9bb413a1ed3db65f8776da55e9085c9b.ppt
- Количество слайдов: 37
 Unit 5: Stoichiometry The measurements/math of chemistry
Unit 5: Stoichiometry The measurements/math of chemistry
 Chemical reactions l. A chemical reaction: One or more substances are made into one or more NEW substances (bonds are broken and/or bonds are made). Ag. I (aq) + F 2 (g) Ag. F (s) + I 2 (s) Aqueous Silver iodide plus fluorine gas yields solid silver fluoride plus solid iodine The arrow is read “reactants yields products.
Chemical reactions l. A chemical reaction: One or more substances are made into one or more NEW substances (bonds are broken and/or bonds are made). Ag. I (aq) + F 2 (g) Ag. F (s) + I 2 (s) Aqueous Silver iodide plus fluorine gas yields solid silver fluoride plus solid iodine The arrow is read “reactants yields products.
 Types of Reactions (Rxns) l Synthesis Reactions: ¡ Reactants and Products: 2 elements or cmpds 1 cmpd ¡ General Equation: A + B AB (ex) 2 H 2 + O 2 2 H 2 O l Decomposition Reactions: ¡ Reactants and Products: 1 cmpd 2 elements or cmpds ¡ General Equation: AB A + B (ex) 2 Na. Cl 2 Na + Cl 2 l Combustion Reactions: ¡ Reactants and Products (Complete Combustion): Organic cmpd + O 2 CO 2 + H 2 O Incomplete combustion: Organic + O 2 CO + H 2 O (ex) C 6 H 12 O 6 + O 2 CO 2 + H 2 O (ex) C 6 H 12 O 6 + O 2 CO + H 2 O **Only identify
Types of Reactions (Rxns) l Synthesis Reactions: ¡ Reactants and Products: 2 elements or cmpds 1 cmpd ¡ General Equation: A + B AB (ex) 2 H 2 + O 2 2 H 2 O l Decomposition Reactions: ¡ Reactants and Products: 1 cmpd 2 elements or cmpds ¡ General Equation: AB A + B (ex) 2 Na. Cl 2 Na + Cl 2 l Combustion Reactions: ¡ Reactants and Products (Complete Combustion): Organic cmpd + O 2 CO 2 + H 2 O Incomplete combustion: Organic + O 2 CO + H 2 O (ex) C 6 H 12 O 6 + O 2 CO 2 + H 2 O (ex) C 6 H 12 O 6 + O 2 CO + H 2 O **Only identify
 Types of Reactions (Rxns) l Single Replacement Reactions: ¡Reactants and Products: ¡General Equation: element + ionic cmpd (aq) (or acid) element + ionic cmpd A + BY (aq) B + AY (ex) Au. Cl (aq) + Cs Cs. Cl + Au **All other reactions happen automatically single replacement reactions DO NOT ALWAYS REACT. **They only occur if the element that is by itself is more reactive than the ELEMENT IN THE SAME COLUMN ON TABLE J in the compound. (ex) Na. Cl (aq) + Li (ex) Na. F (aq) + Cl 2 No reaction
Types of Reactions (Rxns) l Single Replacement Reactions: ¡Reactants and Products: ¡General Equation: element + ionic cmpd (aq) (or acid) element + ionic cmpd A + BY (aq) B + AY (ex) Au. Cl (aq) + Cs Cs. Cl + Au **All other reactions happen automatically single replacement reactions DO NOT ALWAYS REACT. **They only occur if the element that is by itself is more reactive than the ELEMENT IN THE SAME COLUMN ON TABLE J in the compound. (ex) Na. Cl (aq) + Li (ex) Na. F (aq) + Cl 2 No reaction
 Types of Reactions (Rxns) l Double Replacement Reactions: ¡ Reactants and Products: 2 ionic cmpds (aq) (or acid) 2 ionic cmpds ¡ General Equation: AX (aq) + BY(aq) BX + AY (ex) KCl (aq) + Na. NO 3 (aq) Na. Cl + KNO 3
Types of Reactions (Rxns) l Double Replacement Reactions: ¡ Reactants and Products: 2 ionic cmpds (aq) (or acid) 2 ionic cmpds ¡ General Equation: AX (aq) + BY(aq) BX + AY (ex) KCl (aq) + Na. NO 3 (aq) Na. Cl + KNO 3
 Predicting Phases l We should know the phase of any element- we labeled them on our Periodic Tables. l H 2 O and CO 2 are both gases in combustion reactions due to the heat produced in the reaction. l Ionic compounds where there is NO WATER present are solids. l Ionic compounds where there IS WATER present, check Table F.
Predicting Phases l We should know the phase of any element- we labeled them on our Periodic Tables. l H 2 O and CO 2 are both gases in combustion reactions due to the heat produced in the reaction. l Ionic compounds where there is NO WATER present are solids. l Ionic compounds where there IS WATER present, check Table F.
