db3413b3575bb1ee348380859497ca76.ppt
- Количество слайдов: 25
 Transcriptome and analysis of gene transcription
Transcriptome and analysis of gene transcription
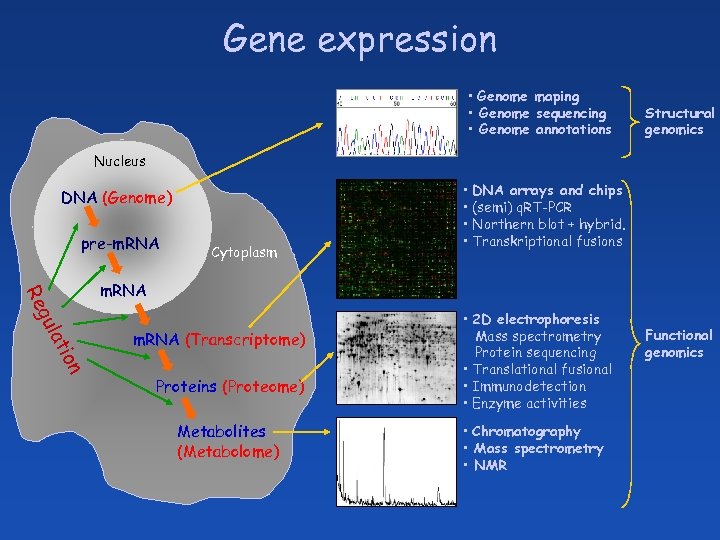 Gene expression • Genome maping • Genome sequencing • Genome annotations Structural genomics Nucleus DNA (Genome) pre-m. RNA Cytoplasm • DNA arrays and chips • (semi) q. RT-PCR • Northern blot + hybrid. • Transkriptional fusions a gul Re m. RNA n tio m. RNA (Transcriptome) Proteins (Proteome) Metabolites (Metabolome) • 2 D electrophoresis Mass spectrometry Protein sequencing • Translational fusional • Immunodetection • Enzyme activities • Chromatography • Mass spectrometry • NMR Functional genomics
Gene expression • Genome maping • Genome sequencing • Genome annotations Structural genomics Nucleus DNA (Genome) pre-m. RNA Cytoplasm • DNA arrays and chips • (semi) q. RT-PCR • Northern blot + hybrid. • Transkriptional fusions a gul Re m. RNA n tio m. RNA (Transcriptome) Proteins (Proteome) Metabolites (Metabolome) • 2 D electrophoresis Mass spectrometry Protein sequencing • Translational fusional • Immunodetection • Enzyme activities • Chromatography • Mass spectrometry • NMR Functional genomics
 Transcriptome - set of all m. RNAs present in certain cell, tissue, organ, … - m. RNA level results from intensity of transcription and m. RNA stability Transcriptomics – expression analysis of populations of genes - analysis of differences in expression of gene populations (under different conditions, treatments, developmental stages)
