85c8fe34c4be8f8c28950d03f8a9fa91.ppt
- Количество слайдов: 78
 Understanding HIV/AIDS Testing and Its Impact on Treatment Sponsored for CME credit by Rush University Medical Center Supported by an independent educational grant from Gilead Sciences Medical Affairs
Understanding HIV/AIDS Testing and Its Impact on Treatment Sponsored for CME credit by Rush University Medical Center Supported by an independent educational grant from Gilead Sciences Medical Affairs
 CPT CODES FOR HIV TESTING AMA and AAHIVM Guidelines for Coding Routine HIV Testing: http: //www. aahivm. org/codingguide 2
CPT CODES FOR HIV TESTING AMA and AAHIVM Guidelines for Coding Routine HIV Testing: http: //www. aahivm. org/codingguide 2
 Educator Derrick Butler, MD, MPH Associate Medical Director T. H. E. Clinic, Inc Los Angeles, CA 3
Educator Derrick Butler, MD, MPH Associate Medical Director T. H. E. Clinic, Inc Los Angeles, CA 3
 Disclosure Information: Educator ● Derrick Butler, MD, MPH - Consultant • - Speakers’ Bureau • 4 Tibotec Therapuetics Gilead Sciences
Disclosure Information: Educator ● Derrick Butler, MD, MPH - Consultant • - Speakers’ Bureau • 4 Tibotec Therapuetics Gilead Sciences
 Quiz 1) Magic Johnson tested positive for HIV in 1992 and is now cured of the virus. a) True, he is rich and can afford the best medicine. b) False, he is still infected, but is controlled on medication. c) Don’t know, I don’t follow football. 5
Quiz 1) Magic Johnson tested positive for HIV in 1992 and is now cured of the virus. a) True, he is rich and can afford the best medicine. b) False, he is still infected, but is controlled on medication. c) Don’t know, I don’t follow football. 5
 New Sign-In Process ● Please clearly print all information on the sign-in sheet ● You must indicate your NAME, DEGREE, MAILING ADDRESS, EMAIL and SIGNATURE (NAPB # ADDRESS, EMAIL and SIGNATURE pharmacists only) in order to attend this lecture ● Completion is required for all healthcare providers ● Failure to provide complete information will result in removal from attending future lectures 6
New Sign-In Process ● Please clearly print all information on the sign-in sheet ● You must indicate your NAME, DEGREE, MAILING ADDRESS, EMAIL and SIGNATURE (NAPB # ADDRESS, EMAIL and SIGNATURE pharmacists only) in order to attend this lecture ● Completion is required for all healthcare providers ● Failure to provide complete information will result in removal from attending future lectures 6
 Accreditation and Designation Rush University Medical Center is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians. Rush University Medical Center designates this live activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only credit commensurate with the extent of their participation in the activity. ANAC is an approved provider of continuing nursing education (CNE) by the Virginia Nurses Association, an accredited approver by the American Nurses Credentialing Center’s Commission on Accreditation. This activity is approved for 1. 0 contact hour by the Association of Nurses in AIDS Care. The University of Florida College of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education (UAN #0012 -9999 -13 -034 -L 02 -P). This activity is accredited for 1 hour of continuing pharmacy education (CPE) credit. The University of Florida College of Pharmacy will report all credit to CPE Monitor within 30 working days after receiving evidence of successful completion of the course. Successful completion means that you must attend the entire program and complete an evaluation form. Commission for Case Manager Certification (CCMC) This program (Understanding HIV/AIDS Testing and Its Impact on Treatment) has been pre-approved by the Commission for Case Manager Certification to provide continuing education credit to Certified Case Managers (CCMs). Approved for 1. 0 CEUs. Approval number: 20131552. Activity Code: S 0001105. Supported by an independent educational grant from Gilead Sciences Medical Affairs. 7
Accreditation and Designation Rush University Medical Center is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians. Rush University Medical Center designates this live activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only credit commensurate with the extent of their participation in the activity. ANAC is an approved provider of continuing nursing education (CNE) by the Virginia Nurses Association, an accredited approver by the American Nurses Credentialing Center’s Commission on Accreditation. This activity is approved for 1. 0 contact hour by the Association of Nurses in AIDS Care. The University of Florida College of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education (UAN #0012 -9999 -13 -034 -L 02 -P). This activity is accredited for 1 hour of continuing pharmacy education (CPE) credit. The University of Florida College of Pharmacy will report all credit to CPE Monitor within 30 working days after receiving evidence of successful completion of the course. Successful completion means that you must attend the entire program and complete an evaluation form. Commission for Case Manager Certification (CCMC) This program (Understanding HIV/AIDS Testing and Its Impact on Treatment) has been pre-approved by the Commission for Case Manager Certification to provide continuing education credit to Certified Case Managers (CCMs). Approved for 1. 0 CEUs. Approval number: 20131552. Activity Code: S 0001105. Supported by an independent educational grant from Gilead Sciences Medical Affairs. 7
 Faculty CME Course Director Harold A. Kessler, MD Professor of Medicine and Immunology/Microbiology Associate Director, Section of Infectious Diseases Rush University Medical Center Chicago, Illinois Professor of Medicine Harvard Medical School Director, HIV Prevention Research Beth Israel Deaconess Medical Center Medical Research Director The Fenway Institute, Fenway Health Boston, Massachusetts CME Reviewer David M. Simon, MD, Ph. D CNE Reviewer Allison R. Webel, RN, Ph. D Associate Professor of Medicine Section of Infectious Diseases Rush University Medical Center Chicago, Illinois 8 Content Development and Training Kenneth H. Mayer, MD Instructor and KL 2 Clinical Research Scholar Frances Payne Bolton School of Nursing Case Western Reserve University Cleveland, Ohio
Faculty CME Course Director Harold A. Kessler, MD Professor of Medicine and Immunology/Microbiology Associate Director, Section of Infectious Diseases Rush University Medical Center Chicago, Illinois Professor of Medicine Harvard Medical School Director, HIV Prevention Research Beth Israel Deaconess Medical Center Medical Research Director The Fenway Institute, Fenway Health Boston, Massachusetts CME Reviewer David M. Simon, MD, Ph. D CNE Reviewer Allison R. Webel, RN, Ph. D Associate Professor of Medicine Section of Infectious Diseases Rush University Medical Center Chicago, Illinois 8 Content Development and Training Kenneth H. Mayer, MD Instructor and KL 2 Clinical Research Scholar Frances Payne Bolton School of Nursing Case Western Reserve University Cleveland, Ohio
 Faculty Disclosures CME Course Director: Harold A. Kessler, MD Content Development and Training: Kenneth H. Mayer, MD Grants/research support None Bristol-Myers Squibb, Gilead Sciences, Merck Consultant None Speakers’ bureau None Abb. Vie, Glaxo. Smith. Kline, Merck None Stock shareholder Other financial or material support 9
Faculty Disclosures CME Course Director: Harold A. Kessler, MD Content Development and Training: Kenneth H. Mayer, MD Grants/research support None Bristol-Myers Squibb, Gilead Sciences, Merck Consultant None Speakers’ bureau None Abb. Vie, Glaxo. Smith. Kline, Merck None Stock shareholder Other financial or material support 9
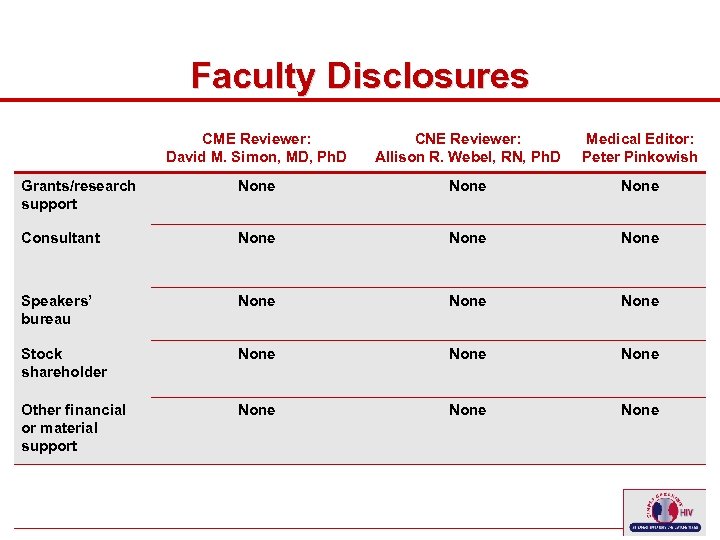 Faculty Disclosures CME Reviewer: David M. Simon, MD, Ph. D Medical Editor: Peter Pinkowish Grants/research support None Consultant None Speakers’ bureau None Stock shareholder None Other financial or material support 10 CNE Reviewer: Allison R. Webel, RN, Ph. D None
Faculty Disclosures CME Reviewer: David M. Simon, MD, Ph. D Medical Editor: Peter Pinkowish Grants/research support None Consultant None Speakers’ bureau None Stock shareholder None Other financial or material support 10 CNE Reviewer: Allison R. Webel, RN, Ph. D None
 Opinions and Off-Label Discussions The opinions or views expressed in this educational program are those of the participants and do not necessarily reflect the opinions or recommendations of Gilead Sciences Medical Affairs, Rush University Medical Center, the Association of Nurses in AIDS Care, the University of Florida College of Pharmacy, or the Commission for Case Manager Certification The faculty may have included discussion on unlabeled uses of a commercial product or an investigational use of a product not yet approved for this purpose Please consult the full prescribing information before using any medication mentioned in this program 11