 Precipitates: Precipiates: (usually from single or double replacement reactions) A solid formed as a product of a reaction that does not dissolve in water (insoluble) “if you’re not part of the solution, you’re part of the precipitate” F Table ______ (ex) Cu(NO 3) (aq) (ex) Pb. Cl 2 (s) (ex) Ca 3(PO 4)2 (s) (ex) Ba(OH)2 (aq)
Precipitates: Precipiates: (usually from single or double replacement reactions) A solid formed as a product of a reaction that does not dissolve in water (insoluble) “if you’re not part of the solution, you’re part of the precipitate” F Table ______ (ex) Cu(NO 3) (aq) (ex) Pb. Cl 2 (s) (ex) Ca 3(PO 4)2 (s) (ex) Ba(OH)2 (aq)
 Predicting Products Practice 1. Li 3(PO 4)(aq) + Mg(NO 3)2(aq) 2. Sr(s) + Na. Cl(aq) 3. C 4 H 12(g) + O 2(g) 4. Al(s) + HCl(aq) 5. CH 3 OH(g) + O 2(g) 6. Na. OH(aq) + Mg. SO 4(aq) 7. K (s) + Br 2(l) 8. Al 2 O 3 1. Determine the type of reaction. 2. Write the products based on the reaction type. 3. Determine NEW subscripts for each compound based on charges. 4. Determine phase of each product. 5. BALANCE
Predicting Products Practice 1. Li 3(PO 4)(aq) + Mg(NO 3)2(aq) 2. Sr(s) + Na. Cl(aq) 3. C 4 H 12(g) + O 2(g) 4. Al(s) + HCl(aq) 5. CH 3 OH(g) + O 2(g) 6. Na. OH(aq) + Mg. SO 4(aq) 7. K (s) + Br 2(l) 8. Al 2 O 3 1. Determine the type of reaction. 2. Write the products based on the reaction type. 3. Determine NEW subscripts for each compound based on charges. 4. Determine phase of each product. 5. BALANCE
 Law of Conservation of Mass l Law of Conservation of Mass: Mass is neither created nor destroyed in a chemical change. ~ 1789 Antoine Lavoisier This is shown by a balanced reaction, or having the same mass on the reactants side as the products side. 0. 28 g 13 x (ex) 2 H 2 O 2 H 2 + O 2 2. 54 g x = 2. 26 g
Law of Conservation of Mass l Law of Conservation of Mass: Mass is neither created nor destroyed in a chemical change. ~ 1789 Antoine Lavoisier This is shown by a balanced reaction, or having the same mass on the reactants side as the products side. 0. 28 g 13 x (ex) 2 H 2 O 2 H 2 + O 2 2. 54 g x = 2. 26 g
 Balancing Reactions Therefore, the mass of the reactants = the mass of the products (no atoms can disappear or appear out of nowhere) The total number of atoms of each element must be equal on both sides of the reaction arrow. **Balance the elements one at a time using the coefficients. After you have written the correct formulas for all compounds – NEVER, NEVER change the subscripts.
Balancing Reactions Therefore, the mass of the reactants = the mass of the products (no atoms can disappear or appear out of nowhere) The total number of atoms of each element must be equal on both sides of the reaction arrow. **Balance the elements one at a time using the coefficients. After you have written the correct formulas for all compounds – NEVER, NEVER change the subscripts.
