7d9652782ccc8ad44e818502c4f4a90b.ppt
- Количество слайдов: 24
 The Medtronic Ventor Engager. TM TAVI System Hendrik Treede, MD University Heart Center Hamburg
The Medtronic Ventor Engager. TM TAVI System Hendrik Treede, MD University Heart Center Hamburg
 Hendrik Treede, MD I have no real or apparent conflicts of interest to report.
Hendrik Treede, MD I have no real or apparent conflicts of interest to report.
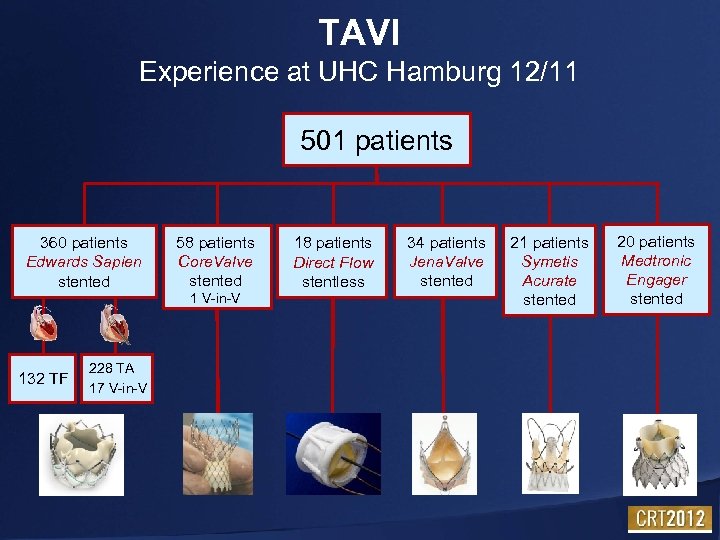 TAVI Experience at UHC Hamburg 12/11 501 patients 360 patients Edwards Sapien stented 58 patients Core. Valve stented 1 V-in-V 132 TF 228 TA 17 V-in-V 18 patients Direct Flow stentless 34 patients Jena. Valve stented 21 patients Symetis Acurate stented 20 patients Medtronic Engager stented
TAVI Experience at UHC Hamburg 12/11 501 patients 360 patients Edwards Sapien stented 58 patients Core. Valve stented 1 V-in-V 132 TF 228 TA 17 V-in-V 18 patients Direct Flow stentless 34 patients Jena. Valve stented 21 patients Symetis Acurate stented 20 patients Medtronic Engager stented
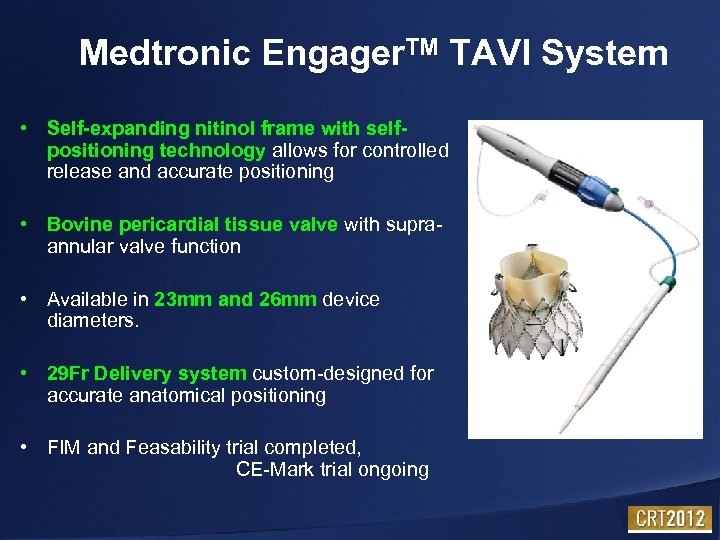 Medtronic Engager. TM TAVI System • Self-expanding nitinol frame with selfpositioning technology allows for controlled release and accurate positioning • Bovine pericardial tissue valve with supraannular valve function • Available in 23 mm and 26 mm device diameters. • 29 Fr Delivery system custom-designed for accurate anatomical positioning • FIM and Feasability trial completed, CE-Mark trial ongoing
