метаболизм англ.ppt
- Количество слайдов: 52
 THE MEDICAL UNIVERSITY “ASTANA” THE DEPARTMENT OF GENERAL AND BIOLOGICAL CHEMISTRY BIOENERGETICS AND METABOLISM The Citric Acid Cycle. Oxidative phosphorylation Associate professor Niyazova RE
THE MEDICAL UNIVERSITY “ASTANA” THE DEPARTMENT OF GENERAL AND BIOLOGICAL CHEMISTRY BIOENERGETICS AND METABOLISM The Citric Acid Cycle. Oxidative phosphorylation Associate professor Niyazova RE
 • Metabolism, the sum of all the chemical transformations taking place in a cell or organism, occurs through a series of enzyme-catalyzed reactions that constitute metabolic pathways. • The precursor is converted into a product through a series of metabolic intermediates called metabolites.
• Metabolism, the sum of all the chemical transformations taking place in a cell or organism, occurs through a series of enzyme-catalyzed reactions that constitute metabolic pathways. • The precursor is converted into a product through a series of metabolic intermediates called metabolites.
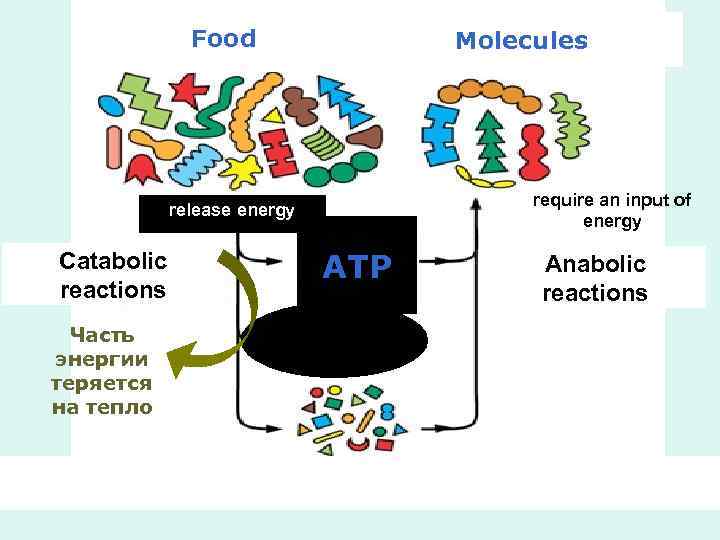
 • Catabolism is the degradative phase of metabolism in which organic nutrient molecules (carbohydrates, fats, and proteins) are converted into smaller, simpler end products (such as lactic acid, CO 2, NH 3). • Catabolic pathways release energy, some of which is conserved in the formation of ATP and reduced electron carriers (NADH, NADPH, and FADH 2)
• Catabolism is the degradative phase of metabolism in which organic nutrient molecules (carbohydrates, fats, and proteins) are converted into smaller, simpler end products (such as lactic acid, CO 2, NH 3). • Catabolic pathways release energy, some of which is conserved in the formation of ATP and reduced electron carriers (NADH, NADPH, and FADH 2)
 • In anabolism, also called biosynthesis, small, simple precursors are built up into larger and more complex molecules, including lipids, polysaccharides, proteins, and nucleic acids. • Anabolic reactions require an input of energy, generally in the form of the phosphoryl group transfer potential of ATP and the reducing power of NADH, NADPH, and FADH 2
• In anabolism, also called biosynthesis, small, simple precursors are built up into larger and more complex molecules, including lipids, polysaccharides, proteins, and nucleic acids. • Anabolic reactions require an input of energy, generally in the form of the phosphoryl group transfer potential of ATP and the reducing power of NADH, NADPH, and FADH 2
 Food Molecules require an input of energy release energy Catabolic reactions Часть энергии теряется на тепло АТP Anabolic reactions
Food Molecules require an input of energy release energy Catabolic reactions Часть энергии теряется на тепло АТP Anabolic reactions
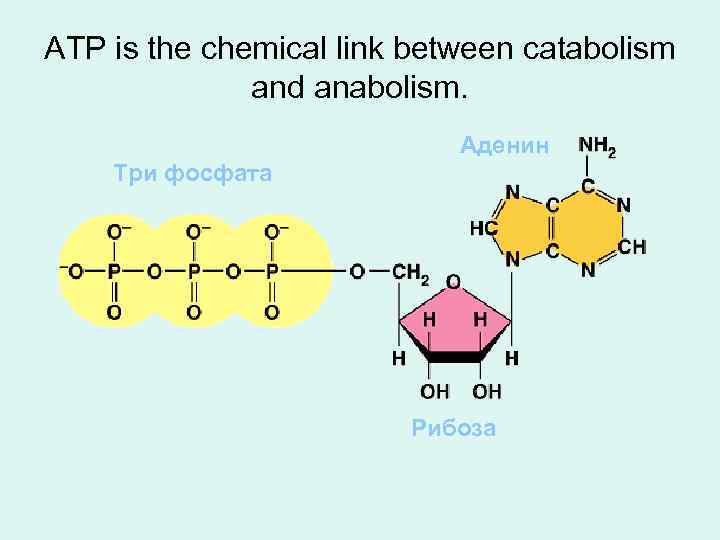 ATP is the chemical link between catabolism and anabolism. Аденин Три фосфата Рибоза
ATP is the chemical link between catabolism and anabolism. Аденин Три фосфата Рибоза
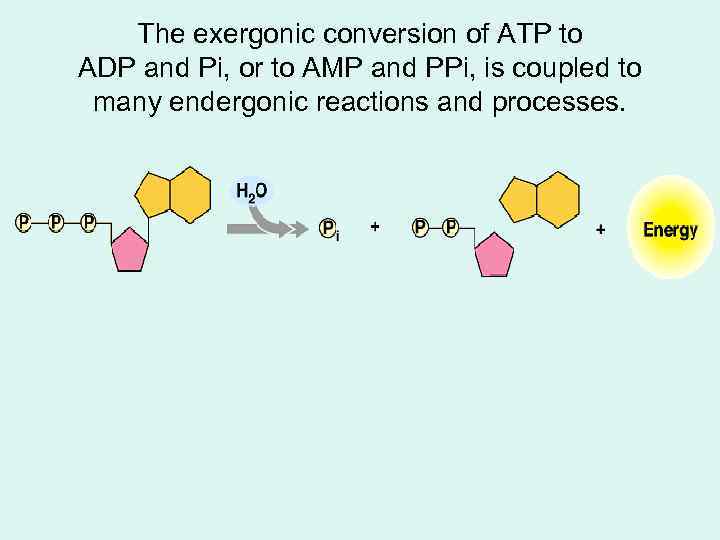 The exergonic conversion of ATP to ADP and Pi, or to AMP and PPi, is coupled to many endergonic reactions and processes.
