391f46666fffb51b4d557ffbe0a68c53.ppt
- Количество слайдов: 15
 The Late. TIME Randomized Trial: Effect of Intracoronary Delivery of Autologous Bone Marrow Mononuclear Cells 2 -3 Weeks following AMI Jay H Traverse, MD Principal Investigator, Late. TIME Study Minneapolis Heart Institute at Abbott Northwestern Hospital University of Minnesota Medical School Cardiovascular Cell Therapy Research Network 2011 Scientific Sessions of the AHA JAMA 2011; 306: 2110 -19
The Late. TIME Randomized Trial: Effect of Intracoronary Delivery of Autologous Bone Marrow Mononuclear Cells 2 -3 Weeks following AMI Jay H Traverse, MD Principal Investigator, Late. TIME Study Minneapolis Heart Institute at Abbott Northwestern Hospital University of Minnesota Medical School Cardiovascular Cell Therapy Research Network 2011 Scientific Sessions of the AHA JAMA 2011; 306: 2110 -19
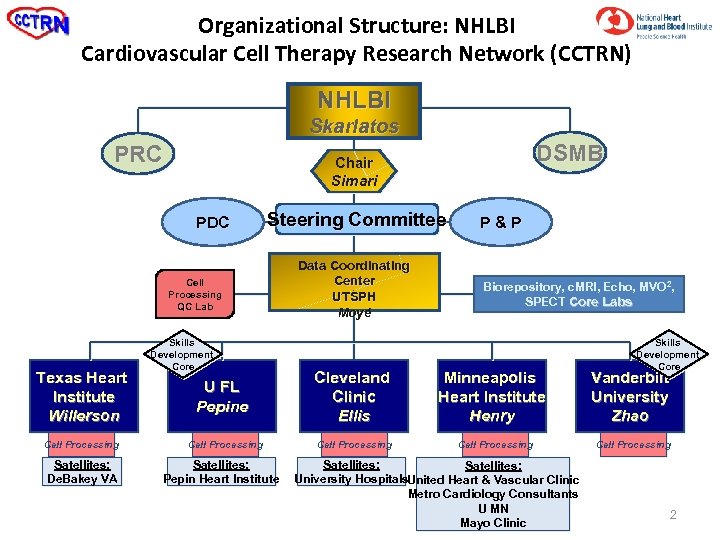 Organizational Structure: NHLBI Cardiovascular Cell Therapy Research Network (CCTRN) NHLBI Skarlatos PRC PDC Steering Committee Cell Processing QC Lab Texas Heart Institute Willerson Cell Processing Satellites: De. Bakey VA DSMB Chair Simari Skills Development Core Data Coordinating Center UTSPH Moyé P&P Biorepository, c. MRI, Echo, MVO 2, SPECT Core Labs Skills Development Core U FL Pepine Cleveland Clinic Ellis Minneapolis Heart Institute Henry Vanderbilt University Zhao Cell Processing Satellites: Pepin Heart Institute Satellites: University Hospitals. United Heart & Vascular Clinic Metro Cardiology Consultants U MN Mayo Clinic 2
