68c552380af4196b11ee1a9c5c3df56e.ppt
- Количество слайдов: 24
 U. S. Department of Health and Human Services Closed-chest Direct Transthoracic Access Ports to the Heart and Closure Without Surgery National Institutes of Health Robert J. Lederman, MD Cardiovascular and Pulmonary Branch Division of Intramural Research National Heart, Lung, and Blood Institute National Institutes of Health Bethesda, MD, USA lederman@nih. gov National Heart, Lung, and Blood Institute Cardiovascular Intervention Program at NHLBI These are non-clinical or investigational procedures. Lederman
U. S. Department of Health and Human Services Closed-chest Direct Transthoracic Access Ports to the Heart and Closure Without Surgery National Institutes of Health Robert J. Lederman, MD Cardiovascular and Pulmonary Branch Division of Intramural Research National Heart, Lung, and Blood Institute National Institutes of Health Bethesda, MD, USA lederman@nih. gov National Heart, Lung, and Blood Institute Cardiovascular Intervention Program at NHLBI These are non-clinical or investigational procedures. Lederman
 Disclosures n NIH and Siemens have a collaborative research and development agreement on cardiovascular interventional MRI n NIH and Neat-Stitch have a collaborative research and development agreement on suture closure of cardiac access ports n AGA Medical provided implants Lederman
Disclosures n NIH and Siemens have a collaborative research and development agreement on cardiovascular interventional MRI n NIH and Neat-Stitch have a collaborative research and development agreement on suture closure of cardiac access ports n AGA Medical provided implants Lederman