The exergonic conversion of ATP to ADP and Pi, or to AMP and PPi, is coupled to many endergonic reactions and processes.
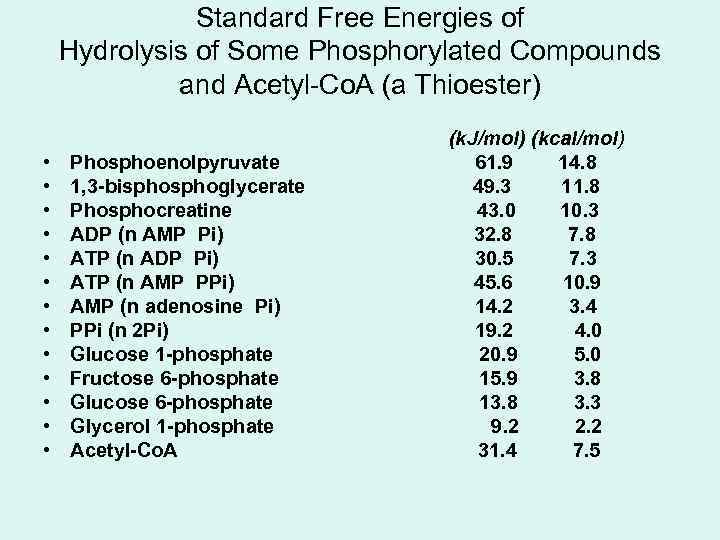 Standard Free Energies of Hydrolysis of Some Phosphorylated Compounds and Acetyl-Co. A (a Thioester) • • • • Phosphoenolpyruvate 1, 3 -bisphoglycerate Phosphocreatine ADP (n AMP Pi) ATP (n ADP Pi) ATP (n AMP PPi) AMP (n adenosine Pi) PPi (n 2 Pi) Glucose 1 -phosphate Fructose 6 -phosphate Glucose 6 -phosphate Glycerol 1 -phosphate Acetyl-Co. A (k. J/mol) (kcal/mol) 61. 9 14. 8 49. 3 11. 8 43. 0 10. 3 32. 8 7. 8 30. 5 7. 3 45. 6 10. 9 14. 2 3. 4 19. 2 4. 0 20. 9 5. 0 15. 9 3. 8 13. 8 3. 3 9. 2 2. 2 31. 4 7. 5