Organizational Structure: NHLBI Cardiovascular Cell Therapy Research Network (CCTRN) NHLBI Skarlatos PRC PDC Steering Committee Cell Processing QC Lab Texas Heart Institute Willerson Cell Processing Satellites: De. Bakey VA DSMB Chair Simari Skills Development Core Data Coordinating Center UTSPH Moyé P&P Biorepository, c. MRI, Echo, MVO 2, SPECT Core Labs Skills Development Core U FL Pepine Cleveland Clinic Ellis Minneapolis Heart Institute Henry Vanderbilt University Zhao Cell Processing Satellites: Pepin Heart Institute Satellites: University Hospitals. United Heart & Vascular Clinic Metro Cardiology Consultants U MN Mayo Clinic 2
 Rationale for Late. TIME • Optimal timing for cell delivery post-AMI is unknown • Almost all BMC Trials delivered cells ≤ 7 days post-AMI • Cell delivery 2 -3 wks post-MI may be an important time period for patients: -initially too sick, or -presenting to hospitals without these capabilities 3
Rationale for Late. TIME • Optimal timing for cell delivery post-AMI is unknown • Almost all BMC Trials delivered cells ≤ 7 days post-AMI • Cell delivery 2 -3 wks post-MI may be an important time period for patients: -initially too sick, or -presenting to hospitals without these capabilities 3
 Late. TIME Study Design • Study Aim: assess safety and efficacy of autologous BMCs delivered 2 -3 wks post-MI on LV function • Target Population: 87 patients w/first large Infuse product via LAD MI, reperfused by PCI + stent, with residual LV dysfunction (EF≤ 45%) • Treatment: 150 x 106 autologous BMCs or placebo delivered by intracoronary infusion (Stop Flow) • 1⁰ Endpoints: change in global and regional LV function from baseline to 6 months by cardiac MRI. • Secondary Endpoints: change in infarct size and LV Volumes 4