![Acknowledgements Collaborators – Michael Slack, MD, [CNMC] – Renu Virmani, MD [CVPath] Alumni Clinical Acknowledgements Collaborators – Michael Slack, MD, [CNMC] – Renu Virmani, MD [CVPath] Alumni Clinical](https://present5.com/presentation/68c552380af4196b11ee1a9c5c3df56e/image-3.jpg) Acknowledgements Collaborators – Michael Slack, MD, [CNMC] – Renu Virmani, MD [CVPath] Alumni Clinical – – – – Amish Raval, MD [Univ. Wisconsin] June-Hong Kim, MD [Pusan U, Korea] Cengizhan Ozturk, MD/Ph. D [Bogazici, Turkey] Alexander J. Dick, MD [Ottowa Heart] Ronnier J. Aviles, MD [Seattle, WA] Ranil De. Silva, FRCP [Brompton, London] James Telep, MD [Fairfax, VA] Abdalla Elagha, MD [U. Cairo] Alumni Scientists – – – – Elliot Mc. Veigh, Ph. D [Johns Hopkins U] Michael Guttman, MSc [Surgi-Vision, Inc] Parag Karmarkar, MS [Surgi-Vision, Inc] Richard Thompson, Ph. D [U Edmonton] Dana Peters, Ph. D [Harvard BIDMC] Luis Gutierrez, Ph. D [Philips Research] Smita Sampath, Ph. D [Yale U] Vincent Wu, BS [NYU SOM] Lederman Clinicians – – – Israel M. Barbash, MD Kanishka Ratnayaka, MD [Children’s, DC] Victor J. Wright, BS Bill Schenke, BS Annette M. Stine, RN Laurie Grant, RN Scientists & Engineers – – – – Ozgur Kocaturk, Ph. D Anthony Z Faranesh, Ph. D Christina E. Saikus, Ph. D MSTP [Emory/GA tech] Michael S. Hansen, Ph. D J. Andrew Derbyshire, Ph. D Jamie A. Bell, BS Merdim Sonmez, MS