Medtronic Engager. TM TAVI System • Self-expanding nitinol frame with selfpositioning technology allows for controlled release and accurate positioning • Bovine pericardial tissue valve with supraannular valve function • Available in 23 mm and 26 mm device diameters. • 29 Fr Delivery system custom-designed for accurate anatomical positioning • FIM and Feasability trial completed, CE-Mark trial ongoing
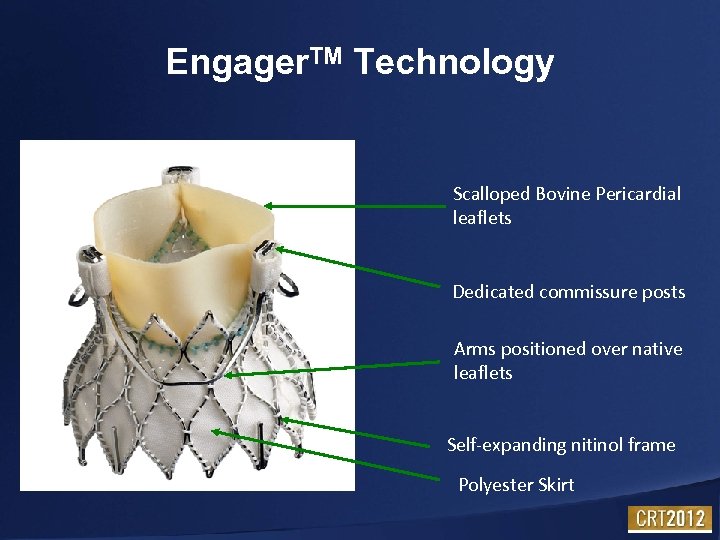 Engager. TM Technology Scalloped Bovine Pericardial leaflets Dedicated commissure posts Arms positioned over native leaflets Self-expanding nitinol frame Polyester Skirt
Engager. TM Technology Scalloped Bovine Pericardial leaflets Dedicated commissure posts Arms positioned over native leaflets Self-expanding nitinol frame Polyester Skirt
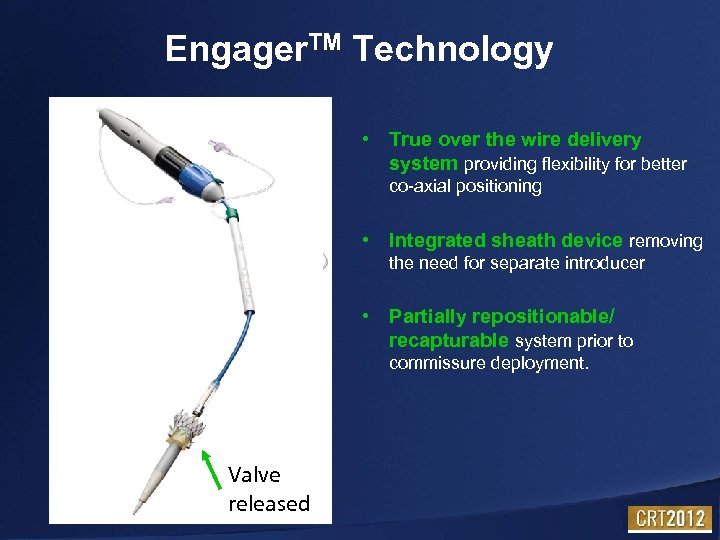 Engager. TM Technology • True over the wire delivery system providing flexibility for better co-axial positioning • Integrated sheath device removing Improved Handle & DS Ergonomics the need for separate introducer • Partially repositionable/ recapturable system prior to commissure deployment. Soft Tip Valve released
