77f55d2733a28340b111a8a5e7c6e315.ppt
- Количество слайдов: 24

The r. Amylase Project Faster, Better Biotech Ellyn Daugherty SMBCP, San Mateo CA www. Biotech. Ed. com www. SMBiotech. com www. emcp. com/biotech www. sargentwelch. com/biotech

Tulsa Community College February 22 -23, 2013 The r. Amylase Project (Faster, Better Biotech) Friday 2/22/12 5 -8 pm 5: 00 pm Amylase collection (before you eat – spit into a tube) 5: 15 pm Welcomes/Light dinner/Intro to The r. Amylase Project 5: 45 pm Set up Lab 6 c Amylase Assay 6: 45 pm Part I - Amylase ELISA Kit (Prepare/plate samples) 7: 45 pm Plan for Saturday (9 am sharp!) Sat 2/23/12 9 -4 pm 9: 00 am Part II - Amylase ELISA Kit (add Ab, colorimetric visualization) 10: 30 am Lab 8 b p. Amylase Restriction Digestion (kit) Prepare samples & set up for gel electrophoresis Run gels and visualize (1 XTAE gels vs 1 X LB gels) 11: 30 am Lab 5 f Amylase PAGE (set up gel, load, run gels during lunch) 12: 30 pm working Lunch (results & Intro to Column Chromatography) 1: 15 pm Lab 9 c Amylase Chromatography (kit) 3: 45 pm "What now? "

In Biotech, companies find a product of interest (amylase) and make sure to have assays to show it’s presence, activity, & concentration Amylase is an enzyme that catalyzes starch digestion. It is used commercially in several ways including: 1) Remove starch in products 2) Produce sugar from starch 3) Processing of cellulostic biofuel

The r. Amylase Project is the focus of the 2 nd semester of the SMBCP • Learn how to assay for a protein of commercial interest (amylase) • Transform cells to produce that protein (using p. Amylase and C 2523 cells) • Scale-up cells to volumes where amylase can be purified and assayed Ion Exchange Chromatography Absorbance (a. u) 0. 35 0. 3 0. 25 0. 2 0. 15 0. 1 0. 05 0 0 5 Fractions (#) 10
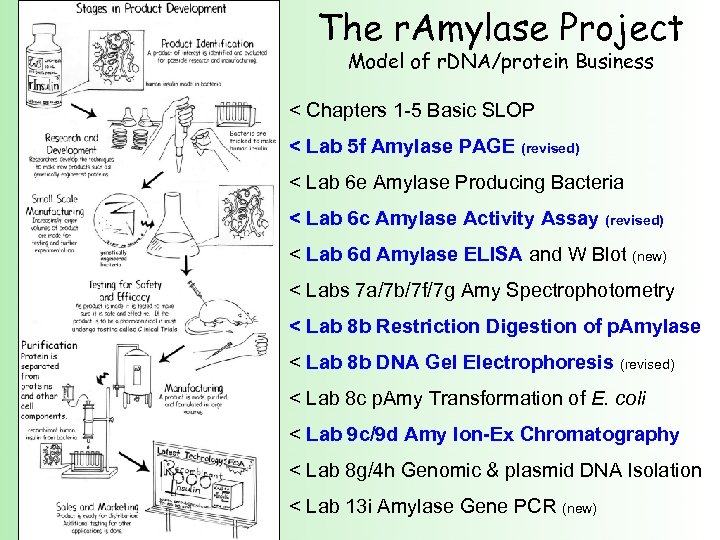
The r. Amylase Project Model of r. DNA/protein Business < Chapters 1 -5 Basic SLOP < Lab 5 f Amylase PAGE (revised) < Lab 6 e Amylase Producing Bacteria < Lab 6 c Amylase Activity Assay (revised) < Lab 6 d Amylase ELISA and W Blot (new) < Labs 7 a/7 b/7 f/7 g Amy Spectrophotometry < Lab 8 b Restriction Digestion of p. Amylase < Lab 8 b DNA Gel Electrophoresis (revised) < Lab 8 c p. Amy Transformation of E. coli < Lab 9 c/9 d Amy Ion-Ex Chromatography < Lab 8 g/4 h Genomic & plasmid DNA Isolation < Lab 13 i Amylase Gene PCR (new)
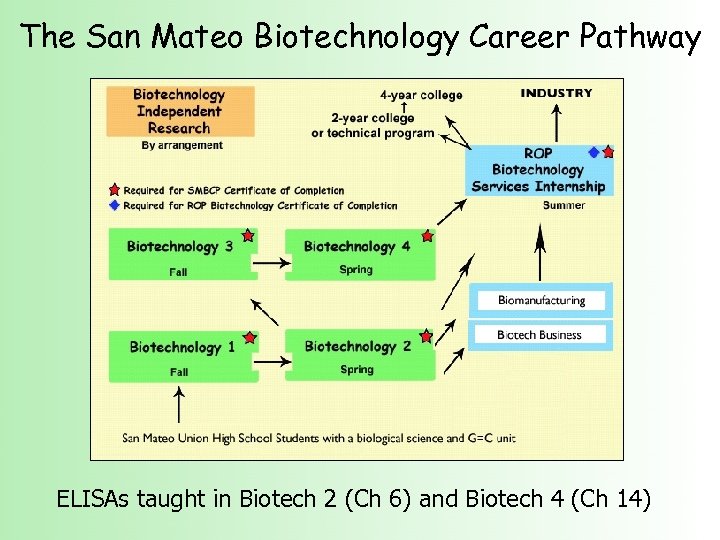
The San Mateo Biotechnology Career Pathway ELISAs taught in Biotech 2 (Ch 6) and Biotech 4 (Ch 14)