Opinions and Off-Label Discussions The opinions or views expressed in this educational program are those of the participants and do not necessarily reflect the opinions or recommendations of Gilead Sciences Medical Affairs, Rush University Medical Center, the Association of Nurses in AIDS Care, the University of Florida College of Pharmacy, or the Commission for Case Manager Certification The faculty may have included discussion on unlabeled uses of a commercial product or an investigational use of a product not yet approved for this purpose Please consult the full prescribing information before using any medication mentioned in this program 11
 New Electronic Evaluation Process ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 12
New Electronic Evaluation Process ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 12
 Learning Objectives (CME/CNE and CPE) CPE CME/CNE ● Upon completion of this activity, the participant intends to incorporate the following objectives into their practice of medicine and/or advance practice nursing: ● Upon completion of this activity, the pharmacist should be able to: - Screen and test my patients for HIV infection based on the recommendations from the Centers for Disease Control and Prevention Describe the screening and testing processes for patients for HIV infection based on the recommendations from the Centers for Disease Control and Prevention - - Counsel my HIV-infected patients on the benefits and risks associated with antiretroviral therapy - - Appropriately select antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services Recommend antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services - 13
Learning Objectives (CME/CNE and CPE) CPE CME/CNE ● Upon completion of this activity, the participant intends to incorporate the following objectives into their practice of medicine and/or advance practice nursing: ● Upon completion of this activity, the pharmacist should be able to: - Screen and test my patients for HIV infection based on the recommendations from the Centers for Disease Control and Prevention Describe the screening and testing processes for patients for HIV infection based on the recommendations from the Centers for Disease Control and Prevention - - Counsel my HIV-infected patients on the benefits and risks associated with antiretroviral therapy - - Appropriately select antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services Recommend antiretroviral therapy for my HIV-infected patients according to the guideline recommendations by the Department of Health and Human Services - 13
 We’ve Come a Long Way, Baby 14
We’ve Come a Long Way, Baby 14
 Yes, We Have…. 15
Yes, We Have…. 15
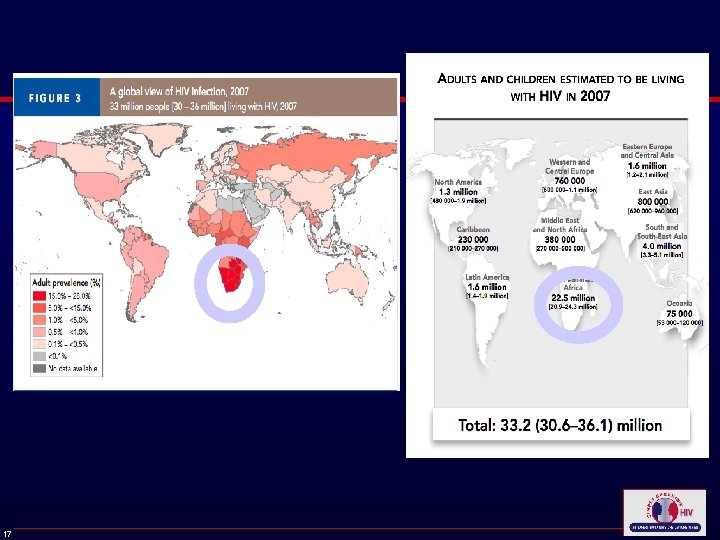
 Evidence for Revising Recommendations ● Many HIV-infected persons access health care but are not tested for HIV until symptomatic ● Routine HIV screening is cost effective, and effective treatment is available ● Opt-out screening increases testing rates ● Awareness of HIV infection leads to substantial reductions in high-risk sexual behavior ● Inconclusive evidence about prevention benefits from typical counseling for persons who test negative ● Great deal of experience with HIV testing, including rapid tests Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 16
Evidence for Revising Recommendations ● Many HIV-infected persons access health care but are not tested for HIV until symptomatic ● Routine HIV screening is cost effective, and effective treatment is available ● Opt-out screening increases testing rates ● Awareness of HIV infection leads to substantial reductions in high-risk sexual behavior ● Inconclusive evidence about prevention benefits from typical counseling for persons who test negative ● Great deal of experience with HIV testing, including rapid tests Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 16
 17
17
 18 18
18 18
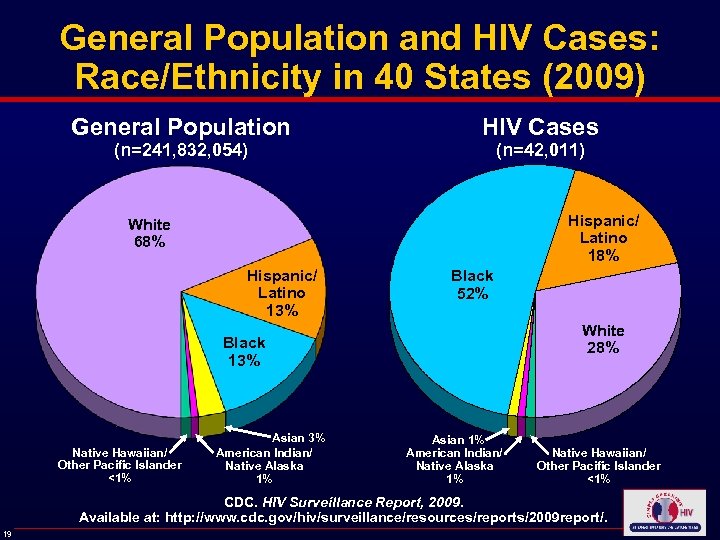 General Population and HIV Cases: Race/Ethnicity in 40 States (2009) General Population (n=241, 832, 054) HIV Cases (n=42, 011) Hispanic/ Latino 18% White 68% Hispanic/ Latino 13% Black 52% White 28% Black 13% Native Hawaiian/ Other Pacific Islander <1% Asian 3% American Indian/ Native Alaska 1% Asian 1% American Indian/ Native Alaska 1% Native Hawaiian/ Other Pacific Islander <1% CDC. HIV Surveillance Report, 2009. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2009 report/. 19
General Population and HIV Cases: Race/Ethnicity in 40 States (2009) General Population (n=241, 832, 054) HIV Cases (n=42, 011) Hispanic/ Latino 18% White 68% Hispanic/ Latino 13% Black 52% White 28% Black 13% Native Hawaiian/ Other Pacific Islander <1% Asian 3% American Indian/ Native Alaska 1% Asian 1% American Indian/ Native Alaska 1% Native Hawaiian/ Other Pacific Islander <1% CDC. HIV Surveillance Report, 2009. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2009 report/. 19
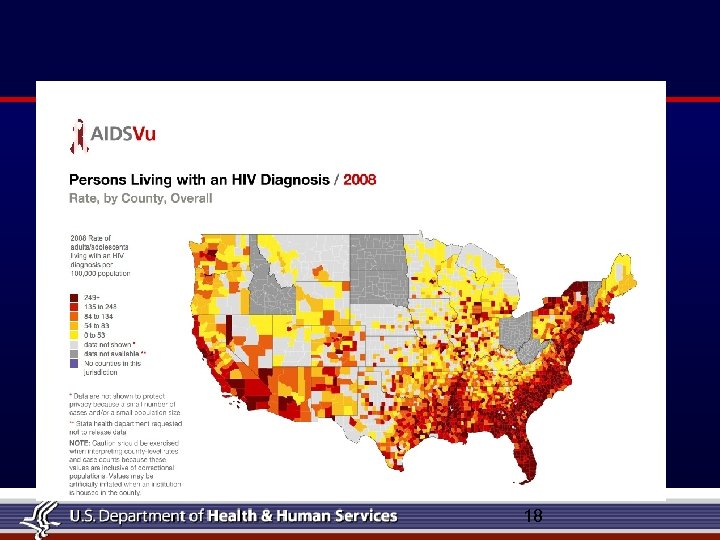
 HIV in Houston ● Houston ranks 8 th nationally in the number of total reported AIDS cases ● African American and Hispanic women together represent less than 25% of all U. S. women, yet they account for more than 78% of AIDS cases reported to date among women. ● Today, 40 million people worldwide are estimated to be living with HIV/AIDS ● AIDS is the leading cause of death among African-American women ages 25 to 34 and African-American men ages 35 to 44 ● CDC estimates that over a million Americans are currently living with HIV. ● 40, 000 Americans are newly infected with HIV each year. ● 6 out of every 10 diagnosed HIV infections in the Houston/Harris County are African American. ● An estimated 1 in 90 Houstonians is living with HIV/AIDS. 20
HIV in Houston ● Houston ranks 8 th nationally in the number of total reported AIDS cases ● African American and Hispanic women together represent less than 25% of all U. S. women, yet they account for more than 78% of AIDS cases reported to date among women. ● Today, 40 million people worldwide are estimated to be living with HIV/AIDS ● AIDS is the leading cause of death among African-American women ages 25 to 34 and African-American men ages 35 to 44 ● CDC estimates that over a million Americans are currently living with HIV. ● 40, 000 Americans are newly infected with HIV each year. ● 6 out of every 10 diagnosed HIV infections in the Houston/Harris County are African American. ● An estimated 1 in 90 Houstonians is living with HIV/AIDS. 20
 HIV in Houston 21
HIV in Houston 21
 Electron micrographic picture 22
Electron micrographic picture 22
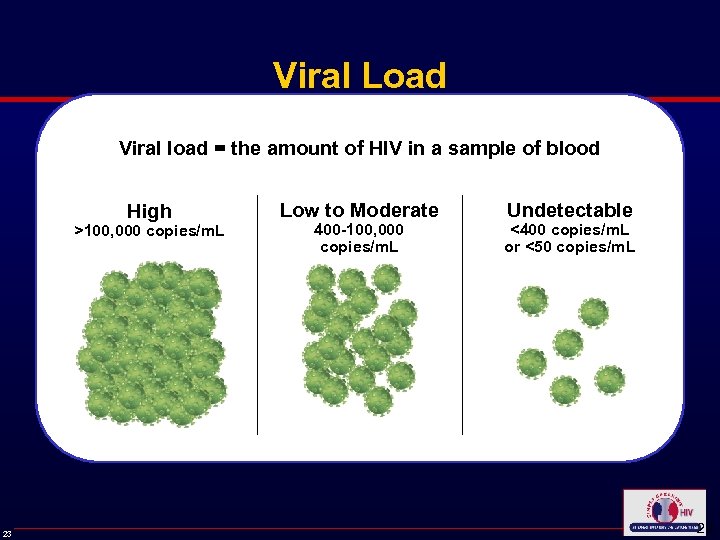 Viral Load Viral load = the amount of HIV in a sample of blood High >100, 000 copies/m. L 23 Low to Moderate 400 -100, 000 copies/m. L Undetectable <400 copies/m. L or <50 copies/m. L 23
Viral Load Viral load = the amount of HIV in a sample of blood High >100, 000 copies/m. L 23 Low to Moderate 400 -100, 000 copies/m. L Undetectable <400 copies/m. L or <50 copies/m. L 23
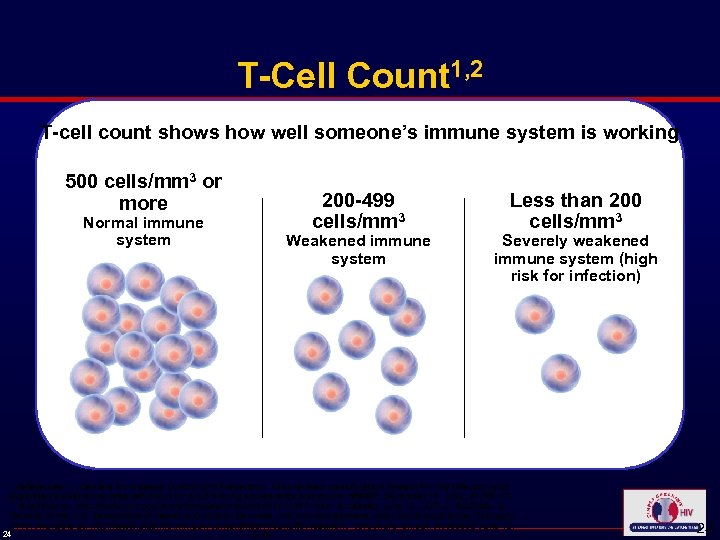 T-Cell Count 1, 2 T-cell count shows how well someone’s immune system is working 500 cells/mm 3 or more Normal immune system 200 -499 cells/mm 3 Weakened immune system Less than 200 cells/mm 3 Severely weakened immune system (high risk for infection) References: 1. Centers for Disease Control and Prevention. 1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR, December 18, 1992; 41(RR-17). Available at: http: //www. cdc. gov/mmwr/preview/mmwrhtml/00018871. htm. Accessed June 12, 2008. 2. AIDSinfo: A Service of the U. S. Department of Health and Human Services. HIV and its treatment: what you should know. February 2008. Available at: http: //www. aidsinfo. nih. gov/contentfiles/HIVand. Its. Treatment_cbrochure_en. pdf. Accessed June 12, 24 2008. 24
T-Cell Count 1, 2 T-cell count shows how well someone’s immune system is working 500 cells/mm 3 or more Normal immune system 200 -499 cells/mm 3 Weakened immune system Less than 200 cells/mm 3 Severely weakened immune system (high risk for infection) References: 1. Centers for Disease Control and Prevention. 1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR, December 18, 1992; 41(RR-17). Available at: http: //www. cdc. gov/mmwr/preview/mmwrhtml/00018871. htm. Accessed June 12, 2008. 2. AIDSinfo: A Service of the U. S. Department of Health and Human Services. HIV and its treatment: what you should know. February 2008. Available at: http: //www. aidsinfo. nih. gov/contentfiles/HIVand. Its. Treatment_cbrochure_en. pdf. Accessed June 12, 24 2008. 24
