d6cd32c308ed6f8d56dccb6d686a6431.ppt
- Количество слайдов: 33
 The UK Approach to Procurement and Innovation Six Countries Programme – Innovation and Procurement Wednesday 16 November 2005 Margaret Horton Head of Research and Innovation
The UK Approach to Procurement and Innovation Six Countries Programme – Innovation and Procurement Wednesday 16 November 2005 Margaret Horton Head of Research and Innovation
 What I am going to talk about…… • UK Government policy • The NHS and innovation • The NHS supply network • A little bit of theory…… • Key NHS changes • Barriers to Innovation
What I am going to talk about…… • UK Government policy • The NHS and innovation • The NHS supply network • A little bit of theory…… • Key NHS changes • Barriers to Innovation
 UK government taking strong interest • Ex DTI So. S Patricia Hewitt met stakeholders Sept 2003 to discuss ways of opening up public procurement (PP) to encourage creativity from suppliers. Key issues emerged: – – • Better forward planning Greater transparency Increasing professionalism of those involved in PP Building in scope to encourage innovation DTI Innovation Report December 2003 – Called for action across the public sector to boost innovation – Stressed the vital role PP can play as a lever for stimulating and enabling supplier innovation – Tasked OGC to develop guidance that identifies ways in which govt can stimulate innovation from suppliers through the procurement process • Patricia Hewitt chairs the “Innovation in the Knowledge Economy” xgovt group to drive report forward. Immediate focus is on breaking down the £ 109 bn spend to identify the opportunities
UK government taking strong interest • Ex DTI So. S Patricia Hewitt met stakeholders Sept 2003 to discuss ways of opening up public procurement (PP) to encourage creativity from suppliers. Key issues emerged: – – • Better forward planning Greater transparency Increasing professionalism of those involved in PP Building in scope to encourage innovation DTI Innovation Report December 2003 – Called for action across the public sector to boost innovation – Stressed the vital role PP can play as a lever for stimulating and enabling supplier innovation – Tasked OGC to develop guidance that identifies ways in which govt can stimulate innovation from suppliers through the procurement process • Patricia Hewitt chairs the “Innovation in the Knowledge Economy” xgovt group to drive report forward. Immediate focus is on breaking down the £ 109 bn spend to identify the opportunities
 “Capturing Innovation – nurturing suppliers’ ideas in the public sector” OGC guidance 2004 • Challenges govt procurers to “think innovation” and become intelligent procurers • Innovation should be encouraged throughout but greatest potential arises from the earliest stages: – When policy is being formulated – When programmes and projects are being shaped – In the formulation of procurement strategy
“Capturing Innovation – nurturing suppliers’ ideas in the public sector” OGC guidance 2004 • Challenges govt procurers to “think innovation” and become intelligent procurers • Innovation should be encouraged throughout but greatest potential arises from the earliest stages: – When policy is being formulated – When programmes and projects are being shaped – In the formulation of procurement strategy
 “Capturing Innovation – nurturing suppliers’ ideas in the public sector” OGC guidance 2004 • Why we don’t capture innovation: – – Inadequate warning Risk aversion Client capability shortfalls Regulation • What we can do to capture innovation: – Challenge (ESI, procurement strategy, SME involvement, output/outcome specifications, acceptance of variants, contract management) – Channel (communicate long-term plans to the market, unsolicited proposals, early design contests and IPR) – Reward (cost and value for money, incentives, risk and reward sharing, payment not the only reward, ownership of IP
“Capturing Innovation – nurturing suppliers’ ideas in the public sector” OGC guidance 2004 • Why we don’t capture innovation: – – Inadequate warning Risk aversion Client capability shortfalls Regulation • What we can do to capture innovation: – Challenge (ESI, procurement strategy, SME involvement, output/outcome specifications, acceptance of variants, contract management) – Channel (communicate long-term plans to the market, unsolicited proposals, early design contests and IPR) – Reward (cost and value for money, incentives, risk and reward sharing, payment not the only reward, ownership of IP
 WHAT HAPPENS IN THE NHS?
