a2cb9f4701ab0bd5ca055b98c15eb6c9.ppt
- Количество слайдов: 13
 The Laws of Thermodynamics and Air Conditioners (your best friends during this summer)
The Laws of Thermodynamics and Air Conditioners (your best friends during this summer)
 What a Fridge and an Air Conditioner does ? • move heat from a hot place to a cold place • these machines generically called a ‘heat pump’ Are there rules/laws that govern this Motion of Heat ? • Yes, these are called ‘laws of thermodynamics’ • Establish relationships between Thermal quantities (Temperature T, Internal Energy U, Entropy) and Mechanical quantities (Work) • There are 4 laws
What a Fridge and an Air Conditioner does ? • move heat from a hot place to a cold place • these machines generically called a ‘heat pump’ Are there rules/laws that govern this Motion of Heat ? • Yes, these are called ‘laws of thermodynamics’ • Establish relationships between Thermal quantities (Temperature T, Internal Energy U, Entropy) and Mechanical quantities (Work) • There are 4 laws
 Zeroth Law of Thermodynamics (definition of temperature) Two objects that are each in thermal equilibrium with a third object are also in thermal equilibrium with one another. A 28 C T=28 C B 28 C There is no way you can do a net transfer heat from house A to B if both are in equilibrium with the environment. i. e. heat will not flow from A to B.
Zeroth Law of Thermodynamics (definition of temperature) Two objects that are each in thermal equilibrium with a third object are also in thermal equilibrium with one another. A 28 C T=28 C B 28 C There is no way you can do a net transfer heat from house A to B if both are in equilibrium with the environment. i. e. heat will not flow from A to B.
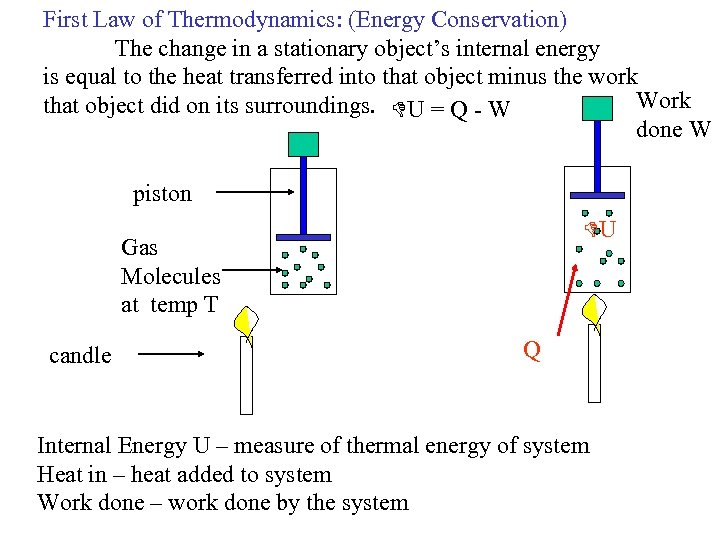 First Law of Thermodynamics: (Energy Conservation) The change in a stationary object’s internal energy is equal to the heat transferred into that object minus the work Work that object did on its surroundings. U = Q - W done W piston U Gas Molecules at temp T candle Q Internal Energy U – measure of thermal energy of system Heat in – heat added to system Work done – work done by the system
First Law of Thermodynamics: (Energy Conservation) The change in a stationary object’s internal energy is equal to the heat transferred into that object minus the work Work that object did on its surroundings. U = Q - W done W piston U Gas Molecules at temp T candle Q Internal Energy U – measure of thermal energy of system Heat in – heat added to system Work done – work done by the system
