a2a2b37b62e3286329d31bd59be31de8.ppt
- Количество слайдов: 42

The Plague (Y. Pestis) Eti Mezei, Elodi Dielubanza, Clara Boyd

Overview - 3 Types of Plague Bubonic Pneumonic - Most common form - Generally secondary complication of - Responsible for most bubonic plague though can be a plague epidemics primary infection - Diagnosis is more difficult Septicemic - Generally secondary complication of bubonic plague though can be a primary infection - Poses the greatest burden to the human system of the 3 types - Easily communicable All types of plague are caused by the gram-negative bacteria Yersinia Pestis

Epidemiology Historical & Modern Aspects Reservoirs, Vectors Transmission World History Discoveries Modern Plague Trends in the USA
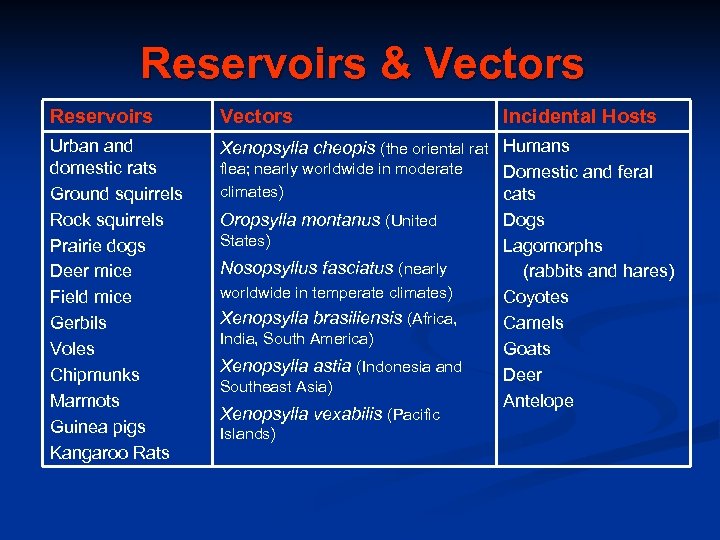
Reservoirs & Vectors Reservoirs Vectors Urban and domestic rats Ground squirrels Rock squirrels Prairie dogs Deer mice Field mice Gerbils Voles Chipmunks Marmots Guinea pigs Kangaroo Rats Xenopsylla cheopis (the oriental rat Humans flea; nearly worldwide in moderate Domestic and feral climates) cats Dogs Oropsylla montanus (United States) Lagomorphs Nosopsyllus fasciatus (nearly (rabbits and hares) worldwide in temperate climates) Coyotes Xenopsylla brasiliensis (Africa, Camels India, South America) Goats Xenopsylla astia (Indonesia and Deer Southeast Asia) Antelope Xenopsylla vexabilis (Pacific Islands) Incidental Hosts

Modes of Transmission ■ Fleabite ■ Inhalation of salivary droplets of infected humans or cats ■ Cat’s scratch ■ Ingestion of the bacillus ■ Contact with infected body fluids

Transmission & Type of Plague Bubonic 1 Septicemic Y. Pestis enters the lymphatic system through the skin Direct inoculation of Y. Direct inhalation or Pestis into ingestion of Y. Pestis bloodstream Bite from infected flea 2 --------- Bite from infected flea; Scratch from infected cat; Contact with Infected Fluids As a complication of bubonic or 1 pneumonic plague Pneumonic Inhaling droplets from cough of infected human/cat; Eating infected animal; Inhaling Y. Pestis in the lab Via hematogenous spread, as a complication of bubonic or septicemic plague
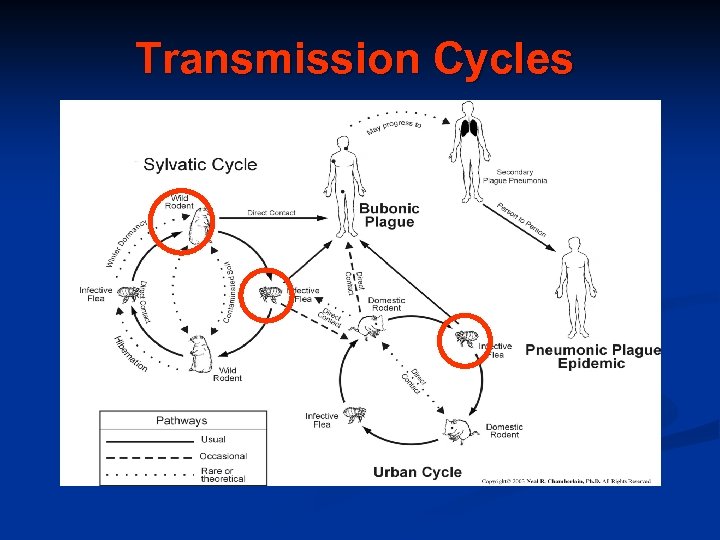
Transmission Cycles