Acknowledgements Collaborators – Michael Slack, MD, [CNMC] – Renu Virmani, MD [CVPath] Alumni Clinical – – – – Amish Raval, MD [Univ. Wisconsin] June-Hong Kim, MD [Pusan U, Korea] Cengizhan Ozturk, MD/Ph. D [Bogazici, Turkey] Alexander J. Dick, MD [Ottowa Heart] Ronnier J. Aviles, MD [Seattle, WA] Ranil De. Silva, FRCP [Brompton, London] James Telep, MD [Fairfax, VA] Abdalla Elagha, MD [U. Cairo] Alumni Scientists – – – – Elliot Mc. Veigh, Ph. D [Johns Hopkins U] Michael Guttman, MSc [Surgi-Vision, Inc] Parag Karmarkar, MS [Surgi-Vision, Inc] Richard Thompson, Ph. D [U Edmonton] Dana Peters, Ph. D [Harvard BIDMC] Luis Gutierrez, Ph. D [Philips Research] Smita Sampath, Ph. D [Yale U] Vincent Wu, BS [NYU SOM] Lederman Clinicians – – – Israel M. Barbash, MD Kanishka Ratnayaka, MD [Children’s, DC] Victor J. Wright, BS Bill Schenke, BS Annette M. Stine, RN Laurie Grant, RN Scientists & Engineers – – – – Ozgur Kocaturk, Ph. D Anthony Z Faranesh, Ph. D Christina E. Saikus, Ph. D MSTP [Emory/GA tech] Michael S. Hansen, Ph. D J. Andrew Derbyshire, Ph. D Jamie A. Bell, BS Merdim Sonmez, MS
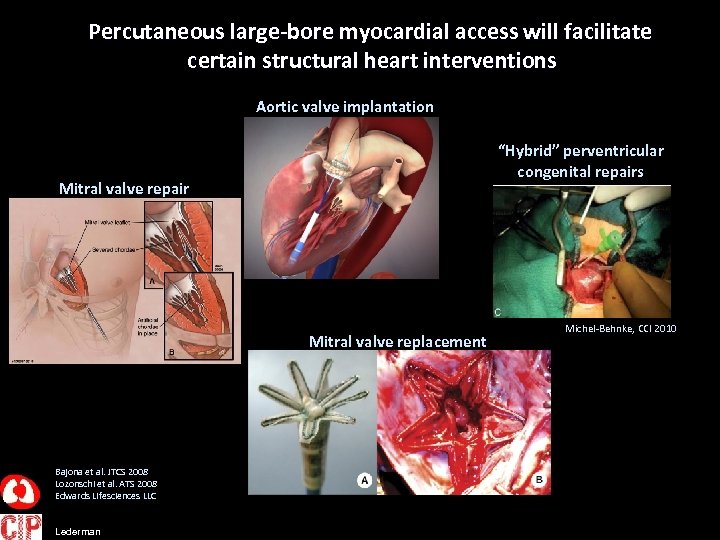 Percutaneous large-bore myocardial access will facilitate certain structural heart interventions Aortic valve implantation “Hybrid” perventricular congenital repairs Mitral valve repair Mitral valve replacement Bajona et al. JTCS 2008 Lozonschi et al. ATS 2008 Edwards Lifesciences LLC Lederman Michel-Behnke, CCI 2010
Percutaneous large-bore myocardial access will facilitate certain structural heart interventions Aortic valve implantation “Hybrid” perventricular congenital repairs Mitral valve repair Mitral valve replacement Bajona et al. JTCS 2008 Lozonschi et al. ATS 2008 Edwards Lifesciences LLC Lederman Michel-Behnke, CCI 2010
