59e40233350399af1a487d78a32f4771.ppt
- Количество слайдов: 15

The Water Cycle Lisa Brown Aerospace Education Specialist Johnson Space Center Houston, Texas

Visit http: //www. nasa-news. org/resources/movies/Water. Cycle. Movie. mpg to view The Water Cycle video. (Link is clickable in ‘Slideshow View’)

What do we use water for? • • • Drinking Shower/Wash Brush Teeth Laundry Toilet Cleaning

How do we get water in space? We take it!

Water Recycling on ISS
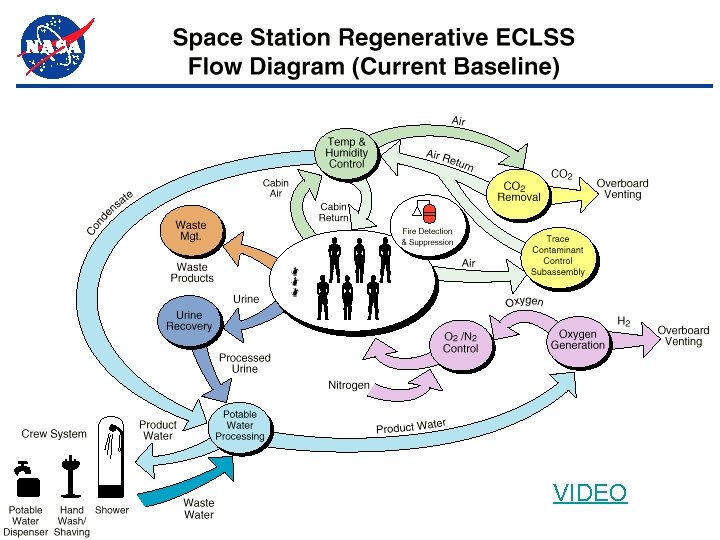
VIDEO

– This is a close-up view of the Environmental Control and Life Support System (ECLSS) Water Recovery System (WRS) racks. The WRS provides clean water through the reclamation of wastewaters, including water obtained from the Space Shuttle’s fuel cells, crewmember urine, used hand wash and oral hygiene water, cabin humidity condensate, and extravehicular activity (EVA) wastes.

Iodine isthesteam the water to use. The process is added to condensedto 265 ºF in It is storedwaterready contaminantsgrowth of form a Then, The. Asand isis Contaminants such a heatedcontrol the are Most the water chlorinethrough to the chemical flowstois added microorganisms -cleanlike The result is pure just liquid, which relatively urine oxygen is then joins thorough. It hasas as hair, skin cells, special drink at home. the undergo the most like reactor using resinsused instead of In space, tubes, be. contaminants removed ofis will sentthat the urine theurineto first is and sorbents water we. Thetypes wastewater to have been Iodine other water, those attractedcommon household water a are founddust, of it, and the steady supply etc. , are toof means to injected. The By following easier astransport treatment. iodine is much a drop chlorine becausehigh temperature kills urine to generated – from suchthese processorin in to itactivities to where is turned helpandcan seedevices. corrosive. materials are support water, less These the all of life isfiltered process orbit, wefiltration thereby housekeeping, because it entire germs inmaterials, space. from Once the brushing and chemical washing, leaving teeth, removing steam the solids behind these processes are completed, and the water passes contaminants humidity fromtubes conditioning wastewaterstream andthem composed of carbon, even from the water air packed in titanium the automated purity inspections hydrogen, and oxygen are broken down system to form carbon dioxide.

Acids and Bases » An acid is any of a class of substances that yields hydrogen ions (H+) when dissolved in water. The greater the concentration of hydrogen ions produced, the more acidic the substance is. Acids are characterized by a sour taste and the ability to react with bases and certain metals to form salts. » A base is any of a class of substances that yields hydroxide ions (OH-) when dissolved in water. The greater the concentration of hydroxide ions produced, the more basic the substance is. Bases are characterized by a bitter taste, a slippery feel, and the ability to react with acids to form salts.

What is p. H? Test p. H using litmus paper, p. H paper, or a p. H sensor.

Conductivity • Dissolved ions. Disassociation

Conductivity

Team Jobs • Principal Investigator • Materials Engineer • Cap. Com • PAO

The Challenge • Briefly design and draw Budget = $50 mil dollars your filtration device Cotton balls = $1 mil each that will yield the purest water. Coffee Filters = $5 mil each • Predict what will Activated Carbon = $15 mil happen. for bag • Build your filtration Macaroni = $10 mil for bag system. Sand = $10 mil for bag • Test systems. • Redesign & repeat Cheese cloth = $5 million • Report results. Rice = $10 mil for bag

So what does all that stuff do? • Activated carbon – used to remove organic containments responsible for taste, odor, color, and clarity problems; remove chlorine and particulates • Sand – natural filter materials; acts as a strainer and traps particulates; aka mechanical or physical filtration • Cotton balls, coffee filters, cheese cloth – traps particulates; absorb water – ions, color, clarity • Macaroni & rice – absorbs; raises p. H because of Carbohydrates
59e40233350399af1a487d78a32f4771.ppt