WHAT HAPPENS IN THE NHS?
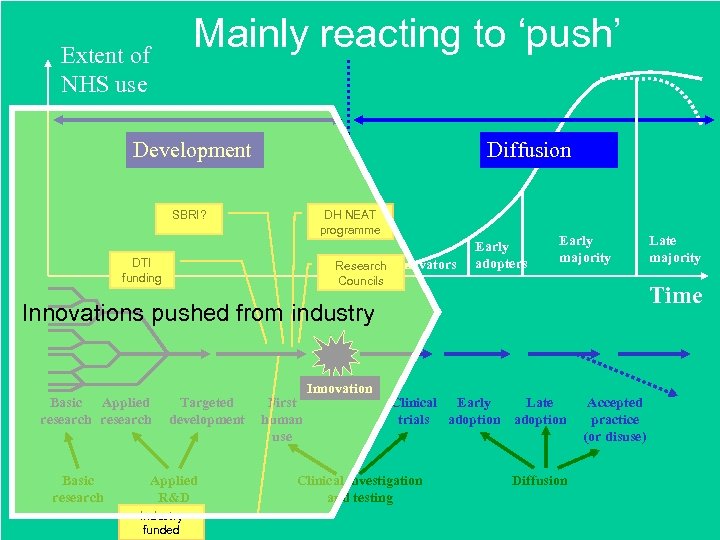 Mainly reacting to ‘push’ Extent of NHS use Development Diffusion SBRI? DH NEAT programme DTI funding Research Innovators Councils Early adopters Early majority Time Innovations pushed from industry Basic Applied research Basic research Targeted development Applied R&D Industry funded First human use Innovation Clinical Early Late trials adoption Clinical investigation and testing Late majority Diffusion Accepted practice (or disuse)
Mainly reacting to ‘push’ Extent of NHS use Development Diffusion SBRI? DH NEAT programme DTI funding Research Innovators Councils Early adopters Early majority Time Innovations pushed from industry Basic Applied research Basic research Targeted development Applied R&D Industry funded First human use Innovation Clinical Early Late trials adoption Clinical investigation and testing Late majority Diffusion Accepted practice (or disuse)
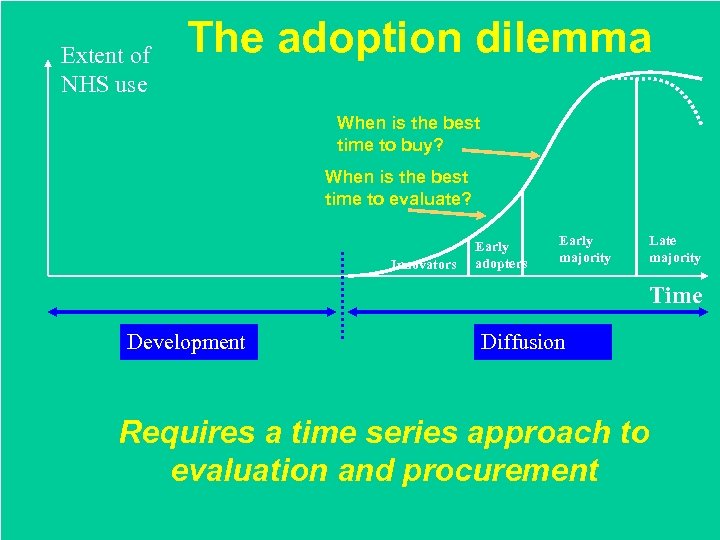 Extent of NHS use The adoption dilemma When is the best time to buy? When is the best time to evaluate? Innovators Early adopters Early majority Late majority Time Development Basic Applied research Basic research Targeted development Diffusion First human use Innovation Clinical Early Late trials adoption Accepted practice (or disuse) Requires a time series approach to evaluation and procurement Applied Clinical investigation Diffusion R&D and testing
Extent of NHS use The adoption dilemma When is the best time to buy? When is the best time to evaluate? Innovators Early adopters Early majority Late majority Time Development Basic Applied research Basic research Targeted development Diffusion First human use Innovation Clinical Early Late trials adoption Accepted practice (or disuse) Requires a time series approach to evaluation and procurement Applied Clinical investigation Diffusion R&D and testing
 But how does the NHS respond? “the UK is a ‘late’ and ‘slow’ adopter of new technology, compared with ‘early’ and ‘rapid’ adoption of technology in the US. Australia, Canada and France tend to be classified as ‘late’ adopters of technology, but once they start to adopt, they do so rapidly” Derek Wanless, 2002 “new technologies currently present themselves to different parts of the NHS in seemingly random order and there is much scope for inconsistency, duplication and delay To negotiate such a system requires either a well-resourced commercial sponsor or a resolute innovator!” Professor Kent Woods, 2002