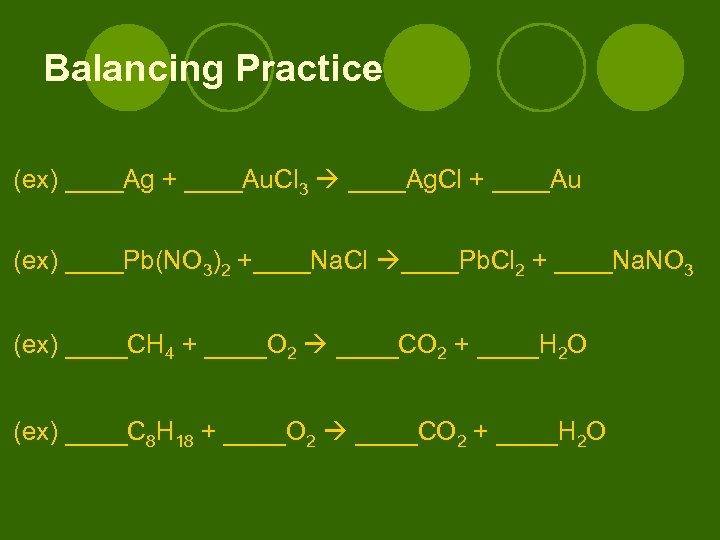 Balancing Practice (ex) ____Ag + ____Au. Cl 3 ____Ag. Cl + ____Au (ex) ____Pb(NO 3)2 +____Na. Cl ____Pb. Cl 2 + ____Na. NO 3 (ex) ____CH 4 + ____O 2 ____CO 2 + ____H 2 O (ex) ____C 8 H 18 + ____O 2 ____CO 2 + ____H 2 O
Balancing Practice (ex) ____Ag + ____Au. Cl 3 ____Ag. Cl + ____Au (ex) ____Pb(NO 3)2 +____Na. Cl ____Pb. Cl 2 + ____Na. NO 3 (ex) ____CH 4 + ____O 2 ____CO 2 + ____H 2 O (ex) ____C 8 H 18 + ____O 2 ____CO 2 + ____H 2 O
 The Mole l Calculating the mass of a compound: ** First you have to know how much of the compound you have and what compound it is to calculate the mass. l The mole (measures how much): Is a word that means a number (6. 02 x 1023) JUST LIKE A DOZEN 12 donuts (Ex) 1 dozen donuts = ________________ 6. 02 x 1023 donuts 1 mole of donuts = _______________ 12 particles (Ex) 1 dozen particles = ________________ 6. 02 x 1023 particles 1 mole of particles = _______________ Particle can mean 1 molecule, 1 atom, 1 ion.
The Mole l Calculating the mass of a compound: ** First you have to know how much of the compound you have and what compound it is to calculate the mass. l The mole (measures how much): Is a word that means a number (6. 02 x 1023) JUST LIKE A DOZEN 12 donuts (Ex) 1 dozen donuts = ________________ 6. 02 x 1023 donuts 1 mole of donuts = _______________ 12 particles (Ex) 1 dozen particles = ________________ 6. 02 x 1023 particles 1 mole of particles = _______________ Particle can mean 1 molecule, 1 atom, 1 ion.
 GFM l Gram Formula Mass (GFM) The mass of 1 mole (6. 02 x 1023 atoms/molecules) of an element or compound 12 amu C atomic mass: (mass of 1 atom) _________ 12 g C molar mass: (mass of 6. 02 x 1023 atoms) _______ 40 amu Ar atomic mass: _________ 40 g Ar molar mass: _________ ¡To find the mass of a mole of a compound you must add the masses of all of the elements in the compound or molecule. (ex) K(NO 3) (ex) CH 4 (ex) Fe 3(PO 4)2 = 39 + 14 + 3(16) = 12 + 4(1) = 101 g = 16 g = 3(56) + 2(31) + 8(16) = 358 g
GFM l Gram Formula Mass (GFM) The mass of 1 mole (6. 02 x 1023 atoms/molecules) of an element or compound 12 amu C atomic mass: (mass of 1 atom) _________ 12 g C molar mass: (mass of 6. 02 x 1023 atoms) _______ 40 amu Ar atomic mass: _________ 40 g Ar molar mass: _________ ¡To find the mass of a mole of a compound you must add the masses of all of the elements in the compound or molecule. (ex) K(NO 3) (ex) CH 4 (ex) Fe 3(PO 4)2 = 39 + 14 + 3(16) = 12 + 4(1) = 101 g = 16 g = 3(56) + 2(31) + 8(16) = 358 g
 Moles of Atoms l If there a dozen cars in the parking lot, how many dozen wheels are there? l If I have a mole of H 2 O and each water molecule has 2 hydrogen atoms, how many moles of hydrogen do I have? l Subscripts actually tell you the number of moles of each atom. (Ex) 1 mole of SO 3 How many moles of S atoms? 1 How many moles of O atoms? 3 How many moles of atoms in 1 4 mole of SO 3? What is the mass of 1 mole of SO 3? = 32 + 3(16) = 80 g (Ex) 1 mole of Ca(OH)2 How many moles of Ca atoms? How many moles of O atoms? How many moles of H atoms? How many moles of atoms in 1 mole of Ca(OH)2? GFM of Ca(OH)2 ? = 40 + 2(16) + 2(1) = 74 g 1 2 2 5
Moles of Atoms l If there a dozen cars in the parking lot, how many dozen wheels are there? l If I have a mole of H 2 O and each water molecule has 2 hydrogen atoms, how many moles of hydrogen do I have? l Subscripts actually tell you the number of moles of each atom. (Ex) 1 mole of SO 3 How many moles of S atoms? 1 How many moles of O atoms? 3 How many moles of atoms in 1 4 mole of SO 3? What is the mass of 1 mole of SO 3? = 32 + 3(16) = 80 g (Ex) 1 mole of Ca(OH)2 How many moles of Ca atoms? How many moles of O atoms? How many moles of H atoms? How many moles of atoms in 1 mole of Ca(OH)2? GFM of Ca(OH)2 ? = 40 + 2(16) + 2(1) = 74 g 1 2 2 5
 Converting moles grams: Table T for mole calculations. (ex) In the lab you are given 96 g of Na. OH. How many moles of Na. OH do you have? (ex) You are asked to measure out 4. 23 mol of K 2 CO 3, how many grams is this?