Standard Free Energies of Hydrolysis of Some Phosphorylated Compounds and Acetyl-Co. A (a Thioester) • • • • Phosphoenolpyruvate 1, 3 -bisphoglycerate Phosphocreatine ADP (n AMP Pi) ATP (n ADP Pi) ATP (n AMP PPi) AMP (n adenosine Pi) PPi (n 2 Pi) Glucose 1 -phosphate Fructose 6 -phosphate Glucose 6 -phosphate Glycerol 1 -phosphate Acetyl-Co. A (k. J/mol) (kcal/mol) 61. 9 14. 8 49. 3 11. 8 43. 0 10. 3 32. 8 7. 8 30. 5 7. 3 45. 6 10. 9 14. 2 3. 4 19. 2 4. 0 20. 9 5. 0 15. 9 3. 8 13. 8 3. 3 9. 2 2. 2 31. 4 7. 5
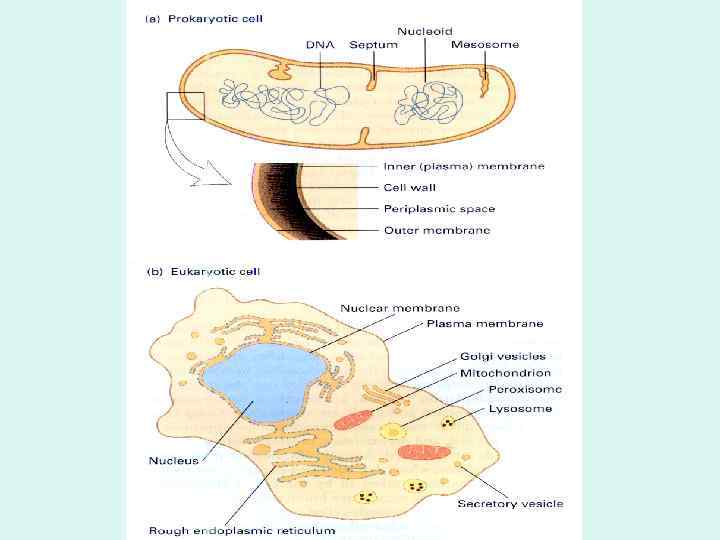
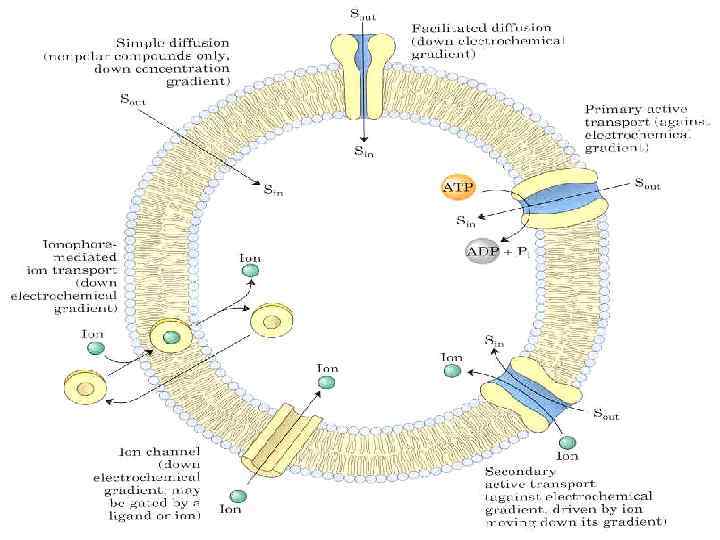
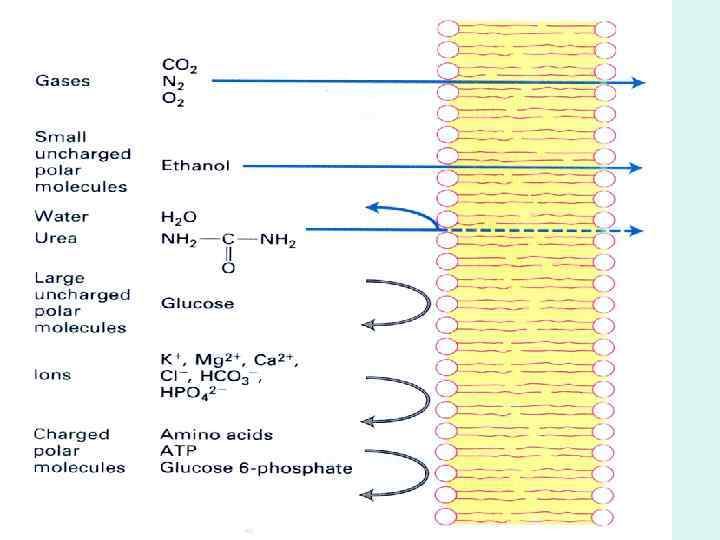
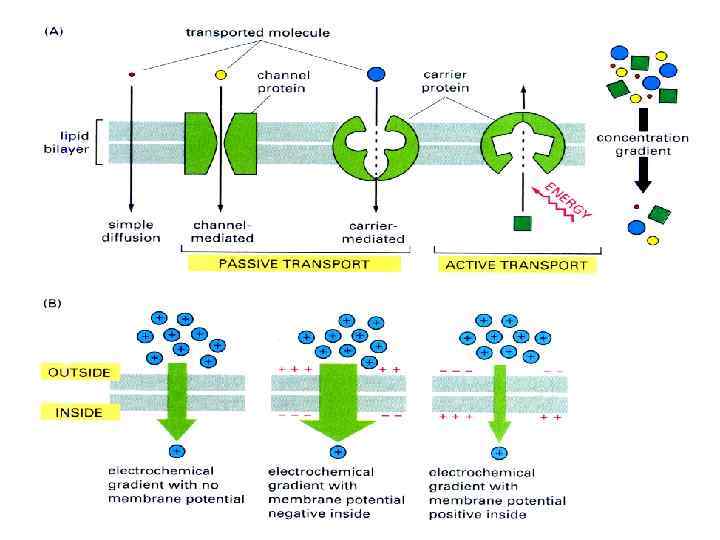
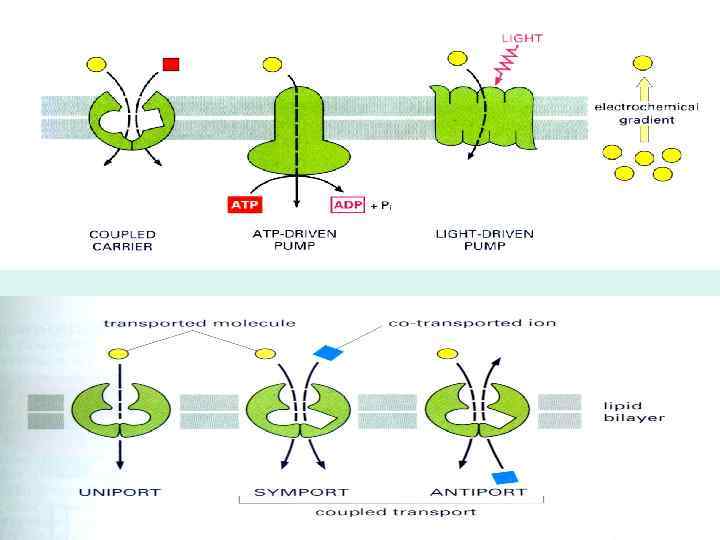
 BIOLOGICAL MEMBRANES AND TRANSPORT
BIOLOGICAL MEMBRANES AND TRANSPORT
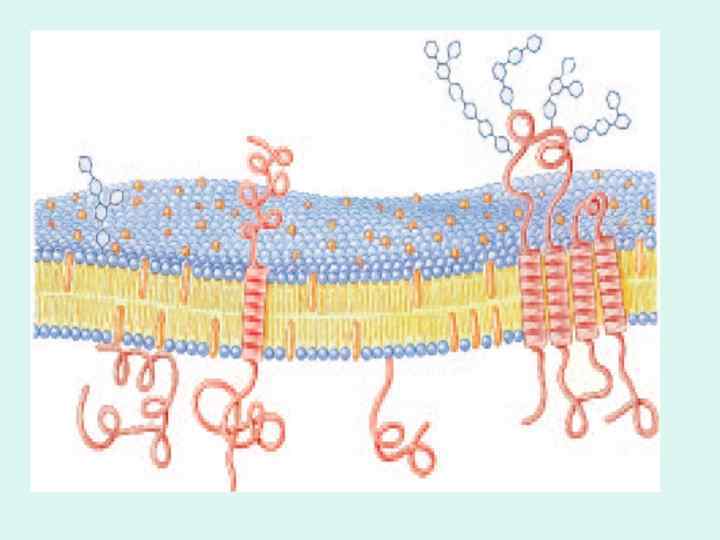
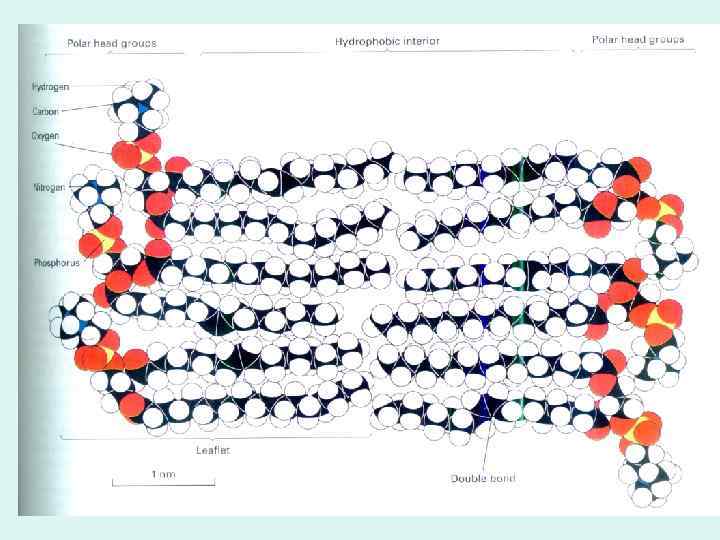