But how does the NHS respond? “the UK is a ‘late’ and ‘slow’ adopter of new technology, compared with ‘early’ and ‘rapid’ adoption of technology in the US. Australia, Canada and France tend to be classified as ‘late’ adopters of technology, but once they start to adopt, they do so rapidly” Derek Wanless, 2002 “new technologies currently present themselves to different parts of the NHS in seemingly random order and there is much scope for inconsistency, duplication and delay To negotiate such a system requires either a well-resourced commercial sponsor or a resolute innovator!” Professor Kent Woods, 2002
 How does the NHS respond? (Co-map report) “Multiple entry routes – central versus local: which is the appropriate entry point for sales effort? PASA, HAs, Acute Trusts, PCTs or social services? ” “Multiple entry routes – clinician versus accountant: what purchasing decisions are driven by clinicians and what by management? How do clinical preferences get incorporated into management purchasing policy? ” “OJEC driven tender process tends to enshrine yesterday’s solution” “NHS purchasing decisions are perceived to be driven by lowest price tenders which results in bias towards old technology and a consequent reduction in longer term benefits to the NHS” “Budgetary silos appear to prevent purchasing decisions which are justifiable in terms of clinical and cost effectiveness and care pathways but cut across organisational boundaries” “There are problems with health economic methodology used because of perceived focus on relative cost of ‘like for like’ replacement products in current systems of practice rather than looking at benefits that may come from change of practice”
How does the NHS respond? (Co-map report) “Multiple entry routes – central versus local: which is the appropriate entry point for sales effort? PASA, HAs, Acute Trusts, PCTs or social services? ” “Multiple entry routes – clinician versus accountant: what purchasing decisions are driven by clinicians and what by management? How do clinical preferences get incorporated into management purchasing policy? ” “OJEC driven tender process tends to enshrine yesterday’s solution” “NHS purchasing decisions are perceived to be driven by lowest price tenders which results in bias towards old technology and a consequent reduction in longer term benefits to the NHS” “Budgetary silos appear to prevent purchasing decisions which are justifiable in terms of clinical and cost effectiveness and care pathways but cut across organisational boundaries” “There are problems with health economic methodology used because of perceived focus on relative cost of ‘like for like’ replacement products in current systems of practice rather than looking at benefits that may come from change of practice”
 WHY DOES IT RESPOND LIKE THIS?
WHY DOES IT RESPOND LIKE THIS?