Engager. TM Technology • True over the wire delivery system providing flexibility for better co-axial positioning • Integrated sheath device removing Improved Handle & DS Ergonomics the need for separate introducer • Partially repositionable/ recapturable system prior to commissure deployment. Soft Tip Valve released
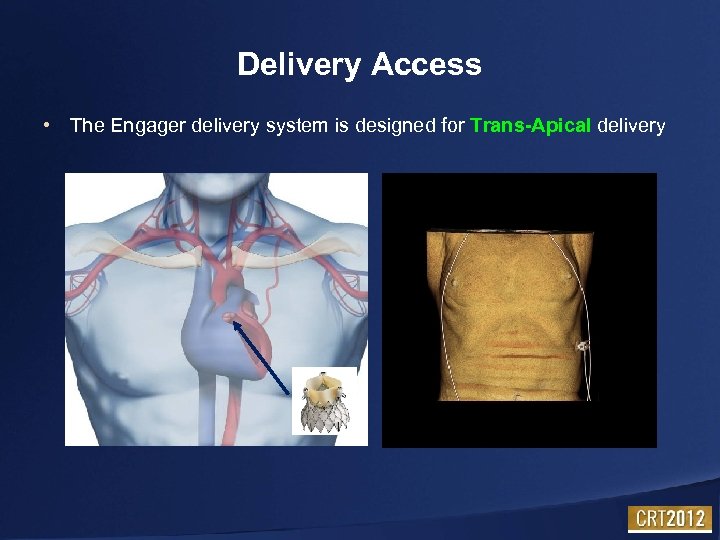 Delivery Access • The Engager delivery system is designed for Trans-Apical delivery
Delivery Access • The Engager delivery system is designed for Trans-Apical delivery
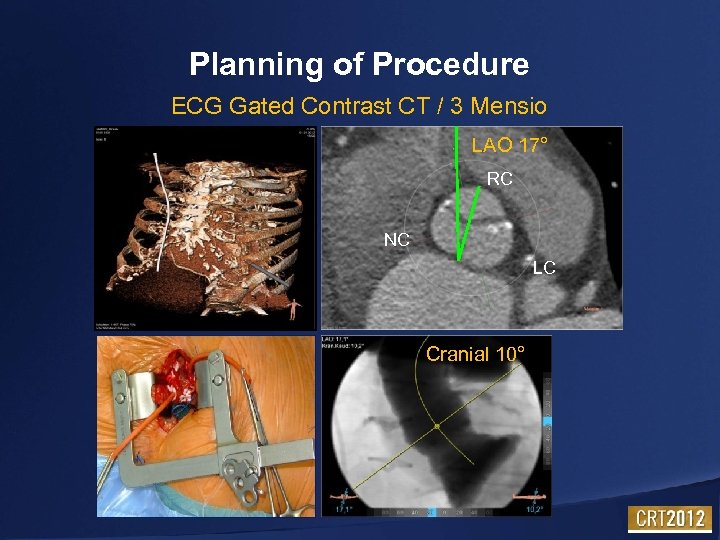 Planning of Procedure ECG Gated Contrast CT / 3 Mensio LAO 17° RC NC LC Cranial 10°
Planning of Procedure ECG Gated Contrast CT / 3 Mensio LAO 17° RC NC LC Cranial 10°
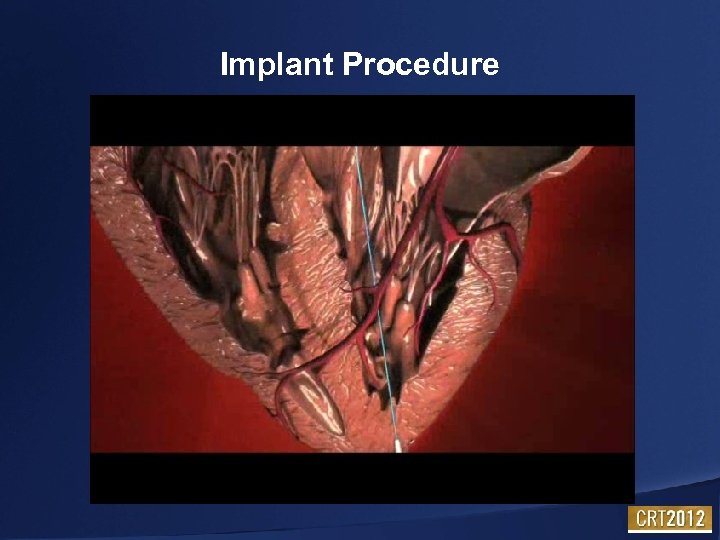 Implant Procedure
Implant Procedure
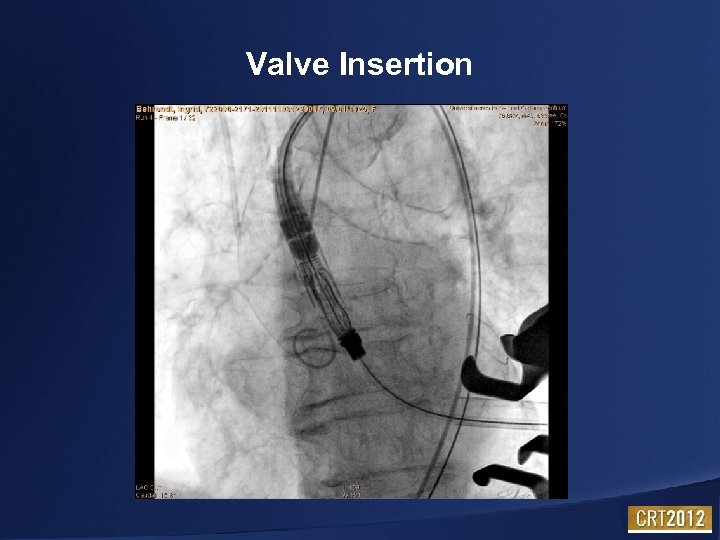 Valve Insertion
Valve Insertion
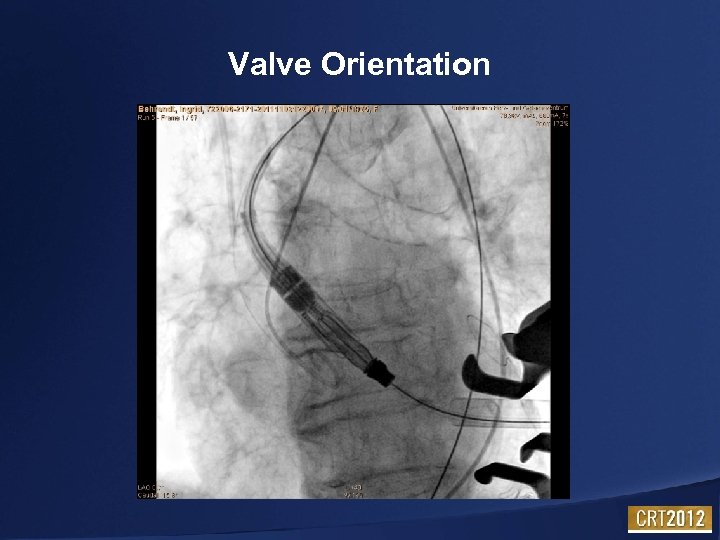 Valve Orientation
Valve Orientation
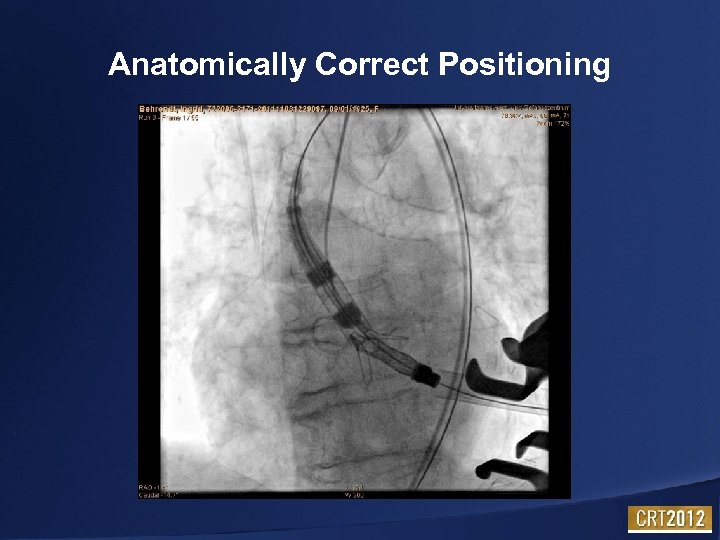 Anatomically Correct Positioning
Anatomically Correct Positioning
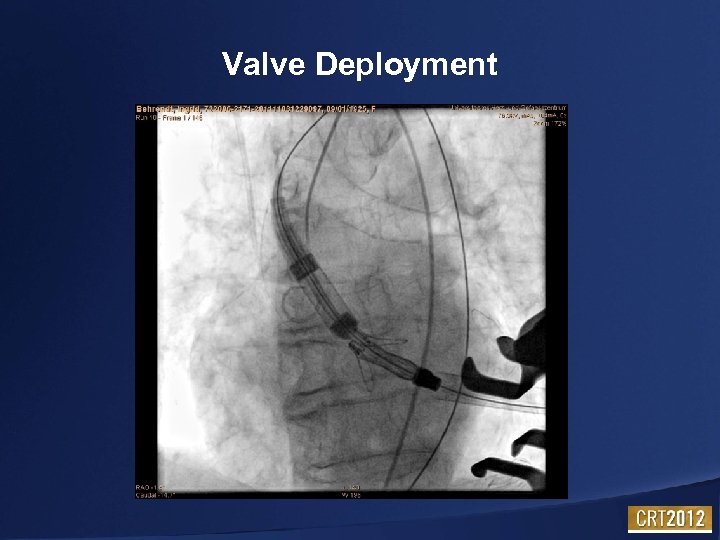 Valve Deployment
Valve Deployment
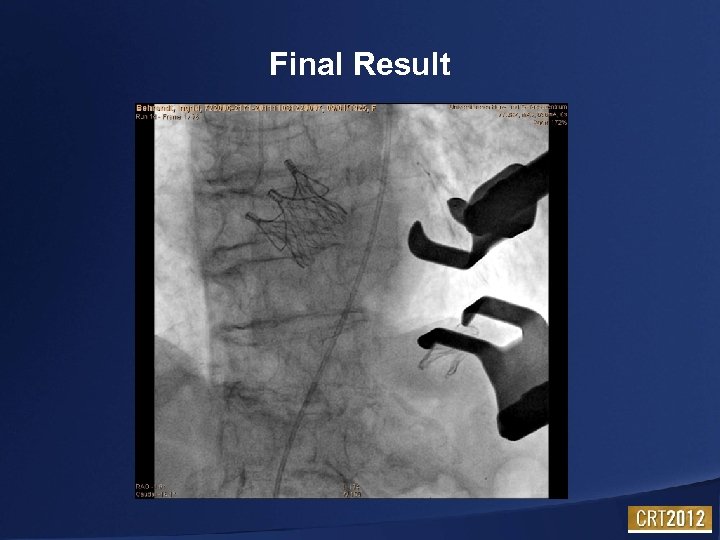 Final Result
Final Result
 Other patients
Other patients