Plague and World History n 1320 B. C. -- 1 st mention of plague is in the Bible Philistines stole the ark of the covenant from the Israelites and plague ensued: [t]he Lord’s hand was heavy upon the people of Ashdod and its vicinity. He brought devastation upon them and afflicted them with tumors. And rats appeared in their land, and death and destruction were throughout the city…[T]he Lord’s hand was upon that city, throwing it into great panic. He afflicted both young and old with and out break of tumors in the groin.

Plague and World History 541 -700 A. D. – 1 st Pandemic, Justinian Plague ■ Began in Pelusium Egypt and spread to the middle East & Europe with estimated population losses of 50 -60% in N. Africa, Europe & S. Asia. ■ A. D. 558 -654: The 2 nd through 11 th epidemics occurred in 8 -12 year cycles.

Plague and. Europe along trade History World Plague believed to have entered 1346 -1666 – 2 nd Pandemic, Black Death n routes from central Asia by fleas in bundles of imported fur. n 1347 -1351: 1 st 5 -year epidemic killed an estimated 17 -28 million people, 30 -40% of Europe’s population at the time. n Epidemics continued throughout the period in 2 -5 year cycles until 1480 and then with less frequency until late 17 th century. n The plague stimulated significant advance in medical practice including the beginnings of clinical research, reorganization of hospitals. A Plague Doctor NOTE: other epidemics existing at the time of the 1 st and 2 nd Plague pandemics may have contributed to depopulation figures

Plague and World History 1855 – 3 rd Pandemic, China ■ Began in the Yünnan province and troops from the war in that area helped to spread plague down the southern coast to Hong Kong and Canton by 1894 and Bombay by 1898. n By 1900 steamships had helped to spread the disease to Africa, Austrailia, Europe Hawaii, India, Japan, the Middle East, the Phillipines, the U. S, and S. America. By 1903, 1 million people/year were dying of plague in India. Mukden, China, 1910 -11: Workers in a plague hospital hose off an autopsy table with carbolic spray

Plague and World History 1855 – 3 rd Pandemic, China (cont’d) n Stable enzootic foci were established on every inhabited continent except Austrailia and outbreaks continue until today though greatly reduced because of advances in public health practices and drugs. n Significantly, it is during the 3 rd Pandemic that the bacterium responsible for plague as well as its vectors and reservoirs were identified.

History of Yersina Pestis n First discovered in 1894 during the Hong Kong Plague by two independent investigators (Alexandre Yersin and Shibasaburo Kitasato) within days of each other. n Kitasato was initially credited for the discovery but Yersin’s bacillus proved to be the infecting bacillus. n Yersin drained buboes of deceased plague victims and identified the bacillus with microscopy. n Confirmed the involvement of the bacillus in infection by injecting healthy rodents with infected lymph isolates. Healthy animals developed plague symptoms and dissection showed blood and organs to be filled with the isolated bacillus.

Y n Nomenclature: the plague bacillus had four name changes. Classifications: Bacterium Pestis until 1900 Bacillus Pestis until 1923 Pasturella Pestis (after Yersin’s mentor) Yersinia Pestis in 1970 until present day

Identification of Vectors and Reservoirs ■ Yersin also confirmed the Black rat (Rattus rattus) as the reservoir after local suspicion implicated its involvement in the transmission of the plague. ■ In 1897 during the Indian outbreak, Paul- Louis Simond and Masanori Ogata identified the oriental rat flea (Xenopsylla Cheopis) as the transmission vector by exposing healthy rodents to fleas collected from the corpses of rats which had died recently from plague.