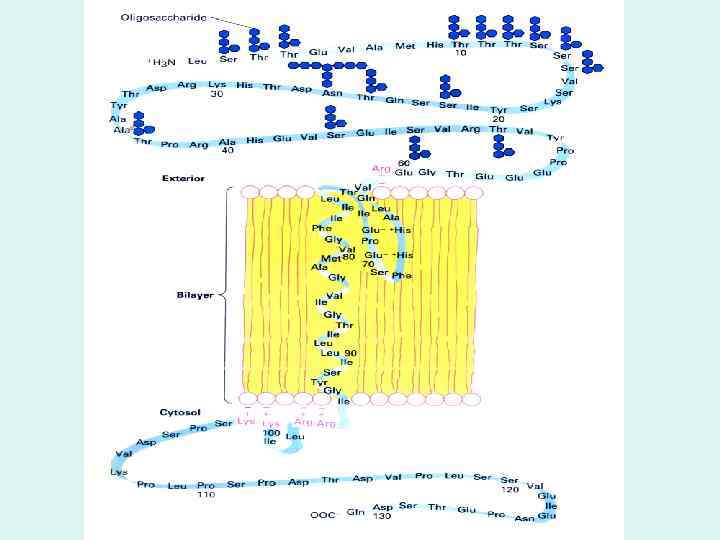
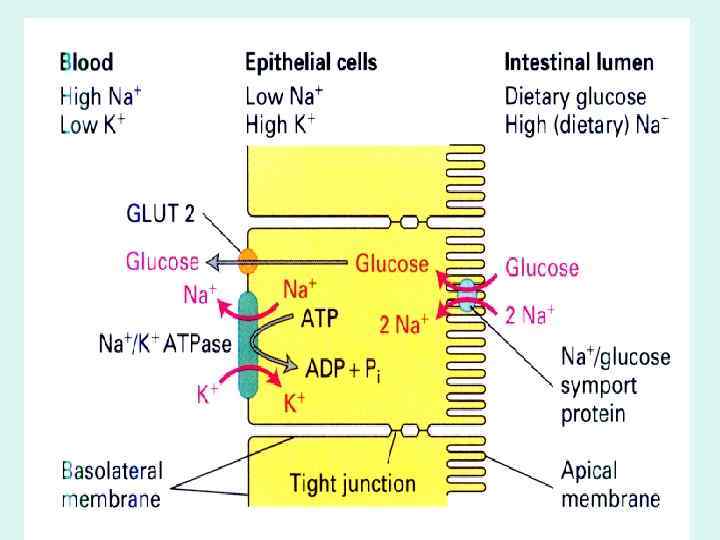
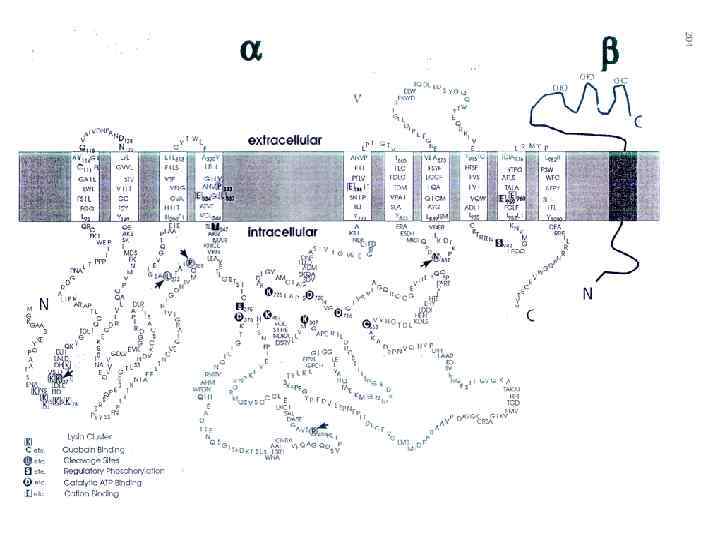
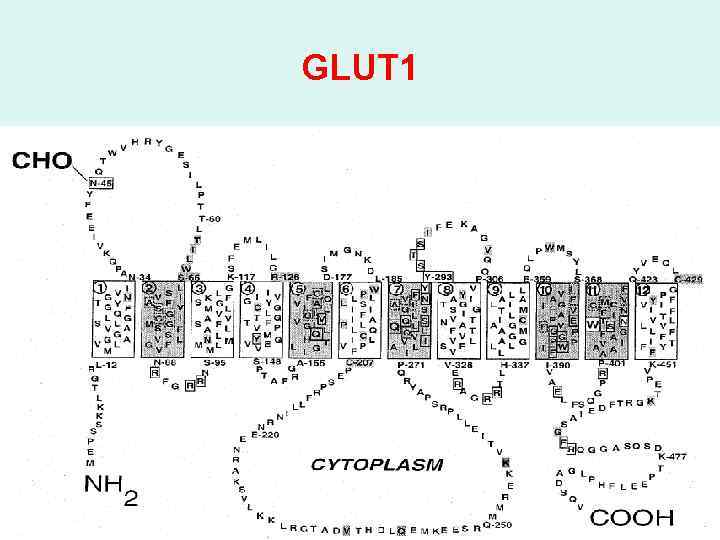 GLUT 1
GLUT 1
 BIOLOGICAL OXYDATION • In many biological oxidations, a compound loses two electrons and two hydrogen ions (that is, two hydrogen atoms); these reactions are commonly called dehydrogenations and the enzymes that catalyze them are called dehydrogenases. • Oxidation reactions generally release energy.
BIOLOGICAL OXYDATION • In many biological oxidations, a compound loses two electrons and two hydrogen ions (that is, two hydrogen atoms); these reactions are commonly called dehydrogenations and the enzymes that catalyze them are called dehydrogenases. • Oxidation reactions generally release energy.