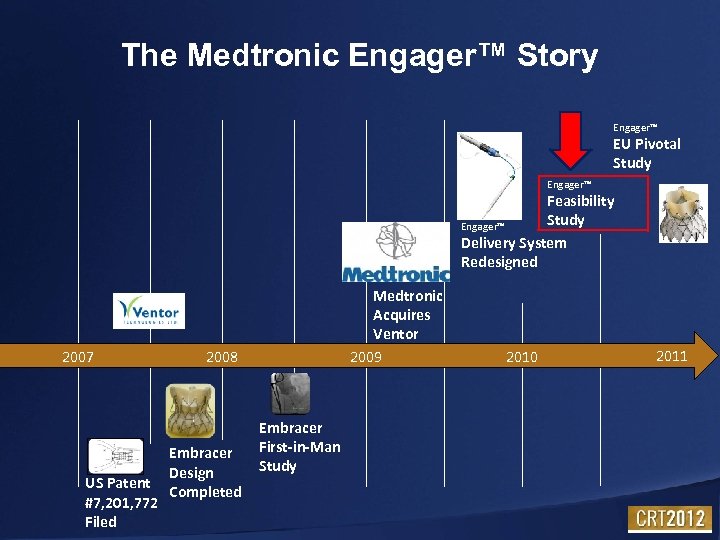 The Medtronic Engager™ Story Engager™ EU Pivotal Study Engager™ Feasibility Study Engager™ Delivery System Redesigned 2007 US Patent #7, 201, 772 Filed Medtronic Acquires Ventor 2009 2008 Embracer Design Completed Embracer First-in-Man Study 2010 2011
The Medtronic Engager™ Story Engager™ EU Pivotal Study Engager™ Feasibility Study Engager™ Delivery System Redesigned 2007 US Patent #7, 201, 772 Filed Medtronic Acquires Ventor 2009 2008 Embracer Design Completed Embracer First-in-Man Study 2010 2011
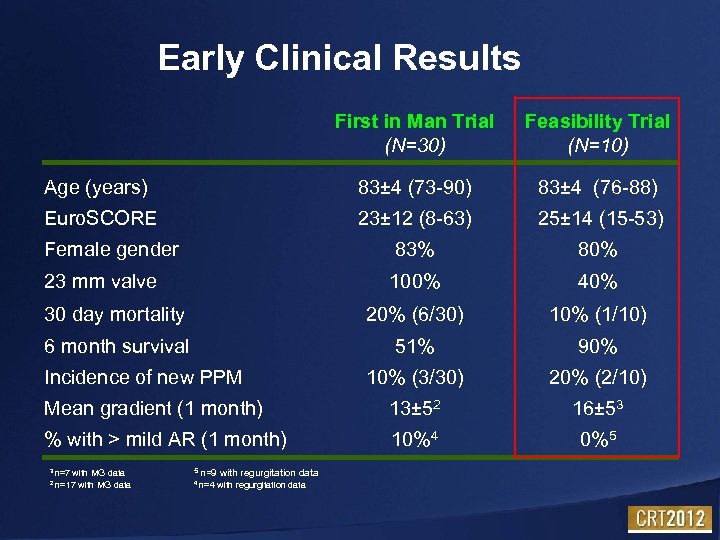 Early Clinical Results First in Man Trial (N=30) Feasibility Trial (N=10) Age (years) 83± 4 (73 -90) 83± 4 (76 -88) Euro. SCORE 23± 12 (8 -63) 25± 14 (15 -53) Female gender 83% 80% 23 mm valve 100% 40% 30 day mortality 20% (6/30) 10% (1/10) 6 month survival 51% 90% 10% (3/30) 20% (2/10) Mean gradient (1 month) 13± 52 16± 53 % with > mild AR (1 month) 10%4 0%5 Incidence of new PPM 3 n=7 5 n=9 with regurgitation data 2 4 n=4 with regurgitation data with MG data n=17 with MG data
Early Clinical Results First in Man Trial (N=30) Feasibility Trial (N=10) Age (years) 83± 4 (73 -90) 83± 4 (76 -88) Euro. SCORE 23± 12 (8 -63) 25± 14 (15 -53) Female gender 83% 80% 23 mm valve 100% 40% 30 day mortality 20% (6/30) 10% (1/10) 6 month survival 51% 90% 10% (3/30) 