57474ab036f3092c2cff609a9c96571a.ppt
- Количество слайдов: 9
 UNITAID actions and main achievements Investing in the quality of medicines XVII International AIDS Conference Satellite Forum WHO – Essential Medicines for HIV/AIDS (Mexico, 4 th August 2008) Jorge Bermudez Executive-Secretary, UNITAID
UNITAID actions and main achievements Investing in the quality of medicines XVII International AIDS Conference Satellite Forum WHO – Essential Medicines for HIV/AIDS (Mexico, 4 th August 2008) Jorge Bermudez Executive-Secretary, UNITAID
 UNITAID added value UNITAID use of funds allows to: 1/ Impact markets and reduce prices: more drugs for same budget ex: price reduction on pediatric (- 40%) and 2 nd line ARVs (-25% to – 50%) 2/ Have manufactured medicines better adapted to patient needs ex: first fixed dose combinations for pediatric ARVs in 2007 3/ Contribute to address quality issues (incentive for manufacturers to invest) ex: support to WHO program for prequalification of products 4/ Deliver rapidly medicines in the countries in need (basic model - medicines instead of funds) ex: treatments already provided in a number of countries for ARV, TB and ACT
UNITAID added value UNITAID use of funds allows to: 1/ Impact markets and reduce prices: more drugs for same budget ex: price reduction on pediatric (- 40%) and 2 nd line ARVs (-25% to – 50%) 2/ Have manufactured medicines better adapted to patient needs ex: first fixed dose combinations for pediatric ARVs in 2007 3/ Contribute to address quality issues (incentive for manufacturers to invest) ex: support to WHO program for prequalification of products 4/ Deliver rapidly medicines in the countries in need (basic model - medicines instead of funds) ex: treatments already provided in a number of countries for ARV, TB and ACT
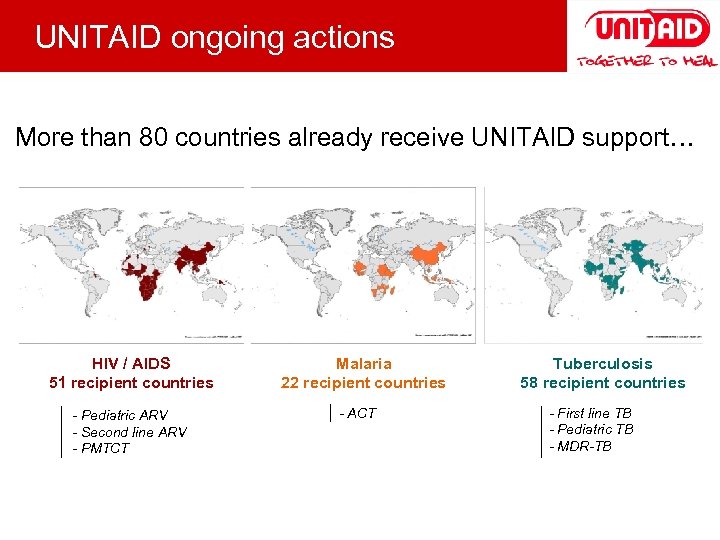 UNITAID ongoing actions More than 80 countries already receive UNITAID support… HIV / AIDS 51 recipient countries - Pediatric ARV - Second line ARV - PMTCT Malaria 22 recipient countries - ACT Tuberculosis 58 recipient countries - First line TB - Pediatric TB - MDR-TB
UNITAID ongoing actions More than 80 countries already receive UNITAID support… HIV / AIDS 51 recipient countries - Pediatric ARV - Second line ARV - PMTCT Malaria 22 recipient countries - ACT Tuberculosis 58 recipient countries - First line TB - Pediatric TB - MDR-TB