Transcriptome - set of all m. RNAs present in certain cell, tissue, organ, … - m. RNA level results from intensity of transcription and m. RNA stability Transcriptomics – expression analysis of populations of genes - analysis of differences in expression of gene populations (under different conditions, treatments, developmental stages)
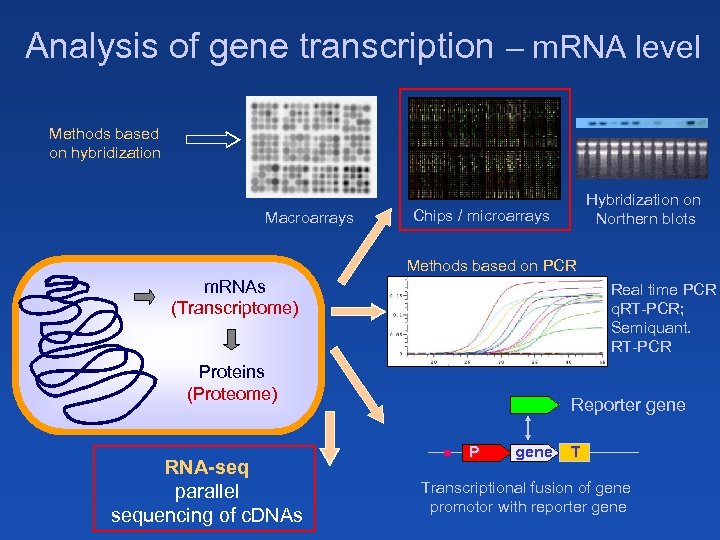 Analysis of gene transcription – m. RNA level Methods based on hybridization Macroarrays Hybridization on Northern blots Chips / microarrays Methods based on PCR m. RNAs (Transcriptome) Real time PCR q. RT-PCR; Semiquant. RT-PCR DNA (Genome) Proteins (Proteome) RNA-seq parallel sequencing of c. DNAs Reporter gene P gene T Transcriptional fusion of gene promotor with reporter gene
Analysis of gene transcription – m. RNA level Methods based on hybridization Macroarrays Hybridization on Northern blots Chips / microarrays Methods based on PCR m. RNAs (Transcriptome) Real time PCR q. RT-PCR; Semiquant. RT-PCR DNA (Genome) Proteins (Proteome) RNA-seq parallel sequencing of c. DNAs Reporter gene P gene T Transcriptional fusion of gene promotor with reporter gene
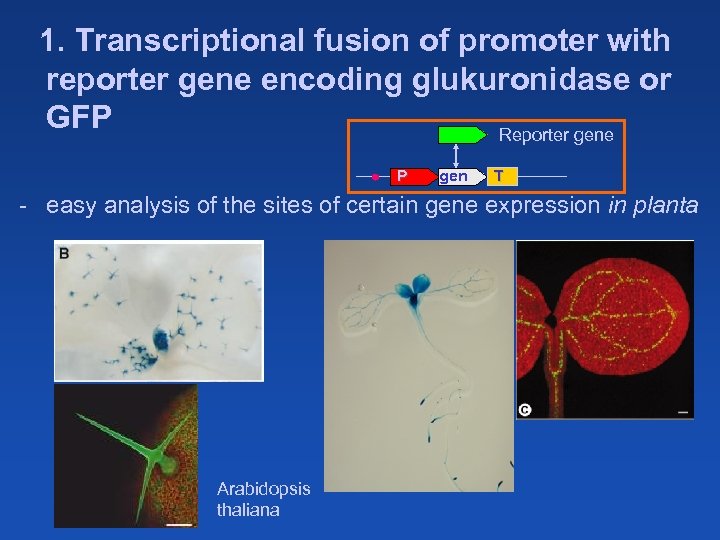 1. Transcriptional fusion of promoter with reporter gene encoding glukuronidase or GFP Reporter gene P gen T - easy analysis of the sites of certain gene expression in planta Arabidopsis thaliana
1. Transcriptional fusion of promoter with reporter gene encoding glukuronidase or GFP Reporter gene P gen T - easy analysis of the sites of certain gene expression in planta Arabidopsis thaliana
