3b9f680f8fe0daa7f5c0dd47841cfe39.ppt
- Количество слайдов: 63

The status of the GCI microprobe M. Folkard, K. M. Prise, B. Vojnovic, G. Schettino, B. D. Michael Gray Cancer Institute, PO Box 100, Mount Vernon Hospital, Northwood, HA 6 2 JR UK

The Gray Cancer Institute LH Gray FRS 1905 -65
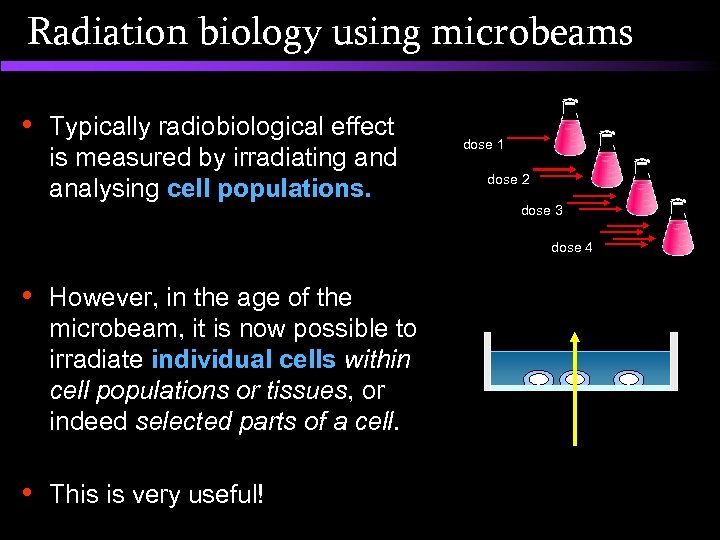
Radiation biology using microbeams • Typically radiobiological effect is measured by irradiating and analysing cell populations. dose 1 dose 2 dose 3 dose 4 • However, in the age of the microbeam, it is now possible to irradiate individual cells within cell populations or tissues, or indeed selected parts of a cell. • This is very useful!

Microbeam development: background • The first (modern) microbeam was developed by Dr Les Braby (Pacific North West, USA) in the late 1980 s. This used vertically collimated light ions • This was dismantled in the mid 1990 s and moved to Texas A&M

Microbeam development: background • The second microbeam was developed by Dr Brenner and Dr Randers-Perhson and colleagues at RARAF, New York in the early 1990 s. This also uses vertically collimated light ions • This microbeam is still being used routinely

Microbeam development: background • The third microbeam was developed by us at GCI in the early 1990 s. Like the RARAF and PNL facilities. It also uses vertically collimated light ions.

Microbeam development: background • The first microbeams were developed to study the effects of very low doses of particles. • A single particle through a cell is the ultimate low dose (of particles). • A microbeam is required to do this.

Microbeam development: background • Then came the bystander effect… Nagasawa and Little, Cancer Res. , 1992. • irradiate cells with a low-dose of a-particles (<1% cells traversed by an a-particle) • • 30% cells exhibited increased chromosome damage Microbeams ideal for the studying the bystander effect

Microbeam: requirements Microbeam ‘shopping list’: • • A ‘micron-sized beam’ of radiation A compatible method of supporting cells A method for identifying (sub-cellular) targets A method for aligning targets to the beam Precise dose delivery Cell revisiting (for assaying damage) Environmental control (temperature, p. H) A high cell throughput (automation)
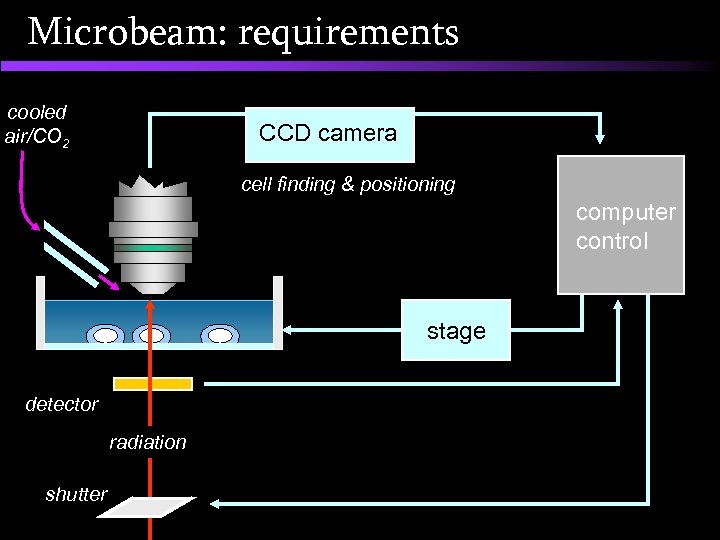
Microbeam: requirements cooled air/CO 2 CCD camera cell finding & positioning computer control stage detector radiation shutter