20% (2/10) Mean gradient (1 month) 13± 52 16± 53 % with > mild AR (1 month) 10%4 0%5 Incidence of new PPM 3 n=7 5 n=9 with regurgitation data 2 4 n=4 with regurgitation data with MG data n=17 with MG data
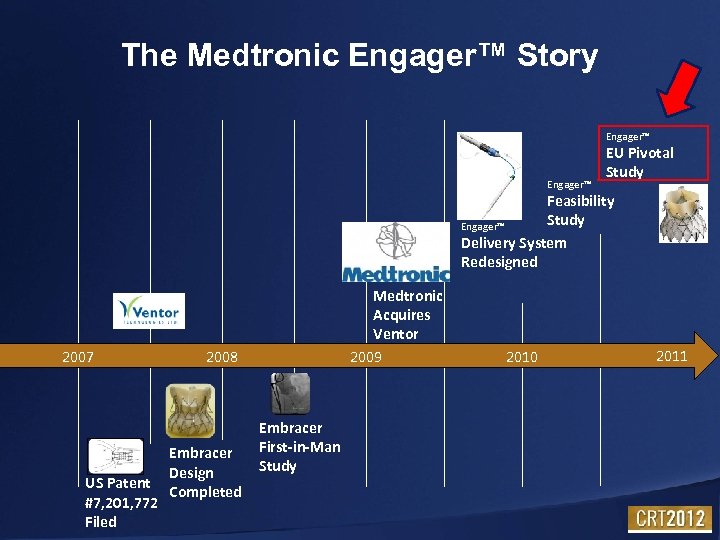 The Medtronic Engager™ Story Engager™ EU Pivotal Study Feasibility Study Engager™ Delivery System Redesigned 2007 US Patent #7, 201, 772 Filed Medtronic Acquires Ventor 2009 2008 Embracer Design Completed Embracer First-in-Man Study 2010 2011
The Medtronic Engager™ Story Engager™ EU Pivotal Study Feasibility Study Engager™ Delivery System Redesigned 2007 US Patent #7, 201, 772 Filed Medtronic Acquires Ventor 2009 2008 Embracer Design Completed Embracer First-in-Man Study 2010 2011
 Engager™ European Pivotal Trial Purpose Evaluate safety & clinical performance of Engager trans-apical aortic valve system Design Prospective, non-randomized, single arm, multicentre Patient Population Patients with symptomatic, severe aortic stenosis at high risk or contra-indicated for open AVR Primary Endpoint All-cause mortality at 30 days post-implantation Sample Size & Scope § § § 150 patients 11 sites in Europe & Israel 5 year follow-up Clinical. Trials. gov identifier: NCT 01348438