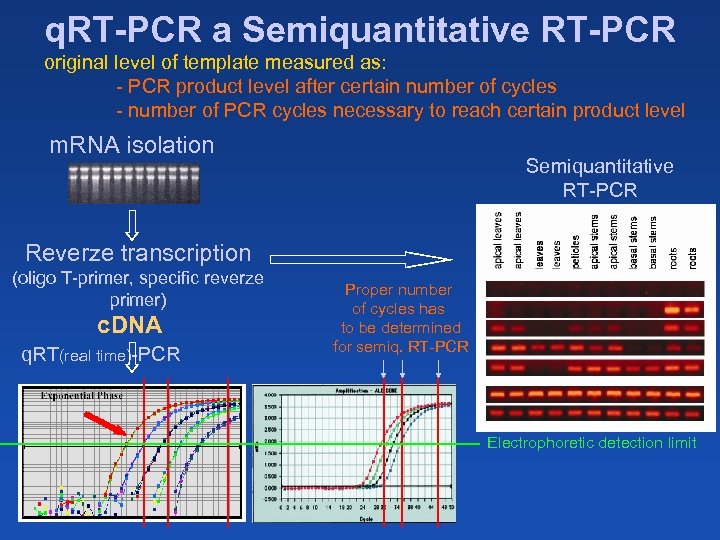 q. RT-PCR a Semiquantitative RT-PCR original level of template measured as: - PCR product level after certain number of cycles - number of PCR cycles necessary to reach certain product level m. RNA isolation Semiquantitative RT-PCR Reverze transcription (oligo T-primer, specific reverze primer) c. DNA q. RT(real time)-PCR Proper number of cycles has to be determined for semiq. RT-PCR Electrophoretic detection limit
q. RT-PCR a Semiquantitative RT-PCR original level of template measured as: - PCR product level after certain number of cycles - number of PCR cycles necessary to reach certain product level m. RNA isolation Semiquantitative RT-PCR Reverze transcription (oligo T-primer, specific reverze primer) c. DNA q. RT(real time)-PCR Proper number of cycles has to be determined for semiq. RT-PCR Electrophoretic detection limit
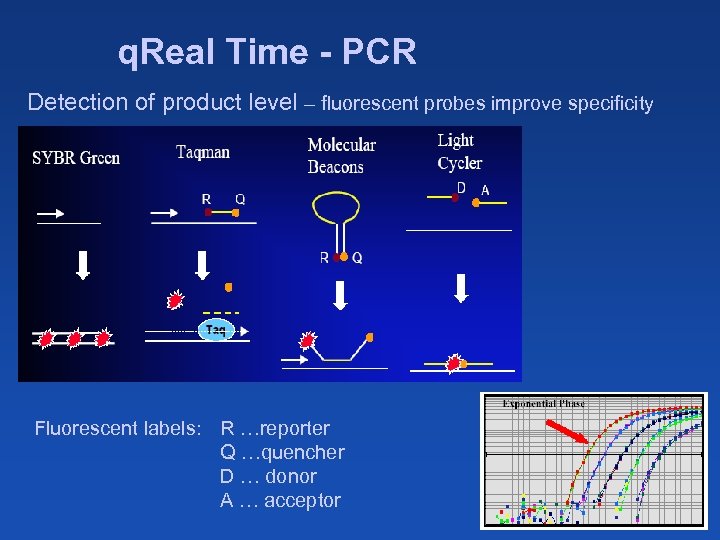 q. Real Time - PCR Detection of product level – fluorescent probes improve specificity Fluorescent labels: R …reporter Q …quencher D … donor A … acceptor
q. Real Time - PCR Detection of product level – fluorescent probes improve specificity Fluorescent labels: R …reporter Q …quencher D … donor A … acceptor
 Principle of detection of nucleic acids by hybridization Probe - strand of NA with known sequence used for detection of complementary strands in a mixture of NAs (e. g. transcripts, c. DNAs, genomic fragments) Two phases system ( ): hybridization of complementary single-stranded NA: immobilized phase (bound on membrane, glass, in tissue) mobile phase (NA in solution)