GCI charged-particle microbeam

GCI charged-particle microbeam Physics Lab 4 MV Van de Graaff 1991 Neutron room
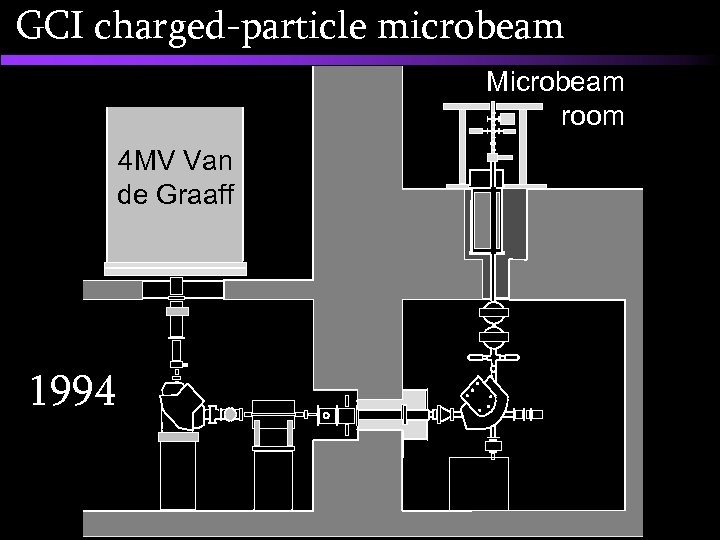
GCI charged-particle microbeam Microbeam room 4 MV Van de Graaff 1994

GCI charged-particle microbeam glass capillary 1 m bore collimator assembly 250 mm optical table

GCI charged-particle microbeam CCD camera + intensifier light source objective stage support and alignment stage support & Z drive collimator assembly optical table
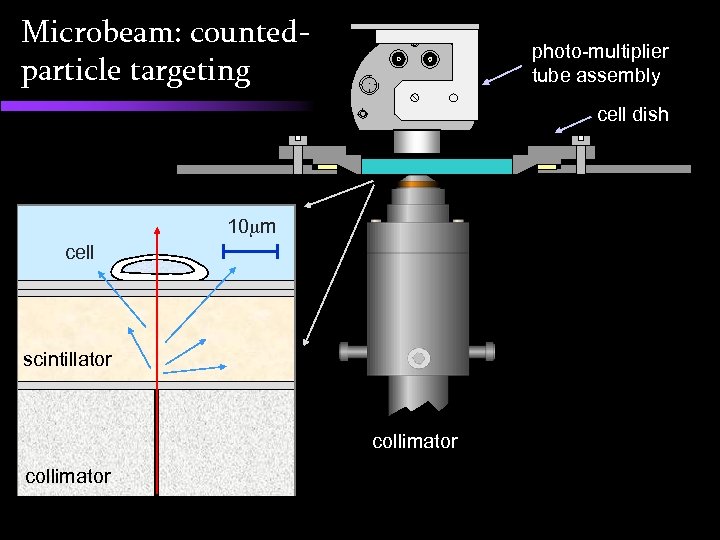
Microbeam: countedparticle targeting photo-multiplier tube assembly cell dish 10 m cell 3 m Mylar scintillator collimator

GCI charged-particle microbeam

GCI charged-particle microbeam

GCI charged-particle microbeam

GCI charged-particle microbeam What is the microbeam targeting accuracy? Targeting accuracy is determined by- • • • Beam size • Target positioning accuracy Accuracy of beam identification and location Accuracy of target identification and location

GCI microbeam- beam size -CR-39 track-etch plastic 3. 5 Me. V 3 He 2+ ions 10 x 10 grid 10 particles per cluster 98% of ions within 2 m of the mean position 50 m
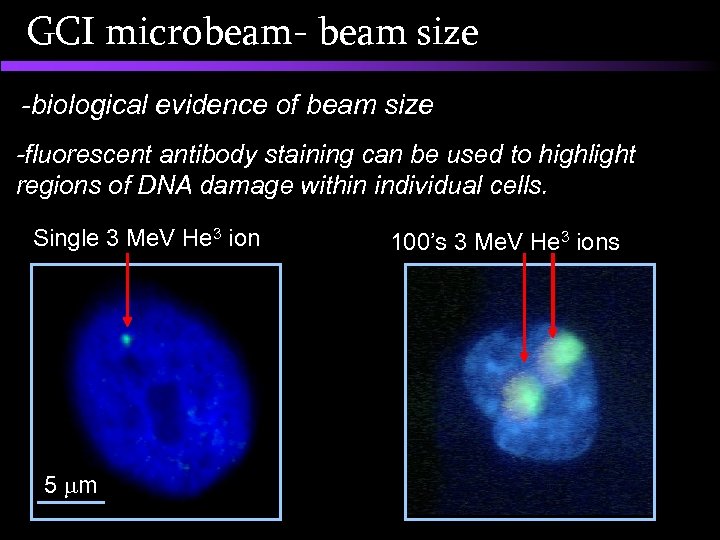
GCI microbeam- beam size -biological evidence of beam size -fluorescent antibody staining can be used to highlight regions of DNA damage within individual cells. Single 3 Me. V He 3 ion 5 m 100’s 3 Me. V He 3 ions