Lab 6 c (modified) Amylase Activity Assay Basic Procedure: • 2% (modified) starch is added to wells. • Amylase known or unknown is added to well. 3 min incubation. • Add Iodine to determine starch remaining.

Introduction to ELISA – short for enzyme-linked immunosorbant assay An ELISA uses antibodies with conjugated enzymes to specifically identify and measure the amount (concentration) of a protein in a solution
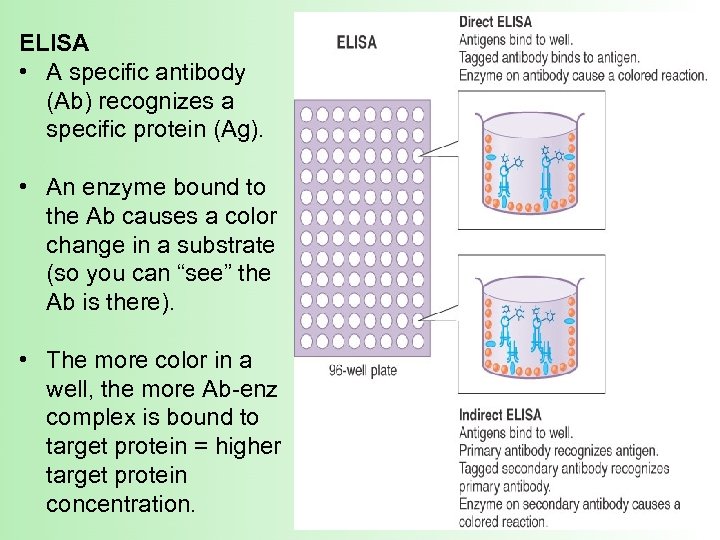
ELISA • A specific antibody (Ab) recognizes a specific protein (Ag). • An enzyme bound to the Ab causes a color change in a substrate (so you can “see” the Ab is there). • The more color in a well, the more Ab-enz complex is bound to target protein = higher target protein concentration.

Di

Lab 6 d Amylase Elisa Kit Expected Results <<<<< The Standards should look like this … …and the unknowns should look something like this >>>

Automated ELISA Plate Reader
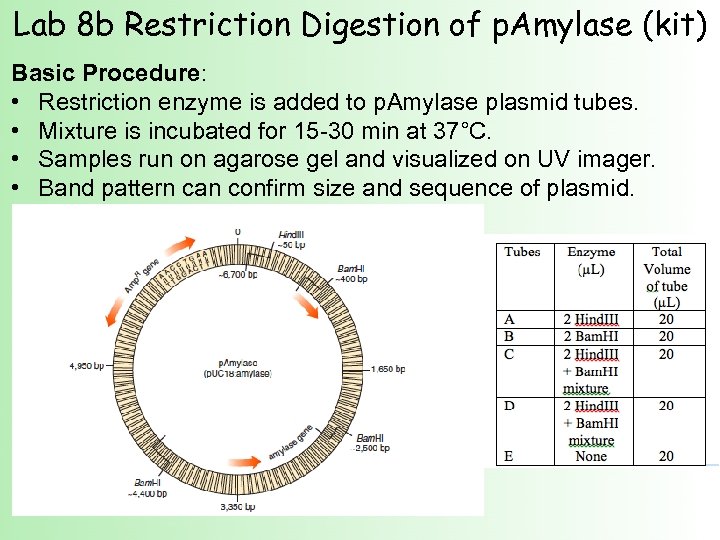
Lab 8 b Restriction Digestion of p. Amylase (kit) Basic Procedure: • Restriction enzyme is added to p. Amylase plasmid tubes. • Mixture is incubated for 15 -30 min at 37°C. • Samples run on agarose gel and visualized on UV imager. • Band pattern can confirm size and sequence of plasmid.