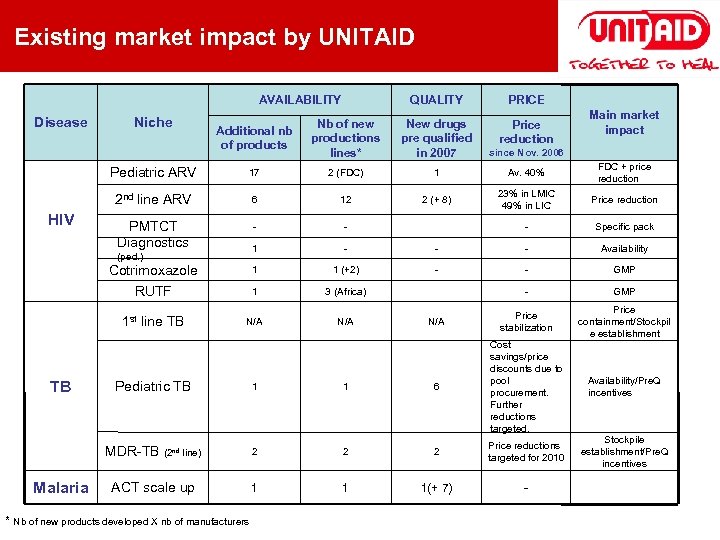 Existing market impact by UNITAID AVAILABILITY Additional nb of products Nb of new productions lines* New drugs pre qualified in 2007 Price reduction since Nov. 2006 17 2 (FDC) 1 Av. 40% FDC + price reduction 2 nd line ARV 6 12 2 (+ 8) 23% in LMIC 49% in LIC Price reduction PMTCT Diagnostics - - - Specific pack 1 - - - Availability Cotrimoxazole 1 1 (+2) - - GMP RUTF 1 3 (Africa) - GMP 1 st line TB HIV PRICE Pediatric ARV Disease QUALITY N/A N/A Price stabilization Price containment/Stockpil e establishment Availability/Pre. Q incentives Stockpile establishment/Pre. Q incentives Niche (ped. ) TB Pediatric TB MDR-TB Malaria (2 nd line) ACT scale up * Nb of new products developed X nb of manufacturers 1 1 6 Cost savings/price discounts due to pool procurement. Further reductions targeted. 2 2 2 Price reductions targeted for 2010 1 1 1(+ 7) - Main market impact
Existing market impact by UNITAID AVAILABILITY Additional nb of products Nb of new productions lines* New drugs pre qualified in 2007 Price reduction since Nov. 2006 17 2 (FDC) 1 Av. 40% FDC + price reduction 2 nd line ARV 6 12 2 (+ 8) 23% in LMIC 49% in LIC Price reduction PMTCT Diagnostics - - - Specific pack 1 - - - Availability Cotrimoxazole 1 1 (+2) - - GMP RUTF 1 3 (Africa) - GMP 1 st line TB HIV PRICE Pediatric ARV Disease QUALITY N/A N/A Price stabilization Price containment/Stockpil e establishment Availability/Pre. Q incentives Stockpile establishment/Pre. Q incentives Niche (ped. ) TB Pediatric TB MDR-TB Malaria (2 nd line) ACT scale up * Nb of new products developed X nb of manufacturers 1 1 6 Cost savings/price discounts due to pool procurement. Further reductions targeted. 2 2 2 Price reductions targeted for 2010 1 1 1(+ 7) - Main market impact
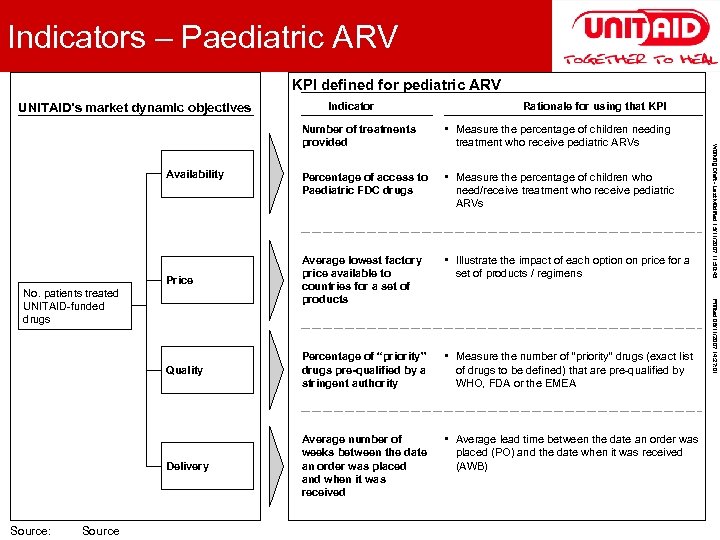 2 Indicators – Paediatric ARV Unit of measure KPI defined for pediatric ARV UNITAID’s market dynamic objectives Indicator Rationale for using that KPI • Measure the percentage of children needing • Measure the percentage of children who • Illustrate the impact of each option on price for a Price Average lowest factory price available to countries for a set of products Percentage of “priority” drugs pre-qualified by a stringent authority • Measure the number of “priority” drugs (exact list Quality • Average lead time between the date an order was Delivery Average number of weeks between the date an order was placed and when it was received No. patients treated UNITAID-funded drugs * Source: Footnote Source need/receive treatment who receive pediatric ARVs set of products / regimens of drugs to be defined) that are pre-qualified by WHO, FDA or the EMEA placed (PO) and the date when it was received (AWB) Printed 06/11/2007 14: 27: 01 Percentage of access to Paediatric FDC drugs Availability treatment who receive pediatric ARVs Working Draft - Last Modified 15/11/2007 11: 50: 45 Number of treatments provided