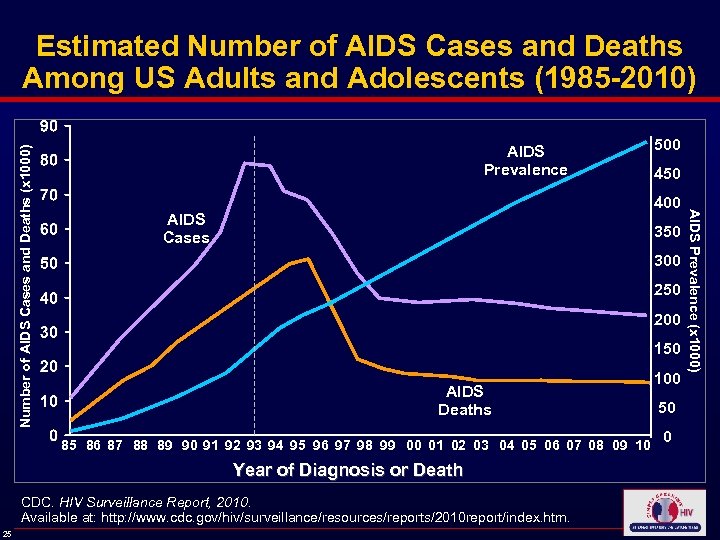 AIDS Prevalence 450 400 AIDS Cases 350 300 250 200 150 AIDS Deaths 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 Year of Diagnosis or Death CDC. HIV Surveillance Report, 2010. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2010 report/index. htm. 25 500 100 50 0 AIDS Prevalence (x 1000) Number of AIDS Cases and Deaths (x 1000) Estimated Number of AIDS Cases and Deaths Among US Adults and Adolescents (1985 -2010)
AIDS Prevalence 450 400 AIDS Cases 350 300 250 200 150 AIDS Deaths 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 Year of Diagnosis or Death CDC. HIV Surveillance Report, 2010. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2010 report/index. htm. 25 500 100 50 0 AIDS Prevalence (x 1000) Number of AIDS Cases and Deaths (x 1000) Estimated Number of AIDS Cases and Deaths Among US Adults and Adolescents (1985 -2010)
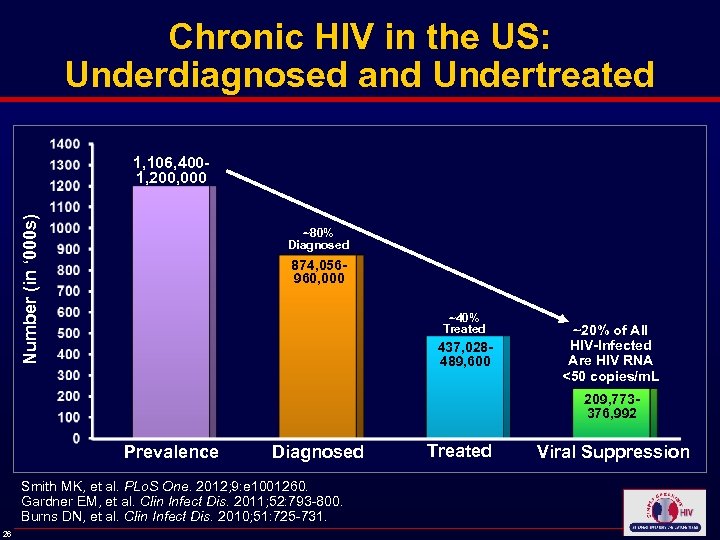 Chronic HIV in the US: Underdiagnosed and Undertreated Number (in ‘ 000 s) 1, 106, 4001, 200, 000 ~80% Diagnosed 874, 056960, 000 ~40% Treated 437, 028489, 600 ~20% of All HIV-Infected Are HIV RNA <50 copies/m. L 209, 773376, 992 Prevalence Diagnosed Smith MK, et al. PLo. S One. 2012; 9: e 1001260. Gardner EM, et al. Clin Infect Dis. 2011; 52: 793 -800. Burns DN, et al. Clin Infect Dis. 2010; 51: 725 -731. 26 Treated Viral Suppression
Chronic HIV in the US: Underdiagnosed and Undertreated Number (in ‘ 000 s) 1, 106, 4001, 200, 000 ~80% Diagnosed 874, 056960, 000 ~40% Treated 437, 028489, 600 ~20% of All HIV-Infected Are HIV RNA <50 copies/m. L 209, 773376, 992 Prevalence Diagnosed Smith MK, et al. PLo. S One. 2012; 9: e 1001260. Gardner EM, et al. Clin Infect Dis. 2011; 52: 793 -800. Burns DN, et al. Clin Infect Dis. 2010; 51: 725 -731. 26 Treated Viral Suppression
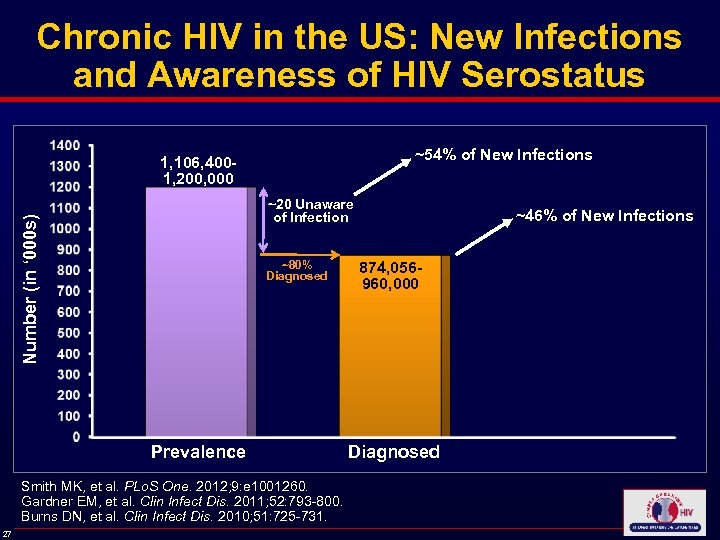 Chronic HIV in the US: New Infections and Awareness of HIV Serostatus ~54% of New Infections 1, 106, 4001, 200, 000 Number (in ‘ 000 s) ~20 Unaware of Infection ~80% Diagnosed Prevalence Smith MK, et al. PLo. S One. 2012; 9: e 1001260. Gardner EM, et al. Clin Infect Dis. 2011; 52: 793 -800. Burns DN, et al. Clin Infect Dis. 2010; 51: 725 -731. 27 ~46% of New Infections 874, 056960, 000 Diagnosed
Chronic HIV in the US: New Infections and Awareness of HIV Serostatus ~54% of New Infections 1, 106, 4001, 200, 000 Number (in ‘ 000 s) ~20 Unaware of Infection ~80% Diagnosed Prevalence Smith MK, et al. PLo. S One. 2012; 9: e 1001260. Gardner EM, et al. Clin Infect Dis. 2011; 52: 793 -800. Burns DN, et al. Clin Infect Dis. 2010; 51: 725 -731. 27 ~46% of New Infections 874, 056960, 000 Diagnosed
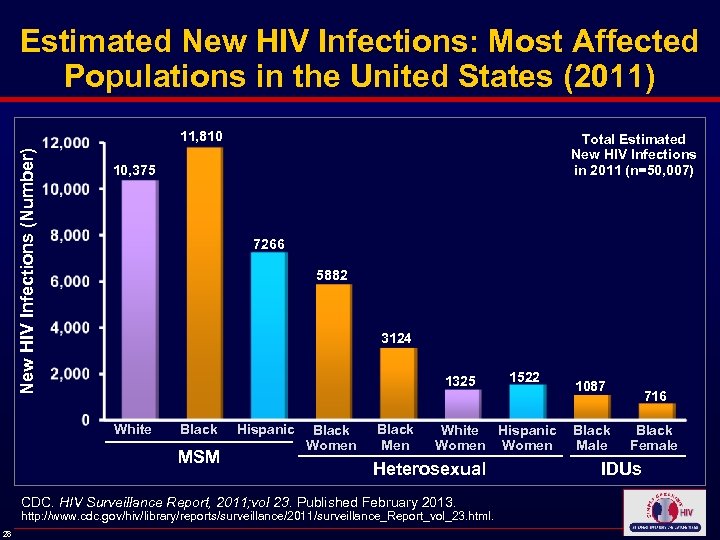 Estimated New HIV Infections: Most Affected Populations in the United States (2011) New HIV Infections (Number) 11, 810 Total Estimated New HIV Infections in 2011 (n=50, 007) 10, 375 7266 5882 3124 1325 White Black MSM Hispanic Black Women Black Men White Hispanic Women Heterosexual CDC. HIV Surveillance Report, 2011; vol 23. Published February 2013. http: //www. cdc. gov/hiv/library/reports/surveillance/2011/surveillance_Report_vol_23. html. 28 1522 1087 Black Male 716 Black Female IDUs
Estimated New HIV Infections: Most Affected Populations in the United States (2011) New HIV Infections (Number) 11, 810 Total Estimated New HIV Infections in 2011 (n=50, 007) 10, 375 7266 5882 3124 1325 White Black MSM Hispanic Black Women Black Men White Hispanic Women Heterosexual CDC. HIV Surveillance Report, 2011; vol 23. Published February 2013. http: //www. cdc. gov/hiv/library/reports/surveillance/2011/surveillance_Report_vol_23. html. 28 1522 1087 Black Male 716 Black Female IDUs
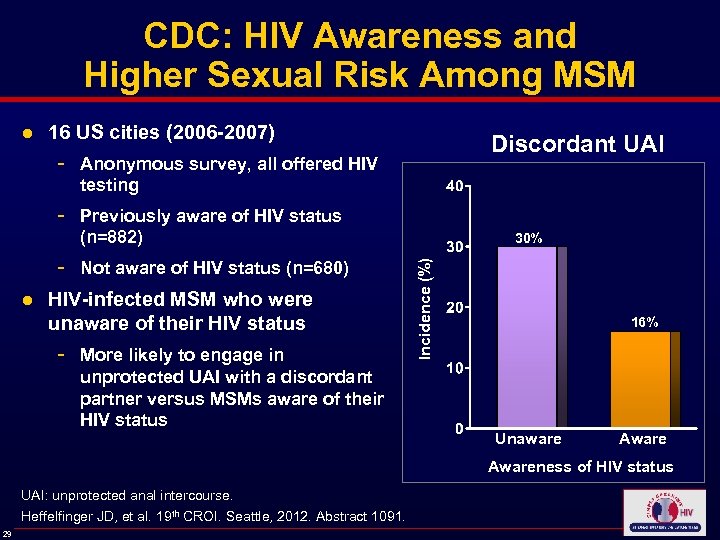 CDC: HIV Awareness and Higher Sexual Risk Among MSM ● 16 US cities (2006 -2007) Anonymous survey, all offered HIV testing - Previously aware of HIV status (n=882) - Not aware of HIV status (n=680) ● HIV-infected MSM who were unaware of their HIV status - More likely to engage in unprotected UAI with a discordant partner versus MSMs aware of their HIV status 30% Incidence (%) - Discordant UAI 16% Unaware Awareness of HIV status UAI: unprotected anal intercourse. Heffelfinger JD, et al. 19 th CROI. Seattle, 2012. Abstract 1091. 29
CDC: HIV Awareness and Higher Sexual Risk Among MSM ● 16 US cities (2006 -2007) Anonymous survey, all offered HIV testing - Previously aware of HIV status (n=882) - Not aware of HIV status (n=680) ● HIV-infected MSM who were unaware of their HIV status - More likely to engage in unprotected UAI with a discordant partner versus MSMs aware of their HIV status 30% Incidence (%) - Discordant UAI 16% Unaware Awareness of HIV status UAI: unprotected anal intercourse. Heffelfinger JD, et al. 19 th CROI. Seattle, 2012. Abstract 1091. 29
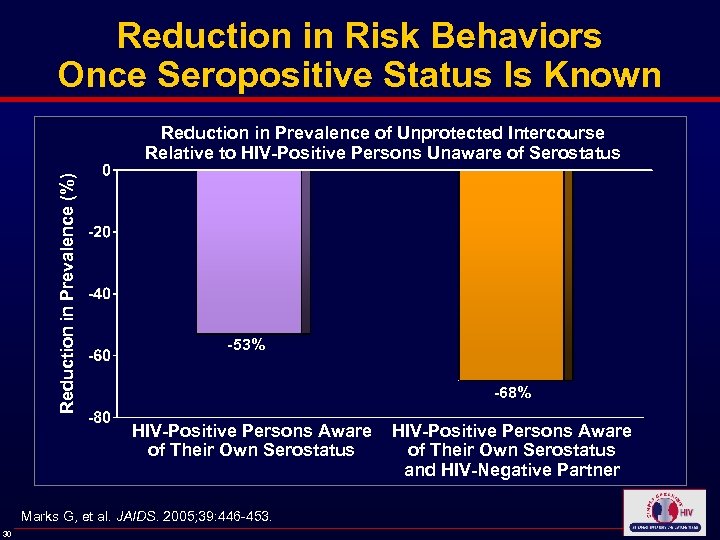 Reduction in Risk Behaviors Once Seropositive Status Is Known Reduction in Prevalence (%) Reduction in Prevalence of Unprotected Intercourse Relative to HIV-Positive Persons Unaware of Serostatus -53% -68% HIV-Positive Persons Aware of Their Own Serostatus Marks G, et al. JAIDS. 2005; 39: 446 -453. 30 HIV-Positive Persons Aware of Their Own Serostatus and HIV-Negative Partner
Reduction in Risk Behaviors Once Seropositive Status Is Known Reduction in Prevalence (%) Reduction in Prevalence of Unprotected Intercourse Relative to HIV-Positive Persons Unaware of Serostatus -53% -68% HIV-Positive Persons Aware of Their Own Serostatus Marks G, et al. JAIDS. 2005; 39: 446 -453. 30 HIV-Positive Persons Aware of Their Own Serostatus and HIV-Negative Partner
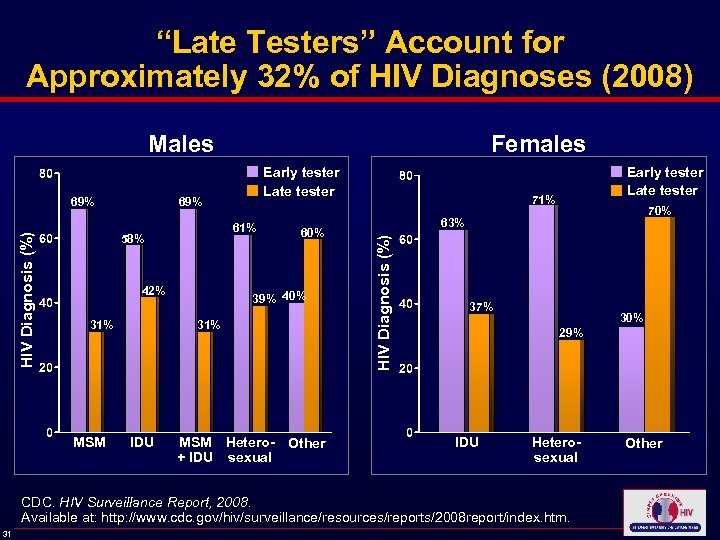 “Late Testers” Account for Approximately 32% of HIV Diagnoses (2008) Males 69% 61% 58% 42% 31% MSM Early tester Late tester 60% 39% 40% 31% IDU MSM Hetero- Other + IDU sexual Early tester Late tester 71% 70% 63% HIV Diagnosis (%) 69% Females 37% 30% 29% IDU Heterosexual CDC. HIV Surveillance Report, 2008. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2008 report/index. htm. 31 Other
“Late Testers” Account for Approximately 32% of HIV Diagnoses (2008) Males 69% 61% 58% 42% 31% MSM Early tester Late tester 60% 39% 40% 31% IDU MSM Hetero- Other + IDU sexual Early tester Late tester 71% 70% 63% HIV Diagnosis (%) 69% Females 37% 30% 29% IDU Heterosexual CDC. HIV Surveillance Report, 2008. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2008 report/index. htm. 31 Other
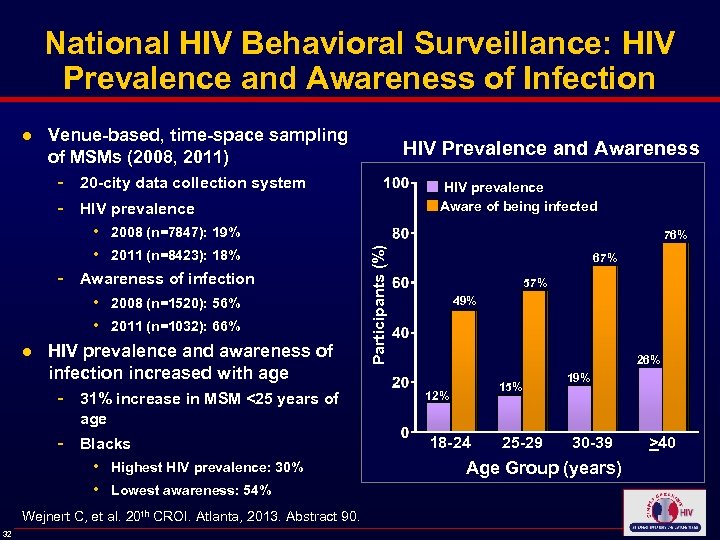 National HIV Behavioral Surveillance: HIV Prevalence and Awareness of Infection ● Venue-based, time-space sampling of MSMs (2008, 2011) 20 -city data collection system HIV prevalence • • - 2008 (n=7847): 19% 2011 (n=8423): 18% Awareness of infection • 2008 (n=1520): 56% • 2011 (n=1032): 66% ● HIV prevalence and awareness of infection increased with age - 31% increase in MSM <25 years of age - Blacks • • Highest HIV prevalence: 30% Lowest awareness: 54% Wejnert C, et al. 20 th CROI. Atlanta, 2013. Abstract 90. 32 HIV prevalence Aware of being infected 76% Participants (%) - HIV Prevalence and Awareness 67% 57% 49% 26% 15% 12% 18 -24 25 -29 19% 30 -39 Age Group (years) >40
National HIV Behavioral Surveillance: HIV Prevalence and Awareness of Infection ● Venue-based, time-space sampling of MSMs (2008, 2011) 20 -city data collection system HIV prevalence • • - 2008 (n=7847): 19% 2011 (n=8423): 18% Awareness of infection • 2008 (n=1520): 56% • 2011 (n=1032): 66% ● HIV prevalence and awareness of infection increased with age - 31% increase in MSM <25 years of age - Blacks • • Highest HIV prevalence: 30% Lowest awareness: 54% Wejnert C, et al. 20 th CROI. Atlanta, 2013. Abstract 90. 32 HIV prevalence Aware of being infected 76% Participants (%) - HIV Prevalence and Awareness 67% 57% 49% 26% 15% 12% 18 -24 25 -29 19% 30 -39 Age Group (years) >40
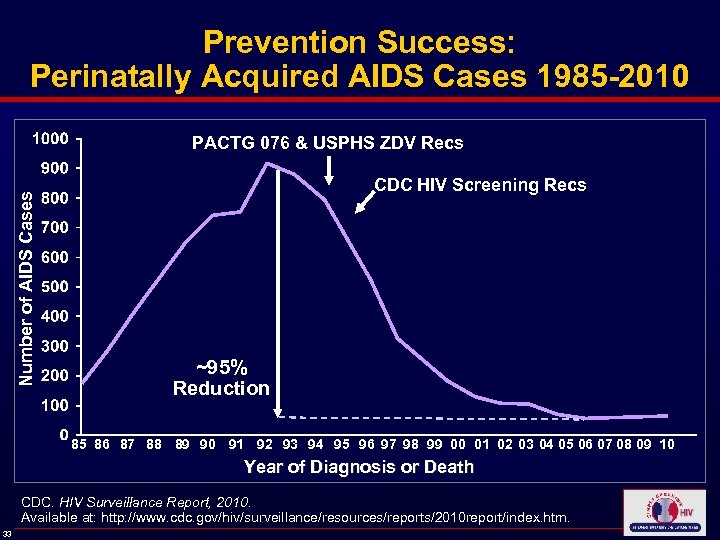 Prevention Success: Perinatally Acquired AIDS Cases 1985 -2010 PACTG 076 & USPHS ZDV Recs Number of AIDS Cases CDC HIV Screening Recs ~95% Reduction 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 Year of Diagnosis or Death CDC. HIV Surveillance Report, 2010. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2010 report/index. htm. 33
Prevention Success: Perinatally Acquired AIDS Cases 1985 -2010 PACTG 076 & USPHS ZDV Recs Number of AIDS Cases CDC HIV Screening Recs ~95% Reduction 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 10 Year of Diagnosis or Death CDC. HIV Surveillance Report, 2010. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2010 report/index. htm. 33