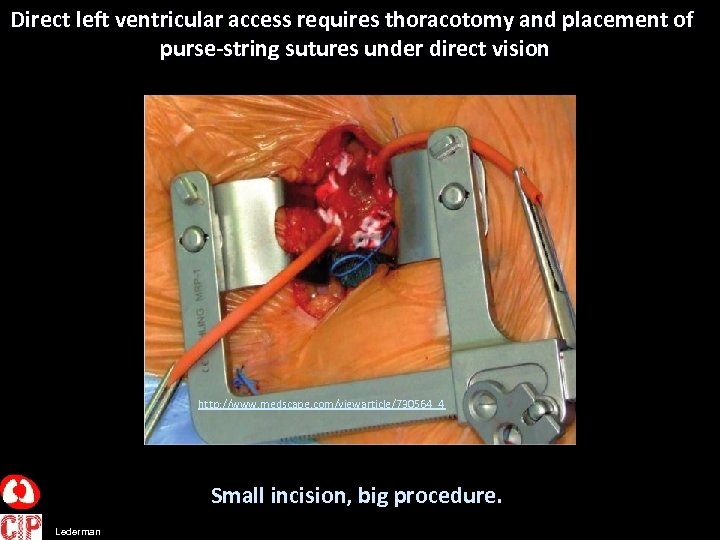 Direct left ventricular access requires thoracotomy and placement of purse-string sutures under direct vision http: //www. medscape. com/viewarticle/730564_4 Small incision, big procedure. Lederman
Direct left ventricular access requires thoracotomy and placement of purse-string sutures under direct vision http: //www. medscape. com/viewarticle/730564_4 Small incision, big procedure. Lederman
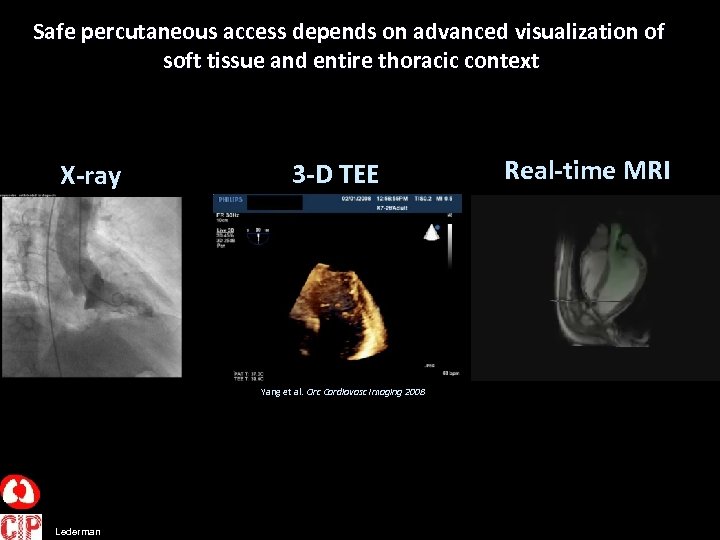 Safe percutaneous access depends on advanced visualization of soft tissue and entire thoracic context X-ray 3 -D TEE Yang et al. Circ Cardiovasc Imaging 2008 Lederman Real-time MRI
Safe percutaneous access depends on advanced visualization of soft tissue and entire thoracic context X-ray 3 -D TEE Yang et al. Circ Cardiovasc Imaging 2008 Lederman Real-time MRI
 Overview of closed-chest direct cardiac access & closure 1. Access to target chamber a. b. c. d. Chest wall Interposed tissue (lung) Myocardial entry Target structure 2. Therapeutic procedure 3. Closure a. b. c. d. Lederman Simple withdrawal: no closure Absorbable implant Suture-device Permanent implant n Intact pericardium? – “Permissive” tamponade to separate pericardial layers
Overview of closed-chest direct cardiac access & closure 1. Access to target chamber a. b. c. d. Chest wall Interposed tissue (lung) Myocardial entry Target structure 2. Therapeutic procedure 3. Closure a. b. c. d. Lederman Simple withdrawal: no closure Absorbable implant Suture-device Permanent implant n Intact pericardium? – “Permissive” tamponade to separate pericardial layers
 Step 1 ACCESS Lederman
Step 1 ACCESS Lederman