GCI microbeam- beam location -intensified CCD camera used to visualise the beam 20 m ~1 m uncertainty in beam location

GCI microbeam- target identification -automated cell finding Typical screenshot during cell-finding step 540 cells in a 1 cm 2 area ~1 m uncertainty in target location
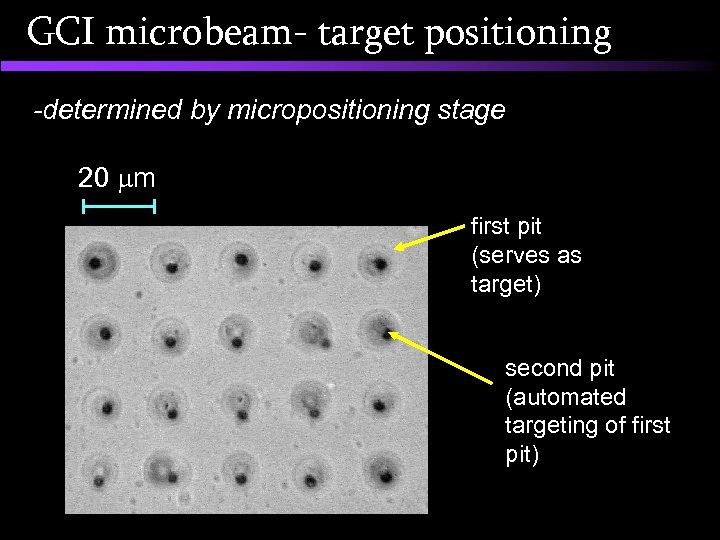
GCI microbeam- target positioning -determined by micropositioning stage 20 m first pit (serves as target) second pit (automated targeting of first pit)
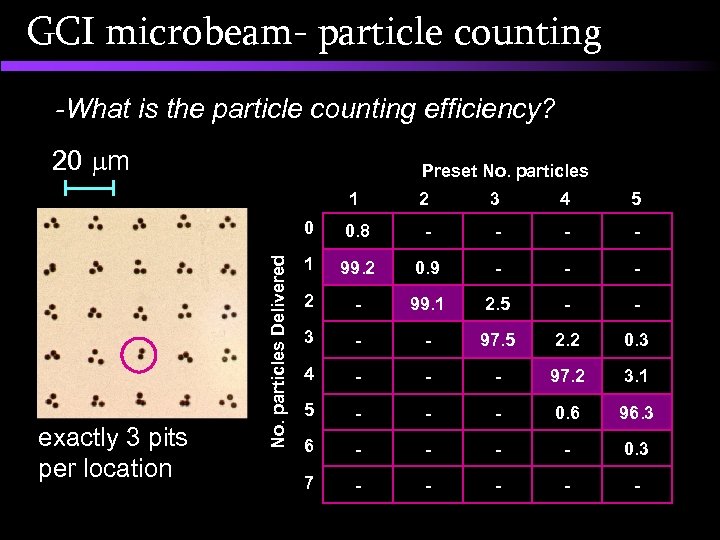
GCI microbeam- particle counting -What is the particle counting efficiency? 20 m Preset No. particles exactly 3 pits per location 2 3 4 5 0 No. particles Delivered 1 0. 8 - - 1 99. 2 0. 9 - - - 2 - 99. 1 2. 5 - - 3 - - 97. 5 2. 2 0. 3 4 - - - 97. 2 3. 1 5 - - - 0. 6 96. 3 6 - - 0. 3 7 - - -
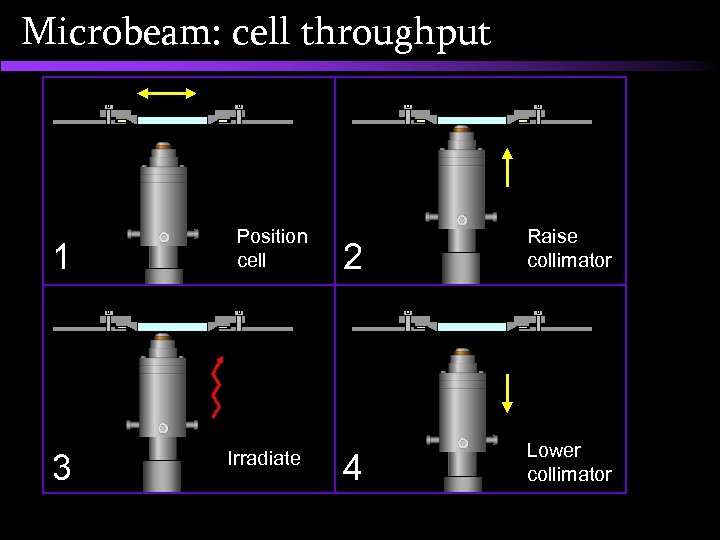
Microbeam: cell throughput 1 3 Position cell Irradiate 2 Raise collimator 4 Lower collimator
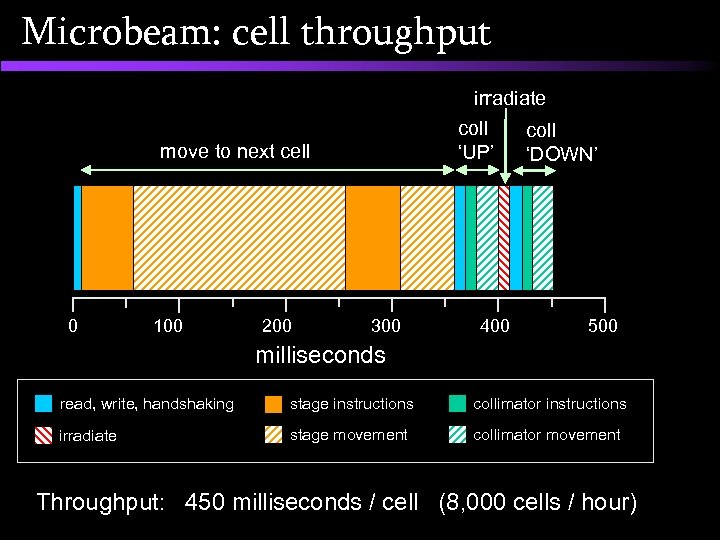
Microbeam: cell throughput irradiate coll ‘UP’ move to next cell 0 100 200 300 400 coll ‘DOWN’ 500 milliseconds read, write, handshaking stage instructions collimator instructions irradiate stage movement collimator movement Throughput: 450 milliseconds / cell (8, 000 cells / hour)

