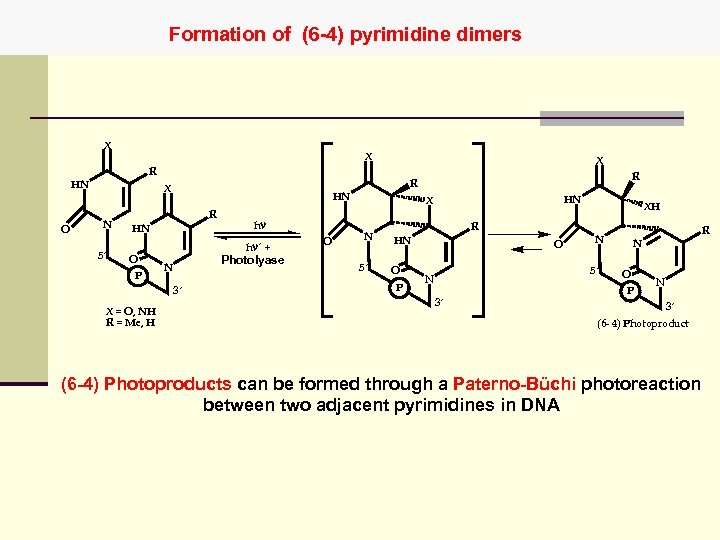 Formation of (6 -4) pyrimidine dimers X X X R HN O N 5´ HN R HN O P hn hn´ + Photolyase N 3´ X = O, NH R = Me, H R R X O HN X N 5´ R HN O P O XH N 5´ N 3´ R N O P N 3´ (6 - 4) Photoproduct (6 -4) Photoproducts can be formed through a Paterno-Büchi photoreaction between two adjacent pyrimidines in DNA
Formation of (6 -4) pyrimidine dimers X X X R HN O N 5´ HN R HN O P hn hn´ + Photolyase N 3´ X = O, NH R = Me, H R R X O HN X N 5´ R HN O P O XH N 5´ N 3´ R N O P N 3´ (6 - 4) Photoproduct (6 -4) Photoproducts can be formed through a Paterno-Büchi photoreaction between two adjacent pyrimidines in DNA
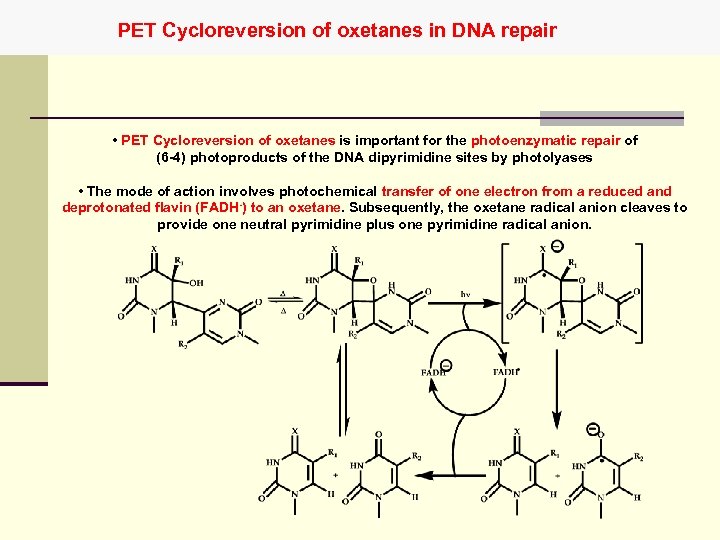 PET Cycloreversion of oxetanes in DNA repair • PET Cycloreversion of oxetanes is important for the photoenzymatic repair of (6 -4) photoproducts of the DNA dipyrimidine sites by photolyases • The mode of action involves photochemical transfer of one electron from a reduced and deprotonated flavin (FADH-) to an oxetane. Subsequently, the oxetane radical anion cleaves to provide one neutral pyrimidine plus one pyrimidine radical anion.
