8bc84f42310cfc06b77e87ef9128ab99.ppt
- Количество слайдов: 25
 The Otto Cycle, The Refrigerator, Polarization of Light
The Otto Cycle, The Refrigerator, Polarization of Light
 The Otto Cycle l Internal Combustion Engine – Uses burning fuel as a heat source. – Can be modeled as a heat engine l Gas Engines usually have 4 to 8 cylinders
The Otto Cycle l Internal Combustion Engine – Uses burning fuel as a heat source. – Can be modeled as a heat engine l Gas Engines usually have 4 to 8 cylinders
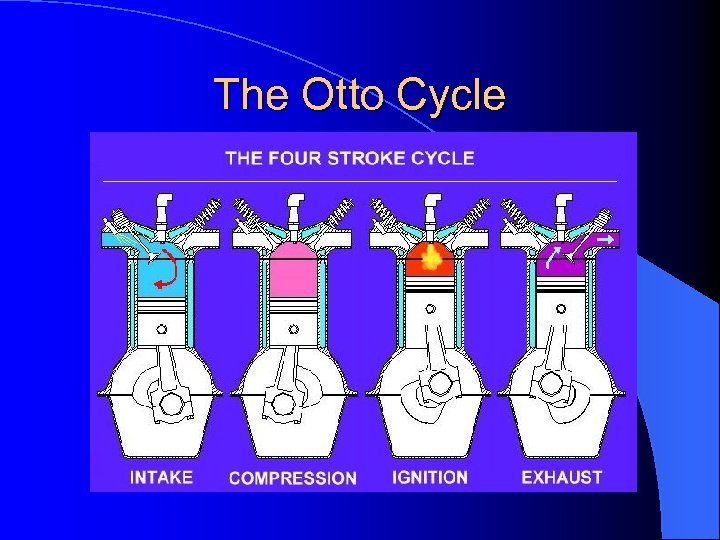 The Otto Cycle
The Otto Cycle
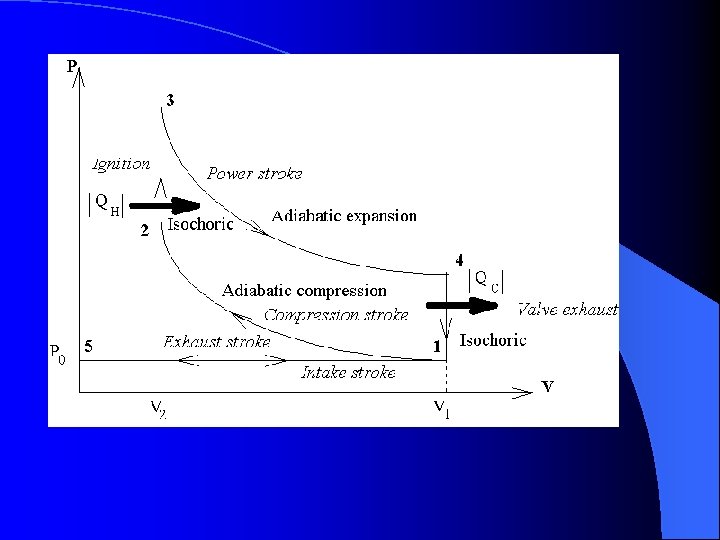
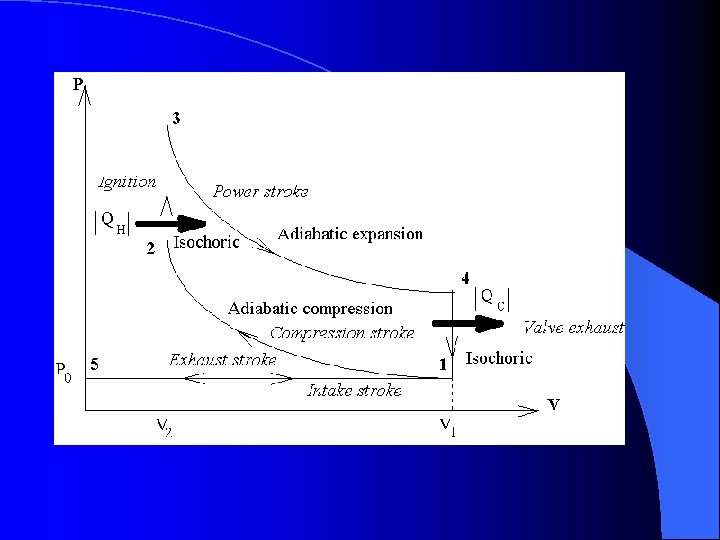
 The Otto Cycle l Step 1 – Intake and Compression – The process is considered adiabatic due to no addition or loss of heat. (Q = 0) – The work done on the gas is equal to the area under the curve.