 HIV Screening: Guidelines From Other Organizations ● US Preventive Services Task Force (2007) - Strongly recommends clinicians screen for HIV in all adolescents and adults at increased risk • - No recommendation for or against routinely screening for HIV adolescents and adults who are not at increased risk for HIV infection Recommends clinicians screen all pregnant women for HIV ● American College of Physicians (2008) (endorsed by the HIV Medicine Association) - Recommends clinicians adopt routine screening for HIV and encourage patients to be tested • - Regardless of whether HIV risk factors are present Recommends clinicians determine the need for repeated screening on an individual basis US Preventive Services Task Force. AHRQ publication number 07 -0597 -EF-2. April 2, 2007. Qaseem A, et al. Ann Intern Med. 2009; 150: 1 -8. 34
HIV Screening: Guidelines From Other Organizations ● US Preventive Services Task Force (2007) - Strongly recommends clinicians screen for HIV in all adolescents and adults at increased risk • - No recommendation for or against routinely screening for HIV adolescents and adults who are not at increased risk for HIV infection Recommends clinicians screen all pregnant women for HIV ● American College of Physicians (2008) (endorsed by the HIV Medicine Association) - Recommends clinicians adopt routine screening for HIV and encourage patients to be tested • - Regardless of whether HIV risk factors are present Recommends clinicians determine the need for repeated screening on an individual basis US Preventive Services Task Force. AHRQ publication number 07 -0597 -EF-2. April 2, 2007. Qaseem A, et al. Ann Intern Med. 2009; 150: 1 -8. 34
 Revised CDC Recommendations for HIV Testing in Healthcare Settings ● Routine voluntary testing for patients ages 13 to 64 years in healthcare settings - Not based on patient risk ● Opt-out testing ● No separate consent for HIV ● Pretest counseling not required ● Repeat HIV testing left to discretion of provider - Based on patient risk Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 35
Revised CDC Recommendations for HIV Testing in Healthcare Settings ● Routine voluntary testing for patients ages 13 to 64 years in healthcare settings - Not based on patient risk ● Opt-out testing ● No separate consent for HIV ● Pretest counseling not required ● Repeat HIV testing left to discretion of provider - Based on patient risk Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 35
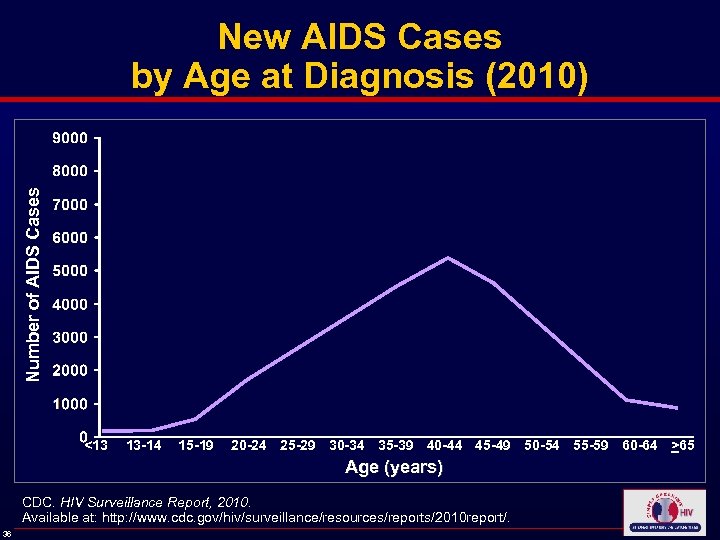 Number of AIDS Cases New AIDS Cases by Age at Diagnosis (2010) <13 13 -14 15 -19 20 -24 25 -29 30 -34 35 -39 40 -44 45 -49 50 -54 55 -59 60 -64 >65 Age (years) CDC. HIV Surveillance Report, 2010. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2010 report/. 36
Number of AIDS Cases New AIDS Cases by Age at Diagnosis (2010) <13 13 -14 15 -19 20 -24 25 -29 30 -34 35 -39 40 -44 45 -49 50 -54 55 -59 60 -64 >65 Age (years) CDC. HIV Surveillance Report, 2010. Available at: http: //www. cdc. gov/hiv/surveillance/resources/reports/2010 report/. 36
 Opt-Out Versus Opt-In Screening Opt-Out Screening ● Implies all patients are considered candidates for screening ● Testing is part of standard panel of tests ● Patients can decline test, but test is performed unless patient specifically refuses Opt-In Screening ● Requires providers to specifically recommend HIV testing and for patients to specifically agree to testing ● May assume that clinicians assess which patient is at-risk for infection ● Greater reluctance on part of patient ● Requires more staff time Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 37
Opt-Out Versus Opt-In Screening Opt-Out Screening ● Implies all patients are considered candidates for screening ● Testing is part of standard panel of tests ● Patients can decline test, but test is performed unless patient specifically refuses Opt-In Screening ● Requires providers to specifically recommend HIV testing and for patients to specifically agree to testing ● May assume that clinicians assess which patient is at-risk for infection ● Greater reluctance on part of patient ● Requires more staff time Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 37
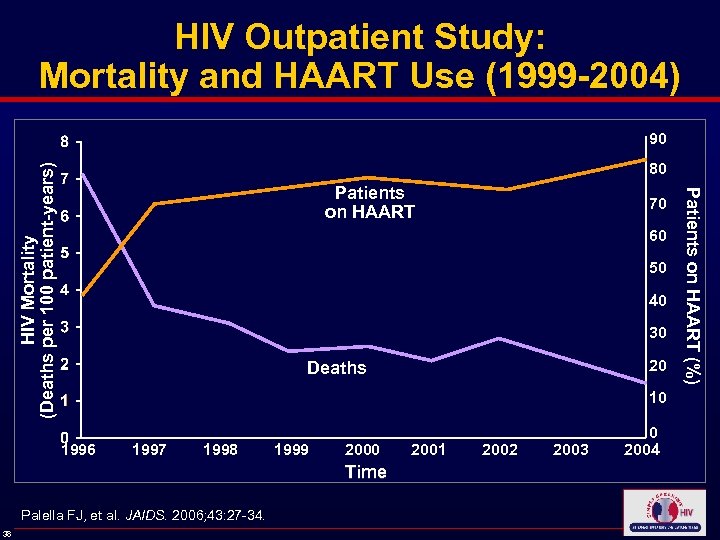 HIV Outpatient Study: Mortality and HAART Use (1999 -2004) 90 Patients on HAART 70 60 50 40 30 Deaths 20 10 0 1996 1997 1998 1999 2000 2001 2002 2003 2004 Time Palella FJ, et al. JAIDS. 2006; 43: 27 -34. 38 Patients on HAART (%) HIV Mortality (Deaths per 100 patient-years) 80
HIV Outpatient Study: Mortality and HAART Use (1999 -2004) 90 Patients on HAART 70 60 50 40 30 Deaths 20 10 0 1996 1997 1998 1999 2000 2001 2002 2003 2004 Time Palella FJ, et al. JAIDS. 2006; 43: 27 -34. 38 Patients on HAART (%) HIV Mortality (Deaths per 100 patient-years) 80
 Adults and Adolescents: Recommendations for HIV-Screening Location ● All primary care settings ● Emergency departments, in-patient services, and urgent care clinics ● Public health settings - Tuberculosis clinics - Sexually transmitted diseases clinics - Substance abuse treatment centers - Correctional facility treatment centers ● Screening may be discontinued in low-prevalence communities with demonstrated yield <1: 1000 Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 39
Adults and Adolescents: Recommendations for HIV-Screening Location ● All primary care settings ● Emergency departments, in-patient services, and urgent care clinics ● Public health settings - Tuberculosis clinics - Sexually transmitted diseases clinics - Substance abuse treatment centers - Correctional facility treatment centers ● Screening may be discontinued in low-prevalence communities with demonstrated yield <1: 1000 Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 39
 American College of Emergency Physicians Policy Statement ● Early HIV diagnosis and treatment - Prolong life, reduce transmission, and a cost-effective public health intervention ● HIV testing in the ED - Should be available in an expeditious and efficient fashion similar to testing and results for other conditions ● HIV screening when deemed appropriate by the ED physician - Must be practical and feasible for emergency settings - Should be integrated with the resources of the entire health care system - All local and state requirements must be met Cannot interfere with the primary acute care mission of ED Should be offered based on the local prevalence and medical needs of the community Policies and procedures must adequately address patient confidentiality, informed consent (state dependent), provider training, significant need for pre and post-test counseling, and linkage to care ACEP Board of Directors. Ann Emerg Med. 2007; 50: 209. 40
American College of Emergency Physicians Policy Statement ● Early HIV diagnosis and treatment - Prolong life, reduce transmission, and a cost-effective public health intervention ● HIV testing in the ED - Should be available in an expeditious and efficient fashion similar to testing and results for other conditions ● HIV screening when deemed appropriate by the ED physician - Must be practical and feasible for emergency settings - Should be integrated with the resources of the entire health care system - All local and state requirements must be met Cannot interfere with the primary acute care mission of ED Should be offered based on the local prevalence and medical needs of the community Policies and procedures must adequately address patient confidentiality, informed consent (state dependent), provider training, significant need for pre and post-test counseling, and linkage to care ACEP Board of Directors. Ann Emerg Med. 2007; 50: 209. 40
 CDC Revised HIV Screening Recommendations for Pregnant Women ● Universal opt-out screening - Included in routine panel of prenatal screening tests Notification and option to decline ● Second test in third trimester for women who are - Known to be at risk for HIV In communities with elevated HIV incidence In high-prevalence health care facilities ● Labor and delivery opt-out rapid testing for women with undocumented HIV status - Initiate antiretroviral prophylaxis on basis of test result ● Rapid testing of newborn if mother’s status unknown at delivery - Initiate antiretroviral prophylaxis within 12 hours of birth on basis of rapid test result Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 41
CDC Revised HIV Screening Recommendations for Pregnant Women ● Universal opt-out screening - Included in routine panel of prenatal screening tests Notification and option to decline ● Second test in third trimester for women who are - Known to be at risk for HIV In communities with elevated HIV incidence In high-prevalence health care facilities ● Labor and delivery opt-out rapid testing for women with undocumented HIV status - Initiate antiretroviral prophylaxis on basis of test result ● Rapid testing of newborn if mother’s status unknown at delivery - Initiate antiretroviral prophylaxis within 12 hours of birth on basis of rapid test result Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 41