Microbeam: cell throughput Märzhäuser stepper motor stage

Microbeam: cell throughput voice coil DC motors optical encoders
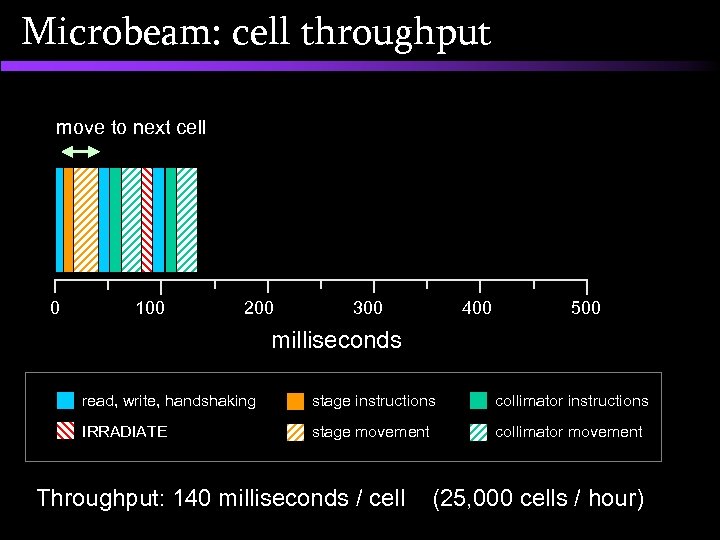
Microbeam: cell throughput move to next cell 0 100 200 300 400 500 milliseconds read, write, handshaking stage instructions collimator instructions IRRADIATE stage movement collimator movement Throughput: 140 milliseconds / cell (25, 000 cells / hour)