Converting moles grams: Table T for mole calculations. (ex) In the lab you are given 96 g of Na. OH. How many moles of Na. OH do you have? (ex) You are asked to measure out 4. 23 mol of K 2 CO 3, how many grams is this?
 Converting moles liters: 1 mole of any gas equals 22. 4 L (ex) How many liters are in 2. 37 mol of H 2? (ex) How many moles are in 65. 9 liters of CO 2?
Converting moles liters: 1 mole of any gas equals 22. 4 L (ex) How many liters are in 2. 37 mol of H 2? (ex) How many moles are in 65. 9 liters of CO 2?
 Converting moles #particles: 1 mole of any substance contains 6. 02 x 1023 particles (ex) How many molecules of water are in 2. 50 moles of water? (ex) How many moles of SO 3 are present if there are 9. 03 x 1023 particles of SO 3?
Converting moles #particles: 1 mole of any substance contains 6. 02 x 1023 particles (ex) How many molecules of water are in 2. 50 moles of water? (ex) How many moles of SO 3 are present if there are 9. 03 x 1023 particles of SO 3?
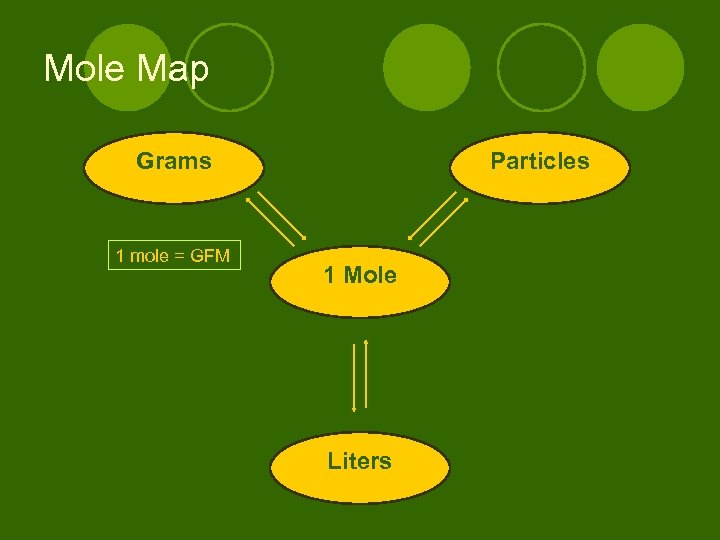 Mole Map Grams 1 mole = GFM Particles 1 Mole Liters
Mole Map Grams 1 mole = GFM Particles 1 Mole Liters
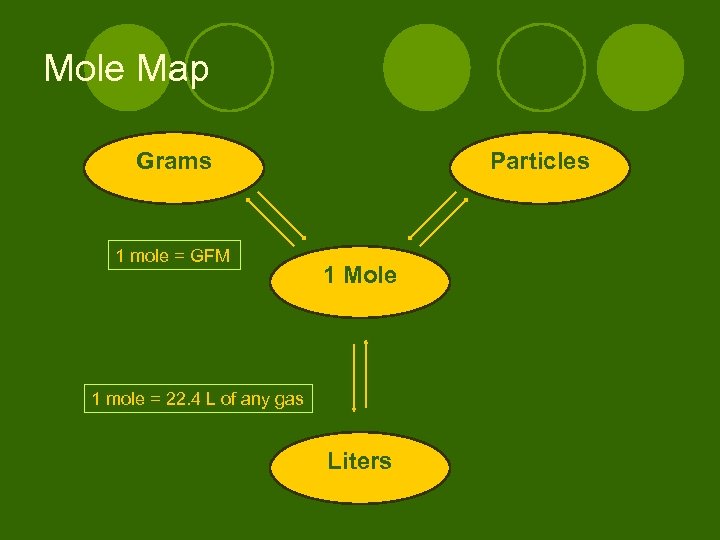 Mole Map Grams 1 mole = GFM Particles 1 Mole 1 mole = 22. 4 L of any gas Liters
Mole Map Grams 1 mole = GFM Particles 1 Mole 1 mole = 22. 4 L of any gas Liters
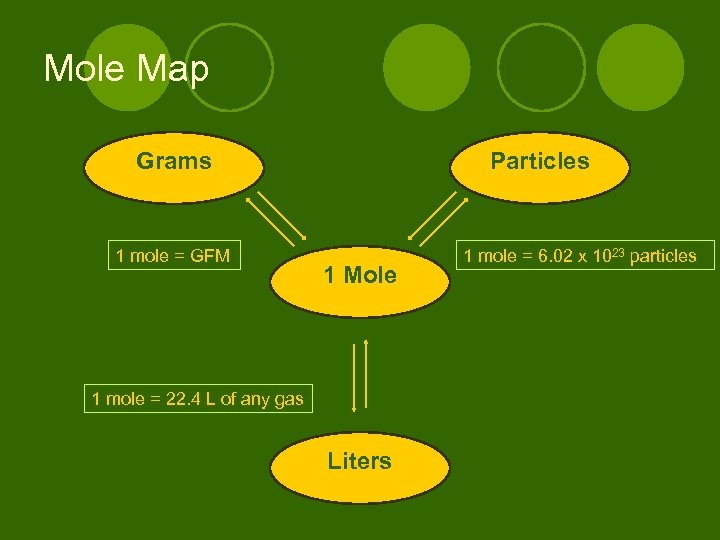 Mole Map Grams 1 mole = GFM Particles 1 Mole 1 mole = 22. 4 L of any gas Liters 1 mole = 6. 02 x 1023 particles
Mole Map Grams 1 mole = GFM Particles 1 Mole 1 mole = 22. 4 L of any gas Liters 1 mole = 6. 02 x 1023 particles
 Avogadro’s Hypothesis l Avogadro’s Hypothesis: Any 2 samples of gas at the same temp, pressure and volume have the same number of particles (ex) Which of the following would occupy the same volume as 2. 5 mol of O 2 at STP? (a) 1. 5 mol of CO 2 at STP (b) 3. 5 mol of CH 4 at STP (c) 3. 0 mol of H 2 at STP (d) 2. 5 mol of Ne at STP (ex) Which of the following would occupy the same volume as 2. 5 mol of O 2 at STP? (a) 33. 6 L of CO 2 at STP (b) 78. 4 L of CH 4 at STP (c) 67. 2 L of H 2 at STP (d) 56. 0 L of Ne at STP