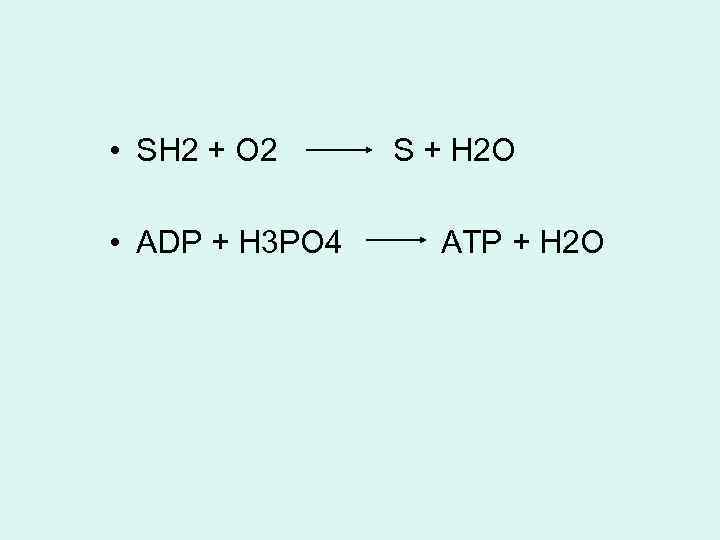 • SH 2 + O 2 • ADP + H 3 PO 4 S + H 2 O ATP + H 2 O
• SH 2 + O 2 • ADP + H 3 PO 4 S + H 2 O ATP + H 2 O
 OXIDATIVE PHOSPHORYLATION • Oxidative phosphorylation occurs in mitochondria; involves the reduction of O 2 to H 2 O with electrons donated by NADH and FADH 2 • Oxidative phosphorylation begins with the entry of electrons into the respiratory chain.
OXIDATIVE PHOSPHORYLATION • Oxidative phosphorylation occurs in mitochondria; involves the reduction of O 2 to H 2 O with electrons donated by NADH and FADH 2 • Oxidative phosphorylation begins with the entry of electrons into the respiratory chain.
 The mitochondrial respiratory chain • In mitochondria, hydride ions removed from substrates by NAD-linked dehydrogenases donate electrons to the respiratory (electron-transfer) chain, which transfers the electrons to molecular O 2, reducing it to H 2 O
The mitochondrial respiratory chain • In mitochondria, hydride ions removed from substrates by NAD-linked dehydrogenases donate electrons to the respiratory (electron-transfer) chain, which transfers the electrons to molecular O 2, reducing it to H 2 O
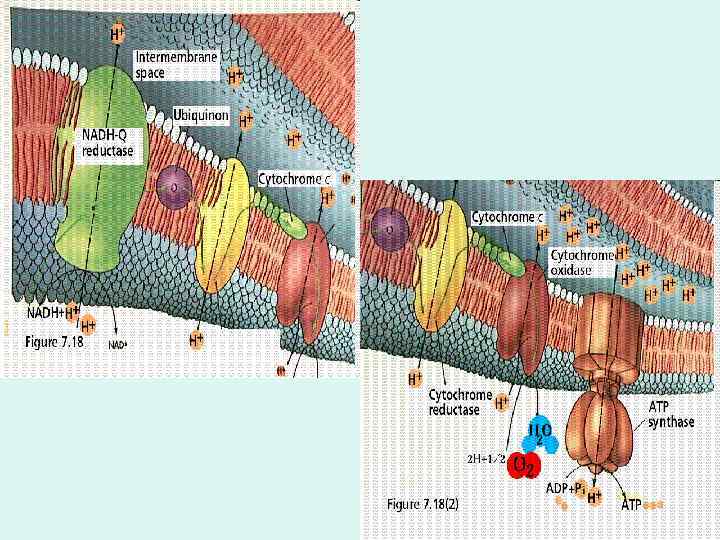
 • The electron carriers of the respiratory chain are organized into membraneembedded supramolecular complexes that can be physically separated. • Complexes I and II catalyze electron transfer to ubiquinone from two different electron donors: NADH (Complex I) and succinate (Complex II).
• The electron carriers of the respiratory chain are organized into membraneembedded supramolecular complexes that can be physically separated. • Complexes I and II catalyze electron transfer to ubiquinone from two different electron donors: NADH (Complex I) and succinate (Complex II).
 • Complex I, also called NADH: ubiquinone oxidoreductase or NADH dehydrogenase including an FMN-containing flavoprotein and at least six ironsulfur centers. • Complex I catalyzes two simultaneous and obligately coupled processes: (1) the exergonic transfer to ubiquinone of a hydride ion from NADH and a proton from the matrix and (2) the endergonic transfer of four protons from the matrix to the intermembrane space.
• Complex I, also called NADH: ubiquinone oxidoreductase or NADH dehydrogenase including an FMN-containing flavoprotein and at least six ironsulfur centers. • Complex I catalyzes two simultaneous and obligately coupled processes: (1) the exergonic transfer to ubiquinone of a hydride ion from NADH and a proton from the matrix and (2) the endergonic transfer of four protons from the matrix to the intermembrane space.
 • Complex II is succinate dehydrogenase, the only membranebound enzyme in the citric acid cycle. The path of electron transfer from the succinate-binding site to FAD, then through the Fe-S centers to the Qbinding site.
• Complex II is succinate dehydrogenase, the only membranebound enzyme in the citric acid cycle. The path of electron transfer from the succinate-binding site to FAD, then through the Fe-S centers to the Qbinding site.
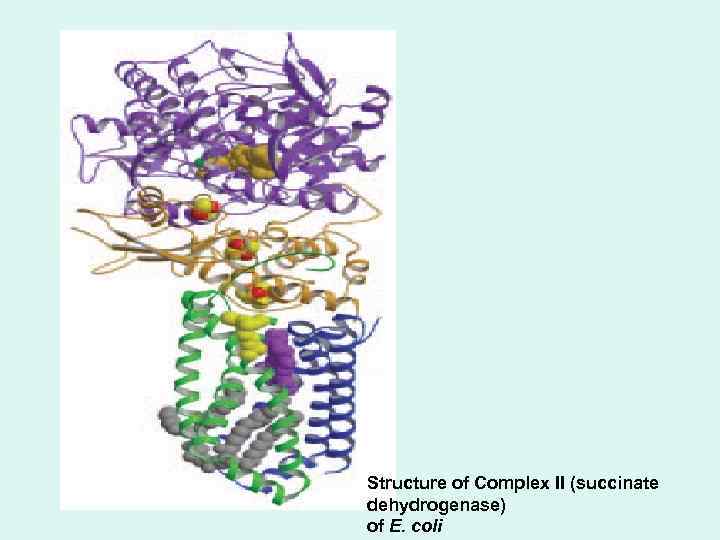 Structure of Complex II (succinate dehydrogenase) of E. coli
Structure of Complex II (succinate dehydrogenase) of E. coli
 • Complex III carries electrons from reduced ubiquinone to cytochrome c • Complex IV completes the sequence by transferring electrons from cytochrome c to O 2.