Principle of detection of nucleic acids by hybridization Probe - strand of NA with known sequence used for detection of complementary strands in a mixture of NAs (e. g. transcripts, c. DNAs, genomic fragments) Two phases system ( ): hybridization of complementary single-stranded NA: immobilized phase (bound on membrane, glass, in tissue) mobile phase (NA in solution)
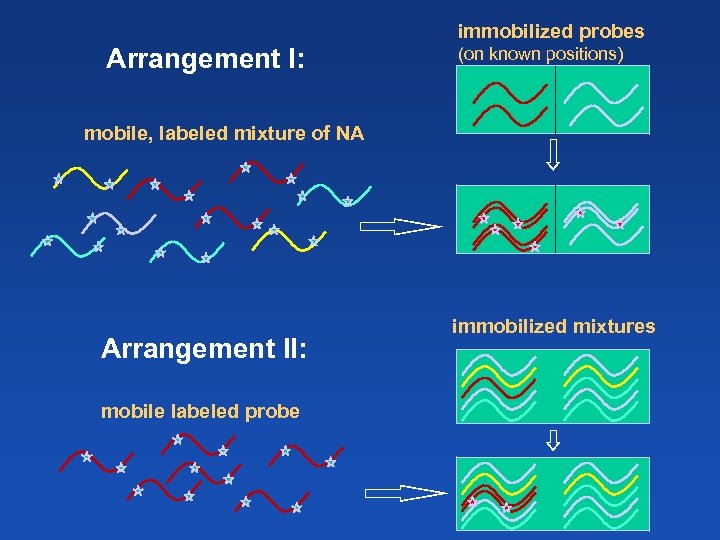 immobilized probes Arrangement I: (on known positions) mobile, labeled mixture of NA Arrangement II: mobile labeled probe immobilized mixtures
immobilized probes Arrangement I: (on known positions) mobile, labeled mixture of NA Arrangement II: mobile labeled probe immobilized mixtures
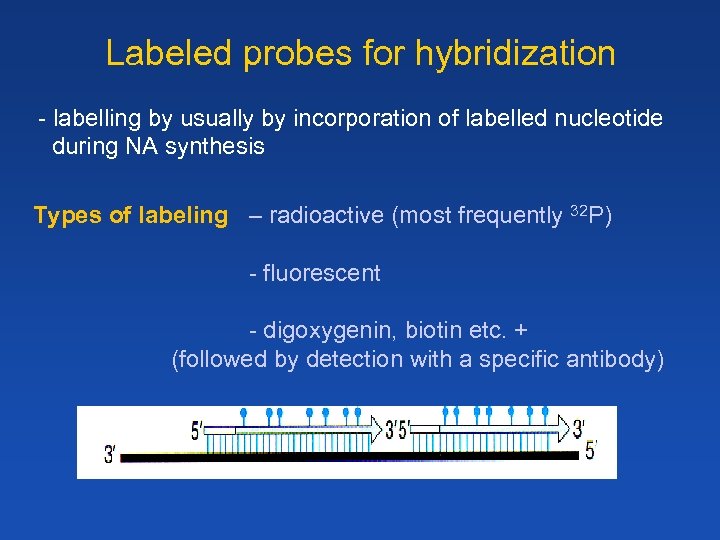 Labeled probes for hybridization - labelling by usually by incorporation of labelled nucleotide during NA synthesis Types of labeling – radioactive (most frequently 32 P) - fluorescent - digoxygenin, biotin etc. + (followed by detection with a specific antibody)
Labeled probes for hybridization - labelling by usually by incorporation of labelled nucleotide during NA synthesis Types of labeling – radioactive (most frequently 32 P) - fluorescent - digoxygenin, biotin etc. + (followed by detection with a specific antibody)
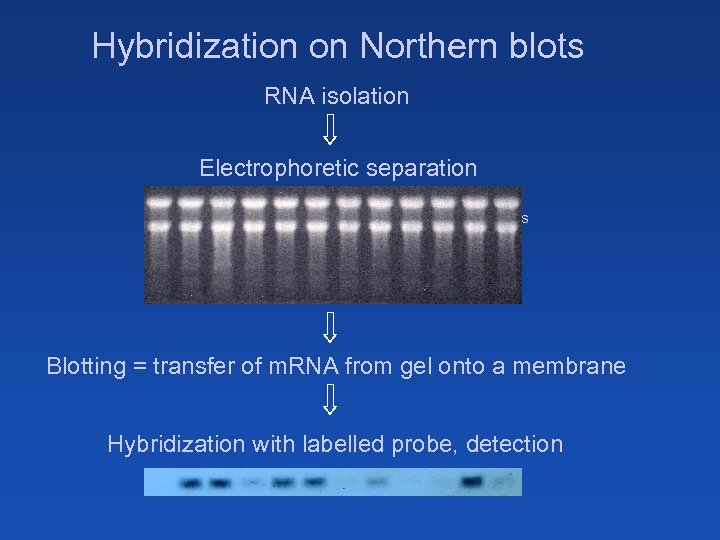 Hybridization on Northern blots RNA isolation Electrophoretic separation Macroarrays Microarrays Blotting = transfer of m. RNA from gel onto a membrane Hybridization with labelled probe, detection
Hybridization on Northern blots RNA isolation Electrophoretic separation Macroarrays Microarrays Blotting = transfer of m. RNA from gel onto a membrane Hybridization with labelled probe, detection