Engager™ European Pivotal Trial Purpose Evaluate safety & clinical performance of Engager trans-apical aortic valve system Design Prospective, non-randomized, single arm, multicentre Patient Population Patients with symptomatic, severe aortic stenosis at high risk or contra-indicated for open AVR Primary Endpoint All-cause mortality at 30 days post-implantation Sample Size & Scope § § § 150 patients 11 sites in Europe & Israel 5 year follow-up Clinical. Trials. gov identifier: NCT 01348438
 Clinical Endpoints Primary Endpoint: All-cause mortality at 30 days Secondary Endpoints: Safety § MACCE free survival at 30 days & 6 months Clinical Performance § Acute device success § Procedural success § MAE at 30 days § § All cause mortality at 6 months Hemodynamic metrics at 30 days & 6 months § D in NYHA: Baseline - 6 mon § D in Distance walked (6 min) § D in Quality of Life (EQ 5 D) § Incidence of hospitalizations
Clinical Endpoints Primary Endpoint: All-cause mortality at 30 days Secondary Endpoints: Safety § MACCE free survival at 30 days & 6 months Clinical Performance § Acute device success § Procedural success § MAE at 30 days § § All cause mortality at 6 months Hemodynamic metrics at 30 days & 6 months § D in NYHA: Baseline - 6 mon § D in Distance walked (6 min) § D in Quality of Life (EQ 5 D) § Incidence of hospitalizations