The Otto Cycle l Step 1 – Intake and Compression – The process is considered adiabatic due to no addition or loss of heat. (Q = 0) – The work done on the gas is equal to the area under the curve.
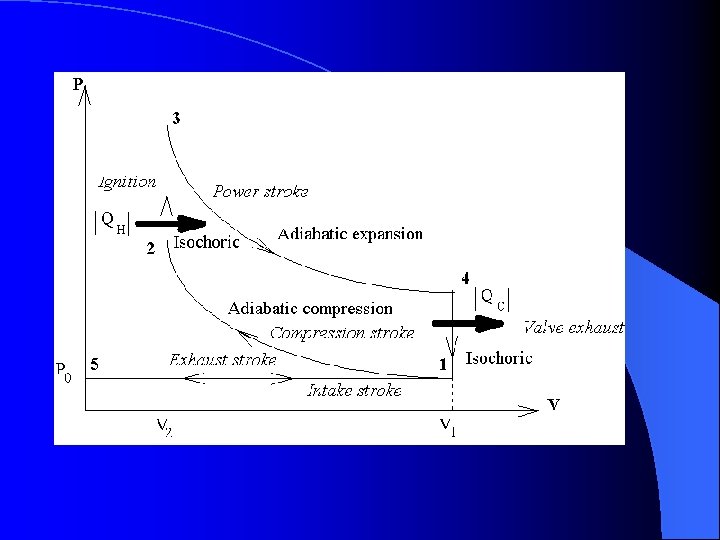
 The Otto Cycle l Now ignition occurs – The gas has been compressed and the spark plug fires – Heat has been added to the gas – Pressure skyrockets but the piston has not moved yet. l Points 2 to 3 are Isochoric/isovolumetric
The Otto Cycle l Now ignition occurs – The gas has been compressed and the spark plug fires – Heat has been added to the gas – Pressure skyrockets but the piston has not moved yet. l Points 2 to 3 are Isochoric/isovolumetric
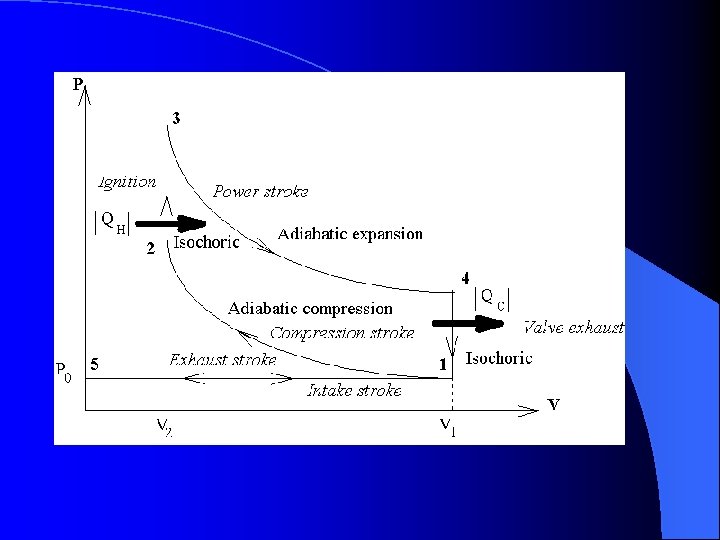
 The Otto Cycle l Expansion occurs – The compressed gas under high pressure pushes the piston downward – The pressure decreases while the volume increases – Points 3 to 4 are adiabatic due to little heat gain or loss
The Otto Cycle l Expansion occurs – The compressed gas under high pressure pushes the piston downward – The pressure decreases while the volume increases – Points 3 to 4 are adiabatic due to little heat gain or loss
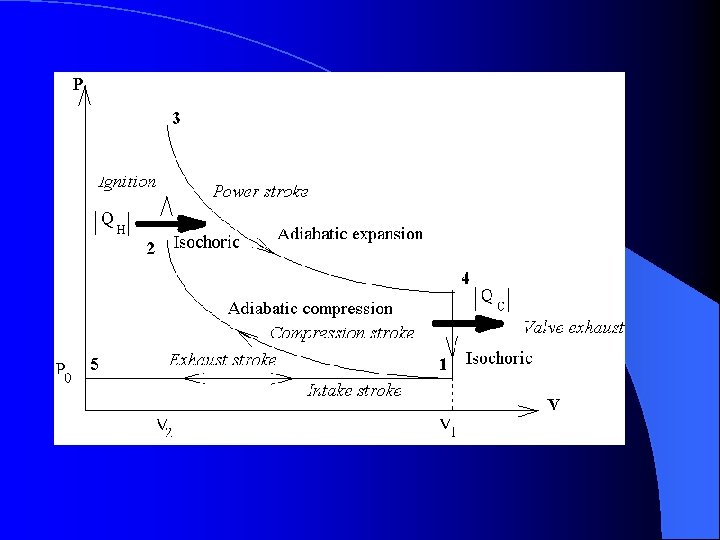