Fast Restriction Digestions (HF)and Fast Agarose Gels (LB) NEB High Fidelity (HF) Restriction Lithium Borate(LB) Buffers Enzymes http: //www. fasterbettermedia. com http: //www. neb. com Lithium borate conducts electricity These engineered enzymes have better than TRIS so you can turn up the same specificity as their the voltage and current on agarose established counterpart with the gels : benefit of reduced star activity and: • Fast gel run times (8 -15 min) • Fast digestion times (5 -15 min) • Same protocol as TAE gels just • Work in same buffer (Buffer 4) substitute 1 X LB buffer for 1 X TAE Bam. HI-HF™ • Purchase as 10 X or 20 X concentrate Hind. III-HF™

Lab 5 f (Modified) Amylase on vertical SDS-PAGE Basic Procedure: • Prepare diluted samples of known amylase concentration with SPLD/BME. • Load and run on Nu. Sep gels at 200 -250 V ! for 30 min. • View immediately on UV imaging system and/or stain with Coomassie blue (long or short method).
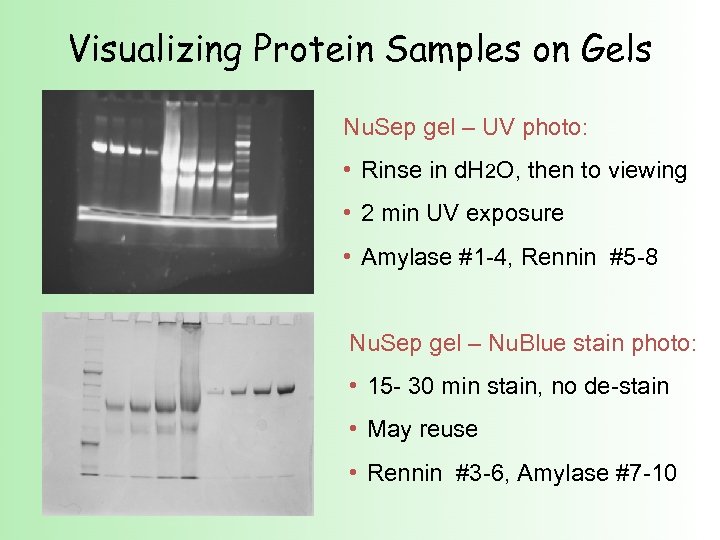
Visualizing Protein Samples on Gels Nu. Sep gel – UV photo: • Rinse in d. H 2 O, then to viewing • 2 min UV exposure • Amylase #1 -4, Rennin #5 -8 Nu. Sep gel – Nu. Blue stain photo: • 15 - 30 min stain, no de-stain • May reuse • Rennin #3 -6, Amylase #7 -10
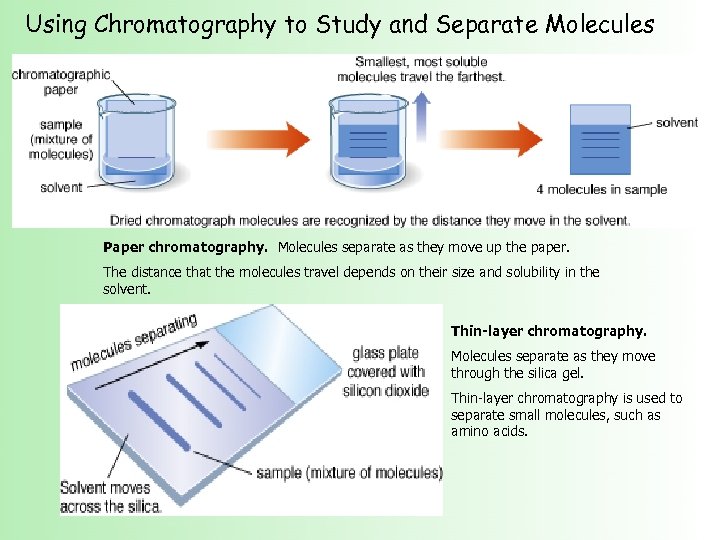