 HIV Screening in Women: American College of Obstetricians and Gynecologists ● Recommends routine HIV screening for women aged 19 to 64 years - Recommends targeted screening for women with risk factors outside this age range ● Supports opt-out HIV screening - Should be aware of and comply with legal requirements regarding HIV testing in their jurisdiction ● Recommends annually review patient’s risk factors for HIV and assess the need for retesting - Repeat HIV testing should be offered at least annually • - IDUs, sex partners who are IDUs or HIV infected, exchange sex for drugs or money, STD, >1 sex partner since last HIV test Encourage women and prospective sex partner to be tested before initiating a new sexual relationship, ACOG Committee Opinion Number 411. Obstet Gynecol. 2008; 114: 401 -403. 42
HIV Screening in Women: American College of Obstetricians and Gynecologists ● Recommends routine HIV screening for women aged 19 to 64 years - Recommends targeted screening for women with risk factors outside this age range ● Supports opt-out HIV screening - Should be aware of and comply with legal requirements regarding HIV testing in their jurisdiction ● Recommends annually review patient’s risk factors for HIV and assess the need for retesting - Repeat HIV testing should be offered at least annually • - IDUs, sex partners who are IDUs or HIV infected, exchange sex for drugs or money, STD, >1 sex partner since last HIV test Encourage women and prospective sex partner to be tested before initiating a new sexual relationship, ACOG Committee Opinion Number 411. Obstet Gynecol. 2008; 114: 401 -403. 42
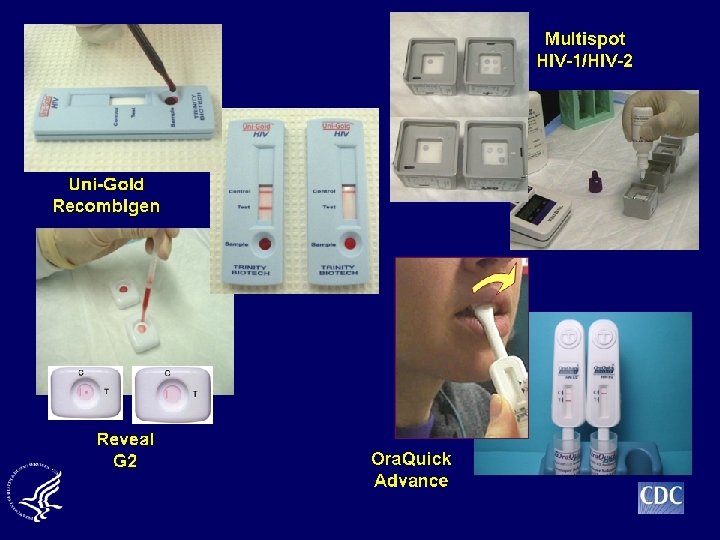
 Types of HIV Screening Tests Available to Primary Care Providers ● Six rapid HIV tests available ● Results available within 15 to 30 minutes ● 4 are CLIA-waived - Ora. Quick Advance Rapid HIV-1/2 Uni-Gold Recombigen HIV Clearview HIV 1/2 Stat-Pak Clearview Complete HIV 1/2 FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 43
Types of HIV Screening Tests Available to Primary Care Providers ● Six rapid HIV tests available ● Results available within 15 to 30 minutes ● 4 are CLIA-waived - Ora. Quick Advance Rapid HIV-1/2 Uni-Gold Recombigen HIV Clearview HIV 1/2 Stat-Pak Clearview Complete HIV 1/2 FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 43
 44
44
 Clinical Laboratory Improvements Amendments (CLIA) Program ● All laboratories (ie, any facility that performs examinations on human specimens) must be certified under the CLIA program - Administered by the Centers for Medicare and Medicaid Services • • Provides oversight of clinical laboratories Issues waiver certificates ● CLIA waiver - Laboratories that perform only tests that are "simple" and that have an "insignificant risk of an erroneous result" must obtain a certificate of waiver before accepting materials derived from the human body for laboratory tests The examinations and procedures [that may be performed by a laboratory with a Certificate of Waiver]… are laboratory examinations and procedures that have been approved by the Food and Drug Administration for home use or that, as determined by the Secretary, are simple laboratory examinations and procedures that have an insignificant risk of an erroneous result, including those that -- (A) employ methodologies that are so simple and accurate as to render the likelihood of erroneous results by the user negligible, or (B) the Secretary has determined pose no unreasonable risk of harm to the patient if Performed incorrectly. CDC. Available at: http: //www. cdc. gov/hiv/topics/testing/resources/reports/labtest_clia. htm. FDA Center for Devices and Radiological Health. Available at: http: //www. accessdata. fda. gov/scripts/cdrh/cfdocs/cf. Clia/Search. cfm. 45
Clinical Laboratory Improvements Amendments (CLIA) Program ● All laboratories (ie, any facility that performs examinations on human specimens) must be certified under the CLIA program - Administered by the Centers for Medicare and Medicaid Services • • Provides oversight of clinical laboratories Issues waiver certificates ● CLIA waiver - Laboratories that perform only tests that are "simple" and that have an "insignificant risk of an erroneous result" must obtain a certificate of waiver before accepting materials derived from the human body for laboratory tests The examinations and procedures [that may be performed by a laboratory with a Certificate of Waiver]… are laboratory examinations and procedures that have been approved by the Food and Drug Administration for home use or that, as determined by the Secretary, are simple laboratory examinations and procedures that have an insignificant risk of an erroneous result, including those that -- (A) employ methodologies that are so simple and accurate as to render the likelihood of erroneous results by the user negligible, or (B) the Secretary has determined pose no unreasonable risk of harm to the patient if Performed incorrectly. CDC. Available at: http: //www. cdc. gov/hiv/topics/testing/resources/reports/labtest_clia. htm. FDA Center for Devices and Radiological Health. Available at: http: //www. accessdata. fda. gov/scripts/cdrh/cfdocs/cf. Clia/Search. cfm. 45
 CLIA-Waived Rapid HIV Antibody Tests: Sensitivity and Specificity Sensitivity (95% CI) Ora. Quick Advance Rapid HIV 1/2 (Orasure)* Oral fluid Whole blood Plasma† Uni-Gold Recombigen HIV (Trinity)‡ Whole blood Serum and plasma† Specificity (95% CI) 99. 3 99. 8 (98. 4 -99. 7) (99. 6 -99. 9) 99. 6 100 (98. 5 -99. 9) (99. 7 -100) 99. 6 99. 9 (98. 9 -99. 8) (99. 6 -99. 9) 100 99. 7 (99. 5 -100) (99. 0 -100) 100 99. 8 (99. 5 -100) *FDA-approved for detection of HIV-2. †Moderate complexity, non-CLIA waived. ‡Not FDA-approved for detection of HIV-2. FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 46 (99. 3 -100)
CLIA-Waived Rapid HIV Antibody Tests: Sensitivity and Specificity Sensitivity (95% CI) Ora. Quick Advance Rapid HIV 1/2 (Orasure)* Oral fluid Whole blood Plasma† Uni-Gold Recombigen HIV (Trinity)‡ Whole blood Serum and plasma† Specificity (95% CI) 99. 3 99. 8 (98. 4 -99. 7) (99. 6 -99. 9) 99. 6 100 (98. 5 -99. 9) (99. 7 -100) 99. 6 99. 9 (98. 9 -99. 8) (99. 6 -99. 9) 100 99. 7 (99. 5 -100) (99. 0 -100) 100 99. 8 (99. 5 -100) *FDA-approved for detection of HIV-2. †Moderate complexity, non-CLIA waived. ‡Not FDA-approved for detection of HIV-2. FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 46 (99. 3 -100)
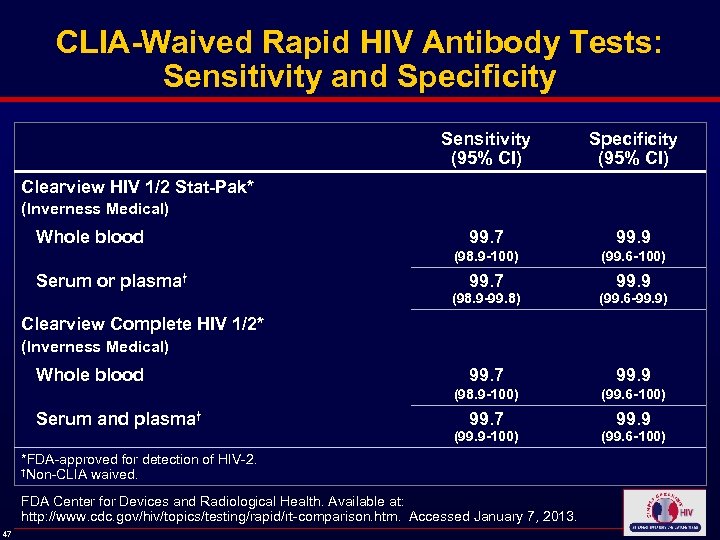 CLIA-Waived Rapid HIV Antibody Tests: Sensitivity and Specificity Sensitivity (95% CI) Specificity (95% CI) 99. 7 99. 9 (98. 9 -100) (99. 6 -100) 99. 7 99. 9 Clearview HIV 1/2 Stat-Pak* (Inverness Medical) Whole blood Serum or plasma† (98. 9 -99. 8) (99. 6 -99. 9) 99. 7 99. 9 (98. 9 -100) (99. 6 -100) 99. 7 99. 9 Clearview Complete HIV 1/2* (Inverness Medical) Whole blood Serum and plasma† (99. 9 -100) *FDA-approved for detection of HIV-2. †Non-CLIA waived. FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 47 (99. 6 -100)
CLIA-Waived Rapid HIV Antibody Tests: Sensitivity and Specificity Sensitivity (95% CI) Specificity (95% CI) 99. 7 99. 9 (98. 9 -100) (99. 6 -100) 99. 7 99. 9 Clearview HIV 1/2 Stat-Pak* (Inverness Medical) Whole blood Serum or plasma† (98. 9 -99. 8) (99. 6 -99. 9) 99. 7 99. 9 (98. 9 -100) (99. 6 -100) 99. 7 99. 9 Clearview Complete HIV 1/2* (Inverness Medical) Whole blood Serum and plasma† (99. 9 -100) *FDA-approved for detection of HIV-2. †Non-CLIA waived. FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 47 (99. 6 -100)
 Non-CLIA-Waived Rapid HIV Antibody Tests: Sensitivity and Specificity Sensitivity (95% CI) Specificity (95% CI) 99. 8 99. 1 (99. 2 -100) (98. 8 -99. 4) 99. 8 98. 6 (99. 0 -100) (98. 4 -98. 8) 100 99. 93 (99. 94 -100) (99. 79 -100) 100 99. 91 (99. 94 -100) (99. 77 -100) Reveal G-3 Rapid HIV-1 (Medmira)* Serum Plasma Multispot HIV-1/HIV-2 Rapid Test (Bio-Rad)† Serum Plasma *Not FDA-approved for detection of HIV-2. †FDA-approved for detection of HIV-2 and can differentiate HIV-1 from HIV-2. FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 48
Non-CLIA-Waived Rapid HIV Antibody Tests: Sensitivity and Specificity Sensitivity (95% CI) Specificity (95% CI) 99. 8 99. 1 (99. 2 -100) (98. 8 -99. 4) 99. 8 98. 6 (99. 0 -100) (98. 4 -98. 8) 100 99. 93 (99. 94 -100) (99. 79 -100) 100 99. 91 (99. 94 -100) (99. 77 -100) Reveal G-3 Rapid HIV-1 (Medmira)* Serum Plasma Multispot HIV-1/HIV-2 Rapid Test (Bio-Rad)† Serum Plasma *Not FDA-approved for detection of HIV-2. †FDA-approved for detection of HIV-2 and can differentiate HIV-1 from HIV-2. FDA Center for Devices and Radiological Health. Available at: http: //www. cdc. gov/hiv/topics/testing/rapid/rt-comparison. htm. Accessed January 7, 2013. 48
 Home HIV Testing Ora. Quick In-Home HIV Test Source: Ora. Sure Technologies: FDA Briefing Materials: May 15, 2012.
