Lecture enzime.ppt
- Количество слайдов: 18
 THE MEDICAL UNIVERSITY ASTANA THE CHAIR OF GENERAL AND BIOLOGICAL CHEMISTRY Enzymes. Properties of enzymes. Regulation of activity of enzymes .
THE MEDICAL UNIVERSITY ASTANA THE CHAIR OF GENERAL AND BIOLOGICAL CHEMISTRY Enzymes. Properties of enzymes. Regulation of activity of enzymes .
 4 Enzymes, the catalysts of biological systems, are remarkable molecular devices that determine the patterns of chemical transformations. They also mediate the transformation of one form of energy into another. 4 The most striking characteristics of enzymes are their catalytic power and specificity. Catalysis takes place at a particular site on the enzyme called the active site. 4 Nearly all known enzymes are proteins.
4 Enzymes, the catalysts of biological systems, are remarkable molecular devices that determine the patterns of chemical transformations. They also mediate the transformation of one form of energy into another. 4 The most striking characteristics of enzymes are their catalytic power and specificity. Catalysis takes place at a particular site on the enzyme called the active site. 4 Nearly all known enzymes are proteins.
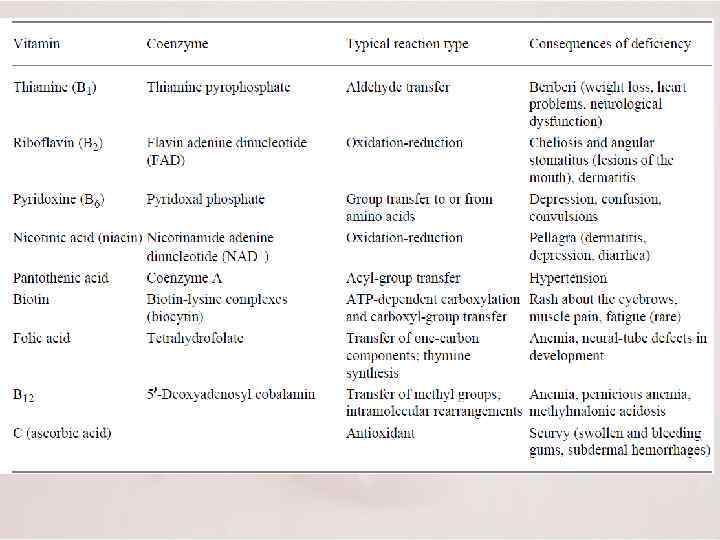
 The catalytic activity of many enzymes depends on the presence of small molecules termed cofactors, although the precise role varies with the cofactor and the enzyme. Such an enzyme without its cofactor is referred to as an apoenzyme; the complete, catalytically active enzyme is called a holoenzyme. 4 Cofactors can be subdivided into two groups: metals and small organic molecules 4 Cofactors that are small organic molecules are called coenzymes. Often derived from vitamins, coenzymes can be either tightly or loosely bound to the enzyme. If tightly bound, they are called