2 Indicators – Paediatric ARV Unit of measure KPI defined for pediatric ARV UNITAID’s market dynamic objectives Indicator Rationale for using that KPI • Measure the percentage of children needing • Measure the percentage of children who • Illustrate the impact of each option on price for a Price Average lowest factory price available to countries for a set of products Percentage of “priority” drugs pre-qualified by a stringent authority • Measure the number of “priority” drugs (exact list Quality • Average lead time between the date an order was Delivery Average number of weeks between the date an order was placed and when it was received No. patients treated UNITAID-funded drugs * Source: Footnote Source need/receive treatment who receive pediatric ARVs set of products / regimens of drugs to be defined) that are pre-qualified by WHO, FDA or the EMEA placed (PO) and the date when it was received (AWB) Printed 06/11/2007 14: 27: 01 Percentage of access to Paediatric FDC drugs Availability treatment who receive pediatric ARVs Working Draft - Last Modified 15/11/2007 11: 50: 45 Number of treatments provided
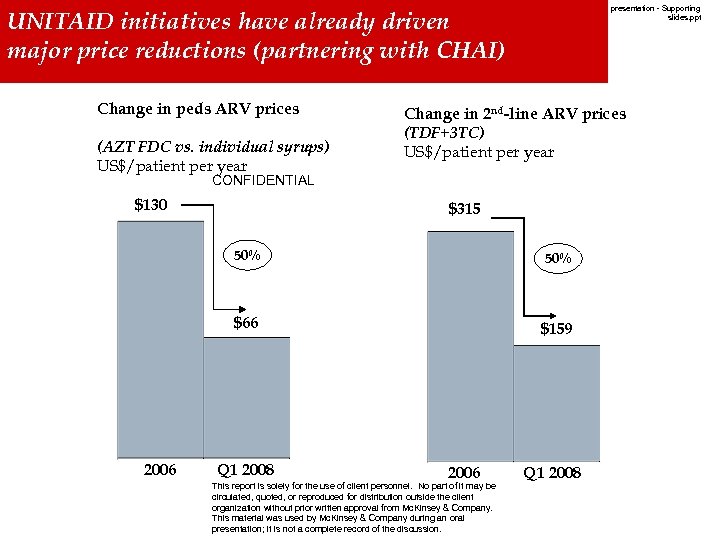 UNITAID initiatives have already driven Working reductions Standard Time major price. Draft W. Europe(partnering with CHAI) Last Modified 15/11/2007 11: 50: 45 20071207 Board presentation - Supporting slides. ppt Printed 06/11/2007 14: 27: 01 Romance Standard Time Change in peds ARV prices (AZT FDC vs. individual syrups) US$/patient per year Change in 2 nd-line ARV prices (TDF+3 TC) US$/patient per year CONFIDENTIAL $130 $315 50% $66 $159 Document Date 2006 Q 1 2008 2006 This report is solely for the use of client personnel. No part of it may be circulated, quoted, or reproduced for distribution outside the client organization without prior written approval from Mc. Kinsey & Company. This material was used by Mc. Kinsey & Company during an oral presentation; it is not a complete record of the discussion. Q 1 2008
UNITAID initiatives have already driven Working reductions Standard Time major price. Draft W. Europe(partnering with CHAI) Last Modified 15/11/2007 11: 50: 45 20071207 Board presentation - Supporting slides. ppt Printed 06/11/2007 14: 27: 01 Romance Standard Time Change in peds ARV prices (AZT FDC vs. individual syrups) US$/patient per year Change in 2 nd-line ARV prices (TDF+3 TC) US$/patient per year CONFIDENTIAL $130 $315 50% $66 $159 Document Date 2006 Q 1 2008 2006 This report is solely for the use of client personnel. No part of it may be circulated, quoted, or reproduced for distribution outside the client organization without prior written approval from Mc. Kinsey & Company. This material was used by Mc. Kinsey & Company during an oral presentation; it is not a complete record of the discussion. Q 1 2008