 Engager™ European Pivotal Trial Participating Study Sites • • • Germany • German Heart Center (Munich) • University Heart Center Hamburg (Hamburg) • Leipzig University Hospital (Leipzig) • Heart & Diabetes Center (Bad Oeynhausen) • Uniklinik Köln Heart Center (Cologne) • Kerckhoff Clinic (Bad Nauheim) • Klinik für Herzchirurgie (Karlsruhe) Switzerland • University Hospital Zurich (Zurich) R. Lange H. Treede F. Mohr J. Börgermann T. Wahlers T. Walther H. Posival V. Falk (PI) Belgium • Cliniques Universitaries Saint-Luc (Brussels) P. Astarci Israel • Sheba Medical Center (Tel Hashomer) U. Raanani France • Hospital Bichat (Paris) P. Nataf
Engager™ European Pivotal Trial Participating Study Sites • • • Germany • German Heart Center (Munich) • University Heart Center Hamburg (Hamburg) • Leipzig University Hospital (Leipzig) • Heart & Diabetes Center (Bad Oeynhausen) • Uniklinik Köln Heart Center (Cologne) • Kerckhoff Clinic (Bad Nauheim) • Klinik für Herzchirurgie (Karlsruhe) Switzerland • University Hospital Zurich (Zurich) R. Lange H. Treede F. Mohr J. Börgermann T. Wahlers T. Walther H. Posival V. Falk (PI) Belgium • Cliniques Universitaries Saint-Luc (Brussels) P. Astarci Israel • Sheba Medical Center (Tel Hashomer) U. Raanani France • Hospital Bichat (Paris) P. Nataf