 The shape of the NHS supply network Other Govt policies e. g. OGC, Sustainability, SMEs DH and its ALBs PASA NPSA R&D NICE MHRA MPIG MA Healthcare Commission 28 Supply Management Confederations 12 NHS IP hubs 1. 2 million staff 600+ organisations 300 Primary Care Trusts 300 Acute Trusts (with own procurement depts 20, 000 suppliers 28 Strategic Health Authorities £ 15 billion non-pay expenditure
The shape of the NHS supply network Other Govt policies e. g. OGC, Sustainability, SMEs DH and its ALBs PASA NPSA R&D NICE MHRA MPIG MA Healthcare Commission 28 Supply Management Confederations 12 NHS IP hubs 1. 2 million staff 600+ organisations 300 Primary Care Trusts 300 Acute Trusts (with own procurement depts 20, 000 suppliers 28 Strategic Health Authorities £ 15 billion non-pay expenditure
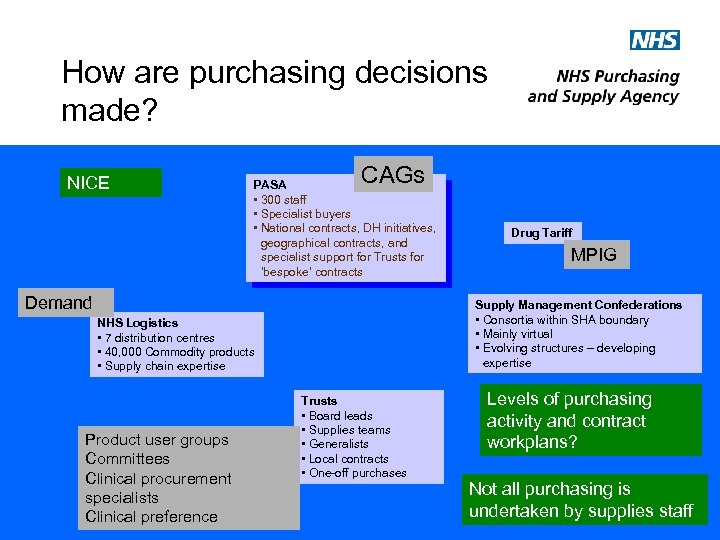 How are purchasing decisions made? NICE CAGs PASA • 300 staff • Specialist buyers • National contracts, DH initiatives, geographical contracts, and specialist support for Trusts for ‘bespoke’ contracts Demand MPIG Supply Management Confederations • Consortia within SHA boundary • Mainly virtual • Evolving structures – developing expertise NHS Logistics • 7 distribution centres • 40, 000 Commodity products • Supply chain expertise Product user groups Committees Clinical procurement specialists Clinical preference Drug Tariff Trusts • Board leads • Supplies teams • Generalists • Local contracts • One-off purchases Levels of purchasing activity and contract workplans? Not all purchasing is undertaken by supplies staff
How are purchasing decisions made? NICE CAGs PASA • 300 staff • Specialist buyers • National contracts, DH initiatives, geographical contracts, and specialist support for Trusts for ‘bespoke’ contracts Demand MPIG Supply Management Confederations • Consortia within SHA boundary • Mainly virtual • Evolving structures – developing expertise NHS Logistics • 7 distribution centres • 40, 000 Commodity products • Supply chain expertise Product user groups Committees Clinical procurement specialists Clinical preference Drug Tariff Trusts • Board leads • Supplies teams • Generalists • Local contracts • One-off purchases Levels of purchasing activity and contract workplans? Not all purchasing is undertaken by supplies staff
 A perception of public procurement ………. • Historical reactive ‘end of the process’ role? • More comfortable buying what has been bought in the past? • Large procurement organisations can become remote • Rigid adherence to EU procurement legislation – to point of using it to justify existence! • Do procurement staff understand their own business and what really drives the NHS? • Struggle to deal with ‘uncertainty of innovations’ – risk averse • We must deliver Value For Money – whatever that means……
A perception of public procurement ………. • Historical reactive ‘end of the process’ role? • More comfortable buying what has been bought in the past? • Large procurement organisations can become remote • Rigid adherence to EU procurement legislation – to point of using it to justify existence! • Do procurement staff understand their own business and what really drives the NHS? • Struggle to deal with ‘uncertainty of innovations’ – risk averse • We must deliver Value For Money – whatever that means……
 Understanding the ‘value’ in VFM • Do procurement staff understand the ‘value’ of new technology?