Using Chromatography to Study and Separate Molecules Paper chromatography. Molecules separate as they move up the paper. The distance that the molecules travel depends on their size and solubility in the solvent. Thin-layer chromatography. Molecules separate as they move through the silica gel. Thin-layer chromatography is used to separate small molecules, such as amino acids.
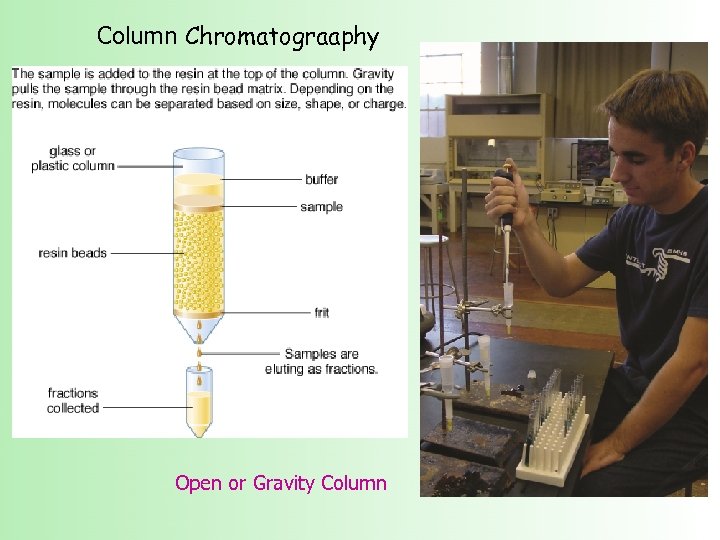
Column Chromatograaphy Open or Gravity Column
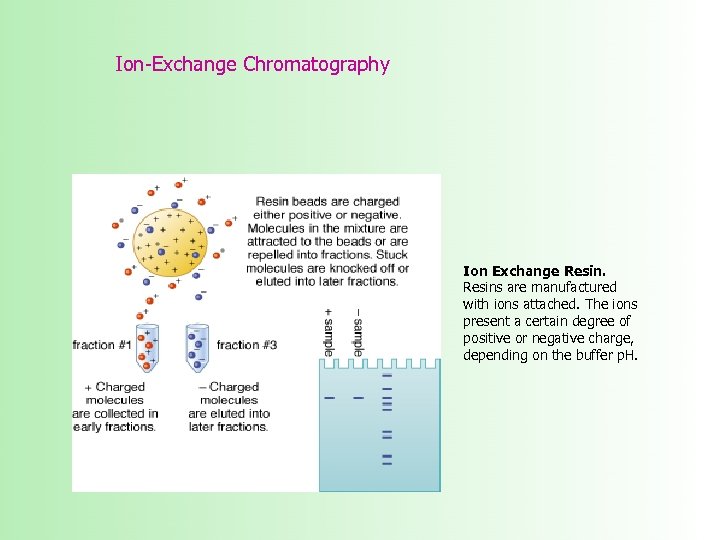
Ion-Exchange Chromatography Ion Exchange Resins are manufactured with ions attached. The ions present a certain degree of positive or negative charge, depending on the buffer p. H.
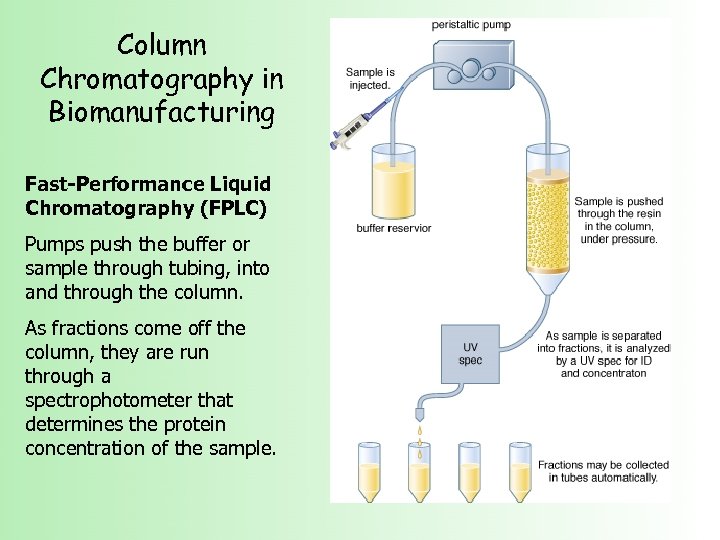
Column Chromatography in Biomanufacturing Fast-Performance Liquid Chromatography (FPLC) Pumps push the buffer or sample through tubing, into and through the column. As fractions come off the column, they are run through a spectrophotometer that determines the protein concentration of the sample.

Column Chromatography in Biomanufacturing Fast-Performance Liquid Chromatography (FPLC)

High-Performance Liquid Chromatography (HPLC) Greatly improved ability to separate, purify, identify, and qualify samples.

Get more help… SMBCP Link IRC Link SW Lists Current Events LN’s workshops New Things/Upd ates All of LN’s PPTs
77f55d2733a28340b111a8a5e7c6e315.ppt