• Complex III carries electrons from reduced ubiquinone to cytochrome c • Complex IV completes the sequence by transferring electrons from cytochrome c to O 2.
 Electron Acceptors • NAD-linked dehydrogenases. NADH and NADPH are water-soluble electron carriers that associate reversibly with dehydrogenases. NADH carries electrons from catabolic reactions to their point of entry into the respiratory chain, NADPH generally supplies electrons to anabolic reactions. • Flavoproteins, bound flavin nucleotide, either FMN or FAD
Electron Acceptors • NAD-linked dehydrogenases. NADH and NADPH are water-soluble electron carriers that associate reversibly with dehydrogenases. NADH carries electrons from catabolic reactions to their point of entry into the respiratory chain, NADPH generally supplies electrons to anabolic reactions. • Flavoproteins, bound flavin nucleotide, either FMN or FAD
 Electrons Pass through a Series of Membrane-Bound Carriers • Ubiquinone (also called coenzyme Q, or simply Q) is a lipid-soluble benzoquinone. • Because ubiquinone is both small and hydrophobic, it is freely diffusible within the lipid bilayer of the inner mitochondrial membrane and can shuttle reducing equivalents between other, less mobile electron carriers in the membrane. And because it carries both electrons and protons, it plays a central role in coupling electron flow to proton movement.
Electrons Pass through a Series of Membrane-Bound Carriers • Ubiquinone (also called coenzyme Q, or simply Q) is a lipid-soluble benzoquinone. • Because ubiquinone is both small and hydrophobic, it is freely diffusible within the lipid bilayer of the inner mitochondrial membrane and can shuttle reducing equivalents between other, less mobile electron carriers in the membrane. And because it carries both electrons and protons, it plays a central role in coupling electron flow to proton movement.
 • The cytochromes are proteins with characteristic strong absorption of visible light, due to their ironcontaining heme prosthetic groups. The cytochromes of type a and b and some of type c are integral proteins of the inner mitochondrial membrane.
• The cytochromes are proteins with characteristic strong absorption of visible light, due to their ironcontaining heme prosthetic groups. The cytochromes of type a and b and some of type c are integral proteins of the inner mitochondrial membrane.
 • In iron-sulfur proteins (Fe-S) the iron is present not in heme but in association with inorganic sulfur atoms or with the sulfur atoms of Cys residues in the protein, or both.
• In iron-sulfur proteins (Fe-S) the iron is present not in heme but in association with inorganic sulfur atoms or with the sulfur atoms of Cys residues in the protein, or both.
 Agents That Interfere with Oxidative Phosphorylation • Amytal (a barbiturate drug), rotenone (a plant product commonly used as an insecticide), and piericidin A (an antibiotic) inhibit electron flow from the Fe-S centers of Complex I to ubiquinone and therefore block the overall process of oxidative phosphorylation. • Antimycin A blocks electron transfer from cytochrome b to cytochrome c 1 (inhibit Complex II). • Cyanide, carbon monoxide inhibit cytochrome oxidase (Complex III).
Agents That Interfere with Oxidative Phosphorylation • Amytal (a barbiturate drug), rotenone (a plant product commonly used as an insecticide), and piericidin A (an antibiotic) inhibit electron flow from the Fe-S centers of Complex I to ubiquinone and therefore block the overall process of oxidative phosphorylation. • Antimycin A blocks electron transfer from cytochrome b to cytochrome c 1 (inhibit Complex II). • Cyanide, carbon monoxide inhibit cytochrome oxidase (Complex III).
 ATP Synthesis How is a concentration gradient of protons transformed into ATP? The chemiosmotic model, proposed by Peter Mitchell, is the paradigm for this mechanism. • The flow of electrons through Complexes I, III, and IV results in pumping of protons across the inner mitochondrial membrane, making the matrix alkaline relative to the intermembrane space. This proton gradient provides the energy (in the form of the proton- motive force) for ATP synthesis from ADP and Pi by ATP synthase (Fo. F 1 complex) in the inner membrane.
ATP Synthesis How is a concentration gradient of protons transformed into ATP? The chemiosmotic model, proposed by Peter Mitchell, is the paradigm for this mechanism. • The flow of electrons through Complexes I, III, and IV results in pumping of protons across the inner mitochondrial membrane, making the matrix alkaline relative to the intermembrane space. This proton gradient provides the energy (in the form of the proton- motive force) for ATP synthesis from ADP and Pi by ATP synthase (Fo. F 1 complex) in the inner membrane.
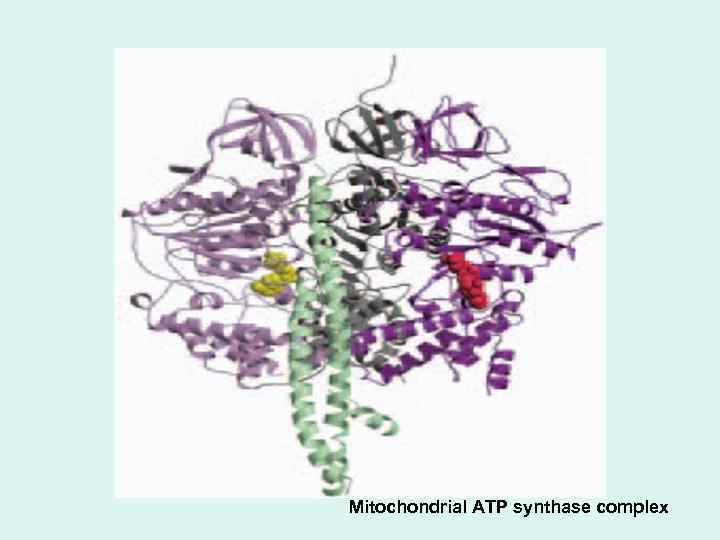 Mitochondrial ATP synthase complex
Mitochondrial ATP synthase complex
 • The ratio of ATP synthesized per ½ O 2 reduced to H 2 O (the P/O ratio) is about 2. 5 when electrons enter the respiratory chain at Complex I, and 1. 5 when electrons enter at Co. Q.