Avogadro’s Hypothesis l Avogadro’s Hypothesis: Any 2 samples of gas at the same temp, pressure and volume have the same number of particles (ex) Which of the following would occupy the same volume as 2. 5 mol of O 2 at STP? (a) 1. 5 mol of CO 2 at STP (b) 3. 5 mol of CH 4 at STP (c) 3. 0 mol of H 2 at STP (d) 2. 5 mol of Ne at STP (ex) Which of the following would occupy the same volume as 2. 5 mol of O 2 at STP? (a) 33. 6 L of CO 2 at STP (b) 78. 4 L of CH 4 at STP (c) 67. 2 L of H 2 at STP (d) 56. 0 L of Ne at STP
 Moles of Compounds l Coefficients tell you how many moles of the compound (the whole thing) there are. (Ex) 2 H 2 O 2 H 2 +O 2 How many moles of water in the above reaction? How many moles of H 2? How many moles of O 2? What is the mole ratio of O 2 to H 2? 2 2 1 1: 2
Moles of Compounds l Coefficients tell you how many moles of the compound (the whole thing) there are. (Ex) 2 H 2 O 2 H 2 +O 2 How many moles of water in the above reaction? How many moles of H 2? How many moles of O 2? What is the mole ratio of O 2 to H 2? 2 2 1 1: 2
 Ratios of moles in reactions: (ex) Given the reaction: CH 4(g) + 2 O 2(g) CO 2(g) + H 2 O(g) How many moles of oxygen are needed for the complete combustion of 7. 0 moles of CH 4? (ex) Given the reaction: 4 Al(s) + 3 O 2(g) 2 Al 2 O 3(s) How many moles of Al 2 O 3 will be formed when 7. 0 mol of Al react completely with O 2?
Ratios of moles in reactions: (ex) Given the reaction: CH 4(g) + 2 O 2(g) CO 2(g) + H 2 O(g) How many moles of oxygen are needed for the complete combustion of 7. 0 moles of CH 4? (ex) Given the reaction: 4 Al(s) + 3 O 2(g) 2 Al 2 O 3(s) How many moles of Al 2 O 3 will be formed when 7. 0 mol of Al react completely with O 2?
 More practice (ex) Given the reaction: N 2(g) + 3 H 2(g) 2 NH 3(g) Calculate the number of moles of NH 3 produced when 2. 50 moles of H 2 are completely reacted. (ex) Given the reaction: CH 4(g) + 2 O 2(g) CO 2(g) + H 2 O(g) Calculate how many moles of CO 2 will be produced when 5. 93 moles of O 2 is completely consumed.