Understanding the ‘value’ in VFM • Do procurement staff understand the ‘value’ of new technology?
 Two worlds of ‘value’ • Problem: how do you define ‘value’ in healthcare? • For medical devices and pharmaceuticals it should be clinical and cost-effectiveness? !? BUT……… Clinical and cost effectiveness (HTA) HM Treasury VFM
Two worlds of ‘value’ • Problem: how do you define ‘value’ in healthcare? • For medical devices and pharmaceuticals it should be clinical and cost-effectiveness? !? BUT……… Clinical and cost effectiveness (HTA) HM Treasury VFM
 Public procurement – the comfort zone • EU regulations based on ‘competition’ • Public procurers only comfortable when they have competition and can test the market • Public procurers therefore intent on ‘commoditising’ • Public procurers have little interest in innovation because public services do not rely on innovation to survive/compete • It’s the law to throw relationships up in the air every few years – therefore little prospect of developing supplier relationships based on innovation!
Public procurement – the comfort zone • EU regulations based on ‘competition’ • Public procurers only comfortable when they have competition and can test the market • Public procurers therefore intent on ‘commoditising’ • Public procurers have little interest in innovation because public services do not rely on innovation to survive/compete • It’s the law to throw relationships up in the air every few years – therefore little prospect of developing supplier relationships based on innovation!
 A BIT OF THEORY…. .
A BIT OF THEORY…. .
 Some of what I have said is backed up by research • Public Technology Procurement (as opposed to regular procurement of existing products where only price/performance are considered) doesn’t fit with ‘auction theory’ of EU public procurement – Reason: EU believe it creates national champions who are not necessarily innovative – The result is ‘perpetuation’: public procurers repeat what they have bought before and create suppliers who become lazy & respond to public supply contracts and do not look to innovate Edquist et al, 2000) (Irony: EU single market set up to strengthen member states rate of innovation but in practice regime has followed a strictly economic & competition approach to public procurement)
Some of what I have said is backed up by research • Public Technology Procurement (as opposed to regular procurement of existing products where only price/performance are considered) doesn’t fit with ‘auction theory’ of EU public procurement – Reason: EU believe it creates national champions who are not necessarily innovative – The result is ‘perpetuation’: public procurers repeat what they have bought before and create suppliers who become lazy & respond to public supply contracts and do not look to innovate Edquist et al, 2000) (Irony: EU single market set up to strengthen member states rate of innovation but in practice regime has followed a strictly economic & competition approach to public procurement)
 Public technology procurement • Key aspect of PTP is dimension of time versus buyer competence (Edquist et al, 2000) creates problems: – Early & sophisticated demand – procuring agency needs to monitor leading edge technology and needs to understand where innovation is needed – Being locked-in too early – into risky (uncertain) new technological trajectories. Solution is to use PTP to support multiple trajectories to allow choice and strategic alternation between them – Buyers develop bargaining power by selecting the right specifications at the right time (beware delays caused by bad organisation and poor coordination of the procurement process)
Public technology procurement • Key aspect of PTP is dimension of time versus buyer competence (Edquist et al, 2000) creates problems: – Early & sophisticated demand – procuring agency needs to monitor leading edge technology and needs to understand where innovation is needed – Being locked-in too early – into risky (uncertain) new technological trajectories. Solution is to use PTP to support multiple trajectories to allow choice and strategic alternation between them – Buyers develop bargaining power by selecting the right specifications at the right time (beware delays caused by bad organisation and poor coordination of the procurement process)
 WHAT IS CHANGING?
WHAT IS CHANGING?