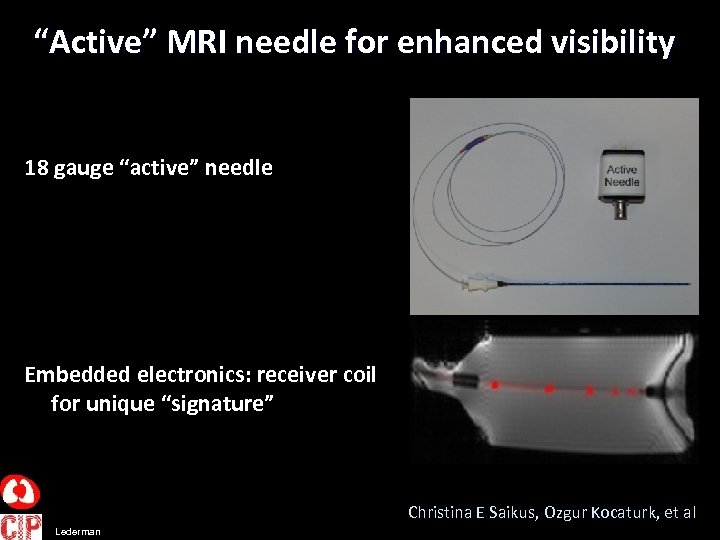 “Active” MRI needle for enhanced visibility 18 gauge “active” needle Embedded electronics: receiver coil for unique “signature” Christina E Saikus, Ozgur Kocaturk, et al Lederman
“Active” MRI needle for enhanced visibility 18 gauge “active” needle Embedded electronics: receiver coil for unique “signature” Christina E Saikus, Ozgur Kocaturk, et al Lederman
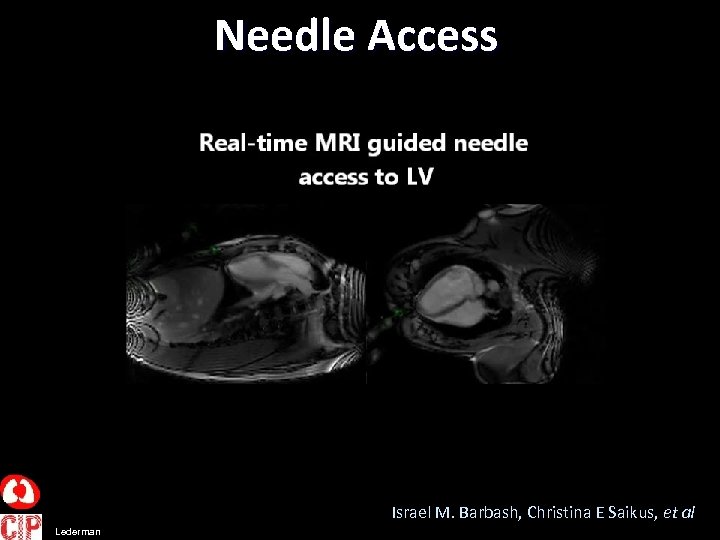 Needle Access Israel M. Barbash, Christina E Saikus, et al Lederman
Needle Access Israel M. Barbash, Christina E Saikus, et al Lederman
 Step 2 THERAPEUTIC PROCEDURE Lederman
Step 2 THERAPEUTIC PROCEDURE Lederman
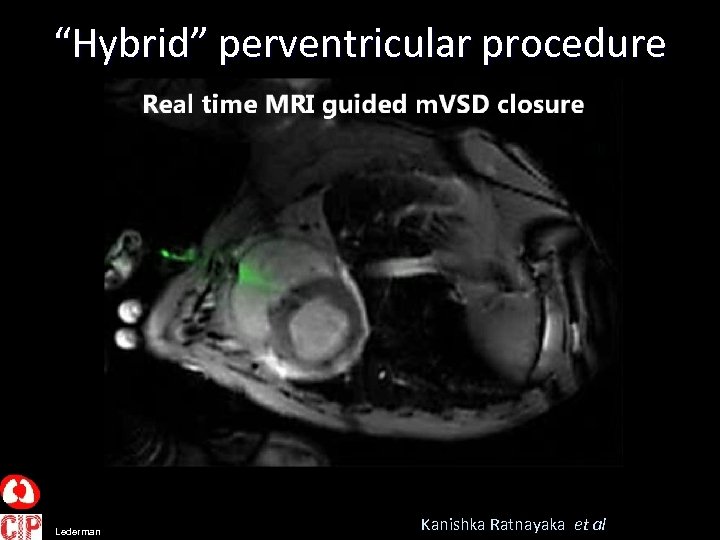 “Hybrid” perventricular procedure Lederman Kanishka Ratnayaka et al
“Hybrid” perventricular procedure Lederman Kanishka Ratnayaka et al
 Step 3 PORT CLOSURE Lederman
Step 3 PORT CLOSURE Lederman
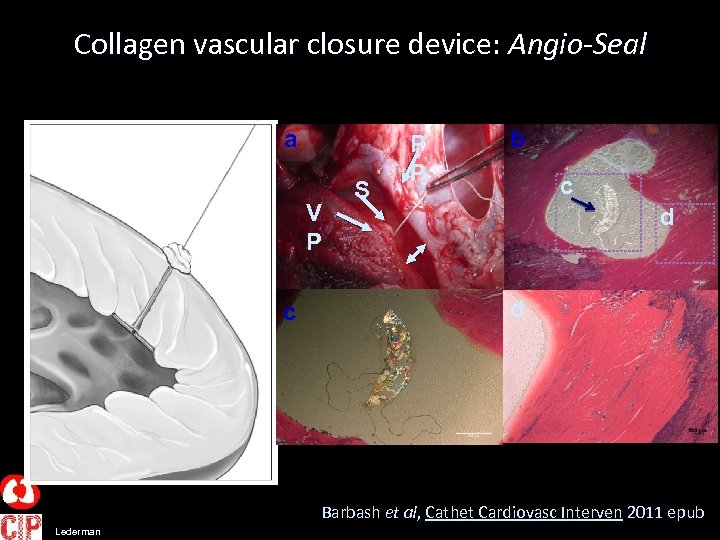 Collagen vascular closure device: Angio-Seal a V P c S P P b c d d Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
Collagen vascular closure device: Angio-Seal a V P c S P P b c d d Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
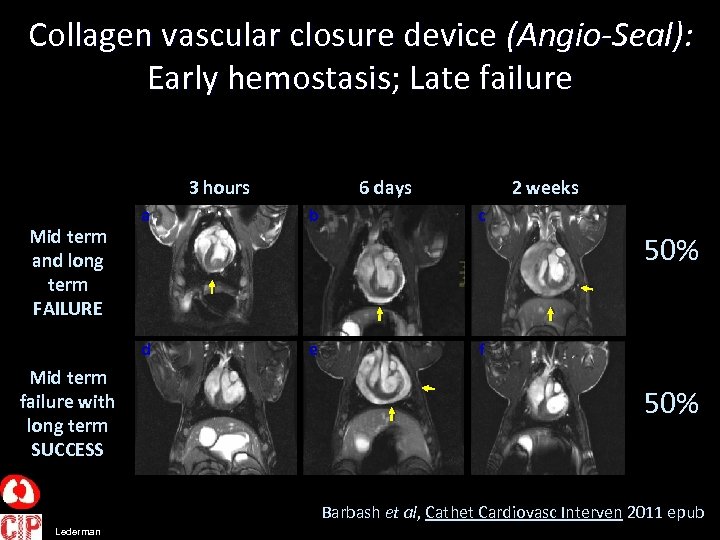 Collagen vascular closure device (Angio-Seal): Early hemostasis; Late failure 3 hours Mid term and long term FAILURE a b 2 weeks c k d Mid term failure with long term SUCCESS 6 days e 50% f 50% Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
Collagen vascular closure device (Angio-Seal): Early hemostasis; Late failure 3 hours Mid term and long term FAILURE a b 2 weeks c k d Mid term failure with long term SUCCESS 6 days e 50% f 50% Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