Early Prevention and Treatment Methods n Yersin isolated serum from horses immunized with the bacillus for treatment of human plague in 1896. n W. M Haffkine created and used an effective preventative vaccine for the Plague containing killed bacteria during the Manchurian outbreak (1910). n L. T Wu characterized pneumonic plague during the 1910 Manchurian outbreak and helped to institute protective measures against aerosol spread of the disease.

Modern Plague n For most of the 20 th century the occurrence of plague has sharply decreased though not disappeared. Plague remains an enzootic infection of rats, ground squirrels and other rodents in every inhabited continent except Austrailia. n Why has Plague decreased so sharply? The WHO credits public health prevention protocols and the development of effective antibiotics.

Modern Plague n WHO reports 1000 -3000 cases of plague worldwide annually (avg. 1700 cases/year for the last 50 years) (conservative estimate) n Plague is largely underreported in countries with limited surveillance and laboratory capabilities. n Modern plague is primarily occurrent in rural areas with poor sanitation and large rodent populations. n Urban cases are increasingly rare. Los Angeles 1924 was the last U. S urban outbreak.
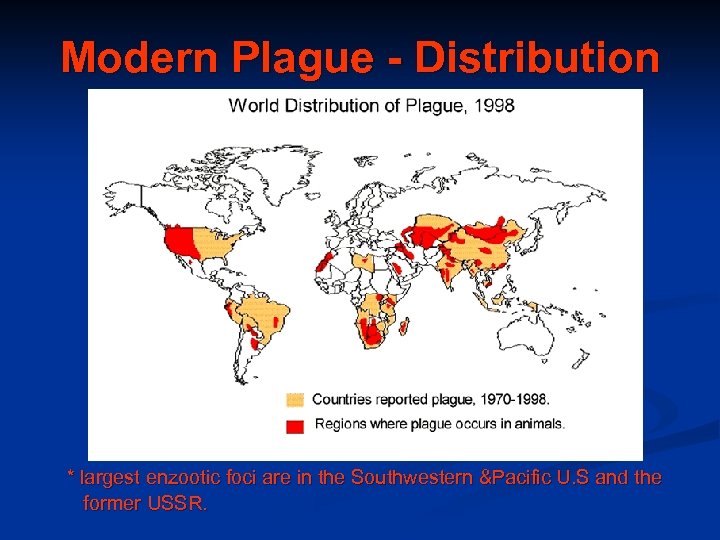
Modern Plague - Distribution * largest enzootic foci are in the Southwestern &Pacific U. S and the former USSR.
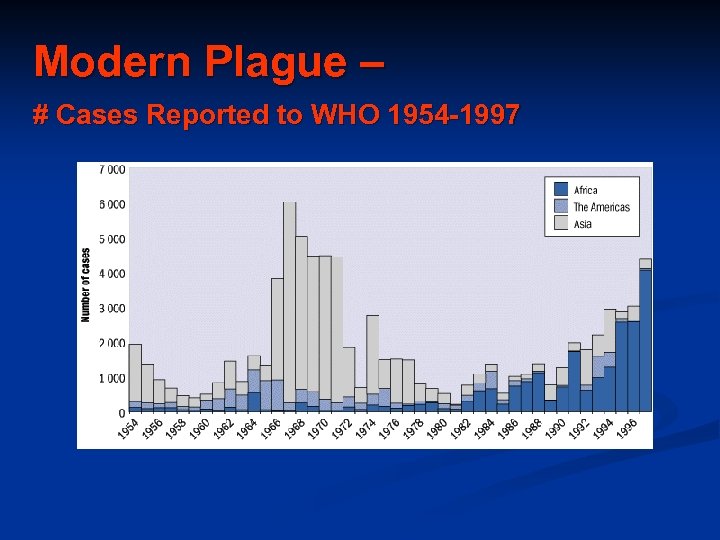
Modern Plague – # Cases Reported to WHO 1954 -1997

Modern Plague - Timeline n 1900 - Plague officially arrives in the U. S. Infected corpse of Chinese Laborer found in San Francisco hotel basement. n 1924 - Los Angeles. 33 cases, 31 fatal. 1 st case, Mexican American male, misdiagnosed with STD, family and neighbors contract plague and die with in two weeks. Many die before public health measures were taken. n 1967 -72 - Vietnam. Defoliation, disturbance of the ecosystem and economic losses during the war contributed to outbreak. Singular contributor to the 1967 jump in worldwide plague vases. n 1992 - Arizona. 1 confirmed case, 1 fatality. Misdiagnosis of pneumonia. Post-mortem lab tests confirmed Y. Pestis. Family cat identified as source.