The catalytic activity of many enzymes depends on the presence of small molecules termed cofactors, although the precise role varies with the cofactor and the enzyme. Such an enzyme without its cofactor is referred to as an apoenzyme; the complete, catalytically active enzyme is called a holoenzyme. 4 Cofactors can be subdivided into two groups: metals and small organic molecules 4 Cofactors that are small organic molecules are called coenzymes. Often derived from vitamins, coenzymes can be either tightly or loosely bound to the enzyme. If tightly bound, they are called
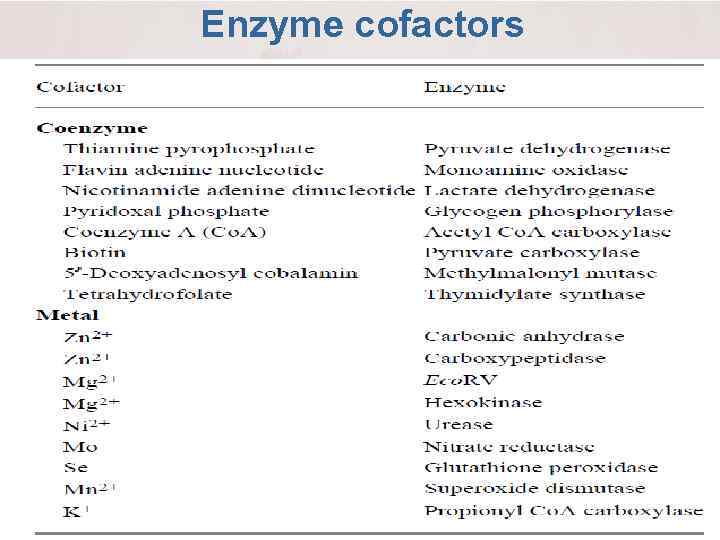 Enzyme cofactors
Enzyme cofactors
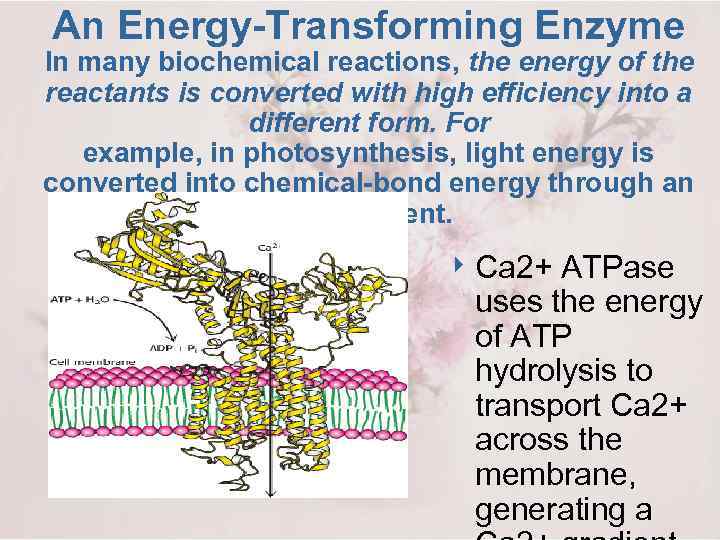 An Energy-Transforming Enzyme In many biochemical reactions, the energy of the reactants is converted with high efficiency into a different form. For example, in photosynthesis, light energy is converted into chemical-bond energy through an ion gradient. 4 Ca 2+ ATPase uses the energy of ATP hydrolysis to transport Ca 2+ across the membrane, generating a
An Energy-Transforming Enzyme In many biochemical reactions, the energy of the reactants is converted with high efficiency into a different form. For example, in photosynthesis, light energy is converted into chemical-bond energy through an ion gradient. 4 Ca 2+ ATPase uses the energy of ATP hydrolysis to transport Ca 2+ across the membrane, generating a
 Six major classes of enzymes are classified on the basis of the types of reactions that they catalyze Specific reactions of each type appear repeatedly, further reducing the number of reactions necessary for the student to learn .
Six major classes of enzymes are classified on the basis of the types of reactions that they catalyze Specific reactions of each type appear repeatedly, further reducing the number of reactions necessary for the student to learn .
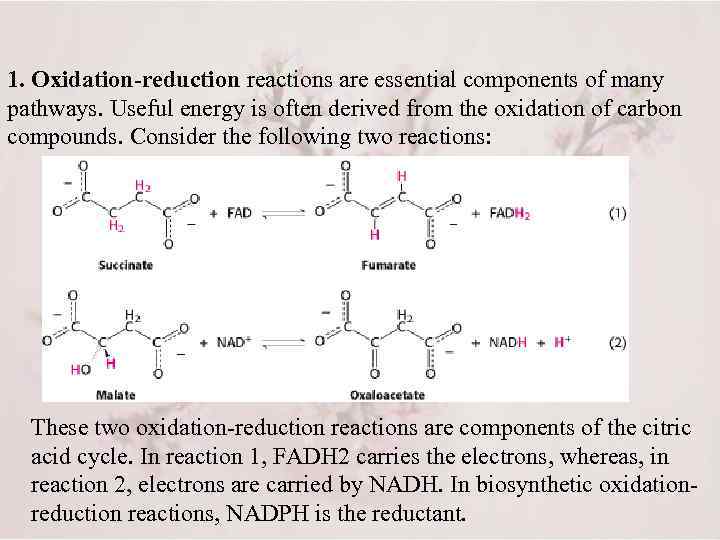 1. Oxidation-reduction reactions are essential components of many pathways. Useful energy is often derived from the oxidation of carbon compounds. Consider the following two reactions: These two oxidation-reduction reactions are components of the citric acid cycle. In reaction 1, FADH 2 carries the electrons, whereas, in reaction 2, electrons are carried by NADH. In biosynthetic oxidationreduction reactions, NADPH is the reductant.