PET Cycloreversion of oxetanes in DNA repair • PET Cycloreversion of oxetanes is important for the photoenzymatic repair of (6 -4) photoproducts of the DNA dipyrimidine sites by photolyases • The mode of action involves photochemical transfer of one electron from a reduced and deprotonated flavin (FADH-) to an oxetane. Subsequently, the oxetane radical anion cleaves to provide one neutral pyrimidine plus one pyrimidine radical anion.
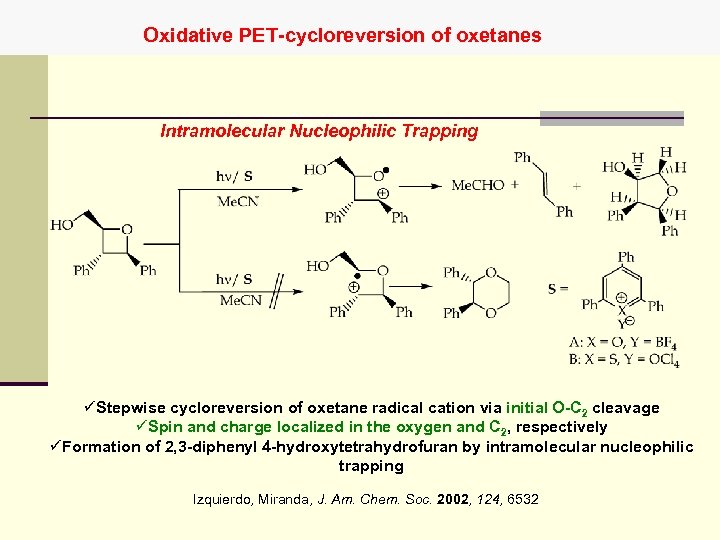 Oxidative PET-cycloreversion of oxetanes Intramolecular Nucleophilic Trapping üStepwise cycloreversion of oxetane radical cation via initial O-C 2 cleavage üSpin and charge localized in the oxygen and C 2, respectively üFormation of 2, 3 -diphenyl 4 -hydroxytetrahydrofuran by intramolecular nucleophilic trapping Izquierdo, Miranda, J. Am. Chem. Soc. 2002, 124, 6532
Oxidative PET-cycloreversion of oxetanes Intramolecular Nucleophilic Trapping üStepwise cycloreversion of oxetane radical cation via initial O-C 2 cleavage üSpin and charge localized in the oxygen and C 2, respectively üFormation of 2, 3 -diphenyl 4 -hydroxytetrahydrofuran by intramolecular nucleophilic trapping Izquierdo, Miranda, J. Am. Chem. Soc. 2002, 124, 6532
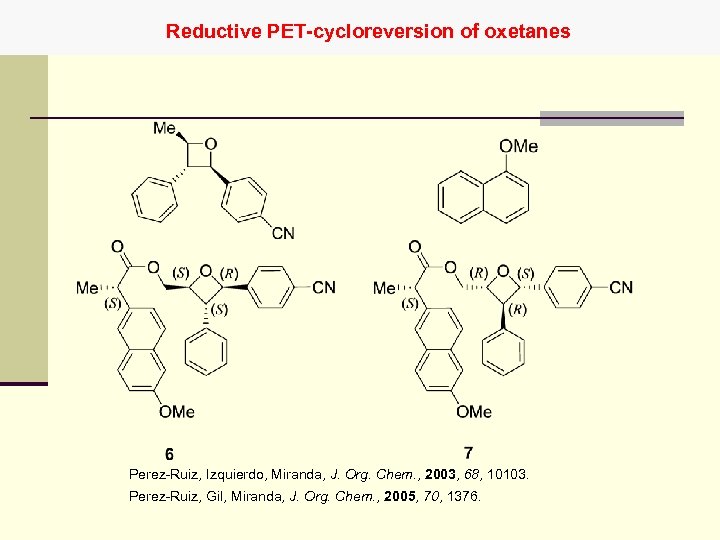 Reductive PET-cycloreversion of oxetanes Perez-Ruiz, Izquierdo, Miranda, J. Org. Chem. , 2003, 68, 10103. Perez-Ruiz, Gil, Miranda, J. Org. Chem. , 2005, 70, 1376.