 The Otto Cycle l Cooling occurs – The gas cools and is expelled out through the exhaust valve. – The intake valve opens and fresh fuel/air mix is taken in and the process repeats – Steps 4 to 5 are isochoric due to no change in volume
The Otto Cycle l Cooling occurs – The gas cools and is expelled out through the exhaust valve. – The intake valve opens and fresh fuel/air mix is taken in and the process repeats – Steps 4 to 5 are isochoric due to no change in volume
 Example I l. A particular heat engine does 20, 000 J of work on a gas during the compression stroke, and the gas does 80, 000 J of work during the power stroke. Draw a PV diagram that indicated the net work done by the heat engine in one complete cycle
Example I l. A particular heat engine does 20, 000 J of work on a gas during the compression stroke, and the gas does 80, 000 J of work during the power stroke. Draw a PV diagram that indicated the net work done by the heat engine in one complete cycle
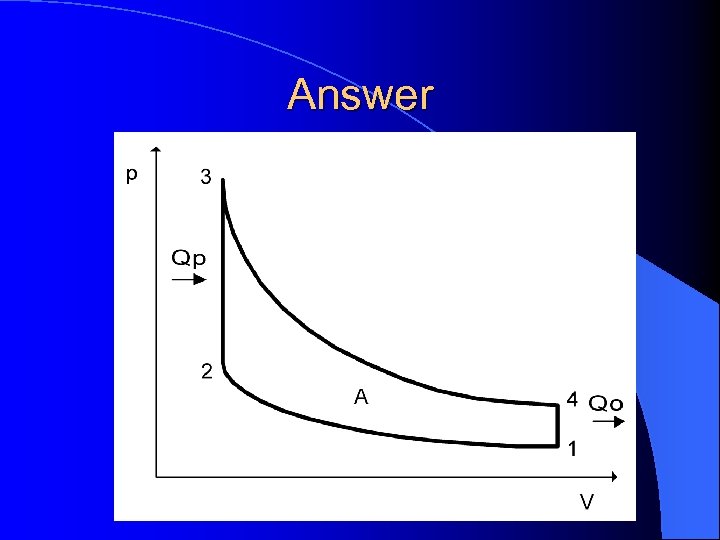 Answer
Answer
 The Refrigerator l Heat engines work between heat flow between TH and TC l Some of the input is converted to work while the rest is exhausted as wasted heat
The Refrigerator l Heat engines work between heat flow between TH and TC l Some of the input is converted to work while the rest is exhausted as wasted heat
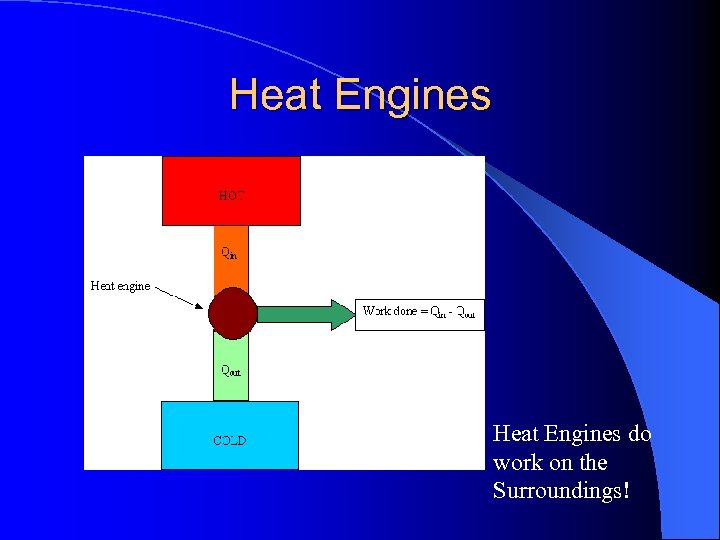 Heat Engines do work on the Surroundings!