1. Oxidation-reduction reactions are essential components of many pathways. Useful energy is often derived from the oxidation of carbon compounds. Consider the following two reactions: These two oxidation-reduction reactions are components of the citric acid cycle. In reaction 1, FADH 2 carries the electrons, whereas, in reaction 2, electrons are carried by NADH. In biosynthetic oxidationreduction reactions, NADPH is the reductant.
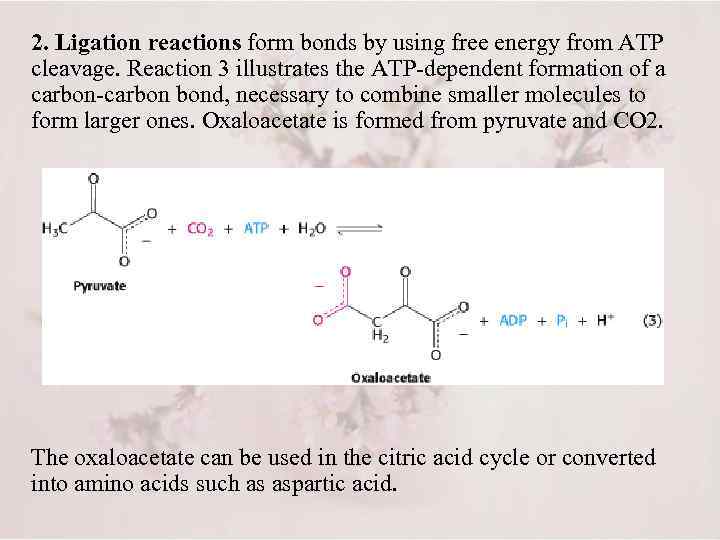 2. Ligation reactions form bonds by using free energy from ATP cleavage. Reaction 3 illustrates the ATP-dependent formation of a carbon-carbon bond, necessary to combine smaller molecules to form larger ones. Oxaloacetate is formed from pyruvate and CO 2. The oxaloacetate can be used in the citric acid cycle or converted into amino acids such as aspartic acid.
2. Ligation reactions form bonds by using free energy from ATP cleavage. Reaction 3 illustrates the ATP-dependent formation of a carbon-carbon bond, necessary to combine smaller molecules to form larger ones. Oxaloacetate is formed from pyruvate and CO 2. The oxaloacetate can be used in the citric acid cycle or converted into amino acids such as aspartic acid.
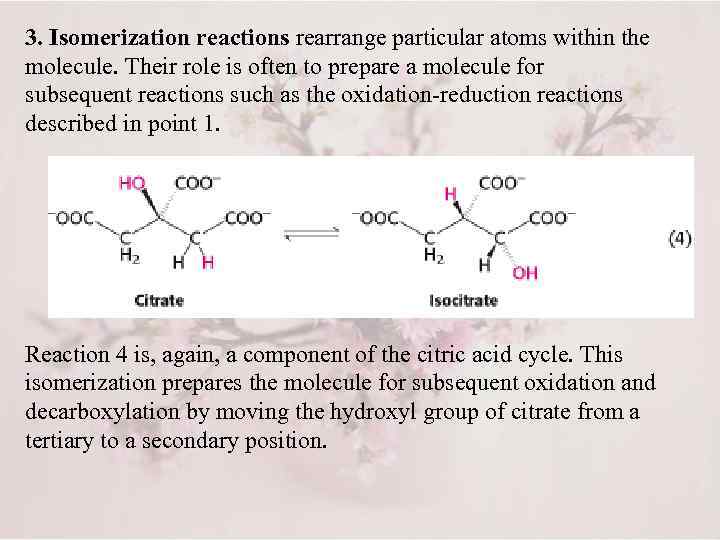 3. Isomerization reactions rearrange particular atoms within the molecule. Their role is often to prepare a molecule for subsequent reactions such as the oxidation-reduction reactions described in point 1. Reaction 4 is, again, a component of the citric acid cycle. This isomerization prepares the molecule for subsequent oxidation and decarboxylation by moving the hydroxyl group of citrate from a tertiary to a secondary position.