 Hybridization on Northern blots Immobilized phase – analyzed mixture of m. RNAs Mobile phase = labeled probe of certain gene (signal = presence of certain transcript + info about the transcript size) x Hybridization on DNA arrays or chips Immobilized phase – multiple probes with known sequences bound on certain places of the solid support Mobile phase = labeled mixture of analyzed NAs (simultaneous detection of presence and quantity of many sequences)
Hybridization on Northern blots Immobilized phase – analyzed mixture of m. RNAs Mobile phase = labeled probe of certain gene (signal = presence of certain transcript + info about the transcript size) x Hybridization on DNA arrays or chips Immobilized phase – multiple probes with known sequences bound on certain places of the solid support Mobile phase = labeled mixture of analyzed NAs (simultaneous detection of presence and quantity of many sequences)
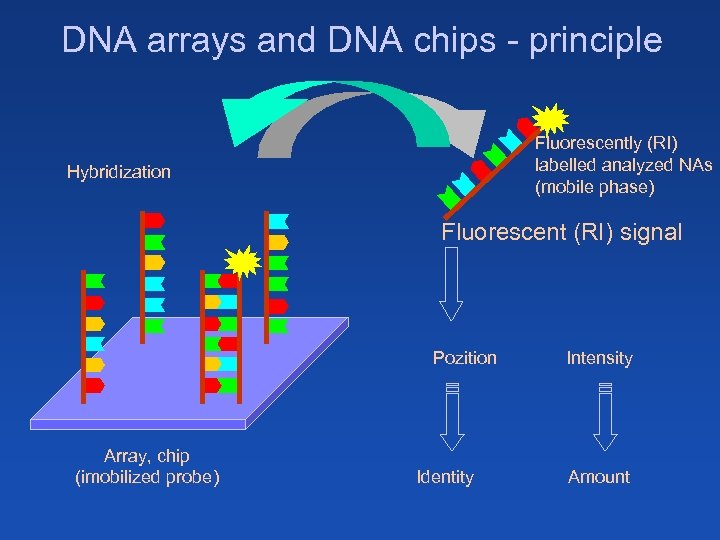 DNA arrays and DNA chips - principle Fluorescently (RI) labelled analyzed NAs (mobile phase) Hybridization Fluorescent (RI) signal Pozition Array, chip (imobilized probe) Identity Intensity Amount
DNA arrays and DNA chips - principle Fluorescently (RI) labelled analyzed NAs (mobile phase) Hybridization Fluorescent (RI) signal Pozition Array, chip (imobilized probe) Identity Intensity Amount
![Terminology: arrays, chips Preparation Support Density [probes/cm 2] Macroarray (High Density Array) Printing of Terminology: arrays, chips Preparation Support Density [probes/cm 2] Macroarray (High Density Array) Printing of](https://present5.com/presentation/db3413b3575bb1ee348380859497ca76/image-14.jpg) Terminology: arrays, chips Preparation Support Density [probes/cm 2] Macroarray (High Density Array) Printing of oligonucleotids or PCR fragments Membrane max. 64 e. g. glass up to 104 Microarray Printing of oligonucleotids or PCR fragments Direct synthesis on the support e. g. glass Chip up to 2. 5 *105