• The ratio of ATP synthesized per ½ O 2 reduced to H 2 O (the P/O ratio) is about 2. 5 when electrons enter the respiratory chain at Complex I, and 1. 5 when electrons enter at Co. Q.
 • The electron transfer and ATP synthesis are obligately coupled; neither reaction occurs without the other. • Certain conditions and reagents, however, can uncouple oxidation from phosphorylation.
• The electron transfer and ATP synthesis are obligately coupled; neither reaction occurs without the other. • Certain conditions and reagents, however, can uncouple oxidation from phosphorylation.
 • Chemical uncouplers include 2, 4 -dinitrophenol (DNP) and carbonylcyanide-ptrifluoromethoxyphenylhydrazone (FCCP) weak acids with hydrophobic properties that permit them to diffuse readily across mitochondrial membranes. After entering the matrix in the protonated form, they can release a proton, thus dissipating the proton gradient. • Ionophores such as valinomycin allow inorganic ions to pass easily through membranes. • Thermogenin, also called the uncoupling protein, provides a path for protons to return to the matrix without passing through the Fo. F 1 complex.
• Chemical uncouplers include 2, 4 -dinitrophenol (DNP) and carbonylcyanide-ptrifluoromethoxyphenylhydrazone (FCCP) weak acids with hydrophobic properties that permit them to diffuse readily across mitochondrial membranes. After entering the matrix in the protonated form, they can release a proton, thus dissipating the proton gradient. • Ionophores such as valinomycin allow inorganic ions to pass easily through membranes. • Thermogenin, also called the uncoupling protein, provides a path for protons to return to the matrix without passing through the Fo. F 1 complex.
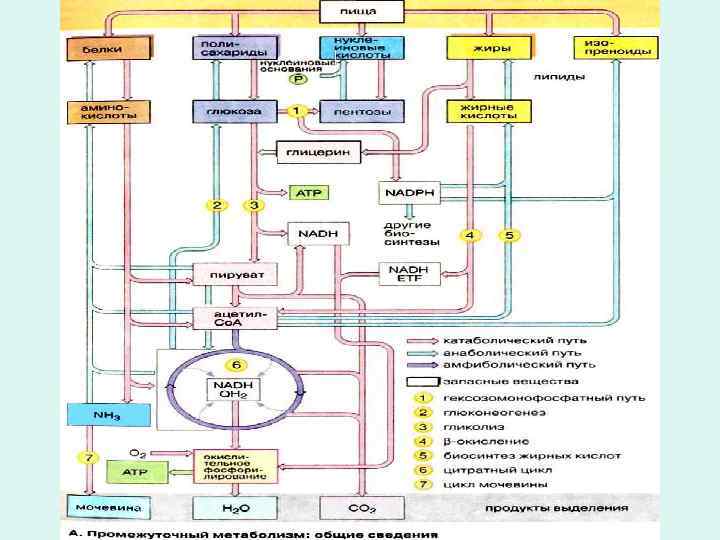
 • Glucose and other sugars, fatty acids, and most amino acids are ultimately oxidized to CO 2 and H 2 O via the citric acid cycle and the respiratory chain. • Before entering the citric acid cycle, the carbon skeletons of sugars and fatty acids are degraded to the acetyl group of acetyl-Co. A, the form in which the cycle accepts most of its fuel input. • Many amino acid carbons also enter the cycle this way, although several amino acids are degraded to other cycle intermediates. • Here we focus on how pyruvate, derived from glucose and other sugars by glycolysis, is oxidized to acetyl-Co. A and CO 2 by the pyruvate dehydrogenase (PDH) complex, located in the mitochondria.
• Glucose and other sugars, fatty acids, and most amino acids are ultimately oxidized to CO 2 and H 2 O via the citric acid cycle and the respiratory chain. • Before entering the citric acid cycle, the carbon skeletons of sugars and fatty acids are degraded to the acetyl group of acetyl-Co. A, the form in which the cycle accepts most of its fuel input. • Many amino acid carbons also enter the cycle this way, although several amino acids are degraded to other cycle intermediates. • Here we focus on how pyruvate, derived from glucose and other sugars by glycolysis, is oxidized to acetyl-Co. A and CO 2 by the pyruvate dehydrogenase (PDH) complex, located in the mitochondria.
 • The PDH complex contains three enzymes— pyruvate dehydrogenase (E 1), dihydrolipoyl transacetylase (E 2), and dihydrolipoyl dehydrogenase (E 3)—each present in multiple copies.
• The PDH complex contains three enzymes— pyruvate dehydrogenase (E 1), dihydrolipoyl transacetylase (E 2), and dihydrolipoyl dehydrogenase (E 3)—each present in multiple copies.
 • thiamine pyrophosphate (TPP), • flavin adenine dinucleotide (FAD), coenzyme A (Co. A, sometimes denoted Co. A-SH, to emphasize the role of the OSH group), • nicotinamide adenine dinucleotide (NAD), • lipoate
• thiamine pyrophosphate (TPP), • flavin adenine dinucleotide (FAD), coenzyme A (Co. A, sometimes denoted Co. A-SH, to emphasize the role of the OSH group), • nicotinamide adenine dinucleotide (NAD), • lipoate
 • E 1 catalyzes first the decarboxylation of pyruvate. • E 2 catalyzes the transfer of the acetyl group to coenzyme A, forming acetyl-Co. A. • E 3 catalyzes the regeneration of the disulfide (oxidized) form of lipoate; electrons pass first to FAD, then to NAD.
• E 1 catalyzes first the decarboxylation of pyruvate. • E 2 catalyzes the transfer of the acetyl group to coenzyme A, forming acetyl-Co. A. • E 3 catalyzes the regeneration of the disulfide (oxidized) form of lipoate; electrons pass first to FAD, then to NAD.