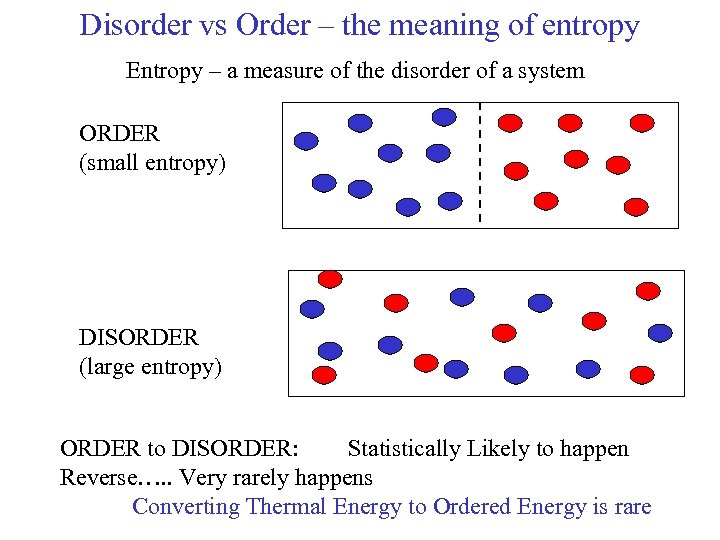 Disorder vs Order – the meaning of entropy Entropy – a measure of the disorder of a system ORDER (small entropy) DISORDER (large entropy) ORDER to DISORDER: Statistically Likely to happen Reverse…. . Very rarely happens Converting Thermal Energy to Ordered Energy is rare
Disorder vs Order – the meaning of entropy Entropy – a measure of the disorder of a system ORDER (small entropy) DISORDER (large entropy) ORDER to DISORDER: Statistically Likely to happen Reverse…. . Very rarely happens Converting Thermal Energy to Ordered Energy is rare
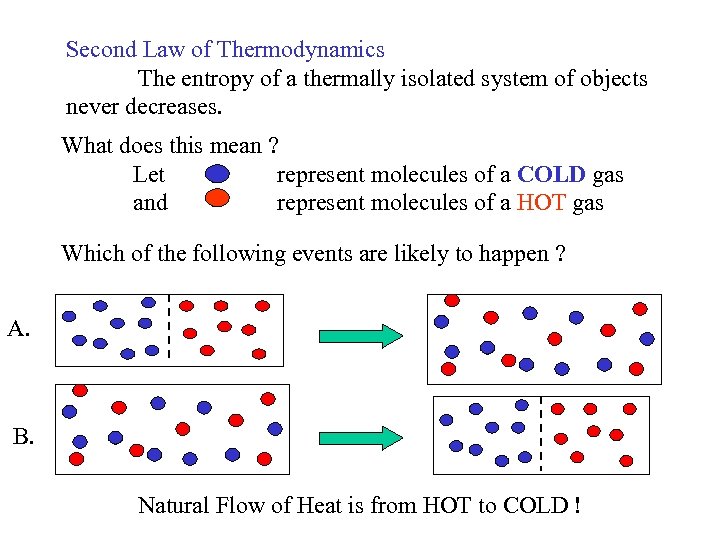 Second Law of Thermodynamics The entropy of a thermally isolated system of objects never decreases. What does this mean ? Let represent molecules of a COLD gas and represent molecules of a HOT gas Which of the following events are likely to happen ? A. B. Natural Flow of Heat is from HOT to COLD !
Second Law of Thermodynamics The entropy of a thermally isolated system of objects never decreases. What does this mean ? Let represent molecules of a COLD gas and represent molecules of a HOT gas Which of the following events are likely to happen ? A. B. Natural Flow of Heat is from HOT to COLD !
 Third Law of Thermodynamics As an object’s temperature approaches zero, its entropy approaches zero.
Third Law of Thermodynamics As an object’s temperature approaches zero, its entropy approaches zero.
 What does a Fridge or Air conditioner do ? • It does the Un-natural: Moves heat from HOT to COLD • Uses a circulating fluid, usually freon, to move heat Key Features of an Air conditioner 1. Evaporator • located in hot room • transfers heat from hot room air to fluid 2. Condenser • located outside room • Transfers heat from fluid to outside air 3. Compressor • located outside room • does work on the fluid by compressing it and creating entropy
What does a Fridge or Air conditioner do ? • It does the Un-natural: Moves heat from HOT to COLD • Uses a circulating fluid, usually freon, to move heat Key Features of an Air conditioner 1. Evaporator • located in hot room • transfers heat from hot room air to fluid 2. Condenser • located outside room • Transfers heat from fluid to outside air 3. Compressor • located outside room • does work on the fluid by compressing it and creating entropy
 A Buffet of Air Conditioners Residential Compressor/ Condenser Wall-mounted air conditioner outside Apartments: compressor/condenser Outside hospitals, etc
A Buffet of Air Conditioners Residential Compressor/ Condenser Wall-mounted air conditioner outside Apartments: compressor/condenser Outside hospitals, etc