Late. TIME Study Design • Study Aim: assess safety and efficacy of autologous BMCs delivered 2 -3 wks post-MI on LV function • Target Population: 87 patients w/first large Infuse product via LAD MI, reperfused by PCI + stent, with residual LV dysfunction (EF≤ 45%) • Treatment: 150 x 106 autologous BMCs or placebo delivered by intracoronary infusion (Stop Flow) • 1⁰ Endpoints: change in global and regional LV function from baseline to 6 months by cardiac MRI. • Secondary Endpoints: change in infarct size and LV Volumes 4
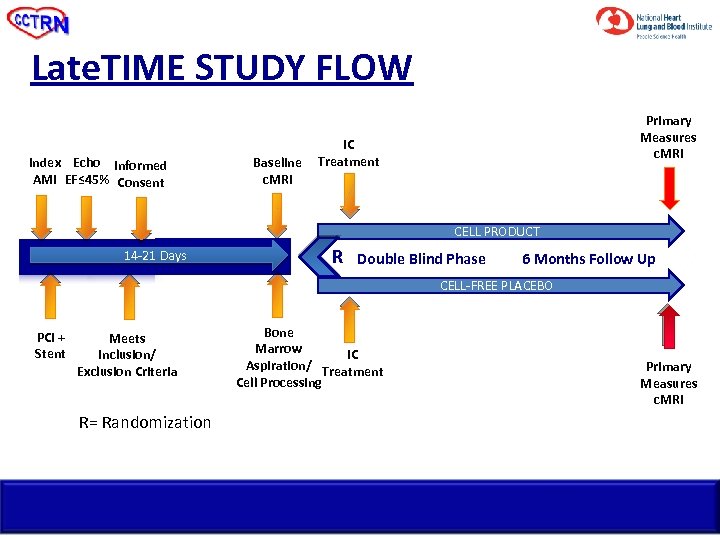 Late. TIME STUDY FLOW Index Echo Informed AMI EF≤ 45% Consent Baseline c. MRI Primary Measures c. MRI IC Treatment CELL PRODUCT 14 -21 Days R Double Blind Phase 6 Months Follow Up CELL-FREE PLACEBO PCI + Stent Meets Inclusion/ Exclusion Criteria R= Randomization Bone Marrow IC Aspiration/ Treatment Cell Processing Primary Measures c. MRI
Late. TIME STUDY FLOW Index Echo Informed AMI EF≤ 45% Consent Baseline c. MRI Primary Measures c. MRI IC Treatment CELL PRODUCT 14 -21 Days R Double Blind Phase 6 Months Follow Up CELL-FREE PLACEBO PCI + Stent Meets Inclusion/ Exclusion Criteria R= Randomization Bone Marrow IC Aspiration/ Treatment Cell Processing Primary Measures c. MRI
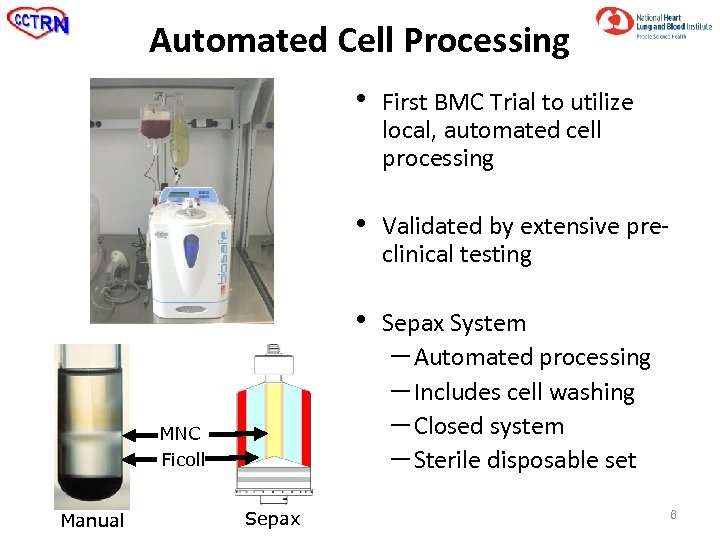 Automated Cell Processing • • Manual Sepax Validated by extensive preclinical testing • MNC Ficoll First BMC Trial to utilize local, automated cell processing Sepax System – Automated processing – Includes cell washing – Closed system – Sterile disposable set 6
Automated Cell Processing • • Manual Sepax Validated by extensive preclinical testing • MNC Ficoll First BMC Trial to utilize local, automated cell processing Sepax System – Automated processing – Includes cell washing – Closed system – Sterile disposable set 6
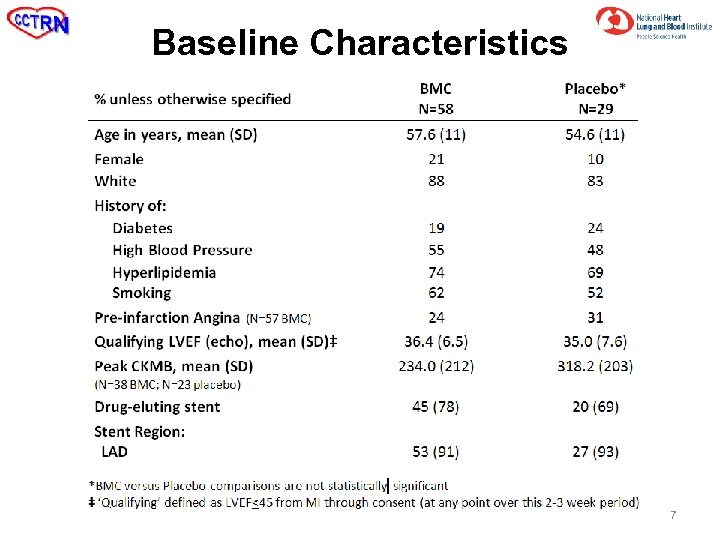 Baseline Characteristics 7
Baseline Characteristics 7