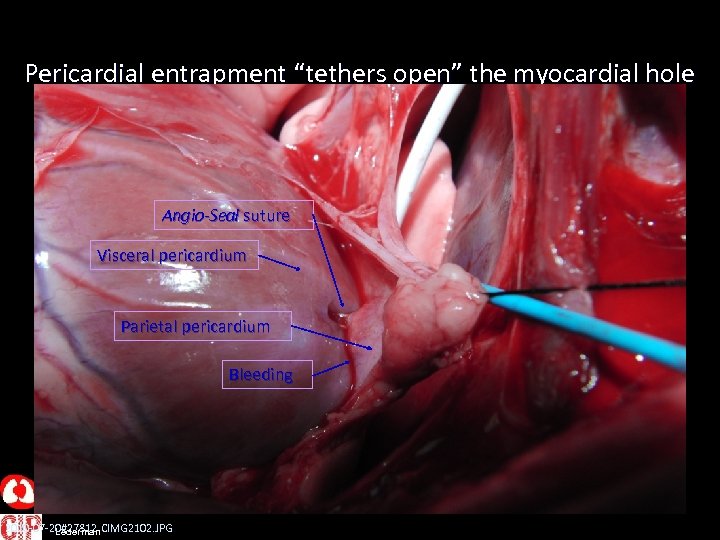 Pericardial entrapment “tethers open” the myocardial hole Angio-Seal suture Visceral pericardium Parietal pericardium Bleeding 2010 -07 -20#27812 CIMG 2102. JPG Lederman
Pericardial entrapment “tethers open” the myocardial hole Angio-Seal suture Visceral pericardium Parietal pericardium Bleeding 2010 -07 -20#27812 CIMG 2102. JPG Lederman
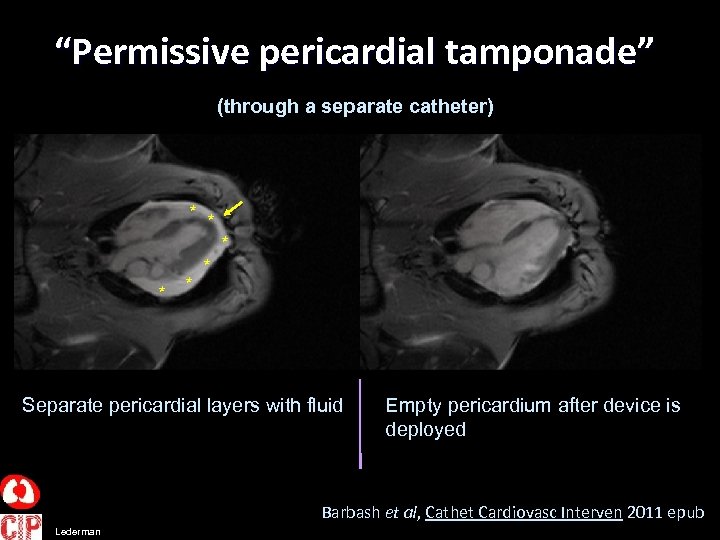 “Permissive pericardial tamponade” (through a separate catheter) * * * Separate pericardial layers with fluid Empty pericardium after device is deployed Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
“Permissive pericardial tamponade” (through a separate catheter) * * * Separate pericardial layers with fluid Empty pericardium after device is deployed Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
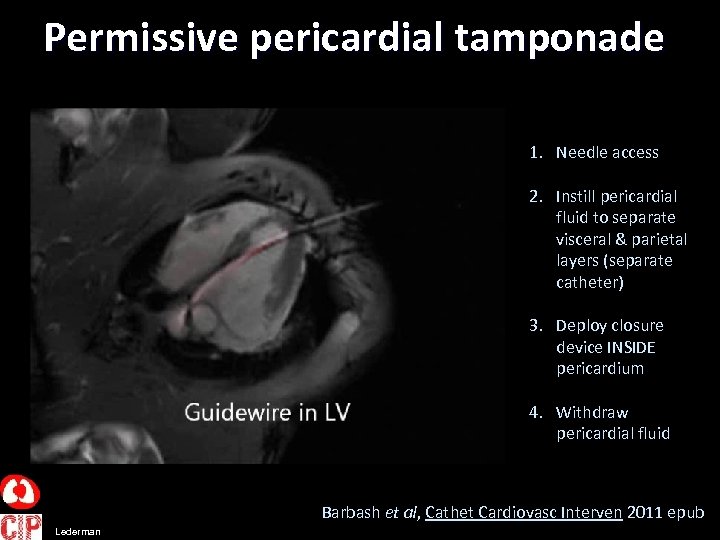 Permissive pericardial tamponade 1. Needle access 2. Instill pericardial fluid to separate visceral & parietal layers (separate catheter) 3. Deploy closure device INSIDE pericardium 4. Withdraw pericardial fluid Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
Permissive pericardial tamponade 1. Needle access 2. Instill pericardial fluid to separate visceral & parietal layers (separate catheter) 3. Deploy closure device INSIDE pericardium 4. Withdraw pericardial fluid Barbash et al, Cathet Cardiovasc Interven 2011 epub Lederman