3. Isomerization reactions rearrange particular atoms within the molecule. Their role is often to prepare a molecule for subsequent reactions such as the oxidation-reduction reactions described in point 1. Reaction 4 is, again, a component of the citric acid cycle. This isomerization prepares the molecule for subsequent oxidation and decarboxylation by moving the hydroxyl group of citrate from a tertiary to a secondary position.
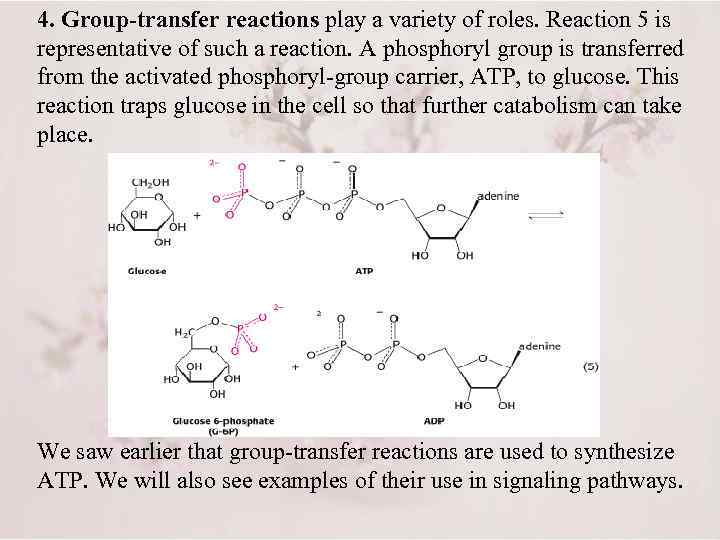 4. Group-transfer reactions play a variety of roles. Reaction 5 is representative of such a reaction. A phosphoryl group is transferred from the activated phosphoryl-group carrier, ATP, to glucose. This reaction traps glucose in the cell so that further catabolism can take place. We saw earlier that group-transfer reactions are used to synthesize ATP. We will also see examples of their use in signaling pathways.
4. Group-transfer reactions play a variety of roles. Reaction 5 is representative of such a reaction. A phosphoryl group is transferred from the activated phosphoryl-group carrier, ATP, to glucose. This reaction traps glucose in the cell so that further catabolism can take place. We saw earlier that group-transfer reactions are used to synthesize ATP. We will also see examples of their use in signaling pathways.
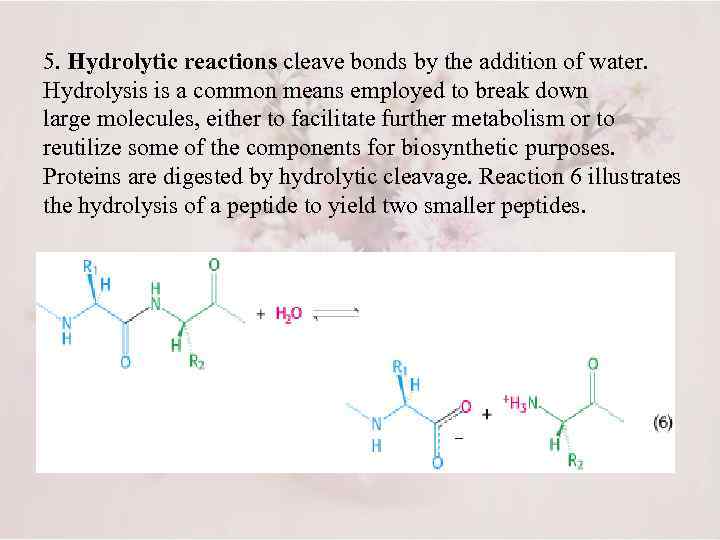 5. Hydrolytic reactions cleave bonds by the addition of water. Hydrolysis is a common means employed to break down large molecules, either to facilitate further metabolism or to reutilize some of the components for biosynthetic purposes. Proteins are digested by hydrolytic cleavage. Reaction 6 illustrates the hydrolysis of a peptide to yield two smaller peptides.