Timeline Cont’d ■ 1994 – India. Bubonic plague begins when rats arrive because of stockpiles of relief grain. Pneumonic form shortly follows. 5150 suspected pneumonic and bubonic cases from 26 states. 163 confirmed with serology, 53 confirmed deaths, 300 suspected. Mass panic; 600, 00 flee Surat including 110 plague cases.

Timeline Cont’d ■ 1997 Zambia- Jan. Namwala region 90 cases of bubonic plague, 22 fatal. Heavy rains drove rats into inhabited regions. Mozambique - Aug. Tete province. 115 reported cases of bubonic. No reported deaths. Malawi - Oct. Southern region, 43 reported cases, 17 of which were seropostive. >60% cases were children. No reported deaths. Mozambique - Nov. August outbreak continues and extends to 225 cases. No reported deaths. ■ 1999 Namibia - May Northwest region, 39 reported cases, 8 deaths. Malawi - July Reported cases in 22 villages, 74 suspected cases total. No reported deaths.

Timeline Cont’d ■ 2001 Zambia - Mar. Nyanje and Petauke regions 436 suspected cases, 11 deaths. Y. Pestis postively identified. ■ 2002 India - Feb. Hat Koti village. 16 cases of pneumonic plague including 4 deaths. Cases linked to one villager. Malawi- June. Nsanje region and 26 nearby villages. 71 reported cases of bubonic plague. ■ 2003 New York - Nov. 2 cases. Couple contracts plague in Santa Fe, NM and travels to New York. Neither case fatal but man required double foot amputation.

Plague Patient Returns Home NEW YORK, Feb. 10, 2003 A New Mexico man who was hospitalized in New York City for more than three months with bubonic plague left the hospital to fly home on Monday, a spokesman said. John Tull left Beth Israel Medical Center at about 7 a. m. , hospital spokesman Mike Quane said. Tull was admitted to Beth Israel on Nov. 5. Tull, whose feet were amputated due to extensive tissue damage, will begin physical therapy in Albuquerque, N. M. , Quane said. John Tull at the Beth Israel Hospital in New York City with his wife Lucinda Marker. Disease investigators believe Tull and his wife, Lucinda Marker, contracted plague from infected fleas on their Santa Fe, N. M. , ranch. They became ill after arriving in New York on Nov. 1 for vacation. CBSNEWS. com

Trends in Human Plague in the U. S. n From 1899 -1926 Plague in the U. S. was an urban epidemic involving domesticated rats with cases most prevalent in California and Hawaii. n Late 1940’s saw a jump in infection in the Southwestern states which remain hotspots for infection. n Static annual infection levels from 1925 -1964 (avg. <2 cases/year). 1965 saw increases that would carry into the 80’s.

Trends in Human Plague in the U. S n Seasonal distribution: 1926 -1979 81. 5% of plague cases occurred in May-September. Prior to 1926 most were Sept. -Oct. n 1970 -1980 53% cases ♀ 59% were < 20 years old. Similar age distribution from 1926 -1969 n Racial dist. : 1970 -79 35% of cases were in Indians (1. 4 Indians per 100, 00 compared to 0. 1 non-Indians).

Simplified Pathogenesis (Focus on Bubonic Plague)