Home HIV Testing Ora. Quick In-Home HIV Test Source: Ora. Sure Technologies: FDA Briefing Materials: May 15, 2012.
 Patient Acceptance of Rapid Testing ● Rapid testing versus “traditional” HIV screening - Tested at anonymous HIV testing site and STD clinic 88% of previously tested preferred rapid results ● Results - Anonymous testing site • - 4% increase in uninfected and 16% increase in HIV-infected individuals learning of their serostatus STD Clinic • 210% increase in uninfected and 23% increase in HIV-infected individuals learning of their serostatus Kassler WJ, et al. AIDS. 1997; 11: 1045 -1051. 50
Patient Acceptance of Rapid Testing ● Rapid testing versus “traditional” HIV screening - Tested at anonymous HIV testing site and STD clinic 88% of previously tested preferred rapid results ● Results - Anonymous testing site • - 4% increase in uninfected and 16% increase in HIV-infected individuals learning of their serostatus STD Clinic • 210% increase in uninfected and 23% increase in HIV-infected individuals learning of their serostatus Kassler WJ, et al. AIDS. 1997; 11: 1045 -1051. 50
 Rapid Testing in a Large Urban Emergency Department ● Rapid HIV test offered to patients seen in the ED (n=1348) ● Opted out (n=348) - Common reasons: recent negative test (46%), low risk (19%) No reason given (27%) ● Opted in (n=1000) - Positive test (n=12) Negative test (n=988) • Never tested before (33%), rest had a previous negative test ● Conclusions - High acceptance of rapid testing Rapid HIV test performed well (no false positive results) Seroprevalence rate meets CDC criteria for routine universal testing Lowman E, et al. 5 th IAS Conference. Cape Town, 2009. Abstract Mo. PEB 012. 51
Rapid Testing in a Large Urban Emergency Department ● Rapid HIV test offered to patients seen in the ED (n=1348) ● Opted out (n=348) - Common reasons: recent negative test (46%), low risk (19%) No reason given (27%) ● Opted in (n=1000) - Positive test (n=12) Negative test (n=988) • Never tested before (33%), rest had a previous negative test ● Conclusions - High acceptance of rapid testing Rapid HIV test performed well (no false positive results) Seroprevalence rate meets CDC criteria for routine universal testing Lowman E, et al. 5 th IAS Conference. Cape Town, 2009. Abstract Mo. PEB 012. 51
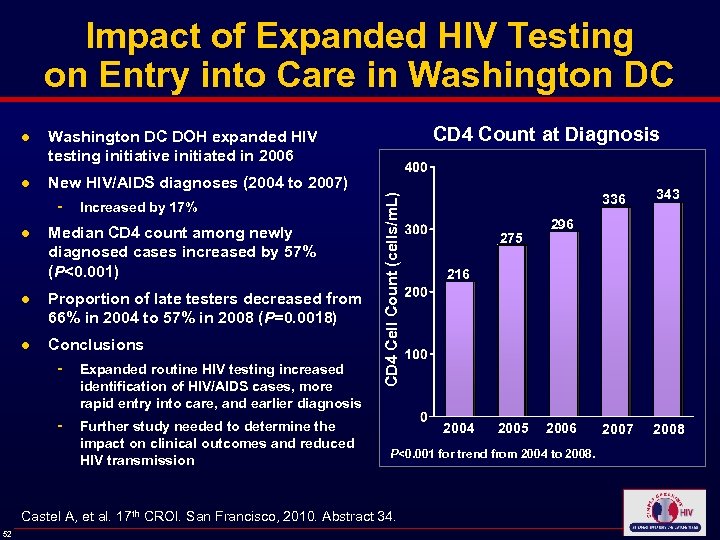 Impact of Expanded HIV Testing on Entry into Care in Washington DC ● CD 4 Count at Diagnosis Washington DC DOH expanded HIV testing initiative initiated in 2006 New HIV/AIDS diagnoses (2004 to 2007) ● Increased by 17% Median CD 4 count among newly diagnosed cases increased by 57% (P<0. 001) ● Proportion of late testers decreased from 66% in 2004 to 57% in 2008 (P=0. 0018) ● Conclusions - Expanded routine HIV testing increased identification of HIV/AIDS cases, more rapid entry into care, and earlier diagnosis - Further study needed to determine the impact on clinical outcomes and reduced HIV transmission CD 4 Cell Count (cells/m. L) ● 275 343 2007 2008 296 216 2004 2005 2006 P<0. 001 for trend from 2004 to 2008. Castel A, et al. 17 th CROI. San Francisco, 2010. Abstract 34. 52 336
Impact of Expanded HIV Testing on Entry into Care in Washington DC ● CD 4 Count at Diagnosis Washington DC DOH expanded HIV testing initiative initiated in 2006 New HIV/AIDS diagnoses (2004 to 2007) ● Increased by 17% Median CD 4 count among newly diagnosed cases increased by 57% (P<0. 001) ● Proportion of late testers decreased from 66% in 2004 to 57% in 2008 (P=0. 0018) ● Conclusions - Expanded routine HIV testing increased identification of HIV/AIDS cases, more rapid entry into care, and earlier diagnosis - Further study needed to determine the impact on clinical outcomes and reduced HIV transmission CD 4 Cell Count (cells/m. L) ● 275 343 2007 2008 296 216 2004 2005 2006 P<0. 001 for trend from 2004 to 2008. Castel A, et al. 17 th CROI. San Francisco, 2010. Abstract 34. 52 336
 HIV Prevalence Among Patients Who Decline Rapid HIV Testing ● Routine “opt-out” HIV testing program at the emergency Department at George Washington University - Approximately 44% of persons approached opted out or declined testing Discarded blood samples were tested for HIV • Ora-Quick rapid HIV test, positive results underwent Western Blot confirmation ● Results - Opt-out versus opt-in positives: 2% versus 0. 7% (P=0. 0038) Demographics of opt-out positives • • • - Women: 67% (P=0. 0014) African Americans: 83. 3% Uninsured: 40% Most common complaint: abdominal pain Most common reason for declining: patients believed they were not at risk ● Interventions are needed to decrease the opt-out rate Czarnogorski M, et al. AIDS Res Treat. 2011; 2011: 879065. Epub 2011. 53
HIV Prevalence Among Patients Who Decline Rapid HIV Testing ● Routine “opt-out” HIV testing program at the emergency Department at George Washington University - Approximately 44% of persons approached opted out or declined testing Discarded blood samples were tested for HIV • Ora-Quick rapid HIV test, positive results underwent Western Blot confirmation ● Results - Opt-out versus opt-in positives: 2% versus 0. 7% (P=0. 0038) Demographics of opt-out positives • • • - Women: 67% (P=0. 0014) African Americans: 83. 3% Uninsured: 40% Most common complaint: abdominal pain Most common reason for declining: patients believed they were not at risk ● Interventions are needed to decrease the opt-out rate Czarnogorski M, et al. AIDS Res Treat. 2011; 2011: 879065. Epub 2011. 53
 Adolescents: Special Considerations for HIV Screening ● HIV screening - Discuss with all adolescents and encourage HIV testing for those who are sexually active ● Laws concerning HIV testing and confidentiality differ among states ● Laws and legal precedents allow for evaluation and treatment of minors for STDs without parental knowledge or consent - Not every state has defined HIV infection explicitly as a condition for which testing or treatment may proceed without parental consent Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 54
Adolescents: Special Considerations for HIV Screening ● HIV screening - Discuss with all adolescents and encourage HIV testing for those who are sexually active ● Laws concerning HIV testing and confidentiality differ among states ● Laws and legal precedents allow for evaluation and treatment of minors for STDs without parental knowledge or consent - Not every state has defined HIV infection explicitly as a condition for which testing or treatment may proceed without parental consent Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 54
 Confirmatory Tests for Patients Who Test HIV Positive ● Positive rapid antibody tests are considered preliminary ● Confirmatory testing is needed to diagnose HIV infection - ELISA and Western blot test ● HIV-positive test results - Communicated confidentially through personal contact by a clinician, nurse, mid-level practitioner, counselor, or other skilled staff Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 55
Confirmatory Tests for Patients Who Test HIV Positive ● Positive rapid antibody tests are considered preliminary ● Confirmatory testing is needed to diagnose HIV infection - ELISA and Western blot test ● HIV-positive test results - Communicated confidentially through personal contact by a clinician, nurse, mid-level practitioner, counselor, or other skilled staff Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 55
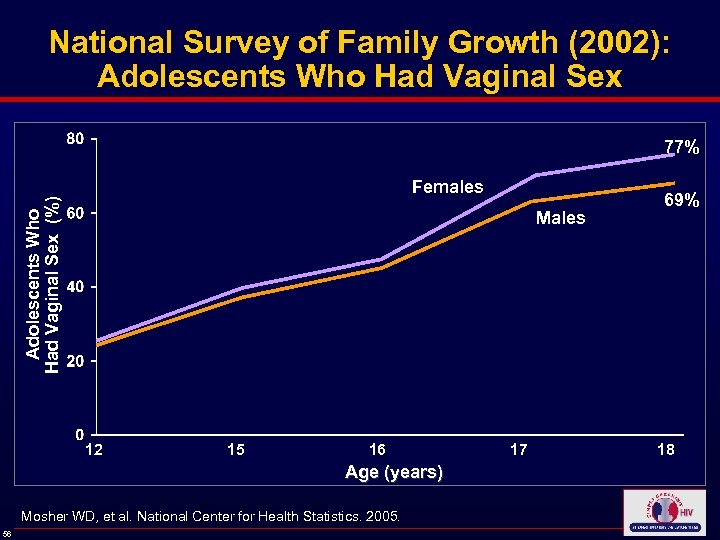 National Survey of Family Growth (2002): Adolescents Who Had Vaginal Sex 77% Adolescents Who Had Vaginal Sex (%) Females Males 69% 12 15 16 17 18 Age (years) Mosher WD, et al. National Center for Health Statistics. 2005. 56
National Survey of Family Growth (2002): Adolescents Who Had Vaginal Sex 77% Adolescents Who Had Vaginal Sex (%) Females Males 69% 12 15 16 17 18 Age (years) Mosher WD, et al. National Center for Health Statistics. 2005. 56
 HIV-Negative Persons: Prevention Services ● Risk screening - Assessment of risk for infection with HIV and other STDs and provision of prevention information should be incorporated into routine primary care of all sexually active persons - Refer at-risk patients to appropriate risk-reduction services (drug treatment, STD counseling) ● Prevention counseling - Does not need to be linked to HIV screening Should be offered or made available in facilities that service high-risk populations Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 57
HIV-Negative Persons: Prevention Services ● Risk screening - Assessment of risk for infection with HIV and other STDs and provision of prevention information should be incorporated into routine primary care of all sexually active persons - Refer at-risk patients to appropriate risk-reduction services (drug treatment, STD counseling) ● Prevention counseling - Does not need to be linked to HIV screening Should be offered or made available in facilities that service high-risk populations Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 57
 Public Health Issues: HIV/AIDS Surveillance ● Risk factor assessment - Recommended in order to target community-wide prevention efforts ● HIV/AIDS case reporting - Mandatory reporting of AIDS cases and HIV infection diagnoses required by all states ● Pediatric exposure reporting - Recommended by the CDC Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 58
Public Health Issues: HIV/AIDS Surveillance ● Risk factor assessment - Recommended in order to target community-wide prevention efforts ● HIV/AIDS case reporting - Mandatory reporting of AIDS cases and HIV infection diagnoses required by all states ● Pediatric exposure reporting - Recommended by the CDC Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 58
 Partner Counseling and Referral ● Encourage patients who test positive for HIV to notify current and prior sex partners ● Local health departments can make notification without disclosing patient’s identity ● Providers should notify patients they may be approached by local health departments for voluntary interviews regarding partner notification Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 59