 Healthcare Industries Task Force • Outcome 1: Device Evaluation • Outcome 2: Innovation • Outcome 3: Procurement Processes
Healthcare Industries Task Force • Outcome 1: Device Evaluation • Outcome 2: Innovation • Outcome 3: Procurement Processes
 9 KEY OUTPUTS • Development of new Device Evaluation Service (DES) • Development of new NHS Innovation Centre • Procurement • Building R&D capacity • Work towards the development of Healthcare Technology Co-operatives • Regulatory issues • Export strategy • Communication on regulation & safety of devices • Training & education
9 KEY OUTPUTS • Development of new Device Evaluation Service (DES) • Development of new NHS Innovation Centre • Procurement • Building R&D capacity • Work towards the development of Healthcare Technology Co-operatives • Regulatory issues • Export strategy • Communication on regulation & safety of devices • Training & education
 Device evaluation (1) • inform procurement decisions, and encourage and support the uptake of useful, safe, innovative products and procedures used in health and social care: – Develop a new device evaluation service to integrate and strengthen horizon scanning, and the assessment of value and effective performance of new and enhanced healthcare technologies, devices and related procedures – Develop nationally accepted methodologies and toolkits for device evaluation that can be used locally to ensure consistency of approach whilst facilitating decision-making at the appropriate level – Consider how best to ensure speed of evaluation, a ‘once-only’ approach and prompt sharing of outputs with stakeholders throughout the health/social care system and industry • To help effect these changes existing DES will move to PASA w. e. f. 1. 4. 05
Device evaluation (1) • inform procurement decisions, and encourage and support the uptake of useful, safe, innovative products and procedures used in health and social care: – Develop a new device evaluation service to integrate and strengthen horizon scanning, and the assessment of value and effective performance of new and enhanced healthcare technologies, devices and related procedures – Develop nationally accepted methodologies and toolkits for device evaluation that can be used locally to ensure consistency of approach whilst facilitating decision-making at the appropriate level – Consider how best to ensure speed of evaluation, a ‘once-only’ approach and prompt sharing of outputs with stakeholders throughout the health/social care system and industry • To help effect these changes existing DES will move to PASA w. e. f. 1. 4. 05
 What will it be called and what will its mission be? “NHS PASA - Centre for Evidence-based Purchasing” • Underpin purchasing decisions and provide objective evidence to support the uptake of useful, safe, innovative products and related procedures in health and social care
What will it be called and what will its mission be? “NHS PASA - Centre for Evidence-based Purchasing” • Underpin purchasing decisions and provide objective evidence to support the uptake of useful, safe, innovative products and related procedures in health and social care
 Innovation (2) • Stimulate more innovation and encourage a more entrepreneurial culture in industry and the NHS: – Work towards the development of a new Innovation Centre in an appropriate organisation to promote and support the rapid development, dissemination and commercialisation of a pipeline of innovations coming from the NHS, academia or industry. The role would be to: • Coordinate and develop the activity of the existing network of NHS Innovation Hubs • Improve interactions and promote the exchange of knowledge between the NHS , industry, financiers and others, utilising online knowledge exchange/comms tools • Play a brokerage role between industry, financiers and the NHS, fostering partnership and collaboration opportunities • Promote successes and facilitate innovation uptake in the NHS • Introduce an ‘innovation fund’ to promote the development and exploitation by the NHS of innovative products and procedures • Establish collaboration between Med Devices Faraday Partnership and others to deliver coordination, brokerage and routemap, and increase translational research