Heat Engines do work on the Surroundings!
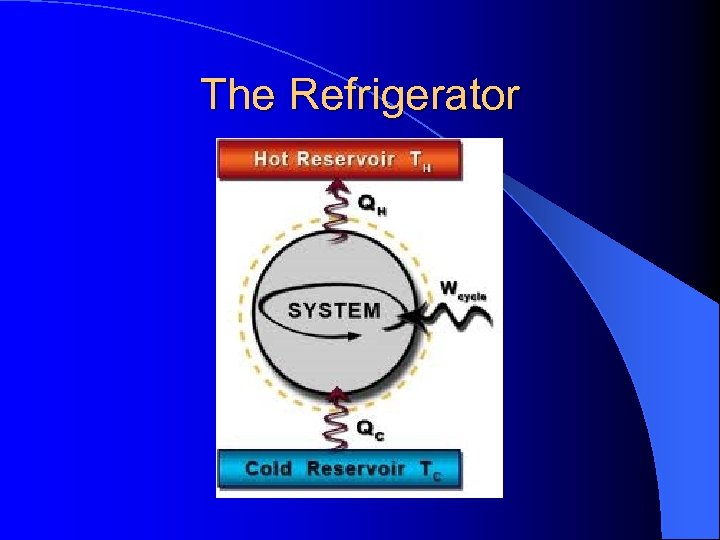
 The Refrigerator l Is the backward process of a heat engine! – The surroundings do work on the fluid in a heat engine. – Energy flows backwards or uphill from cold to hot.
The Refrigerator l Is the backward process of a heat engine! – The surroundings do work on the fluid in a heat engine. – Energy flows backwards or uphill from cold to hot.
 The Refrigerator
The Refrigerator
 The Refrigerator l The low temperature reservoir is the inside of the refrigerator and the high temperature reservoir is the air outside the refrigerator. l For an air conditioner, the cold reservoir is inside the house and the hot reservoir is outside the house.
The Refrigerator l The low temperature reservoir is the inside of the refrigerator and the high temperature reservoir is the air outside the refrigerator. l For an air conditioner, the cold reservoir is inside the house and the hot reservoir is outside the house.
 The Refrigerator l The key components – Pump and expansion valve – Work is done by compressing the freon and blowing air across the coils releasing heat to the kitchen or outside the house
The Refrigerator l The key components – Pump and expansion valve – Work is done by compressing the freon and blowing air across the coils releasing heat to the kitchen or outside the house
 The Refrigerator l The freon enters to the inside of the refrigerator and passes through an expansion valve. l The freon expands (endothermic process) and absorbs heat from the inside of the fridge. Thus the inside of the fridge cools off.
The Refrigerator l The freon enters to the inside of the refrigerator and passes through an expansion valve. l The freon expands (endothermic process) and absorbs heat from the inside of the fridge. Thus the inside of the fridge cools off.
 Coefficient of Performance l CP = Qc/W = Qc/(QH-Qc) l CPideal = Tc/(TH-TC)
Coefficient of Performance l CP = Qc/W = Qc/(QH-Qc) l CPideal = Tc/(TH-TC)
 The reverse? ? l Called a heat pump? ? – Your house heater – CP = QH/W = QH/(QH-QC)
The reverse? ? l Called a heat pump? ? – Your house heater – CP = QH/W = QH/(QH-QC)
 Example II l An ideal air conditioner requires 1500 W of power to remove 3000 J of heat from the inside of a house each second. The outside temperature is 28°C. a) How much heat is moved to the outside each second? B) What is the CP value of the AC unit? C) What is the lowest temperature the AC could possibly make the inside of the house? D) If the AC is turned around in the winter and used as a heat pump, what would be the coefficient of performance for it, if all else remains the same?
Example II l An ideal air conditioner requires 1500 W of power to remove 3000 J of heat from the inside of a house each second. The outside temperature is 28°C. a) How much heat is moved to the outside each second? B) What is the CP value of the AC unit? C) What is the lowest temperature the AC could possibly make the inside of the house? D) If the AC is turned around in the winter and used as a heat pump, what would be the coefficient of performance for it, if all else remains the same?
 Answer l 4500 J, 2. 0, 201 K or -72°C, 3. 0
Answer l 4500 J, 2. 0, 201 K or -72°C, 3. 0