Terminology: arrays, chips Preparation Support Density [probes/cm 2] Macroarray (High Density Array) Printing of oligonucleotids or PCR fragments Membrane max. 64 e. g. glass up to 104 Microarray Printing of oligonucleotids or PCR fragments Direct synthesis on the support e. g. glass Chip up to 2. 5 *105
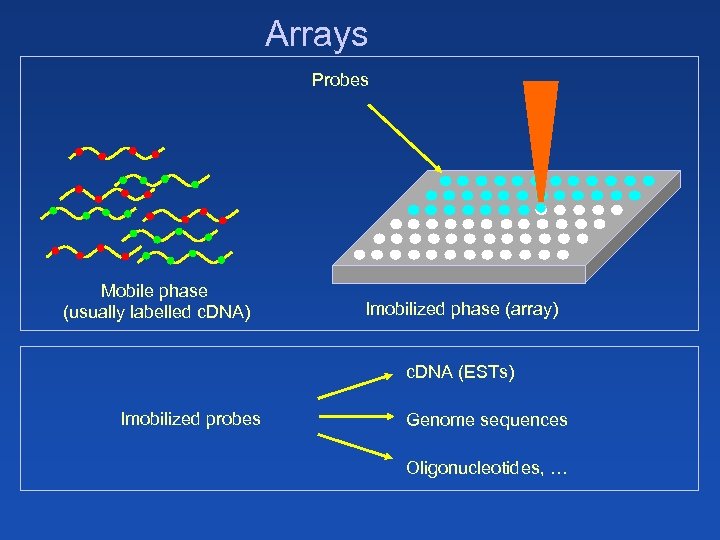 Arrays Probes Mobile phase (usually labelled c. DNA) Imobilized phase (array) c. DNA (ESTs) Imobilized probes Genome sequences Oligonucleotides, …
Arrays Probes Mobile phase (usually labelled c. DNA) Imobilized phase (array) c. DNA (ESTs) Imobilized probes Genome sequences Oligonucleotides, …
 Automated preparation of macroarrays contact printing 4. 5 mm
Automated preparation of macroarrays contact printing 4. 5 mm
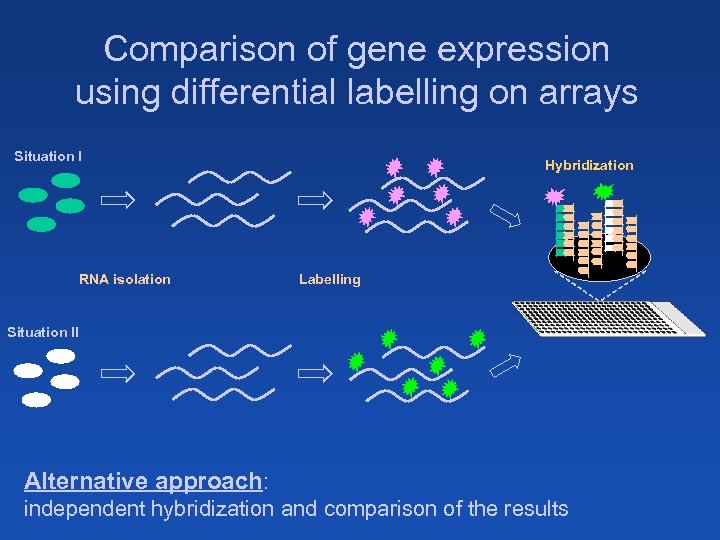 Comparison of gene expression using differential labelling on arrays Situation I RNA isolation Hybridization Labelling Situation II Alternative approach: independent hybridization and comparison of the results
Comparison of gene expression using differential labelling on arrays Situation I RNA isolation Hybridization Labelling Situation II Alternative approach: independent hybridization and comparison of the results
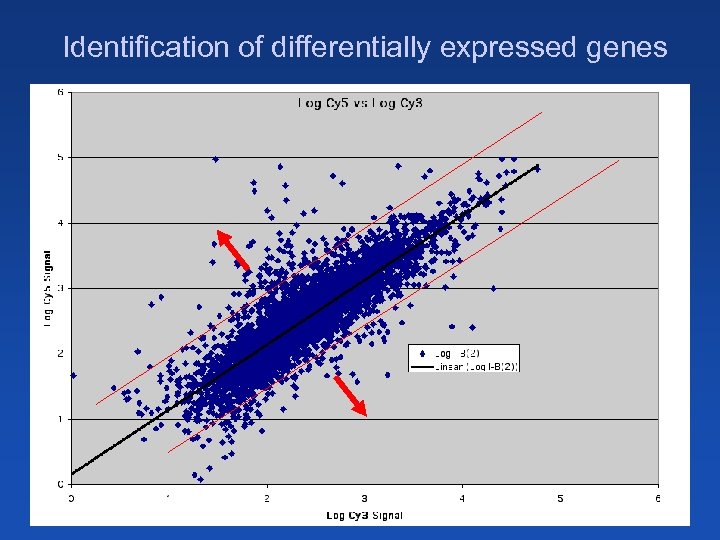 Identification of differentially expressed genes
Identification of differentially expressed genes