GCI charged-particle microbeam Performance summary: • Targeting accuracy: 2 m • Detection efficiency: ~99% • Dose rate <1000 particles s-1 • Cell throughput: 9000 cells / hour
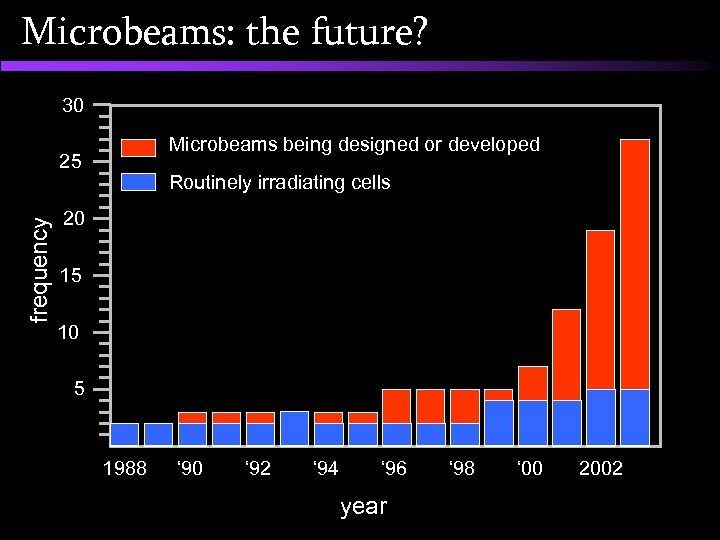
Microbeams: the future? 30 Microbeams being designed or developed frequency 25 Routinely irradiating cells 20 15 10 5 1988 ‘ 90 ‘ 92 ‘ 94 ‘ 96 year ‘ 98 ‘ 00 2002
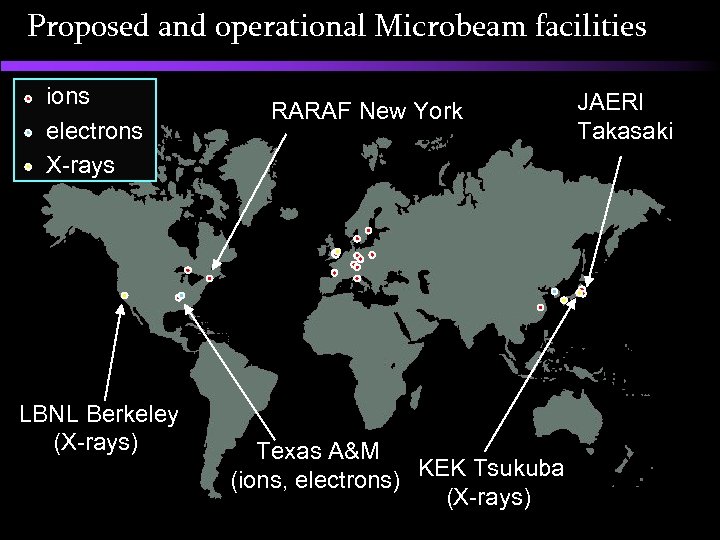
Proposed and operational Microbeam facilities ions electrons X-rays LBNL Berkeley (X-rays) RARAF New York Texas A&M (ions, electrons) KEK Tsukuba (X-rays) JAERI Takasaki
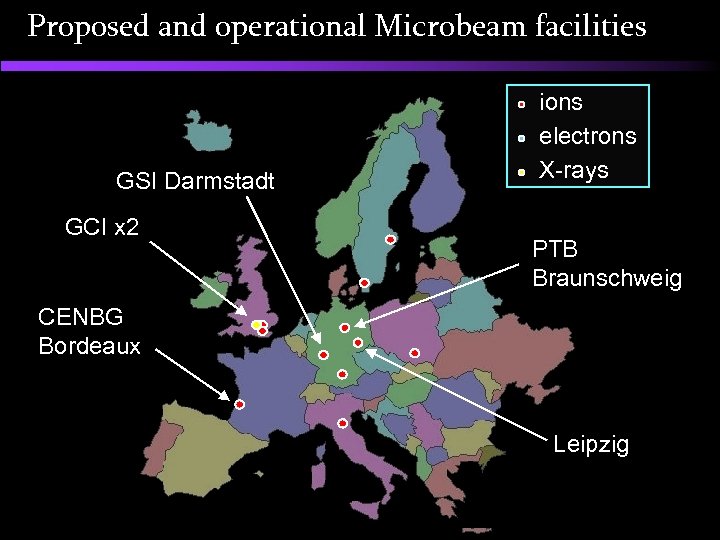
Proposed and operational Microbeam facilities GSI Darmstadt GCI x 2 ions electrons X-rays PTB Braunschweig CENBG Bordeaux Leipzig

Microbeams: the future? • What are the important specifications for a microbeam of the future? - Ask the biologist (she/he is your customer)! • Radiation biology is a ‘moving target’ • Funding priorities in radiation science for radiation science changes year by year • Currently (in the UK) ‘clinically relevant’ research has a high priority

Microbeams: the future? • Microbeam performance specified by: - Radiation quality (type, energy) Targeting accuracy Dose delivery (particle counting) Cell throughput Cell environment

Radiation quality Ion microbeams can be loosely divided into two categories: - Light ion microbeams (H, He) - ‘Heavy’ ion microbeams (>He)

Radiation quality Light ions (H, He) • protons ü most penetrating for given accelerator ü useful LET range (10 – 40 ke. V mm-1) ü relevant to proton radiotherapy û easily scattered • helium ions ü radiobiolically relevant to risk (radon) ü good penetration, less readily scattered

Radiation quality Heavy ions (>He) ü (very) high LET studies ü particle radiotherapy, other than protons ü reduced scattering û limited radiobiolical relevance

Radiation quality The competition… • Low-energy X-rays ü relatively easy to generate (tabletop) ü optimum for low LET studies ü no scattering- nanometre probes possible û (so far) limited penetration • electrons ü easy to generate and focus û secondary electrons disperse energy

