02763f705b54fd904db61ca69d410626.ppt
- Количество слайдов: 13

The Use of Intravenous (IV) Bisphonates at Nightingale House Hospice (NHH) March 2007 – March 2009 A Re-Audit Assessing Appropriate and Complete Documentation of Clinical Information Dr Nigel Martin Dr Nicola Browne 9 th Annual Palliative Care Conference 5 -6 November 2009

Overview • Introduction • The Bisphonate Therapy Booklet • The Audit – Method of Audit – Results of Audit • Discussion • What next?

Introduction • A bit about us……. • A bit about our subject – Bisphonates

Audit Method • Retrospective data collection • Included all patients on the NHH bisphonate register who were receiving bisphonates between March 2007 - March 2009 • 25 patient in total. • 2 patients were excluded • Information collected was based on NHH medicines policy on the ‘Use of Bisphonates in Palliative Medicine – 2007’ • Analysed using excel • Compare to results of previous audit to see if progress had been made

Audit Results • 65% of our cohort were female and 35% were male • Median age was 65 yrs • Most patients had either breast/prostate cancer • Reason for treatment documented in 79% • Pain (VA score) was documented in 96% initially and 91% on atleast 1 further occasion
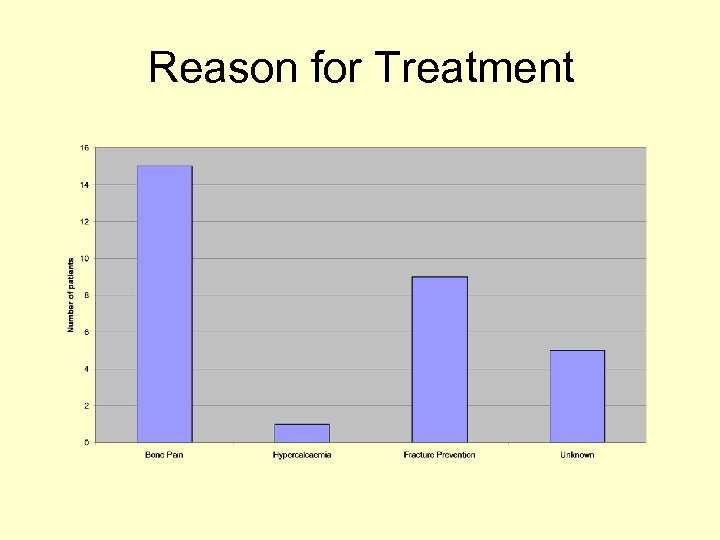
Reason for Treatment