 Nitinol Implant; “Active” delivery cable Stainless steel n Titanium Amplatzer muscular VSD occluder: nitinol mesh – Stainless steel screw replaced with titanium. n “Active” delivery cable incorporating loopless antenna. n Antenna tuning to maximize signal at occluder fully deployed state. Jamie A. Bell, Christina E. Saikus, Ozgur Kocaturk Lederman
Nitinol Implant; “Active” delivery cable Stainless steel n Titanium Amplatzer muscular VSD occluder: nitinol mesh – Stainless steel screw replaced with titanium. n “Active” delivery cable incorporating loopless antenna. n Antenna tuning to maximize signal at occluder fully deployed state. Jamie A. Bell, Christina E. Saikus, Ozgur Kocaturk Lederman
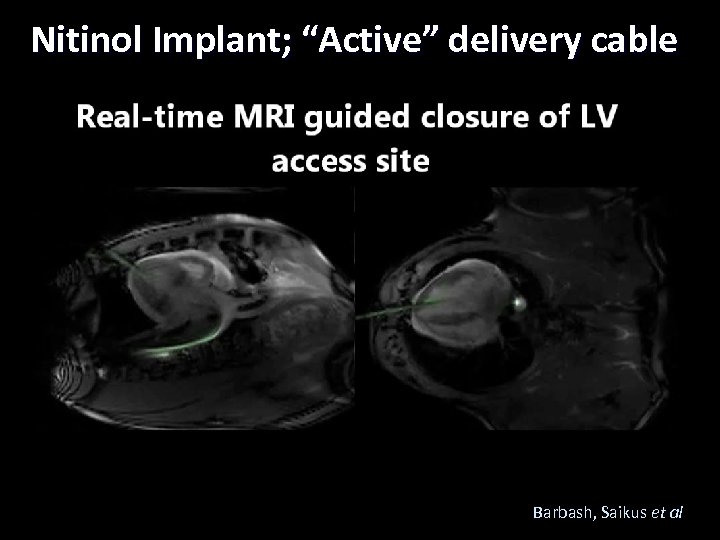 Nitinol Implant; “Active” delivery cable Barbash, Saikus et al Lederman
Nitinol Implant; “Active” delivery cable Barbash, Saikus et al Lederman
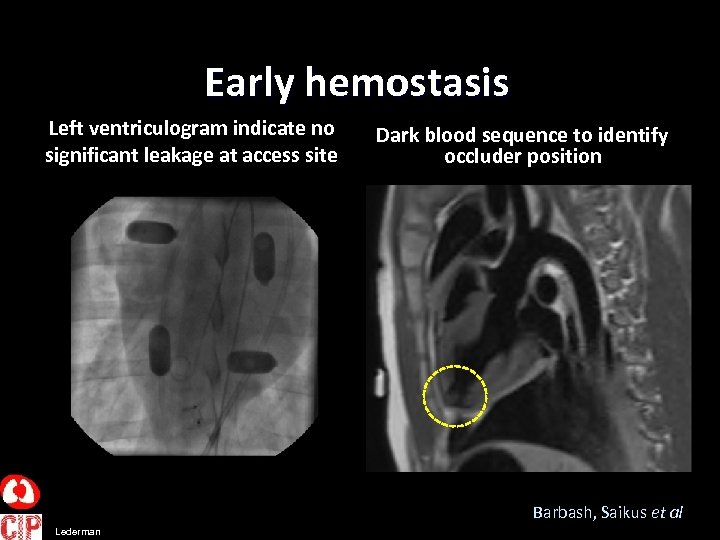 Early hemostasis Left ventriculogram indicate no significant leakage at access site Dark blood sequence to identify occluder position Barbash, Saikus et al Lederman
Early hemostasis Left ventriculogram indicate no significant leakage at access site Dark blood sequence to identify occluder position Barbash, Saikus et al Lederman
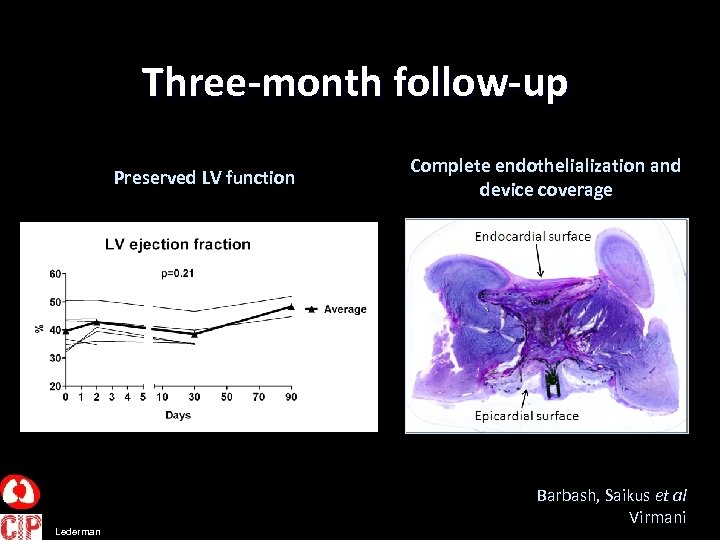 Three-month follow-up Preserved LV function Lederman Complete endothelialization and device coverage Barbash, Saikus et al Virmani
Three-month follow-up Preserved LV function Lederman Complete endothelialization and device coverage Barbash, Saikus et al Virmani