Targeting accuracy • micron-sized beam spot achieved by…. - collimation magnetic focusing electrostatic focusing

Targeting accuracy • collimation ü low-cost technology ü can be compact û scattering limits spot size to a few microns • focusing ü sub-micron spot sizes possible ü steerable beam û greater space requirement û greater cost and complexity û existing microprobes horizontal

Dose delivery (particle counting) Particle detectors are either invasive (between beam and cell), or non-invasive (after cell) • invasive ü places less constraints on sample û introduces scattering • non-invasive ü does not scatter beam û cannot be used with thick samples, or partially penetrating radiations û removal of culture medium required

Cell throughput is determined largely by the speed of beam-cell alignment • moving cell / fixed beam - mandatory for collimated systems ü based on established microscope technology û sample stage accuracy is an issue û stage movement can limit speed

Cell throughput is determined largely by the speed of beam-cell alignment • moving beam / fixed cell ü very high cell throughputs possible ü no moving parts û requires focused beam û verification of targeting difficult û limited scan area may mean that some sample stage movement is still required
Cell environment The cell environment and the means to image cells must be ‘stress free’ • Possible source of stress - insufficient temperature control incorrect p. H use of fluorescent dyes to image cells UV illumination of cells

Cell environment Target identification • If using dyes… - minimize dye concentration (sensitive camera) minimize UV exposure- take ‘snapshots’, not live images use dyes excited by near-visible wavelengths

Cell environment Target identification • target identification without the use of dyes - but very difficult to do automatically - off-line cell finding probably essential this is the ‘holy grail’ for microbeam cell studies identification of targets possible by eye better image processing is NOT the answer the method of sample preparation and microscope illumination optics are much more important

Future priorities for GCI • Non-targeted effects such as the bystander effect are of great interest to the radiation risk community • These effects may also be ‘exploitable’ in radiotherapy • The extent to which these effects are important in ‘real’ biological systems is still largely unknown • Studies that use tissue models and in vivo will be significant

Future priorities for GCI • Comparison and development of cell and tissue models • Microbeams with increased penetration • Investigate dependence on radiation quality • Non-invasive deep tissue imaging
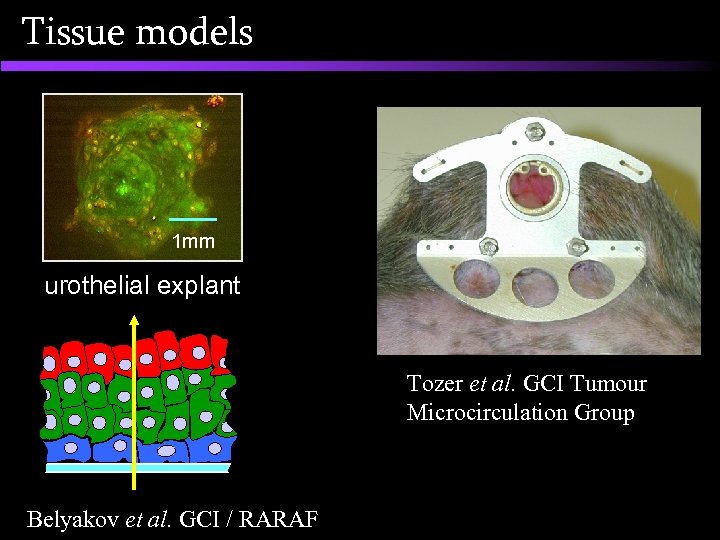
Tissue models 1 mm urothelial explant Tozer et al. GCI Tumour Microcirculation Group Belyakov et al. GCI / RARAF
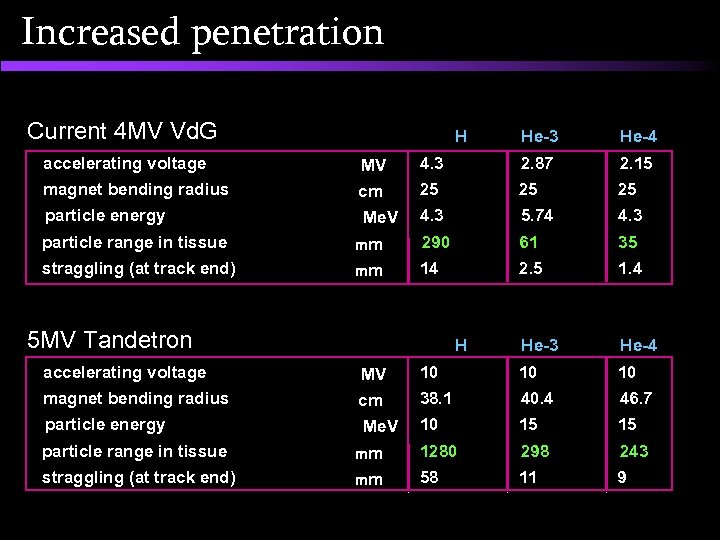
Increased penetration Current 4 MV Vd. G H He-3 He-4 accelerating voltage MV 4. 3 2. 87 2. 15 magnet bending radius cm 25 25 25 particle energy Me. V 4. 3 5. 74 4. 3 290 61 35 14 2. 5 1. 4 He-3 He-4 particle range in tissue straggling (at track end) mm mm 5 MV Tandetron H accelerating voltage MV 10 10 10 magnet bending radius cm 38. 1 40. 4 46. 7 particle energy Me. V 10 15 15 1280 298 243 58 11 9 particle range in tissue straggling (at track end) mm mm
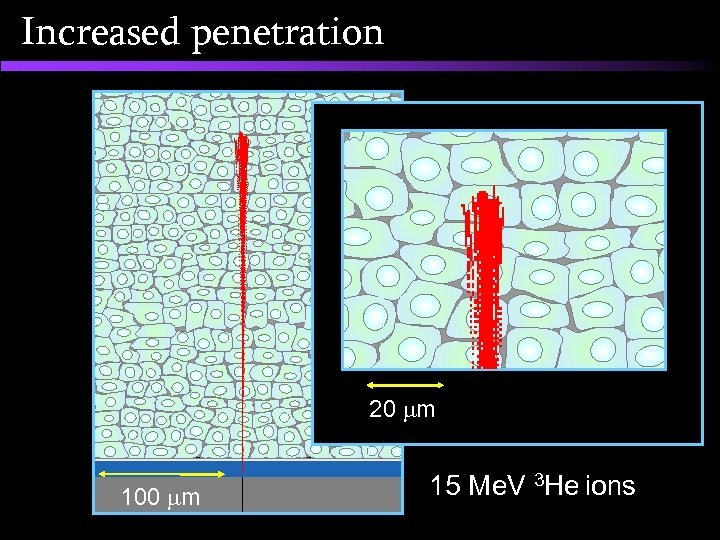
Increased penetration 20 m 100 m 15 Me. V 3 He ions