More practice (ex) Given the reaction: N 2(g) + 3 H 2(g) 2 NH 3(g) Calculate the number of moles of NH 3 produced when 2. 50 moles of H 2 are completely reacted. (ex) Given the reaction: CH 4(g) + 2 O 2(g) CO 2(g) + H 2 O(g) Calculate how many moles of CO 2 will be produced when 5. 93 moles of O 2 is completely consumed.
 Now all together!! l Complete the reaction, balance, and solve. HNO 3 (aq) + Mg (s) If 52. 94 g of HNO 3 are reacted with Mg, how many liters of hydrogen gas will be produced?
Now all together!! l Complete the reaction, balance, and solve. HNO 3 (aq) + Mg (s) If 52. 94 g of HNO 3 are reacted with Mg, how many liters of hydrogen gas will be produced?
 Percent Composition l Percent Composition: the part of the whole compound is made up of a certain element Part %= x 100 Found on Table T Whole What percent of the people in this room have brown eyes? (Ex) Find the percent composition of potassium in potassium sulfide? K 2 S = 2(39) / 110 x 100 = 70. 91 % K (Ex 2) Find the percent composition of nitrogen in ammonium nitrate? NH 4 NO 3 = 2(14) / 80 x 100 = 35. 00 % N (Ex 3) Which of the following compounds has the greatest percent composition by mass, of oxygen: Ba. O, CO, Al 2 O 3, or Na. Cl. O? Ba. O: = 16 / 153 x 100 CO: = 16 / 28 x 100 Al 2 O 3: = 3(16) / 102 x 100 Na. Cl. O: = 16 / 74 x 100 = 10. 46 % O = 57. 14 % O = 47. 06 % O = 21. 62 % O
Percent Composition l Percent Composition: the part of the whole compound is made up of a certain element Part %= x 100 Found on Table T Whole What percent of the people in this room have brown eyes? (Ex) Find the percent composition of potassium in potassium sulfide? K 2 S = 2(39) / 110 x 100 = 70. 91 % K (Ex 2) Find the percent composition of nitrogen in ammonium nitrate? NH 4 NO 3 = 2(14) / 80 x 100 = 35. 00 % N (Ex 3) Which of the following compounds has the greatest percent composition by mass, of oxygen: Ba. O, CO, Al 2 O 3, or Na. Cl. O? Ba. O: = 16 / 153 x 100 CO: = 16 / 28 x 100 Al 2 O 3: = 3(16) / 102 x 100 Na. Cl. O: = 16 / 74 x 100 = 10. 46 % O = 57. 14 % O = 47. 06 % O = 21. 62 % O
 Percent Hydrate l Hydrate: an ionic compound that has water trapped inside the crystal lattice l Anhydrate: a hydrate that has had the water driven off (evaporated) The water is trapped inside the ionic crystal by molecule-ion forces of attraction. The • shows that the water is NOT bonded to the ionic compound; it is just a force of attraction. Find the GFM of each hydrate: (ex) Cu. SO 4 • 5 H 2 O = 64 + 32 + 4(16) + 5(2 + 16) = 250 g = 243 g (ex 2) Ba. Cl 2 • 2 H 2 O = 137 + 2(35) + 2(18) = 359 g (ex 3) Ba. SO 4 • 7 H 2 O = 137 + 32 + 4(16) + 7(18) For each of the examples above find the % H 2 O of hydration (ex) Cu. SO 4 • 5 H 2 O (ex 2) Ba. Cl 2 • 2 H 2 O (ex 3) Ba. SO 4 • 7 H 2 O = 5(18) / 250 x 100 = 36. 00% = 2(18) / 243 x 100 = 14. 81% = 7(18) / 359 x 100 = 35. 10%
Percent Hydrate l Hydrate: an ionic compound that has water trapped inside the crystal lattice l Anhydrate: a hydrate that has had the water driven off (evaporated) The water is trapped inside the ionic crystal by molecule-ion forces of attraction. The • shows that the water is NOT bonded to the ionic compound; it is just a force of attraction. Find the GFM of each hydrate: (ex) Cu. SO 4 • 5 H 2 O = 64 + 32 + 4(16) + 5(2 + 16) = 250 g = 243 g (ex 2) Ba. Cl 2 • 2 H 2 O = 137 + 2(35) + 2(18) = 359 g (ex 3) Ba. SO 4 • 7 H 2 O = 137 + 32 + 4(16) + 7(18) For each of the examples above find the % H 2 O of hydration (ex) Cu. SO 4 • 5 H 2 O (ex 2) Ba. Cl 2 • 2 H 2 O (ex 3) Ba. SO 4 • 7 H 2 O = 5(18) / 250 x 100 = 36. 00% = 2(18) / 243 x 100 = 14. 81% = 7(18) / 359 x 100 = 35. 10%
 Finding % Water in a Hydrate in Lab l If we wanted to remove the water from the ionic compound, we could heat up the hydrate until the molecule-ion forces were broken. l In lab, how do we know all the water has been driven off (evaporated)? When we get the same mass 3 x in a row. (ex) We have 0. 5 mol of Cu. SO 4 5 H 2 O. How many moles of water are present?