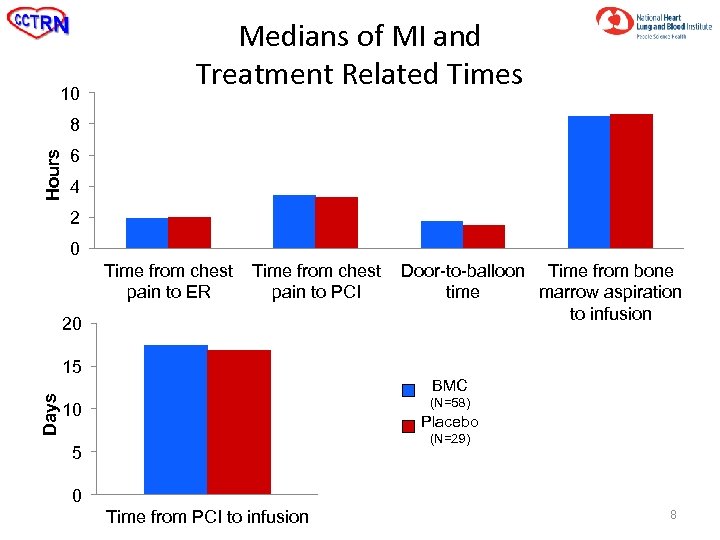 10 Medians of MI and Treatment Related Times Hours 8 6 4 2 0 Time from chest pain to ER Time from chest pain to PCI 20 Door-to-balloon Time from bone time marrow aspiration to infusion Days 15 BMC (N=58) 10 Placebo (N=29) 5 0 Time from PCI to infusion 8
10 Medians of MI and Treatment Related Times Hours 8 6 4 2 0 Time from chest pain to ER Time from chest pain to PCI 20 Door-to-balloon Time from bone time marrow aspiration to infusion Days 15 BMC (N=58) 10 Placebo (N=29) 5 0 Time from PCI to infusion 8
 Cell Characteristics 9
Cell Characteristics 9
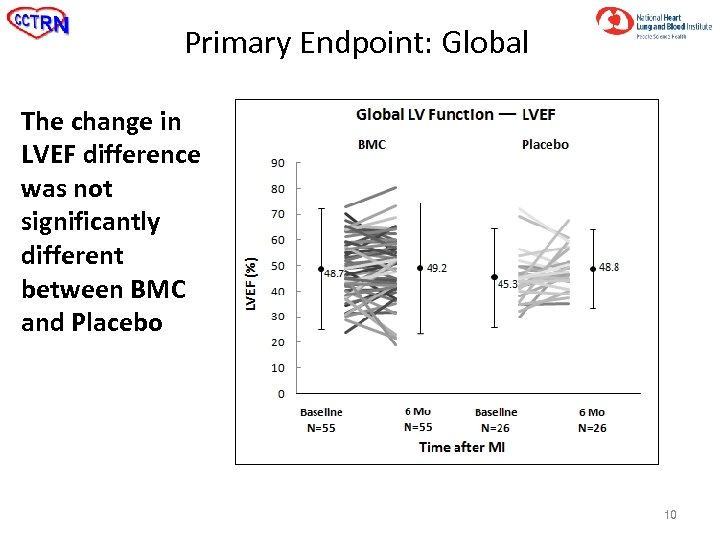 Primary Endpoint: Global The change in LVEF difference was not significantly different between BMC and Placebo 10
Primary Endpoint: Global The change in LVEF difference was not significantly different between BMC and Placebo 10
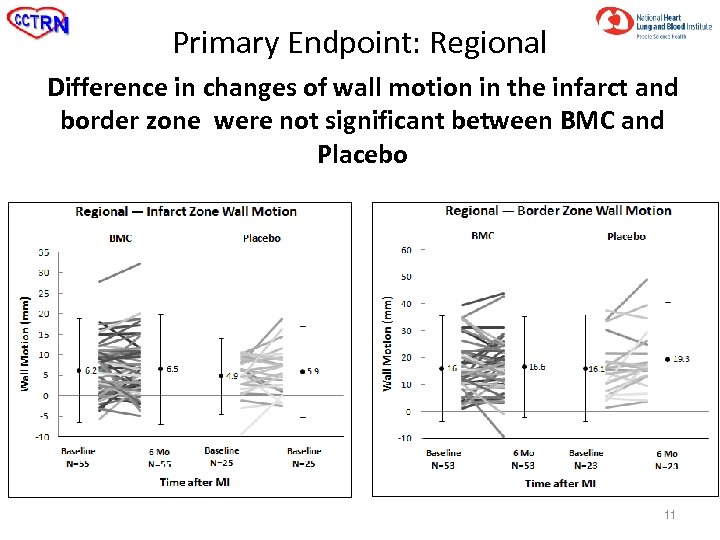 Primary Endpoint: Regional Difference in changes of wall motion in the infarct and border zone were not significant between BMC and Placebo 11
Primary Endpoint: Regional Difference in changes of wall motion in the infarct and border zone were not significant between BMC and Placebo 11
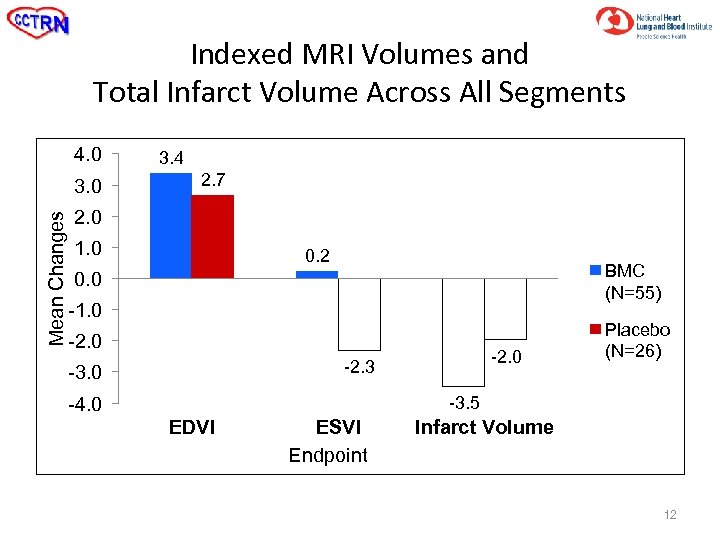 Indexed MRI Volumes and Total Infarct Volume Across All Segments 4. 0 Mean Changes 3. 0 3. 4 2. 7 2. 0 1. 0 0. 2 BMC (N=55) 0. 0 -1. 0 -2. 3 -3. 0 -4. 0 Placebo (N=26) -3. 5 EDVI ESVI Endpoint Infarct Volume 12
Indexed MRI Volumes and Total Infarct Volume Across All Segments 4. 0 Mean Changes 3. 0 3. 4 2. 7 2. 0 1. 0 0. 2 BMC (N=55) 0. 0 -1. 0 -2. 3 -3. 0 -4. 0 Placebo (N=26) -3. 5 EDVI ESVI Endpoint Infarct Volume 12
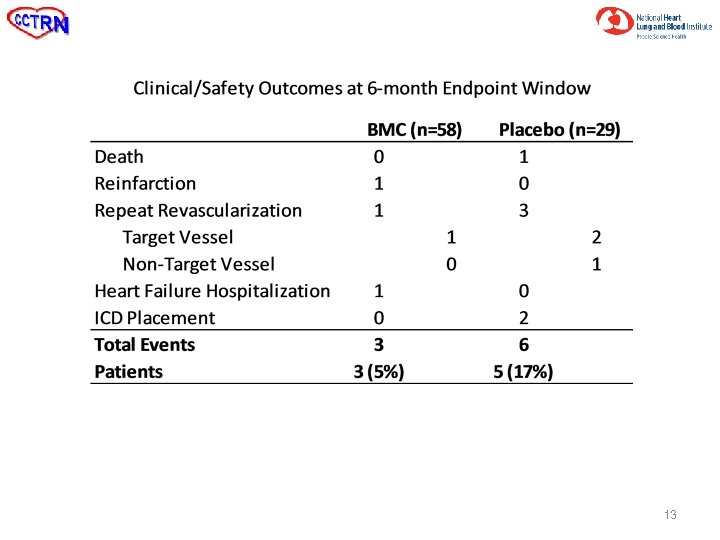 13
13