Innovation (2) • Stimulate more innovation and encourage a more entrepreneurial culture in industry and the NHS: – Work towards the development of a new Innovation Centre in an appropriate organisation to promote and support the rapid development, dissemination and commercialisation of a pipeline of innovations coming from the NHS, academia or industry. The role would be to: • Coordinate and develop the activity of the existing network of NHS Innovation Hubs • Improve interactions and promote the exchange of knowledge between the NHS , industry, financiers and others, utilising online knowledge exchange/comms tools • Play a brokerage role between industry, financiers and the NHS, fostering partnership and collaboration opportunities • Promote successes and facilitate innovation uptake in the NHS • Introduce an ‘innovation fund’ to promote the development and exploitation by the NHS of innovative products and procedures • Establish collaboration between Med Devices Faraday Partnership and others to deliver coordination, brokerage and routemap, and increase translational research
 National Institute for Improvement & Innovation Lord Warner said the NHS Institute will: • “work closely with clinicians, NHS organisations, patients, the public, academia and industry in the UK and world-wide to identify best practice; • Develop the NHS’ capability for service transformation, technology and product innovation, leadership development and learning; • Support the rapid adoption and spread of new ideas by providing guidance on practical change ideas and ways to facilitate local, safe implementation; • Promote a culture of innovation and life-long learning for all NHS staff”
National Institute for Improvement & Innovation Lord Warner said the NHS Institute will: • “work closely with clinicians, NHS organisations, patients, the public, academia and industry in the UK and world-wide to identify best practice; • Develop the NHS’ capability for service transformation, technology and product innovation, leadership development and learning; • Support the rapid adoption and spread of new ideas by providing guidance on practical change ideas and ways to facilitate local, safe implementation; • Promote a culture of innovation and life-long learning for all NHS staff”
 Procurement and Innovation • Aim of combined strategies to provide over time: – single market entry point to NHS – clear pathway & support from early product development to “ready for market” – faster NHS uptake of innovation – informed purchasing decisions – stimulus for innovation – better outcomes for patients Outputs are interdependent!
Procurement and Innovation • Aim of combined strategies to provide over time: – single market entry point to NHS – clear pathway & support from early product development to “ready for market” – faster NHS uptake of innovation – informed purchasing decisions – stimulus for innovation – better outcomes for patients Outputs are interdependent!
 Procurement processes (3) • Embed modern approaches to procurement in the NHS to deliver better value for the service of patients through : – nationally agreed/accepted best practice models, including early communication with industry on workplans, to provide clarity on levels of market access and to ensure capture of innovative solutions – A focus for regional procurement with significant clinician involvement to provide the platform for an informed approach to procurement decision-making – Ensuring that the role of procurement in supporting the timely uptake of new technologies identified as providing benefit to patients is embraced – The above to be incorporated into the redesign of PASA, the CPH model, and continuing SMCs – Ensure training and education for devices are covered – Regular dialogue between NHS and industry to encourage input into policymaking (Pb. R, NSFs)
Procurement processes (3) • Embed modern approaches to procurement in the NHS to deliver better value for the service of patients through : – nationally agreed/accepted best practice models, including early communication with industry on workplans, to provide clarity on levels of market access and to ensure capture of innovative solutions – A focus for regional procurement with significant clinician involvement to provide the platform for an informed approach to procurement decision-making – Ensuring that the role of procurement in supporting the timely uptake of new technologies identified as providing benefit to patients is embraced – The above to be incorporated into the redesign of PASA, the CPH model, and continuing SMCs – Ensure training and education for devices are covered – Regular dialogue between NHS and industry to encourage input into policymaking (Pb. R, NSFs)