Finding % Water in a Hydrate in Lab l If we wanted to remove the water from the ionic compound, we could heat up the hydrate until the molecule-ion forces were broken. l In lab, how do we know all the water has been driven off (evaporated)? When we get the same mass 3 x in a row. (ex) We have 0. 5 mol of Cu. SO 4 5 H 2 O. How many moles of water are present?
 % water in hydrate Lab Data: (ex 1) A hydrate has a mass of 45. 3 g. After being heated to a constant mass, the substance has a mass of 39. 8 g. What is the percent water of hydration? (ex 2) crucible 5. 13 g crucible and hydrate 7. 14 g crucible and anhydrate 6. 29 g Given the lab data above, find the percent water of hydration.
% water in hydrate Lab Data: (ex 1) A hydrate has a mass of 45. 3 g. After being heated to a constant mass, the substance has a mass of 39. 8 g. What is the percent water of hydration? (ex 2) crucible 5. 13 g crucible and hydrate 7. 14 g crucible and anhydrate 6. 29 g Given the lab data above, find the percent water of hydration.
 Writing Empirical Formulas l Empirical Formula: The most reduced ratio of elements (moles) in a chemical formula Ionic compounds are ALWAYS written as empirical formulas. (ex) Na 2 O is an empirical formula because… It is reduced (ionic) (ex 2) C 8 H 18 is not an empirical formula because… It is not reduced (molecular) What would be the empirical formula for C 8 H 18? C 4 H 9 ¡Some molecular formulas are empirical. (ex) H 2 O ¡Sometimes it is important to know the most reduced ratio of elements in a compound (even in molecular compounds)…Do I need twice as much H as O to make a reaction work or three times as much H than O?
Writing Empirical Formulas l Empirical Formula: The most reduced ratio of elements (moles) in a chemical formula Ionic compounds are ALWAYS written as empirical formulas. (ex) Na 2 O is an empirical formula because… It is reduced (ionic) (ex 2) C 8 H 18 is not an empirical formula because… It is not reduced (molecular) What would be the empirical formula for C 8 H 18? C 4 H 9 ¡Some molecular formulas are empirical. (ex) H 2 O ¡Sometimes it is important to know the most reduced ratio of elements in a compound (even in molecular compounds)…Do I need twice as much H as O to make a reaction work or three times as much H than O?
 Empirical Formulas: Steps The steps to writing an empirical formula: 1. 2. Divide by the mass on the PT to get moles. Simplify the mole ratio by dividing by the smallest #. (ex) The percent composition ofa compound is 11. 1% hydrogen and 88. 9% oxygen. Write the empirical formula. 1. = Moles = g/GFM 11. 1 g / 1 g per mol = 11. 1 mol H 88. 9 g / 16 g per mol = 5. 56 mol O 2. = 11. 1 mol / 5. 56 mol = 2 mol H 5. 56 mol / 5. 56 mol = 1 mol O H 2 O
Empirical Formulas: Steps The steps to writing an empirical formula: 1. 2. Divide by the mass on the PT to get moles. Simplify the mole ratio by dividing by the smallest #. (ex) The percent composition ofa compound is 11. 1% hydrogen and 88. 9% oxygen. Write the empirical formula. 1. = Moles = g/GFM 11. 1 g / 1 g per mol = 11. 1 mol H 88. 9 g / 16 g per mol = 5. 56 mol O 2. = 11. 1 mol / 5. 56 mol = 2 mol H 5. 56 mol / 5. 56 mol = 1 mol O H 2 O
 Empirical Formulas: Practice (ex 2) Determine the empirical formula for the compound which when analyzed showed 39. 7% sodium by mass and 60. 3% chlorine by mass. 1. = Moles = g/GFM 39. 7 g / 23 g per mol 60. 3 g / 35 g per mol = 1. 73 mol Na 2. = = 1. 73 mol Cl 1. 73 mol / 1. 73 mol = 1 mol Na 1. 73 mol / 1. 73 mol = 1 mol Cl Na. Cl
Empirical Formulas: Practice (ex 2) Determine the empirical formula for the compound which when analyzed showed 39. 7% sodium by mass and 60. 3% chlorine by mass. 1. = Moles = g/GFM 39. 7 g / 23 g per mol 60. 3 g / 35 g per mol = 1. 73 mol Na 2. = = 1. 73 mol Cl 1. 73 mol / 1. 73 mol = 1 mol Na 1. 73 mol / 1. 73 mol = 1 mol Cl Na. Cl