Radiation quality GCI soft X-ray microprobe
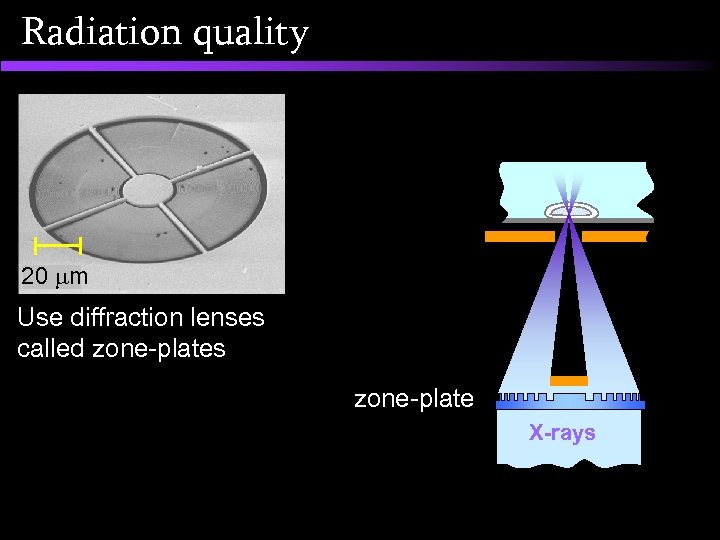
Radiation quality 20 m Use diffraction lenses called zone-plates zone-plate X-rays
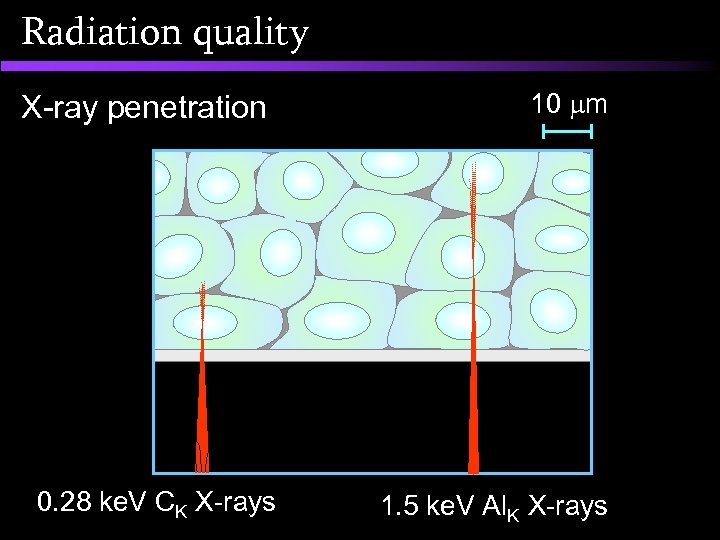
Radiation quality 10 m X-ray penetration 0. 28 ke. V CK X-rays 1. 5 ke. V Al. K X-rays
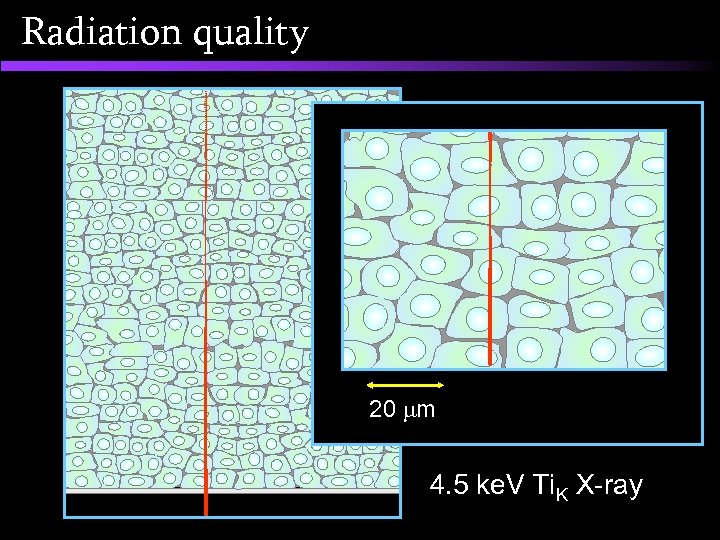
Radiation quality 20 m 4. 5 ke. V Ti. K X-ray
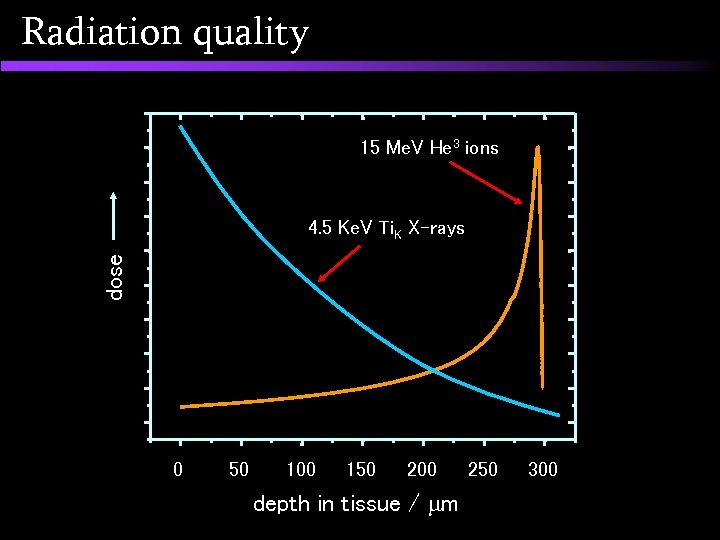
Radiation quality 15 Me. V He 3 ions dose 4. 5 Ke. V Ti. K X-rays 0 50 100 150 200 depth in tissue / m 250 300