Reductive PET-cycloreversion of oxetanes Perez-Ruiz, Izquierdo, Miranda, J. Org. Chem. , 2003, 68, 10103. Perez-Ruiz, Gil, Miranda, J. Org. Chem. , 2005, 70, 1376.
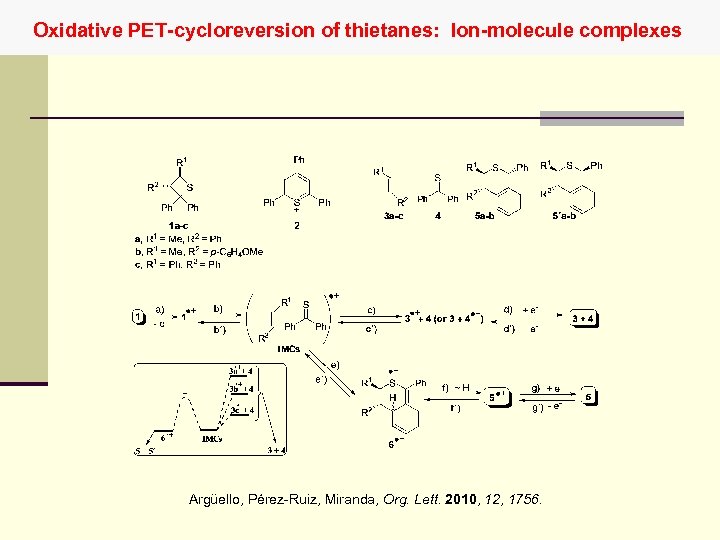 Oxidative PET-cycloreversion of thietanes: Ion-molecule complexes Argüello, Pérez-Ruiz, Miranda, Org. Lett. 2010, 12, 1756.
Oxidative PET-cycloreversion of thietanes: Ion-molecule complexes Argüello, Pérez-Ruiz, Miranda, Org. Lett. 2010, 12, 1756.
 - Introduction - Photodamage to biomolecules - Photoprotection/photorepair - Conclusions
- Introduction - Photodamage to biomolecules - Photoprotection/photorepair - Conclusions
 Conclusions - Solar (and artificial) light may produce both desired and undesired effects on biological systems - It is necessary to achieve a satisfactory understanding of the chemical mechanisms involved in photobiological effects - Based on mechanistic knowledge, it is possible to minimize the adverse effects of light, while enhancing its beneficial effects
Conclusions - Solar (and artificial) light may produce both desired and undesired effects on biological systems - It is necessary to achieve a satisfactory understanding of the chemical mechanisms involved in photobiological effects - Based on mechanistic knowledge, it is possible to minimize the adverse effects of light, while enhancing its beneficial effects
 ACKNOWLEDGEMENTS Ph. D STUDENTS POST-DOCS COLLABORATIONS P. Bartovsky P. Bonancia M. Gómez M. Marin G. Nardi E. Nuin L. Piñero J. Rohacova S. Soldevila R. Alonso I. Andreu M. C. Cuquerella V. Lhiaubet-Vallet R. Pérez UPV-CSIC F. Boscá S. Encinas M. J. Climent M. C. Jiménez M. L. Marin I. M. Morera R. Tormos FUNDING European Union Spanish Government Regional Government UPV and CSIC ISDIN Organon/Merck EXTERNAL J. V. Castell J. Pérez-Prieto T. Gimisis C. Chatgilialoglu
ACKNOWLEDGEMENTS Ph. D STUDENTS POST-DOCS COLLABORATIONS P. Bartovsky P. Bonancia M. Gómez M. Marin G. Nardi E. Nuin L. Piñero J. Rohacova S. Soldevila R. Alonso I. Andreu M. C. Cuquerella V. Lhiaubet-Vallet R. Pérez UPV-CSIC F. Boscá S. Encinas M. J. Climent M. C. Jiménez M. L. Marin I. M. Morera R. Tormos FUNDING European Union Spanish Government Regional Government UPV and CSIC ISDIN Organon/Merck EXTERNAL J. V. Castell J. Pérez-Prieto T. Gimisis C. Chatgilialoglu