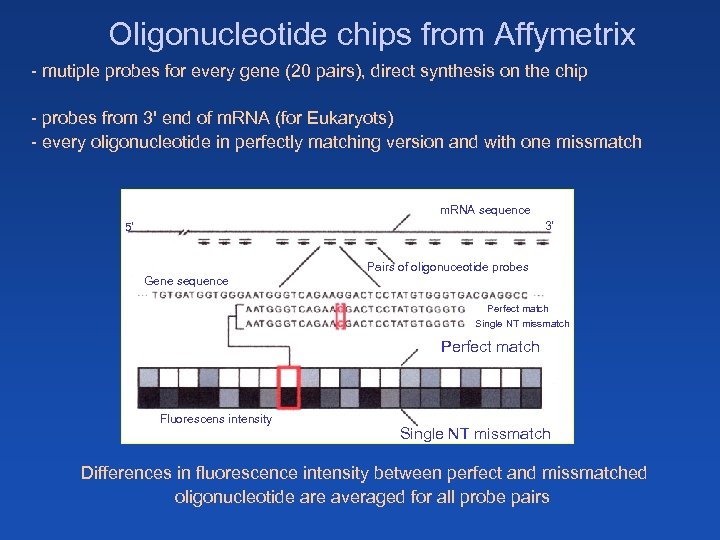 Oligonucleotide chips from Affymetrix - mutiple probes for every gene (20 pairs), direct synthesis on the chip - probes from 3' end of m. RNA (for Eukaryots) - every oligonucleotide in perfectly matching version and with one missmatch m. RNA sequence 3‘ 5‘ Pairs of oligonuceotide probes Gene sequence Perfect match Single NT missmatch Perfect match Fluorescens intensity Single NT missmatch Differences in fluorescence intensity between perfect and missmatched oligonucleotide are averaged for all probe pairs
Oligonucleotide chips from Affymetrix - mutiple probes for every gene (20 pairs), direct synthesis on the chip - probes from 3' end of m. RNA (for Eukaryots) - every oligonucleotide in perfectly matching version and with one missmatch m. RNA sequence 3‘ 5‘ Pairs of oligonuceotide probes Gene sequence Perfect match Single NT missmatch Perfect match Fluorescens intensity Single NT missmatch Differences in fluorescence intensity between perfect and missmatched oligonucleotide are averaged for all probe pairs
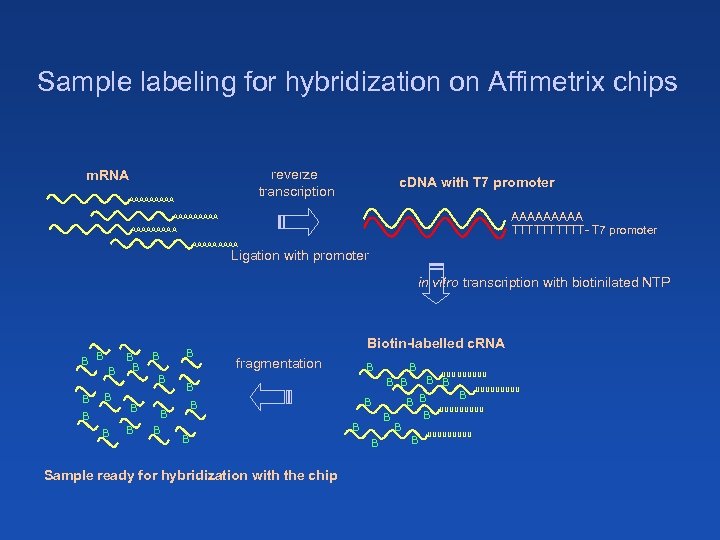 Sample labeling for hybridization on Affimetrix chips reverze transcription m. RNA AAAAA c. DNA with T 7 promoter AAAAA TTTTT- T 7 promoter AAAAAAAAA Ligation with promoter in vitro transcription with biotinilated NTP B B B B Biotin-labelled c. RNA fragmentation B B B Sample ready for hybridization with the chip B B UUUUUUUUU B B B B UUUUU
Sample labeling for hybridization on Affimetrix chips reverze transcription m. RNA AAAAA c. DNA with T 7 promoter AAAAA TTTTT- T 7 promoter AAAAAAAAA Ligation with promoter in vitro transcription with biotinilated NTP B B B B Biotin-labelled c. RNA fragmentation B B B Sample ready for hybridization with the chip B B UUUUUUUUU B B B B UUUUU