 Conclusions • Intracoronary delivery of autologous BMCs 2 -3 weeks following primary PCI + stenting after moderate to large acute MIs is safe. • No benefit of BMC therapy on global and regional LV function is observed at 6 months by c MRI. 14
Conclusions • Intracoronary delivery of autologous BMCs 2 -3 weeks following primary PCI + stenting after moderate to large acute MIs is safe. • No benefit of BMC therapy on global and regional LV function is observed at 6 months by c MRI. 14
 Acknowledgements • • National Heart Lung & Blood Institute • • University of Texas School of Public Health Biosafe Boston Scientific The clinical centers (Cleveland Clinic, Minneapolis Heart Institute, Texas Heart Institute, University of Florida, and Vanderbilt University), their satellites (University Hospitals, St. Paul Heart Clinic, Metropolitan Cardiology Consultants, University of Minnesota, Mayo Clinic, De. Bakey VA, and Pepin Heart Hospital) and their research teams Center for Cell & Gene Therapy, Baylor College of Medicine The University of Florida c. MRI and Cleveland Clinic Echo core labs The University of Minnesota and University of Florida Biorepositories 15
Acknowledgements • • National Heart Lung & Blood Institute • • University of Texas School of Public Health Biosafe Boston Scientific The clinical centers (Cleveland Clinic, Minneapolis Heart Institute, Texas Heart Institute, University of Florida, and Vanderbilt University), their satellites (University Hospitals, St. Paul Heart Clinic, Metropolitan Cardiology Consultants, University of Minnesota, Mayo Clinic, De. Bakey VA, and Pepin Heart Hospital) and their research teams Center for Cell & Gene Therapy, Baylor College of Medicine The University of Florida c. MRI and Cleveland Clinic Echo core labs The University of Minnesota and University of Florida Biorepositories 15


