 The Urgent Need for Industry Guidance in Anti-infective Clinical Development Helen Boucher, MD Tufts - New England Medical Center, Boston, MA On behalf of The Infectious Diseases Society of America 1
The Urgent Need for Industry Guidance in Anti-infective Clinical Development Helen Boucher, MD Tufts - New England Medical Center, Boston, MA On behalf of The Infectious Diseases Society of America 1
 Disclosures i i i Aspreva Pharmaceuticalsa Cubist Pharmaceuticalsa, s, sh Johnson & Johnsona Pfizera, s, sh Schering-Plougha, s Targantaa a = Advisory/Consultation; s = Speaker; sh = Shareholder. 2
Disclosures i i i Aspreva Pharmaceuticalsa Cubist Pharmaceuticalsa, s, sh Johnson & Johnsona Pfizera, s, sh Schering-Plougha, s Targantaa a = Advisory/Consultation; s = Speaker; sh = Shareholder. 2
 Urgent Need Industry Guidance in Anti-infectives i i i Advances in clinical trial design have led to new considerations in anti-infective drug development; e. g. , i. Superiority vs. non-inferiority studies i. Number of studies required per indication Result - growing uncertainty in anti-infective development i Lack of written guidance contributes to this uncertainty Consequence: i Chilling effect on new anti-infective drug research and development 3
Urgent Need Industry Guidance in Anti-infectives i i i Advances in clinical trial design have led to new considerations in anti-infective drug development; e. g. , i. Superiority vs. non-inferiority studies i. Number of studies required per indication Result - growing uncertainty in anti-infective development i Lack of written guidance contributes to this uncertainty Consequence: i Chilling effect on new anti-infective drug research and development 3
 No Drugs!! “Product development in areas crucial to public health goals, such as antibiotics, has slowed significantly during the past decade. ” U. S. Food and Drug Administration. Innovation/Stagnation: Challenge and Opportunity on the Critical Path to New Medical Products. March 2004. 4
No Drugs!! “Product development in areas crucial to public health goals, such as antibiotics, has slowed significantly during the past decade. ” U. S. Food and Drug Administration. Innovation/Stagnation: Challenge and Opportunity on the Critical Path to New Medical Products. March 2004. 4
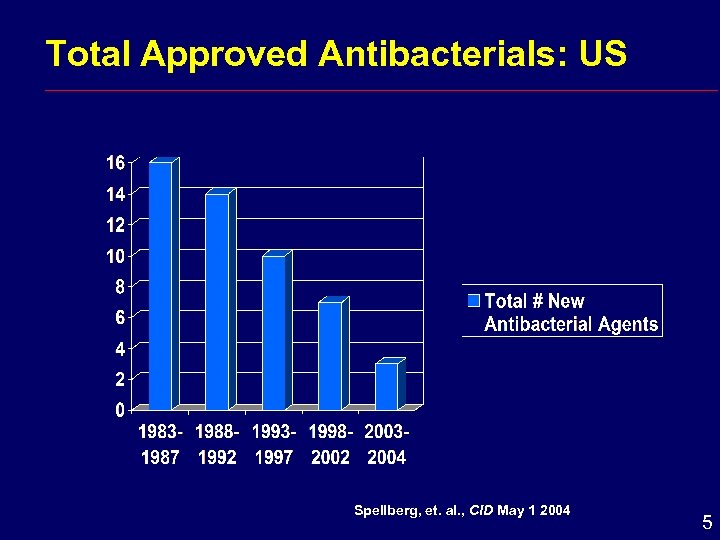 Total Approved Antibacterials: US Spellberg, et. al. , CID May 1 2004 5
Total Approved Antibacterials: US Spellberg, et. al. , CID May 1 2004 5
 Industry Guidance in Anti-infectives History - Overview i 2002 – present i IDSA leadership has urged FDA officials to move quickly to publish adequate industry guidance for anti-infective development i. Adequate guidance for industry i. Adequate scientific basis for determining safety and effectiveness 6
Industry Guidance in Anti-infectives History - Overview i 2002 – present i IDSA leadership has urged FDA officials to move quickly to publish adequate industry guidance for anti-infective development i. Adequate guidance for industry i. Adequate scientific basis for determining safety and effectiveness 6
 Industry Guidance in Anti-infectives History i i November, 2002, FDA/IDSA/Ph. RMA workshop i 5 guidance documents for i Resistant pathogens i Bacterial meningitis i Acute bacterial sinusitis i Acute bacterial otitis media i Acute exacerbation of chronic bronchitis Identified as important in addressing Industry need for clarity 7
Industry Guidance in Anti-infectives History i i November, 2002, FDA/IDSA/Ph. RMA workshop i 5 guidance documents for i Resistant pathogens i Bacterial meningitis i Acute bacterial sinusitis i Acute bacterial otitis media i Acute exacerbation of chronic bronchitis Identified as important in addressing Industry need for clarity 7
 Industry Guidance in Anti-infectives History i i 2004 - present i Collaborative workshops (FDA, IDSA, Ph. RMA, ISAP) i Face-to-face meetings i Legislation – Senate and House Commitment to publish guidance documents in order to address this pressing need 8
Industry Guidance in Anti-infectives History i i 2004 - present i Collaborative workshops (FDA, IDSA, Ph. RMA, ISAP) i Face-to-face meetings i Legislation – Senate and House Commitment to publish guidance documents in order to address this pressing need 8
 Industry Guidance in Anti-infectives IDSA Concern i In the absence of published guidelines during this period of evolution, the Agency appears not to have adopted a uniform approach for communicating nor a consistent timeline for implementing this new thinking; e. g. , i AIDAC meeting September 12, 2006 i Faropenem non-approvable letter October, 2006 i Today’s meeting 9
Industry Guidance in Anti-infectives IDSA Concern i In the absence of published guidelines during this period of evolution, the Agency appears not to have adopted a uniform approach for communicating nor a consistent timeline for implementing this new thinking; e. g. , i AIDAC meeting September 12, 2006 i Faropenem non-approvable letter October, 2006 i Today’s meeting 9
 Industry Guidance in Anti-infectives Conclusion i i Many barriers to the development of safe and effective antibiotics Absence of clear FDA guidance on trial design is consistently identified as a major barrier IDSA finds the delay in release of the 5 completed guideline documents mysterious, detrimental to drug discovery and development and, in the end, harmful to patients and public health IDSA remains willing to help facilitate this process in any way the Agency would find helpful 10
Industry Guidance in Anti-infectives Conclusion i i Many barriers to the development of safe and effective antibiotics Absence of clear FDA guidance on trial design is consistently identified as a major barrier IDSA finds the delay in release of the 5 completed guideline documents mysterious, detrimental to drug discovery and development and, in the end, harmful to patients and public health IDSA remains willing to help facilitate this process in any way the Agency would find helpful 10
 IDSA represents more than 8, 400 physicians and scientists who work in various areas of the field of infectious diseases medicine, including patient care, basic and clinical research, public health, and academia. 11
IDSA represents more than 8, 400 physicians and scientists who work in various areas of the field of infectious diseases medicine, including patient care, basic and clinical research, public health, and academia. 11