Partner Counseling and Referral ● Encourage patients who test positive for HIV to notify current and prior sex partners ● Local health departments can make notification without disclosing patient’s identity ● Providers should notify patients they may be approached by local health departments for voluntary interviews regarding partner notification Branson BM, et al. MMWR Recomm Rep. 2006; 55(RR-14): 1 -17. 59
 Impact of New Guidelines on the Health Care System ● Implementation of routine testing - Dramatically increase funding needs of screening programs, including the cost of the test itself (conventional or rapid HIV tests) ● Effective HIV screening programs - Need to effectively link patients to competent and appropriate care and prevention services ● Approximately 50% of HIV-positive patients have no health insurance - Increased demand on public funding to support the cost of care and medications 60
Impact of New Guidelines on the Health Care System ● Implementation of routine testing - Dramatically increase funding needs of screening programs, including the cost of the test itself (conventional or rapid HIV tests) ● Effective HIV screening programs - Need to effectively link patients to competent and appropriate care and prevention services ● Approximately 50% of HIV-positive patients have no health insurance - Increased demand on public funding to support the cost of care and medications 60
 When to Start Treatment 2/2013 DHHS Guidelines 2012 IAS-USA Guidelines CD 4 Count HIV RNA (cells/mm 3) (copies/m. L) Any value Treat <500 Any value Treat >500* Any value Treat Pregnant women Any value Treat HIV-associated nephropathy Any value Treat HIV/HBV coinfection when HBV treatment is indicated Any value Treat Clinical Category AIDS-defining illness or severe symptoms Asymptomatic *Consideration of transmission risk and willingness to adhere to early therapy are considerations in when to start. The IAS-USA guidelines also recommends initiating antiretroviral therapy in HIV-infected patients with active hepatitis C virus infection, active or high risk for cardiovascular disease, and symptomatic primary HIV infection. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013; Thompson MA, et al. JAMA. 2012; 308: 387 -402. 61
When to Start Treatment 2/2013 DHHS Guidelines 2012 IAS-USA Guidelines CD 4 Count HIV RNA (cells/mm 3) (copies/m. L) Any value Treat <500 Any value Treat >500* Any value Treat Pregnant women Any value Treat HIV-associated nephropathy Any value Treat HIV/HBV coinfection when HBV treatment is indicated Any value Treat Clinical Category AIDS-defining illness or severe symptoms Asymptomatic *Consideration of transmission risk and willingness to adhere to early therapy are considerations in when to start. The IAS-USA guidelines also recommends initiating antiretroviral therapy in HIV-infected patients with active hepatitis C virus infection, active or high risk for cardiovascular disease, and symptomatic primary HIV infection. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013; Thompson MA, et al. JAMA. 2012; 308: 387 -402. 61
 NRTI 62 Current ARV Medications ● Abacavir (ABC) ● Didanosine (dd. I) ● Emtricitabine (FTC) PI ● Lamivudine (3 TC) ● Atazanavir (ATV) ● Darunavir (DRV) ● Stavudine (d 4 T) ● Fosamprenavir (FPV) ● Tenofovir (TDF) ● Indinavir (IDV) ● Zidovudine (AZT, ZDV) ● Lopinavir (LPV) NNRTI ● Nelfinavir (NFV) ● Delavirdine (DLV) ● Ritonavir (RTV) ● Efavirenz (EFV) ● Saquinavir (SQV) ● Etravirine (ETR) ● Tipranavir (TPV) ● Nevirapine (NVP) ●Rilpivirine (RPV) Fusion Inhibitor ● Enfuvirtide (ENF, T-20) CCR 5 Antagonist ● Maraviroc (MVC) Integrase Inhibitor ● Raltegravir (RAL) ●Elvitagravir (EVG)
NRTI 62 Current ARV Medications ● Abacavir (ABC) ● Didanosine (dd. I) ● Emtricitabine (FTC) PI ● Lamivudine (3 TC) ● Atazanavir (ATV) ● Darunavir (DRV) ● Stavudine (d 4 T) ● Fosamprenavir (FPV) ● Tenofovir (TDF) ● Indinavir (IDV) ● Zidovudine (AZT, ZDV) ● Lopinavir (LPV) NNRTI ● Nelfinavir (NFV) ● Delavirdine (DLV) ● Ritonavir (RTV) ● Efavirenz (EFV) ● Saquinavir (SQV) ● Etravirine (ETR) ● Tipranavir (TPV) ● Nevirapine (NVP) ●Rilpivirine (RPV) Fusion Inhibitor ● Enfuvirtide (ENF, T-20) CCR 5 Antagonist ● Maraviroc (MVC) Integrase Inhibitor ● Raltegravir (RAL) ●Elvitagravir (EVG)
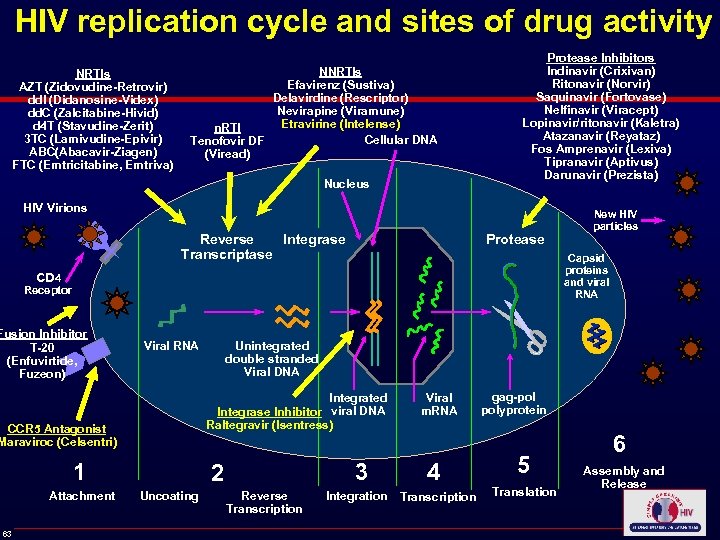 HIV replication cycle and sites of drug activity NRTIs AZT (Zidovudine-Retrovir) dd. I (Didanosine-Videx) dd. C (Zalcitabine-Hivid) d 4 T (Stavudine-Zerit) 3 TC (Lamivudine-Epivir) ABC(Abacavir-Ziagen) FTC (Emtricitabine, Emtriva) NNRTIs Efavirenz (Sustiva) Delavirdine (Rescriptor) Nevirapine (Viramune) Etravirine (Intelense) n. RTI Cellular DNA Tenofovir DF (Viread) Nucleus Protease Inhibitors Indinavir (Crixivan) Ritonavir (Norvir) Saquinavir (Fortovase) Nelfinavir (Viracept) Lopinavir/ritonavir (Kaletra) Atazanavir (Reyataz) Fos Amprenavir (Lexiva) Tipranavir (Aptivus) Darunavir (Prezista) HIV Virions Reverse Integrase Transcriptase Protease Capsid proteins and viral RNA CD 4 Receptor Fusion Inhibitor T-20 (Enfuvirtide, Fuzeon) Viral RNA 1 Attachment 63 Unintegrated double stranded Viral DNA Integrated Integrase Inhibitor viral DNA Raltegravir (Isentress) CCR 5 Antagonist Maraviroc (Celsentri) 3 2 Uncoating New HIV particles Reverse Transcription Integration Viral m. RNA 4 Transcription gag-pol polyprotein 5 Translation 6 Assembly and Release
HIV replication cycle and sites of drug activity NRTIs AZT (Zidovudine-Retrovir) dd. I (Didanosine-Videx) dd. C (Zalcitabine-Hivid) d 4 T (Stavudine-Zerit) 3 TC (Lamivudine-Epivir) ABC(Abacavir-Ziagen) FTC (Emtricitabine, Emtriva) NNRTIs Efavirenz (Sustiva) Delavirdine (Rescriptor) Nevirapine (Viramune) Etravirine (Intelense) n. RTI Cellular DNA Tenofovir DF (Viread) Nucleus Protease Inhibitors Indinavir (Crixivan) Ritonavir (Norvir) Saquinavir (Fortovase) Nelfinavir (Viracept) Lopinavir/ritonavir (Kaletra) Atazanavir (Reyataz) Fos Amprenavir (Lexiva) Tipranavir (Aptivus) Darunavir (Prezista) HIV Virions Reverse Integrase Transcriptase Protease Capsid proteins and viral RNA CD 4 Receptor Fusion Inhibitor T-20 (Enfuvirtide, Fuzeon) Viral RNA 1 Attachment 63 Unintegrated double stranded Viral DNA Integrated Integrase Inhibitor viral DNA Raltegravir (Isentress) CCR 5 Antagonist Maraviroc (Celsentri) 3 2 Uncoating New HIV particles Reverse Transcription Integration Viral m. RNA 4 Transcription gag-pol polyprotein 5 Translation 6 Assembly and Release
 What Is HAART? • • • HAART stands for Highly Active Antiretroviral Therapy HAART combines drugs from different classes, slowing HIV replication down at different stages HAART is also called combination therapy, a “cocktail, ” or a “regimen” Examples of HAART regimens: NNRTI + or PI Reference: AIDSinfo: A Service of the U. S. Department of Health and Human Services. HIV and its treatment: what you should know. February 2008. Available at: http: //www. aidsinfo. nih. gov/contentfiles/HIVand. Its. Treatment_cbrochure_en. pdf. 64 64
What Is HAART? • • • HAART stands for Highly Active Antiretroviral Therapy HAART combines drugs from different classes, slowing HIV replication down at different stages HAART is also called combination therapy, a “cocktail, ” or a “regimen” Examples of HAART regimens: NNRTI + or PI Reference: AIDSinfo: A Service of the U. S. Department of Health and Human Services. HIV and its treatment: what you should know. February 2008. Available at: http: //www. aidsinfo. nih. gov/contentfiles/HIVand. Its. Treatment_cbrochure_en. pdf. 64 64
 Therapy is Easier, More Potent, and Less Toxic in Single-Tablet Regimens 65
Therapy is Easier, More Potent, and Less Toxic in Single-Tablet Regimens 65
 DHHS Guidelines: Preferred Regimens NNRTI PI Efavirenz 1/emtricitabine 2/tenofovir DF 3 Atazanavir 4 + ritonavir + emtricitabine 2/tenofovir DF 3 Darunavir + ritonavir (qd) + emtricitabine 2/tenofovir DF 3 INSTI Raltegravir + emtricitabine 2/tenofovir DF 3 Pregnant Lopinavir/r bid + zidovudine/lamivudine 2 women INSTI: Integrase strand transfer inhibitors. 1 Efavirenz should not be used during the first trimester of pregnancy or in women trying to conceive or not using effective and consistent contraception. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Tenofovir DF should be used with caution in patients with renal insufficiency. 4 Atazanavir + RTV should not be used in patients who require >20 mg omeprazole equivalent/day. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 66
DHHS Guidelines: Preferred Regimens NNRTI PI Efavirenz 1/emtricitabine 2/tenofovir DF 3 Atazanavir 4 + ritonavir + emtricitabine 2/tenofovir DF 3 Darunavir + ritonavir (qd) + emtricitabine 2/tenofovir DF 3 INSTI Raltegravir + emtricitabine 2/tenofovir DF 3 Pregnant Lopinavir/r bid + zidovudine/lamivudine 2 women INSTI: Integrase strand transfer inhibitors. 1 Efavirenz should not be used during the first trimester of pregnancy or in women trying to conceive or not using effective and consistent contraception. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Tenofovir DF should be used with caution in patients with renal insufficiency. 4 Atazanavir + RTV should not be used in patients who require >20 mg omeprazole equivalent/day. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 66
 DHHS Guidelines: Alternative Regimens NNRTI Efavirenz + (abacavir 1 or zidovudine)/lamivudine 2 Rilpivirine 3/emtricitabine 2/tenofovir DF Rilpivirine 3 + abacavir/lamivudine 2 PI Atazanavir + ritonavir + abacavir/lamivudine 2 Darunavir + ritonavir + abacavir/lamivudine 2 Fosamprenavir + ritonavir (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF Lopinavir/r 4 (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF INSTI Raltegravir + abacavir/lamivudine 2 Elvitegravir/cobicistat/emtricitabine/tenofovir DF 5 1 Abacavir should not be used in patients who test positive for HLA-B*5701. Use abacavir with caution in patients with high risk of cardiovascular disease or pretreatment HIV RNA >100, 000 copies/m. L. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Use rilpivirine with caution in patients with pretreatment HIV RNA >100, 000 copies/m. L. 4 Once-daily lopinavir/r is not recommended in pregnant women. 5 Patients with creatinine clearance >70 m. L/min. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 67