 2 Better products at lower price Unit of measure Now (UNITAID-CHAI): Fixed dose combination 3 tablets a day 200 $ per patient per year 60 $per patient per year Working Draft - Last Modified 15/11/2007 11: 50: 45 Before: Single dose syrups 16 bottles of syrup monthly Printed 06/11/2007 14: 27: 01 * Source: Footnote Source
2 Better products at lower price Unit of measure Now (UNITAID-CHAI): Fixed dose combination 3 tablets a day 200 $ per patient per year 60 $per patient per year Working Draft - Last Modified 15/11/2007 11: 50: 45 Before: Single dose syrups 16 bottles of syrup monthly Printed 06/11/2007 14: 27: 01 * Source: Footnote Source
 UNITAID: challenges ahead Consolidate current niches and necessary transitions with partners Identify areas to expand (diagnostics…) VSC (a worldwide citizenship solidarity contribution) Moving towards a patent pool
UNITAID: challenges ahead Consolidate current niches and necessary transitions with partners Identify areas to expand (diagnostics…) VSC (a worldwide citizenship solidarity contribution) Moving towards a patent pool
 20071207 Board presentation - Supporting slides. ppt www. unitaid. eu Working Draft Last Modified 15/11/2007 11: 50: 45 W. Europe Standard Time Printed 06/11/2007 14: 27: 01 Romance Standard Time CONFIDENTIAL Document Date This report is solely for the use of client personnel. No part of it may be circulated, quoted, or reproduced for distribution outside the client organization without prior written approval from Mc. Kinsey & Company. This material was used by Mc. Kinsey & Company during an oral presentation; it is not a complete record of the discussion.
20071207 Board presentation - Supporting slides. ppt www. unitaid. eu Working Draft Last Modified 15/11/2007 11: 50: 45 W. Europe Standard Time Printed 06/11/2007 14: 27: 01 Romance Standard Time CONFIDENTIAL Document Date This report is solely for the use of client personnel. No part of it may be circulated, quoted, or reproduced for distribution outside the client organization without prior written approval from Mc. Kinsey & Company. This material was used by Mc. Kinsey & Company during an oral presentation; it is not a complete record of the discussion.