 THE CITRIC ACID CYCLE • The citric acid cycle, also called the tricarboxylic acid (TCA) cycle or the Krebs cycle (after its discoverer, Hans Krebs). • The citric acid cycle is a nearly universal central catabolic pathway in which compounds derived from the breakdown of carbohydrates, fats, and proteins are oxidized to CO 2, with most of the energy of oxidation temporarily held in the electron carriers FADH 2 and NADH. During aerobic metabolism, these electrons are transferred to O 2 and the energy of electron flow is trapped as ATP. Hans Krebs, 1900– 1981
THE CITRIC ACID CYCLE • The citric acid cycle, also called the tricarboxylic acid (TCA) cycle or the Krebs cycle (after its discoverer, Hans Krebs). • The citric acid cycle is a nearly universal central catabolic pathway in which compounds derived from the breakdown of carbohydrates, fats, and proteins are oxidized to CO 2, with most of the energy of oxidation temporarily held in the electron carriers FADH 2 and NADH. During aerobic metabolism, these electrons are transferred to O 2 and the energy of electron flow is trapped as ATP. Hans Krebs, 1900– 1981
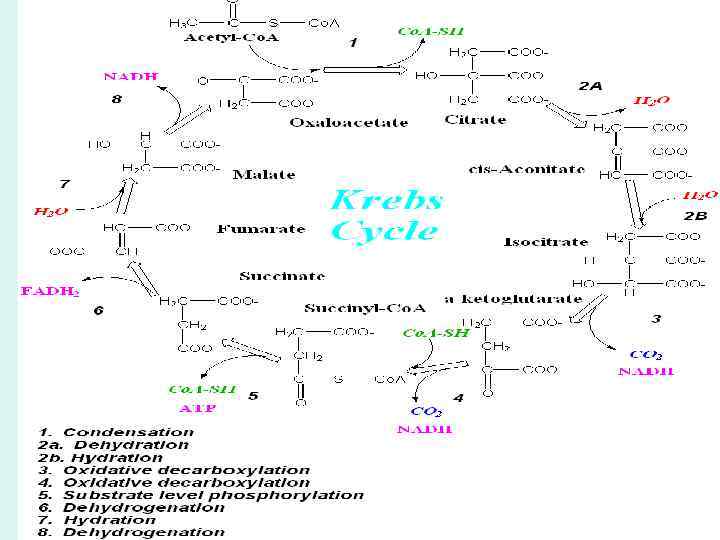
 The Citric Acid Cycle Has Eight Steps 1. The first reaction of the cycle is the condensation of acetyl-Co. A with oxaloacetate to form citrate, catalyzed by citrate synthase 2. The enzyme aconitase catalyzes the reversible transformation of citrate to isocitrate, through the intermediary formation of the tricarboxylic acid cisaconitate 3. Isocitrate dehydrogenase catalyzes oxidative decarboxylation of isocitrate to form –ketoglutarate 4. The next step is another oxidative decarboxylation, in which -ketoglutarate is converted to succinyl-Co. A and CO 2 by the action of the -ketoglutarate dehydrogenase complex.
The Citric Acid Cycle Has Eight Steps 1. The first reaction of the cycle is the condensation of acetyl-Co. A with oxaloacetate to form citrate, catalyzed by citrate synthase 2. The enzyme aconitase catalyzes the reversible transformation of citrate to isocitrate, through the intermediary formation of the tricarboxylic acid cisaconitate 3. Isocitrate dehydrogenase catalyzes oxidative decarboxylation of isocitrate to form –ketoglutarate 4. The next step is another oxidative decarboxylation, in which -ketoglutarate is converted to succinyl-Co. A and CO 2 by the action of the -ketoglutarate dehydrogenase complex.
 5. Succinyl-Co. A synthetase or succinic thiokinase catalyzes the reversible of Succinyl-Co. A to Succinate 6. The succinate formed from succinyl-Co. A is oxidized to fumarate by the flavoprotein succinate dehydrogenase 7. The reversible hydration of fumarate to L-malate is catalyzed by fumarase 8. In the last reaction of the citric acid cycle, NADlinked L-malate dehydrogenase catalyzes the oxidation of L-malate to oxaloacetate
5. Succinyl-Co. A synthetase or succinic thiokinase catalyzes the reversible of Succinyl-Co. A to Succinate 6. The succinate formed from succinyl-Co. A is oxidized to fumarate by the flavoprotein succinate dehydrogenase 7. The reversible hydration of fumarate to L-malate is catalyzed by fumarase 8. In the last reaction of the citric acid cycle, NADlinked L-malate dehydrogenase catalyzes the oxidation of L-malate to oxaloacetate
 • The energy released by these oxidations was conserved in the reduction of three NAD and one FAD and the production of one ATP or GTP • Although the citric acid cycle directly generates only one ATP per turn (in the conversion of succinyl-Co. A to succinate), the four oxidation steps in the cycle provide a large flow of electrons into the respiratory chain via NADH and FADH 2 and thus lead to formation of a large number of ATP molecules during oxidative phosphorylation.
• The energy released by these oxidations was conserved in the reduction of three NAD and one FAD and the production of one ATP or GTP • Although the citric acid cycle directly generates only one ATP per turn (in the conversion of succinyl-Co. A to succinate), the four oxidation steps in the cycle provide a large flow of electrons into the respiratory chain via NADH and FADH 2 and thus lead to formation of a large number of ATP molecules during oxidative phosphorylation.
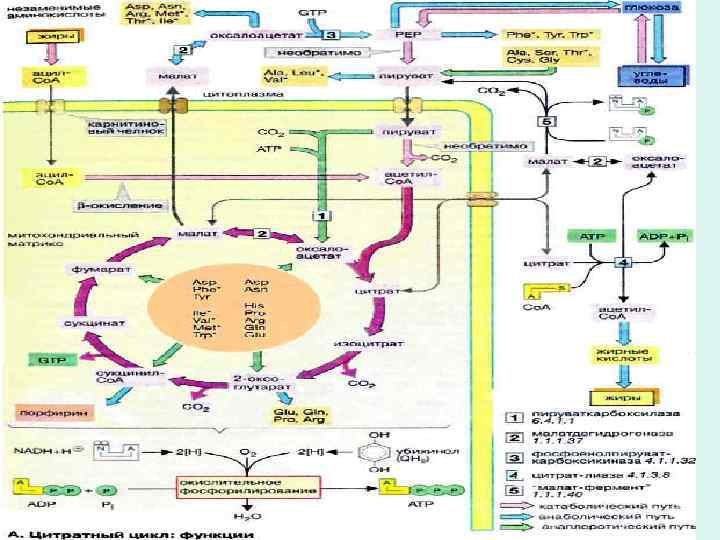
 • The citric acid cycle is amphibolic, serving in both catabolism and anabolism; cycle intermediates can be drawn off and used as the starting material for a variety of biosynthetic products.
• The citric acid cycle is amphibolic, serving in both catabolism and anabolism; cycle intermediates can be drawn off and used as the starting material for a variety of biosynthetic products.