Pathogenesis “Blocked” Flea feeds on Y. Pestis-infected blood Y. Pestis enters flea’s midgut & multiplies logarithmically Clump of Y. Pestis & fibronous material forms in the midgut, blocking flea’s proventriculus During next meal, blood cannot enter the midgut & flea gets very hungry Flea bites vigorously & regurgitates the contents of its midgut into the next wound
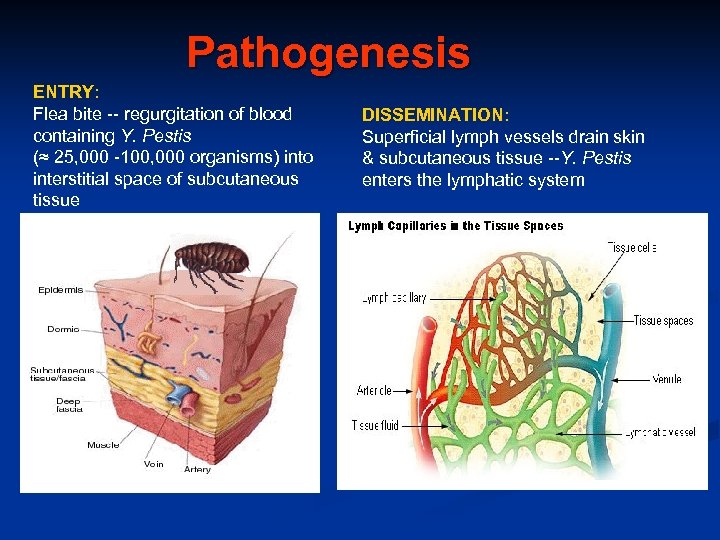
Pathogenesis ENTRY: Flea bite -- regurgitation of blood containing Y. Pestis (≈ 25, 000 -100, 000 organisms) into interstitial space of subcutaneous tissue DISSEMINATION: Superficial lymph vessels drain skin & subcutaneous tissue --Y. Pestis enters the lymphatic system
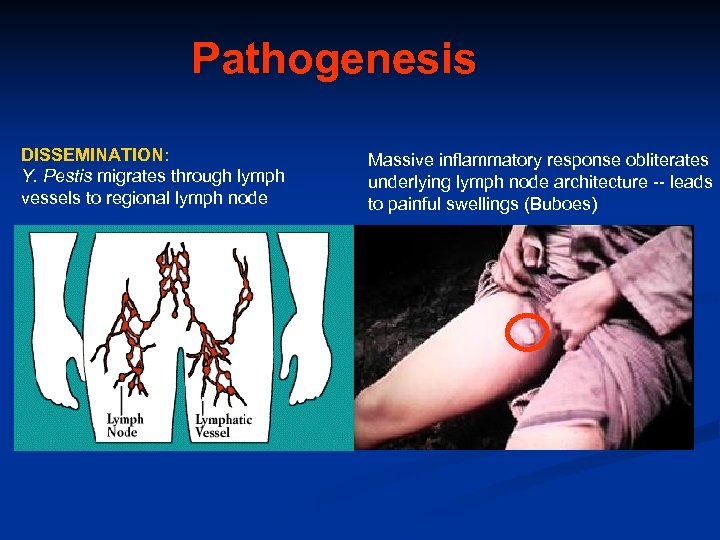
Pathogenesis DISSEMINATION: Y. Pestis migrates through lymph vessels to regional lymph node Massive inflammatory response obliterates underlying lymph node architecture -- leads to painful swellings (Buboes)
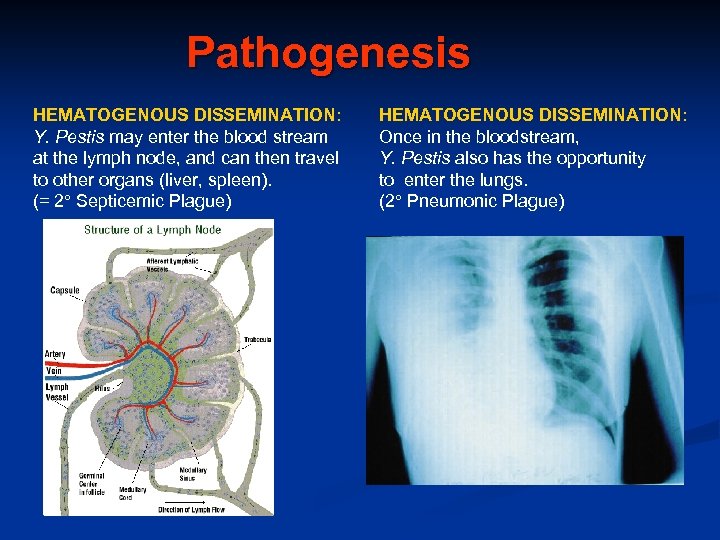
Pathogenesis HEMATOGENOUS DISSEMINATION: Y. Pestis may enter the blood stream at the lymph node, and can then travel to other organs (liver, spleen). (= 2 Septicemic Plague) HEMATOGENOUS DISSEMINATION: Once in the bloodstream, Y. Pestis also has the opportunity to enter the lungs. (2 Pneumonic Plague)
Molecular Biology of Yersinia Pestis Bacteriological Characteristics Evolution Biovars Genome Molecular Pathogenesis Proteins/Virulence Factors/ Immunology