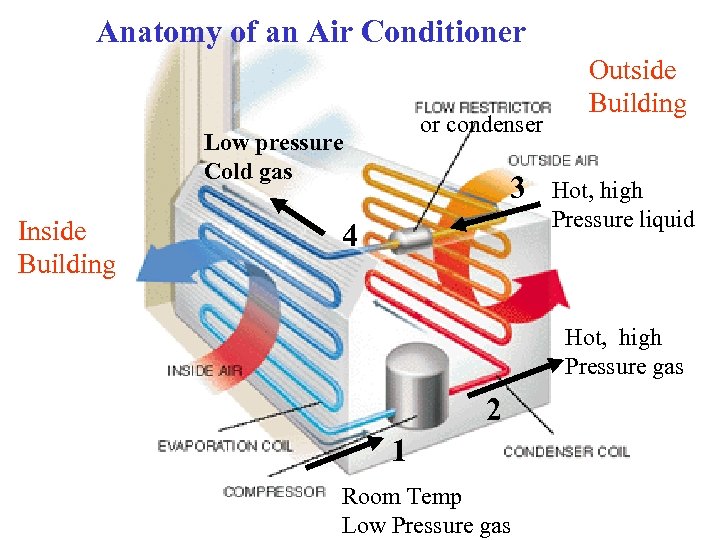 Anatomy of an Air Conditioner or condenser Low pressure Cold gas Inside Building 3 4 Outside Building Hot, high Pressure liquid Hot, high Pressure gas 2 1 Room Temp Low Pressure gas
Anatomy of an Air Conditioner or condenser Low pressure Cold gas Inside Building 3 4 Outside Building Hot, high Pressure liquid Hot, high Pressure gas 2 1 Room Temp Low Pressure gas
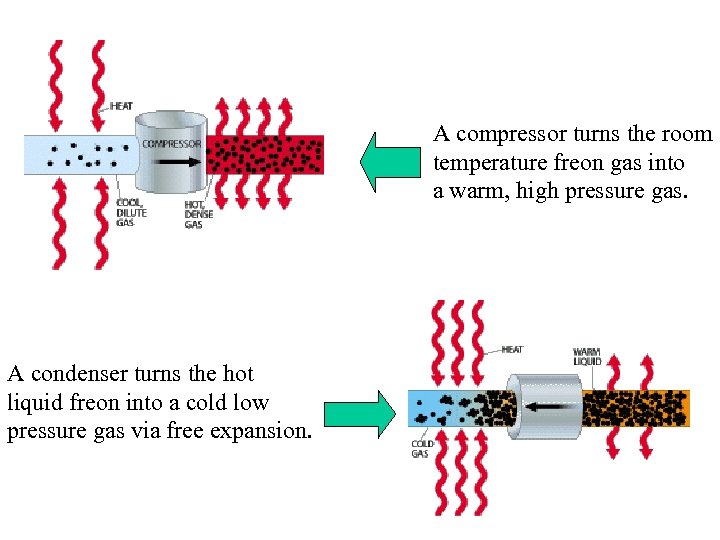 A compressor turns the room temperature freon gas into a warm, high pressure gas. A condenser turns the hot liquid freon into a cold low pressure gas via free expansion.
A compressor turns the room temperature freon gas into a warm, high pressure gas. A condenser turns the hot liquid freon into a cold low pressure gas via free expansion.
 The Refrigeration Sequence in an Air conditioner Process 4 1: Heat is absorbed by freon gas in tubes inside a warm room. Becomes a room temperature low pressure gas before entering compressor. Process 1 2: Room Temp low pressure freon gas enters a compressor and comes out as a high pressure hot gas. Passing thru tubes outside building, hot freon dumps heat to outside air Process 2 3: Warm freon gas enters a constriction and is further pressurized to form a liquid in the condenser. Process 3 4. Freon liquid undergoes free expansion into a gas and cools. The cool gas then takes in heat from the room, becoming a room temperature low pressure gas The cycle is then repeated.
The Refrigeration Sequence in an Air conditioner Process 4 1: Heat is absorbed by freon gas in tubes inside a warm room. Becomes a room temperature low pressure gas before entering compressor. Process 1 2: Room Temp low pressure freon gas enters a compressor and comes out as a high pressure hot gas. Passing thru tubes outside building, hot freon dumps heat to outside air Process 2 3: Warm freon gas enters a constriction and is further pressurized to form a liquid in the condenser. Process 3 4. Freon liquid undergoes free expansion into a gas and cools. The cool gas then takes in heat from the room, becoming a room temperature low pressure gas The cycle is then repeated.
 Although the airconditioner pumps heat from cold to hot regions, it doesn’t violate the 2 nd law of thermodynamics. The compressor adds entropy, so that the total entropy of the system actually increases.
Although the airconditioner pumps heat from cold to hot regions, it doesn’t violate the 2 nd law of thermodynamics. The compressor adds entropy, so that the total entropy of the system actually increases.