 Is there a need for another second generation transapical TAVI device? What are the distinct features of the Engager. TM TAVI system ?
Is there a need for another second generation transapical TAVI device? What are the distinct features of the Engager. TM TAVI system ?
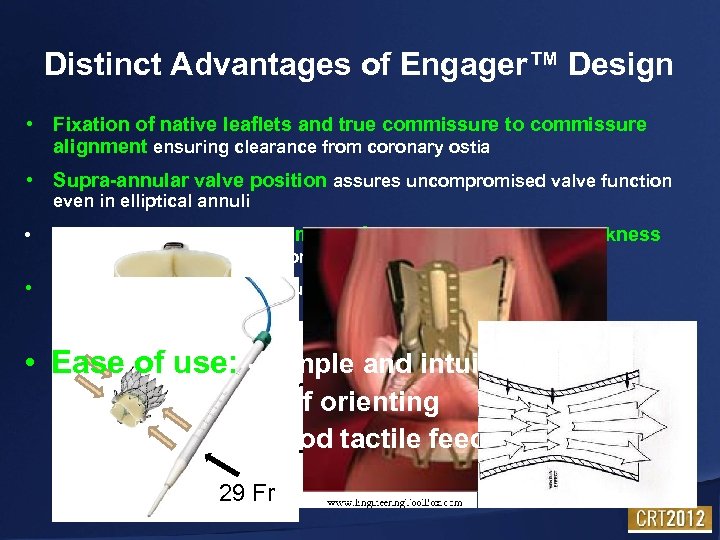 Distinct Advantages of Engager™ Design • Fixation of native leaflets and true commissure to commissure alignment ensuring clearance from coronary ostia • Supra-annular valve position assures uncompromised valve function even in elliptical annuli • Stent Design allows less crimping forces and pericardial thickness equal to surgical valves for maximum durability • Venturi Effect for less turbulent and guided bloodflow • Ease of use: - simple and intuitive delivery - self orienting - good tactile feedback 29 Fr
Distinct Advantages of Engager™ Design • Fixation of native leaflets and true commissure to commissure alignment ensuring clearance from coronary ostia • Supra-annular valve position assures uncompromised valve function even in elliptical annuli • Stent Design allows less crimping forces and pericardial thickness equal to surgical valves for maximum durability • Venturi Effect for less turbulent and guided bloodflow • Ease of use: - simple and intuitive delivery - self orienting - good tactile feedback 29 Fr
 THANK YOU
THANK YOU