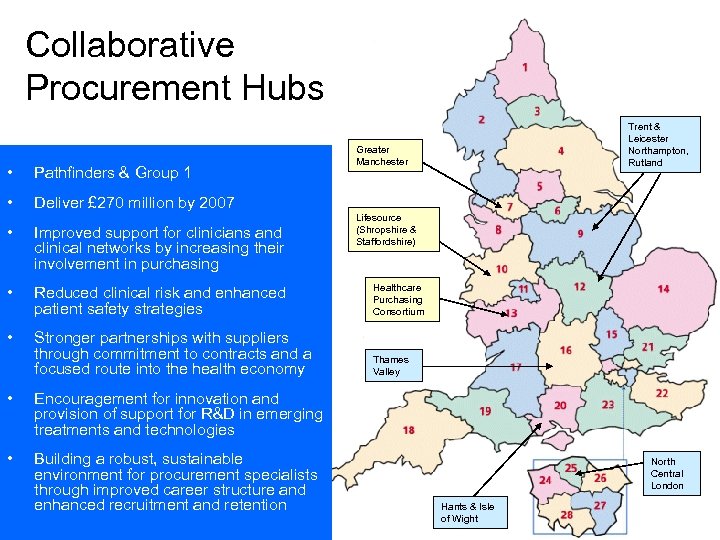 Collaborative Procurement Hubs • Pathfinders & Group 1 • Trent & Leicester Northampton, Rutland Greater Manchester Deliver £ 270 million by 2007 Lifesource (Shropshire & Staffordshire) • Improved support for clinicians and clinical networks by increasing their involvement in purchasing • Reduced clinical risk and enhanced patient safety strategies Healthcare Purchasing Consortium • Stronger partnerships with suppliers through commitment to contracts and a focused route into the health economy Thames Valley • Encouragement for innovation and provision of support for R&D in emerging treatments and technologies • Building a robust, sustainable environment for procurement specialists through improved career structure and enhanced recruitment and retention North Central London Hants & Isle of Wight
Collaborative Procurement Hubs • Pathfinders & Group 1 • Trent & Leicester Northampton, Rutland Greater Manchester Deliver £ 270 million by 2007 Lifesource (Shropshire & Staffordshire) • Improved support for clinicians and clinical networks by increasing their involvement in purchasing • Reduced clinical risk and enhanced patient safety strategies Healthcare Purchasing Consortium • Stronger partnerships with suppliers through commitment to contracts and a focused route into the health economy Thames Valley • Encouragement for innovation and provision of support for R&D in emerging treatments and technologies • Building a robust, sustainable environment for procurement specialists through improved career structure and enhanced recruitment and retention North Central London Hants & Isle of Wight
 So …… Barriers to Innovation? • Organisational boundaries • Lack of forward planning & existing financial frameworks • Understanding Value • Incentives and measures • Attitudes, behaviours and capability • Ethos of public procurement – competition = innovation? • Regulation - ‘One size fits all’ approach
So …… Barriers to Innovation? • Organisational boundaries • Lack of forward planning & existing financial frameworks • Understanding Value • Incentives and measures • Attitudes, behaviours and capability • Ethos of public procurement – competition = innovation? • Regulation - ‘One size fits all’ approach
 Steps to overcome barriers? (1) • Understand real business needs and match to procurement strategy • Create effective networks and clear pathways for innovation which allow ‘pull’ as well as ‘push’ • Create capability • Create intelligent ‘platforms’ for procurement • Define and understand value & communicate value definition • Develop procurement processes which encourage opportunities to harness innovation throughout procurement life-cycles
Steps to overcome barriers? (1) • Understand real business needs and match to procurement strategy • Create effective networks and clear pathways for innovation which allow ‘pull’ as well as ‘push’ • Create capability • Create intelligent ‘platforms’ for procurement • Define and understand value & communicate value definition • Develop procurement processes which encourage opportunities to harness innovation throughout procurement life-cycles
 Steps to overcome barriers? (2) • Develop procurement toolkits which provide options to avoid ‘one size fits all’ approach • Develop new measures for procurement which are focused on ‘value’ and which incentivise them to seek innovations to support business needs • Create body of knowledge to underpin procurement decisions • Challenge organisational boundaries and financial systems which restrict opportunities for Innovation
Steps to overcome barriers? (2) • Develop procurement toolkits which provide options to avoid ‘one size fits all’ approach • Develop new measures for procurement which are focused on ‘value’ and which incentivise them to seek innovations to support business needs • Create body of knowledge to underpin procurement decisions • Challenge organisational boundaries and financial systems which restrict opportunities for Innovation