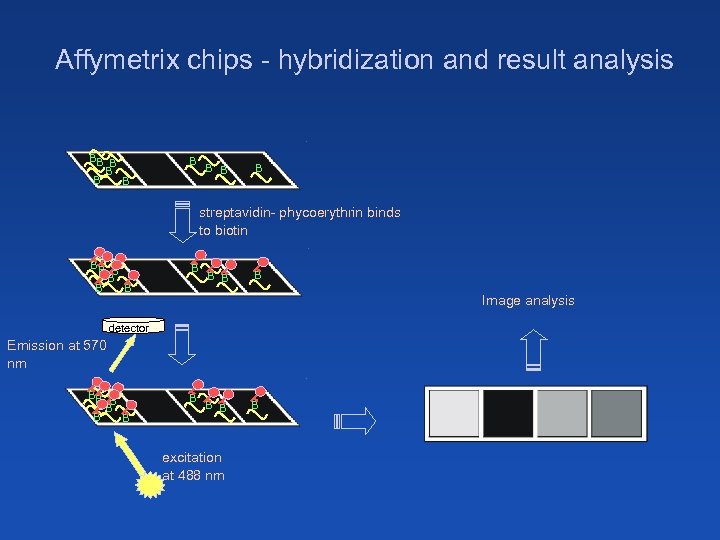 Affymetrix chips - hybridization and result analysis BB B B B B streptavidin- phycoerythrin binds to biotin BB B B B B Image analysis detector Emission at 570 nm BB B B B excitation at 488 nm B
Affymetrix chips - hybridization and result analysis BB B B B B streptavidin- phycoerythrin binds to biotin BB B B B B Image analysis detector Emission at 570 nm BB B B B excitation at 488 nm B
 Genevestigator https: //www. genevestigator. com partially free approach to chip results Selection of: - species - genes - chips (experiments)
Genevestigator https: //www. genevestigator. com partially free approach to chip results Selection of: - species - genes - chips (experiments)
 09. 01. 2003
09. 01. 2003
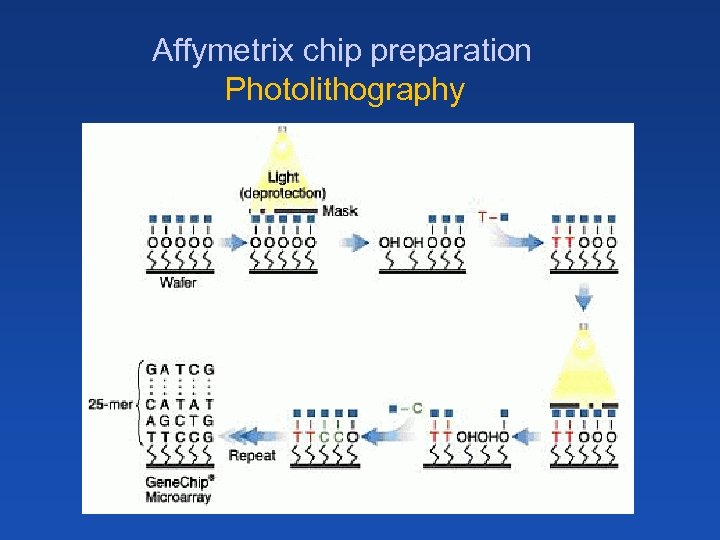 Affymetrix chip preparation Photolithography
Affymetrix chip preparation Photolithography
 Troubles with hybridization on arrays 1. Non-specific (cross-) hybridizations, background 2. Signal intensity depends also on sequence (differences in efficiency of hybridization) 3. Reproducibility Solutions: • every probe on different positions on the array • several different probes for every gene • Affimetrix chips
Troubles with hybridization on arrays 1. Non-specific (cross-) hybridizations, background 2. Signal intensity depends also on sequence (differences in efficiency of hybridization) 3. Reproducibility Solutions: • every probe on different positions on the array • several different probes for every gene • Affimetrix chips