Audit Results Cont. • • Verbal consent was documented in 71% Dental check was documented in 91% Full biochemical details were recorded in 78% Calcium supplement use was documented in 91% • Zoledronate was used in 78% • Batch number/Infusion number documented in 100% • Bisphonates were prescribed correctly 100% (taken into account renal function)
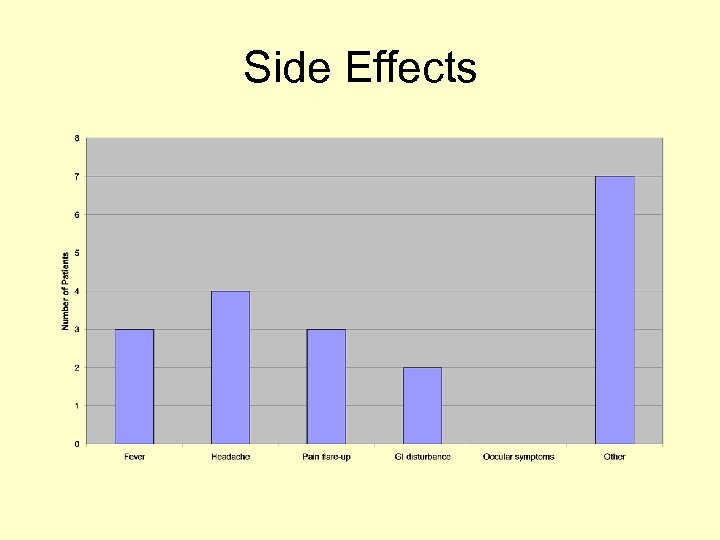
Side Effects
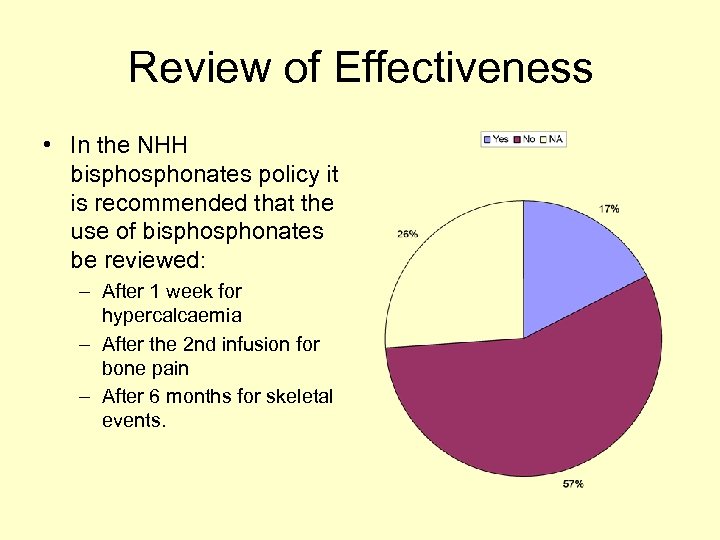
Review of Effectiveness • In the NHH bisphonates policy it is recommended that the use of bisphonates be reviewed: – After 1 week for hypercalcaemia – After the 2 nd infusion for bone pain – After 6 months for skeletal events.
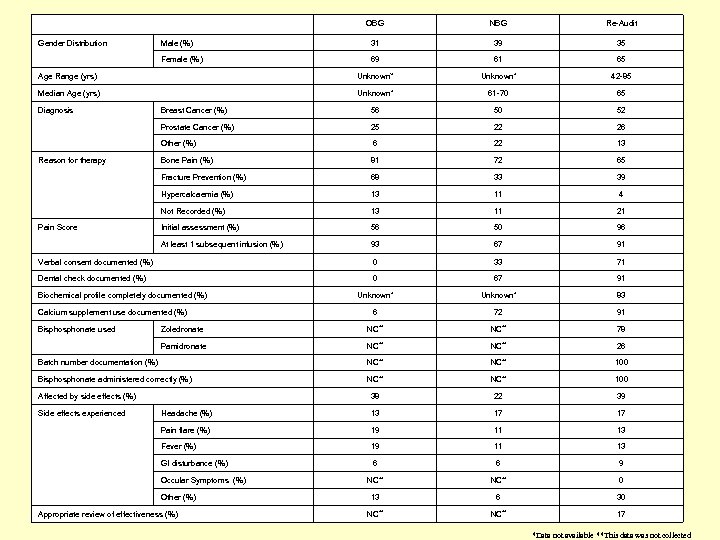
OBG NBG Re-Audit Male (%) 31 39 35 Female (%) 69 61 65 Age Range (yrs) Unknown’* Unknown* 42 -85 Median Age (yrs) Unknown* 61 -70 65 Breast Cancer (%) 56 50 52 Prostate Cancer (%) 25 22 26 Other (%) 6 22 13 Bone Pain (%) 81 72 65 Fracture Prevention (%) 68 33 39 Hypercalcaemia (%) 13 11 4 Not Recorded (%) 13 11 21 Initial assessment (%) 56 50 96 At least 1 subsequent infusion (%) 93 67 91 Verbal consent documented (%) 0 33 71 Dental check documented (%) 0 67 91 Unknown* 83 6 72 91 Zoledronate NC** 78 Pamidronate NC** 26 Batch number documentation (%) NC** 100 Bisphonate administered correctly (%) NC** 100 38 22 39 Headache (%) 13 17 17 Pain flare (%) 19 11 13 Fever (%) 19 11 13 GI disturbance (%) 6 6 9 NC** 0 13 6 30 NC** 17 Gender Distribution Diagnosis Reason for therapy Pain Score Biochemical profile completely documented (%) Calcium supplement use documented (%) Bisphonate used Affected by side effects (%) Side effects experienced Occular Symptoms (%) Other (%) Appropriate review of effectiveness (%) *Data not available **This data was not collected

Discussion • Greater preponderance of female patients remains unchanged • Our use of bisphonates maps that of guidelines (bone pain, skeletal event prevention) • Pain score is the only method of assessing the effectiveness of bisphonates currently and documentation has been increased • Due to side effects there should be documentation of verbal consent, dentitian check, biochemical status and use of calcium supplements prior to therapy – our rates have increased with all of these • Side effects continue to be experienced at a similar rate • Our weakest link currently appears to be documentating a review of effectiveness

Recommendations for Improvement • Space on therapy booklet for – Review of Effectiveness – Documenting Qo. L – Reason for discontinuation of therapy

Any Questions?
02763f705b54fd904db61ca69d410626.ppt