5. Hydrolytic reactions cleave bonds by the addition of water. Hydrolysis is a common means employed to break down large molecules, either to facilitate further metabolism or to reutilize some of the components for biosynthetic purposes. Proteins are digested by hydrolytic cleavage. Reaction 6 illustrates the hydrolysis of a peptide to yield two smaller peptides.
 6. The addition of functional groups to double bonds or the removal of groups to form double bonds constitutes the final class of reactions. The enzymes that catalyze these types of reaction are classified as lyases. An important example, illustrated in reaction 7, is the conversion of the six-carbon molecule fructose 1, 6 -bisphosphate (F-1, 6 -BP) into 2 three-carbon fragments: dihydroxyacetone phosphate and glyceraldehyde 3 -phosphate. This reaction is a key step in glycolysis, a key pathway for extracting energy from glucose. Dehydrations to form double bonds, such as the formation of phosphoenolpyruvate (Table 14. 1) from 2 phosphoglycerate, are important reactions of this type. The dehydration sets up the next step in the pathway, a group-transfer reaction that uses the high phosphoryl transfer potential of the product PEP to form ATP from ADP.
6. The addition of functional groups to double bonds or the removal of groups to form double bonds constitutes the final class of reactions. The enzymes that catalyze these types of reaction are classified as lyases. An important example, illustrated in reaction 7, is the conversion of the six-carbon molecule fructose 1, 6 -bisphosphate (F-1, 6 -BP) into 2 three-carbon fragments: dihydroxyacetone phosphate and glyceraldehyde 3 -phosphate. This reaction is a key step in glycolysis, a key pathway for extracting energy from glucose. Dehydrations to form double bonds, such as the formation of phosphoenolpyruvate (Table 14. 1) from 2 phosphoglycerate, are important reactions of this type. The dehydration sets up the next step in the pathway, a group-transfer reaction that uses the high phosphoryl transfer potential of the product PEP to form ATP from ADP.
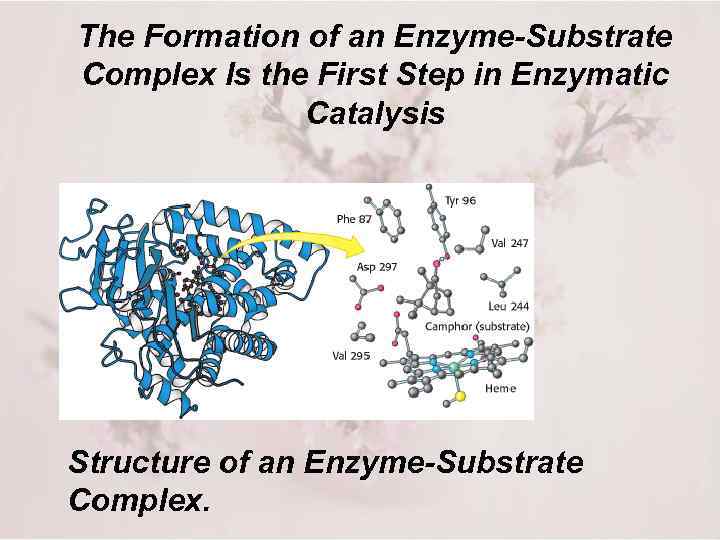 The Formation of an Enzyme-Substrate Complex Is the First Step in Enzymatic Catalysis Structure of an Enzyme-Substrate Complex.
The Formation of an Enzyme-Substrate Complex Is the First Step in Enzymatic Catalysis Structure of an Enzyme-Substrate Complex.