 Empirical Formulas: Step 4 l Step 3: If you don’t end up within a tenth of a whole number, multiply all #’s by a common factor to get whole #’s. (ex 3) A compound of zinc and phosphorus, when analyzed, showed 76. 0%Zn and 24. 0% P by mass. Calculate the simplest formula for the compound. 1. = 2. = 3. = Mol = g/GFM 76. 0 g / 65 g per mol 24. 0 g / 31 g per mol = 1. 17 mol Zn = 0. 77 mol P 1. 17 mol / 0. 77 mol = 1. 5 mol Zn Multiply both numbers by 2 to get = 3 mol Zn whole number ratio 0. 77 mol / 0. 77 mol = 1 mol P = 2 mol P Zn 3 P 2
Empirical Formulas: Step 4 l Step 3: If you don’t end up within a tenth of a whole number, multiply all #’s by a common factor to get whole #’s. (ex 3) A compound of zinc and phosphorus, when analyzed, showed 76. 0%Zn and 24. 0% P by mass. Calculate the simplest formula for the compound. 1. = 2. = 3. = Mol = g/GFM 76. 0 g / 65 g per mol 24. 0 g / 31 g per mol = 1. 17 mol Zn = 0. 77 mol P 1. 17 mol / 0. 77 mol = 1. 5 mol Zn Multiply both numbers by 2 to get = 3 mol Zn whole number ratio 0. 77 mol / 0. 77 mol = 1 mol P = 2 mol P Zn 3 P 2
 Molecular Formulas l Molecular (true) Formula: gives the actual # of moles of elements in a molecule (not reduced) **Only molecular compounds have molecular formulas. (ex) C 6 H 12 O 6
Molecular Formulas l Molecular (true) Formula: gives the actual # of moles of elements in a molecule (not reduced) **Only molecular compounds have molecular formulas. (ex) C 6 H 12 O 6
 Molecular Formulas: Steps The steps to writing a molecular formula: 1. 2. 3. Find the empirical formula Find the mass of the empirical formula Find how many times larger the GFM is than the empirical formula and multiply the subscripts by that number (ex) What is the molecular formula of a compound that has a GFM of 92 g and an empirical formula of NO 2? 1. = 2. = NO 2 3. = 92 g / 46 g = 2 times as larges as the empirical formula 14 + 2(16) = 46 g N 2 O 4
Molecular Formulas: Steps The steps to writing a molecular formula: 1. 2. 3. Find the empirical formula Find the mass of the empirical formula Find how many times larger the GFM is than the empirical formula and multiply the subscripts by that number (ex) What is the molecular formula of a compound that has a GFM of 92 g and an empirical formula of NO 2? 1. = 2. = NO 2 3. = 92 g / 46 g = 2 times as larges as the empirical formula 14 + 2(16) = 46 g N 2 O 4
 Molecular Formulas: Practice (ex 2) A compound of phosphorus and oxygen, when analyzed, showed 39. 24% P and 60. 76% O by mass. Calculate the simplest formula for the compound. 1. = 2. = Mol = g/GFM 39. 24 g P 60. 76 g O 39. 24 g / 31 g per mol 60. 76 g / 16 g per mol = 1. 27 mol P = 3. 80 mol O 1. 27 mol / 1. 27 mol = 1 mol P 3. 80 mol / 1. 27 mol PO 3 = 3 mol O 3. = Find the molecular formula if the GFM of this compound is 158 g. 4. = mass of empirical = 31 + 3(16) = 79 g formula 5. = = 158 g / 79 g = 2 P 2 O 6
Molecular Formulas: Practice (ex 2) A compound of phosphorus and oxygen, when analyzed, showed 39. 24% P and 60. 76% O by mass. Calculate the simplest formula for the compound. 1. = 2. = Mol = g/GFM 39. 24 g P 60. 76 g O 39. 24 g / 31 g per mol 60. 76 g / 16 g per mol = 1. 27 mol P = 3. 80 mol O 1. 27 mol / 1. 27 mol = 1 mol P 3. 80 mol / 1. 27 mol PO 3 = 3 mol O 3. = Find the molecular formula if the GFM of this compound is 158 g. 4. = mass of empirical = 31 + 3(16) = 79 g formula 5. = = 158 g / 79 g = 2 P 2 O 6
 (ex) Bill Nye measures out 62 g of Na. NO 3. How many moles of Na. NO 3 does Bill have? (ex) You are asked to measure out 1. 49 moles of SCl 2. How many grams will you need to measure out on the scale?
(ex) Bill Nye measures out 62 g of Na. NO 3. How many moles of Na. NO 3 does Bill have? (ex) You are asked to measure out 1. 49 moles of SCl 2. How many grams will you need to measure out on the scale?