 Other thoughts Performance requirement n n Acceptable failure rate? Surgical rescue? Simple withdrawal / no device • Case series in post-operative pericardium • Intact pericardium: unattractive Tissue characteristics n n n Infarction Hypertrophy Heterogeneity Subvalvar apparatus • Avoid entrapment Procedure “security” True apex vs para-apical • Intracameral retention sheaths • Apical danger zone: mechanical vulnerability • Apical entrapment/distortion of implants Drains Lederman • Overnight drains • Mid-term effusions?
Other thoughts Performance requirement n n Acceptable failure rate? Surgical rescue? Simple withdrawal / no device • Case series in post-operative pericardium • Intact pericardium: unattractive Tissue characteristics n n n Infarction Hypertrophy Heterogeneity Subvalvar apparatus • Avoid entrapment Procedure “security” True apex vs para-apical • Intracameral retention sheaths • Apical danger zone: mechanical vulnerability • Apical entrapment/distortion of implants Drains Lederman • Overnight drains • Mid-term effusions?
 Conclusions: Myocardial Port Access & Closure n Closed-chest access ports are realistic development targets to introduce large devices into the heart n Real time MRI presents the entire thoracic context to the operator, visualizing targets and complications. n If the pericardium is intact, its walls should be separated; pericardial entrapment causes bleeding lederman@nih. gov Lederman
Conclusions: Myocardial Port Access & Closure n Closed-chest access ports are realistic development targets to introduce large devices into the heart n Real time MRI presents the entire thoracic context to the operator, visualizing targets and complications. n If the pericardium is intact, its walls should be separated; pericardial entrapment causes bleeding lederman@nih. gov Lederman