DHHS Guidelines: Alternative Regimens NNRTI Efavirenz + (abacavir 1 or zidovudine)/lamivudine 2 Rilpivirine 3/emtricitabine 2/tenofovir DF Rilpivirine 3 + abacavir/lamivudine 2 PI Atazanavir + ritonavir + abacavir/lamivudine 2 Darunavir + ritonavir + abacavir/lamivudine 2 Fosamprenavir + ritonavir (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF Lopinavir/r 4 (qd or bid) + abacavir/lamivudine 2 or emtricitabine 2/tenofovir DF INSTI Raltegravir + abacavir/lamivudine 2 Elvitegravir/cobicistat/emtricitabine/tenofovir DF 5 1 Abacavir should not be used in patients who test positive for HLA-B*5701. Use abacavir with caution in patients with high risk of cardiovascular disease or pretreatment HIV RNA >100, 000 copies/m. L. 2 Lamivudine may substitute for emtricitabine or visa versa. 3 Use rilpivirine with caution in patients with pretreatment HIV RNA >100, 000 copies/m. L. 4 Once-daily lopinavir/r is not recommended in pregnant women. 5 Patients with creatinine clearance >70 m. L/min. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 67
 IAS-USA Guidelines: Preferred Regimens NNRTI PI Efavirenz/emtricitabine/tenofovir DF Efavirenz + abacavir/lamivudine 1 -3 Atazanavir + ritonavir + emtricitabine/tenofovir DF Atazanavir + ritonavir + abacavir/lamivudine 3, 4 Darunavir + ritonavir (qd) + emtricitabine/tenofovir DF INSTI Raltegravir + emtricitabine/tenofovir DF INSTI: Integrase strand transfer inhibitors. 1 HLA-B*5701 -negative patients with baseline plasma HIV RNA <100, 000 copies/m. L. 2 Avoiding the use of abacavir or lopinavir/ritonavir might be considered for patients with or at high risk of cardiovascular disease. 3 HLA-B*5701 screening is recommended before abacavir administration to reduce the risk of hypersensitivity reaction. 4 Patients with baseline plasma HIV RNA <100, 000 copies/m. L. Thompson MA, et al. JAMA. 2012; 308: 387 -402. 68
IAS-USA Guidelines: Preferred Regimens NNRTI PI Efavirenz/emtricitabine/tenofovir DF Efavirenz + abacavir/lamivudine 1 -3 Atazanavir + ritonavir + emtricitabine/tenofovir DF Atazanavir + ritonavir + abacavir/lamivudine 3, 4 Darunavir + ritonavir (qd) + emtricitabine/tenofovir DF INSTI Raltegravir + emtricitabine/tenofovir DF INSTI: Integrase strand transfer inhibitors. 1 HLA-B*5701 -negative patients with baseline plasma HIV RNA <100, 000 copies/m. L. 2 Avoiding the use of abacavir or lopinavir/ritonavir might be considered for patients with or at high risk of cardiovascular disease. 3 HLA-B*5701 screening is recommended before abacavir administration to reduce the risk of hypersensitivity reaction. 4 Patients with baseline plasma HIV RNA <100, 000 copies/m. L. Thompson MA, et al. JAMA. 2012; 308: 387 -402. 68
 IAS-USA Guidelines: Alternative Regimens 1 NNRTI Nevirapine + emtricitabine/tenofovir or abacavir/zidovudine 2 Rilpivirine/emtricitabine/tenofovir DF Rilpivirine + abacavir/lamivudine 2 PI Darunavir + ritonavir + abacavir/lamivudine 2 Lopinavir/r 3 (qd or bid) + abacavir/lamivudine or emtricitabine/tenofovir DF INSTI Raltegravir + abacavir/lamivudine 2 Elvitegravir/cobicistat/emtricitabine/tenofovir DF INSTI: Integrase strand transfer inhibitors. 1 Zidovudine/lamivudine is an alternative NRTI component of NNRTI-, PI/r-, and raltegravir-based regimens, but the toxicity profile of zidovudine reduces its utility. 2 HLA-B*5701 screening is recommended before abacavir administration to reduce the risk of hypersensitivity reaction. 3 Avoiding the use of abacavir or lopinavir/ritonavir might be considered for patients with or at high risk of cardiovascular disease. Thompson MA, et al. JAMA. 2012; 308: 387 -402. 69
IAS-USA Guidelines: Alternative Regimens 1 NNRTI Nevirapine + emtricitabine/tenofovir or abacavir/zidovudine 2 Rilpivirine/emtricitabine/tenofovir DF Rilpivirine + abacavir/lamivudine 2 PI Darunavir + ritonavir + abacavir/lamivudine 2 Lopinavir/r 3 (qd or bid) + abacavir/lamivudine or emtricitabine/tenofovir DF INSTI Raltegravir + abacavir/lamivudine 2 Elvitegravir/cobicistat/emtricitabine/tenofovir DF INSTI: Integrase strand transfer inhibitors. 1 Zidovudine/lamivudine is an alternative NRTI component of NNRTI-, PI/r-, and raltegravir-based regimens, but the toxicity profile of zidovudine reduces its utility. 2 HLA-B*5701 screening is recommended before abacavir administration to reduce the risk of hypersensitivity reaction. 3 Avoiding the use of abacavir or lopinavir/ritonavir might be considered for patients with or at high risk of cardiovascular disease. Thompson MA, et al. JAMA. 2012; 308: 387 -402. 69
 Importance of HIV Expertise in Clinical Care ● Multiple studies show expertise in HIV care improves - Survival Rate of hospitalizations Compliance with guidelines Adherence to medications ● DHHS panel recommendation - HIV primary care by a clinician with at least 20, and preferably 50, HIV-infected patients DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 70
Importance of HIV Expertise in Clinical Care ● Multiple studies show expertise in HIV care improves - Survival Rate of hospitalizations Compliance with guidelines Adherence to medications ● DHHS panel recommendation - HIV primary care by a clinician with at least 20, and preferably 50, HIV-infected patients DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision February 12, 2013. 70
 AIDS Education Training Centers ● Network of Training Centers - 11 Regional Centers with more than 130 local sites serving healthcare providers nationwide - 4 National Centers provide support services to the AETC network ● National Resource Center - Provides virtual library of online training resources for adaptation to meet local training needs AETC. Available at: http: //www. aids-ed. org. 71
AIDS Education Training Centers ● Network of Training Centers - 11 Regional Centers with more than 130 local sites serving healthcare providers nationwide - 4 National Centers provide support services to the AETC network ● National Resource Center - Provides virtual library of online training resources for adaptation to meet local training needs AETC. Available at: http: //www. aids-ed. org. 71
 National HIV Consultation Service (Warmline) for Healthcare Professionals ● 1 -800 -933 -3413 ● Provides information and individual case consultation ● Service is offered free of charge to healthcare professionals ● Staffed 6 am to 5 pm Pacific time National HIV/AIDS Clinicians' Consultation Center. Available at: http: //www. nccc. ucsf. edu/. Accessed January 7, 2013. 72
National HIV Consultation Service (Warmline) for Healthcare Professionals ● 1 -800 -933 -3413 ● Provides information and individual case consultation ● Service is offered free of charge to healthcare professionals ● Staffed 6 am to 5 pm Pacific time National HIV/AIDS Clinicians' Consultation Center. Available at: http: //www. nccc. ucsf. edu/. Accessed January 7, 2013. 72
 Summary ● There is a need to increase the proportion of persons who are aware of their HIV-infection status - Routine, voluntary, opt-out screening in health care settings is needed ● Rapid HIV tests - Provide clinicians with preliminary results in 15 to 30 minutes ● Patients at high-risk for HIV - Should be counseled on risk reduction strategies ● Treatment and referral services for those who test HIV positive will benefit patients and society - Prolong life of HIV-infected individuals - Likely reduce risk of transmission 73
Summary ● There is a need to increase the proportion of persons who are aware of their HIV-infection status - Routine, voluntary, opt-out screening in health care settings is needed ● Rapid HIV tests - Provide clinicians with preliminary results in 15 to 30 minutes ● Patients at high-risk for HIV - Should be counseled on risk reduction strategies ● Treatment and referral services for those who test HIV positive will benefit patients and society - Prolong life of HIV-infected individuals - Likely reduce risk of transmission 73
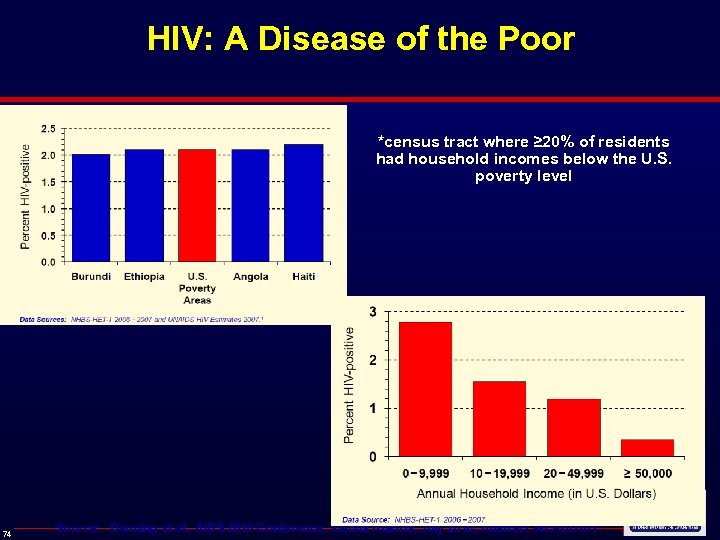 HIV: A Disease of the Poor *census tract where ≥ 20% of residents had household incomes below the U. S. poverty level * 74 Source: Denning at el. , AIDS 2010 Conference, Vienna Austria, July 2010, Abstract WEPDD 101
HIV: A Disease of the Poor *census tract where ≥ 20% of residents had household incomes below the U. S. poverty level * 74 Source: Denning at el. , AIDS 2010 Conference, Vienna Austria, July 2010, Abstract WEPDD 101
 STIGMA Church sign in Birmingham, Alabama 75
STIGMA Church sign in Birmingham, Alabama 75
 Additional Resources ● CDC - http: //www. cdc. gov/hiv/topics/testing/healthcare/index. htm ● HIVInsite - http: //hivinsite. ucsf. edu/ ● AIDS Education Training Centers - www. aids-ed. org 76
Additional Resources ● CDC - http: //www. cdc. gov/hiv/topics/testing/healthcare/index. htm ● HIVInsite - http: //hivinsite. ucsf. edu/ ● AIDS Education Training Centers - www. aids-ed. org 76
 New Electronic Evaluation Process ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 77
New Electronic Evaluation Process ● You will receive an electronic evaluation to the email address provided within 1 business day ● Reminder email communications will be sent up to 5 days post lecture until the evaluation is completed ● Completion Is Required for CME/CNE/CPE credit and future attendance ● Incomplete evaluations will preclude attendees from receiving their CME/CNE/CPE certificate & future communications about lectures in your area 77
 Outcomes Measurement Reminder ● We are required to assess “changes in learners’ competence, performance or patient outcomes achieved as a result of their participation in a CME/CNE/CPE sponsored educational activity” ● As a result of this requirement you will receive a short survey via email 8 to 12 weeks after completing this course - We consider the survey to be an additional component of your overall participation in this educational activity and would urge you to reflect on what you learned in the activity and then complete this survey 78
Outcomes Measurement Reminder ● We are required to assess “changes in learners’ competence, performance or patient outcomes achieved as a result of their participation in a CME/CNE/CPE sponsored educational activity” ● As a result of this requirement you will receive a short survey via email 8 to 12 weeks after completing this course - We consider the survey to be an additional component of your overall participation in this educational activity and would urge you to reflect on what you learned in the activity and then complete this survey 78