Bacteriological Characteristics n Gram negative n Non-motile n Enterobacteriacae n Non-spore forming coccobacillus n Facultative anaerobe n Obligate parasite
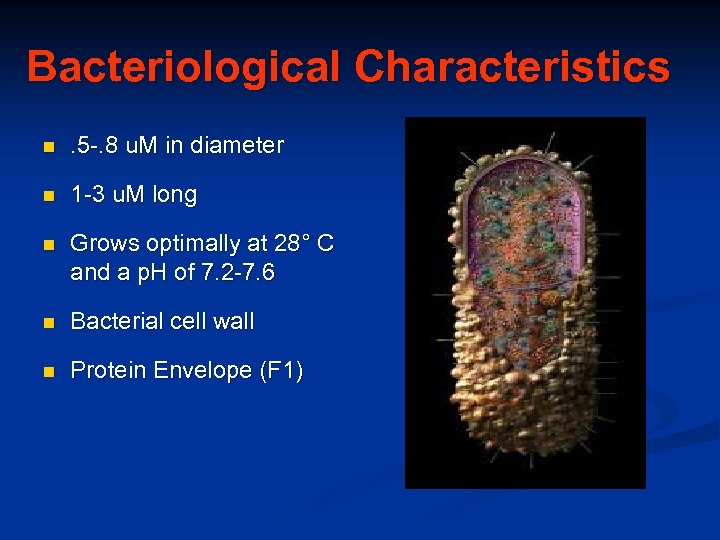
Bacteriological Characteristics n . 5 -. 8 u. M in diameter n 1 -3 u. M long n Grows optimally at 28° C and a p. H of 7. 2 -7. 6 n Bacterial cell wall n Protein Envelope (F 1)

Evolution of Y. Pestis There are 11 species of Yersinia 3 pathogenic species of Yersinia n n Yersinia Pseudotuberculosis - enteropathogen Yersinia Enterocolitis - enteropathogen Yersinia Pestis - systemic pathogen n n Y. Pestis evolved from Y. pseudotuberculosis 1500 -4000 years ago n n 90% chromosomal DNA relatedness Physiological and antigenic similarities

Y. Pseudotuberculosis n Disease n Enteric infection n Transmission n Enters mammals through food and water n n Y. Pestis n Disease n Bubonic Plague n Transmission n Flea, mammal, never found in free environment Genes n Yad. A, Inv, O antigen n Pseudogenes n Yad. A, Inv, O antigen Extrachromosomal DNA n p. CD 1 n p. Fra n p. Pst n Pathogenicity Islands

Biovars of Y. Pestis n 3 biovars named based on their ability to convert nitrate to nitrite and ferment glycerol n n n Glycerol Antiqua (1 st pandemic) + + Medievalis (2 nd pandemic) Orientalis (3 rd pandemic) Nitrite + +

Genome n 4500 genes n One chromosome- 4. 6 Mb n Pathogenicity islands (pgm locus) n Three plasmids n p. Fra – 96. 2 kb n p. Pst 1 - 9. 7 kb n p. CD 1 - 70. 3 kb

Chromosome Pgm locus n 102 kb pathogenicity island in the chromosome n contains three hemin storage genes (Hms) n n n These genes only found in Y. pestis They are essential for flea blockage Allow Y. Pestis to store large quantities of hemin (iron source) in its outer membrane n Bacteria requires iron in order to cause infection n An inorganic iron transport system (ybt) (a sidephore) is encoded within Pgm locus n Ybt chelates iron bound to eukaryotic proteins and transports them to the bacterium

p. Fra n 60 -280 kb (depending on the strain) n Found only in Y. Pestis n Contains gene for murine toxin (Ymt) n Two forms of a protein toxic for mice and rats n Proposed as B-adrenergic antagonist n Ymt transcription is threefold higher at 26°C(flea) than at 37°C(mammal). n This gene is believed to be essential for flea colonization

p. Fra n Contains structural and regulatory genes that encodes F 1 protein capsule (caf 1) n F 1 is a proteinacious capsule that forms at 37°C (mammal) but not 26°C (flea) n F 1 capsule makes bacterium resistant to phagocytosis by monocytes n Resistance to phagocytosis by monocytes allows journey from dermis to viscera
a2a2b37b62e3286329d31bd59be31de8.ppt