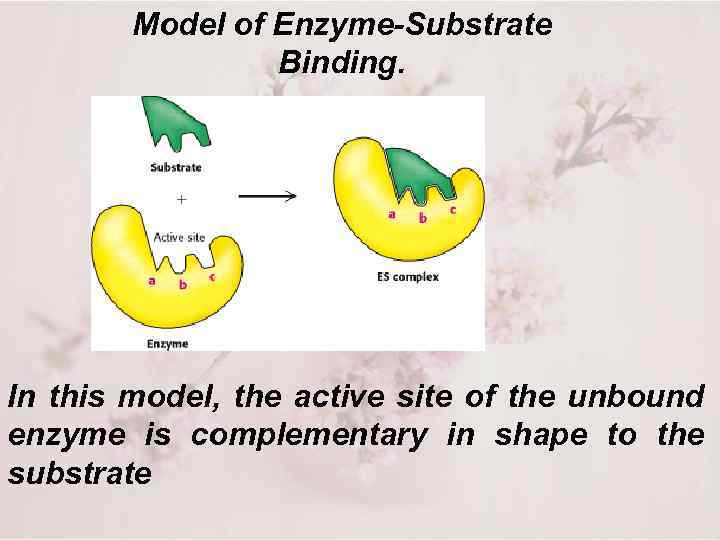 Model of Enzyme-Substrate Binding. In this model, the active site of the unbound enzyme is complementary in shape to the substrate
Model of Enzyme-Substrate Binding. In this model, the active site of the unbound enzyme is complementary in shape to the substrate
 Enzymes Can Be Inhibited by Specific Molecules This means of inhibiting enzyme activity serves as a major control mechanism in biological systems. The regulation of allosteric enzymes typifies this type of control. In addition, many drugs and toxic agents act by inhibiting enzymes Enzyme inhibition can be either reversible or irreversible. An irreversible inhibitor dissociates very slowly from its target enzyme because it has become tightly bound to the enzyme, either covalently or noncovalently. Some irreversible inhibitors are important drugs. Penicillin acts by covalently modifying the enzyme transpeptidase, thereby preventing the synthesis of bacterial cell walls and thus killing the bacteria (Section 8. 5. 5). Aspirin acts by covalently modifying the enzyme cyclooxygenase, reducing the synthesis of inflammatory signals. Reversible inhibition, in contrast with irreversible inhibition, is characterized by a rapid dissociation of the enzymeinhibitor complex.
Enzymes Can Be Inhibited by Specific Molecules This means of inhibiting enzyme activity serves as a major control mechanism in biological systems. The regulation of allosteric enzymes typifies this type of control. In addition, many drugs and toxic agents act by inhibiting enzymes Enzyme inhibition can be either reversible or irreversible. An irreversible inhibitor dissociates very slowly from its target enzyme because it has become tightly bound to the enzyme, either covalently or noncovalently. Some irreversible inhibitors are important drugs. Penicillin acts by covalently modifying the enzyme transpeptidase, thereby preventing the synthesis of bacterial cell walls and thus killing the bacteria (Section 8. 5. 5). Aspirin acts by covalently modifying the enzyme cyclooxygenase, reducing the synthesis of inflammatory signals. Reversible inhibition, in contrast with irreversible inhibition, is characterized by a rapid dissociation of the enzymeinhibitor complex.
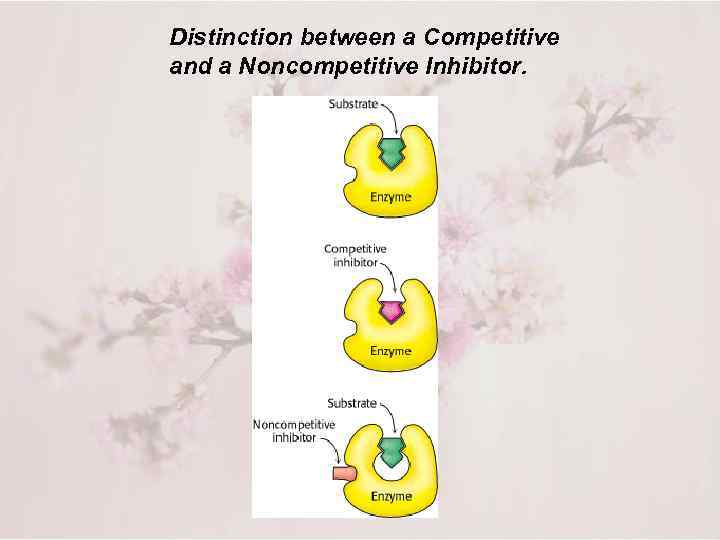 Distinction between a Competitive and a Noncompetitive Inhibitor.
Distinction between a Competitive and a Noncompetitive Inhibitor.
 Thank you for attention !
Thank you for attention !