Radiation quality The new GCI 4. 5 ke. V Ti. K X-ray microprobe

Deep tissue imaging • Scanning confocal system – Bio. Rad 1024 MP • Femtosecond multi-photon system • External detectors, time-resolved detection
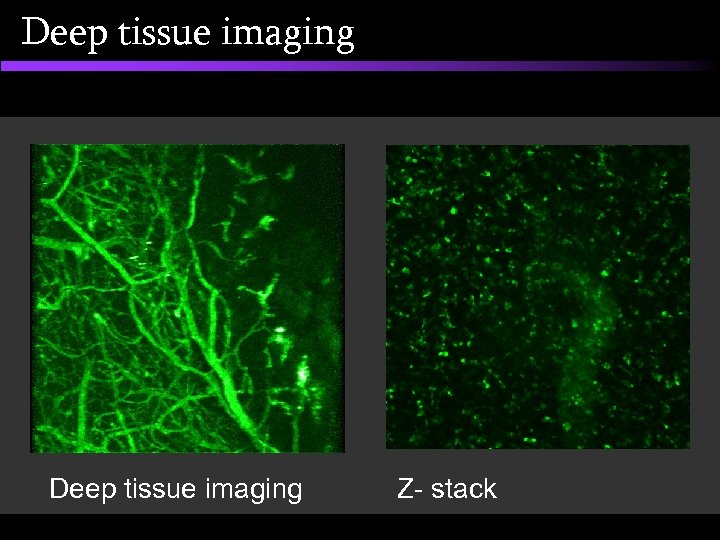
Deep tissue imaging Z- stack

Acknowledgements B. Vojnovic K. M. Prise K. Atkinson G. Schettino C. Shao A. G. Michette* P. Charalambous * S. Pfauntsch * * King’s College London S. Gilchrist R. Sunderland P. Barber R. Locke B. D. Michael Mech. & elec. workshops
3b9f680f8fe0daa7f5c0dd47841cfe39.ppt