40335874605cbe587a720e39f5f07c8d.ppt
- Количество слайдов: 35

The Intersection of Economics and Access: Sustainability Issues Andrew Farlow University of Oxford Conference on Innovation and Technological Transfer for Global Health 9 th-13 th September 2007 1

Overview of Session • Sustainable infrastructure and human resources • Sustainability of vaccine programs • Sustainability of global health funding • Power from the bottom to drive sustainability? 2
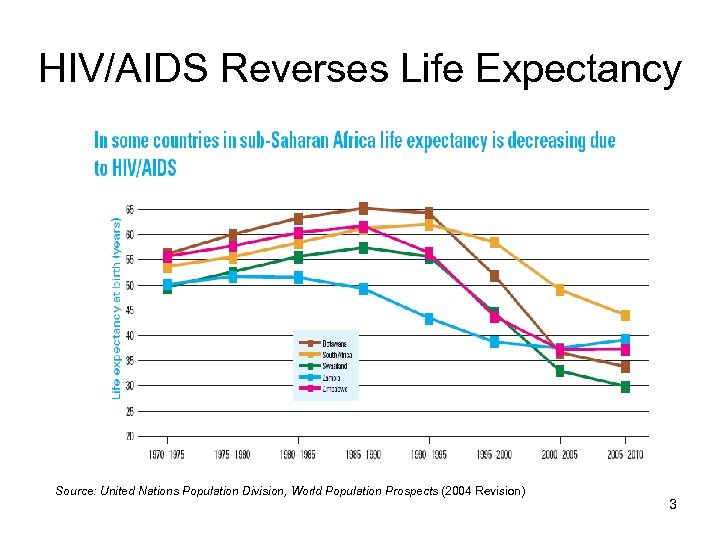
HIV/AIDS Reverses Life Expectancy Source: United Nations Population Division, World Population Prospects (2004 Revision) 3
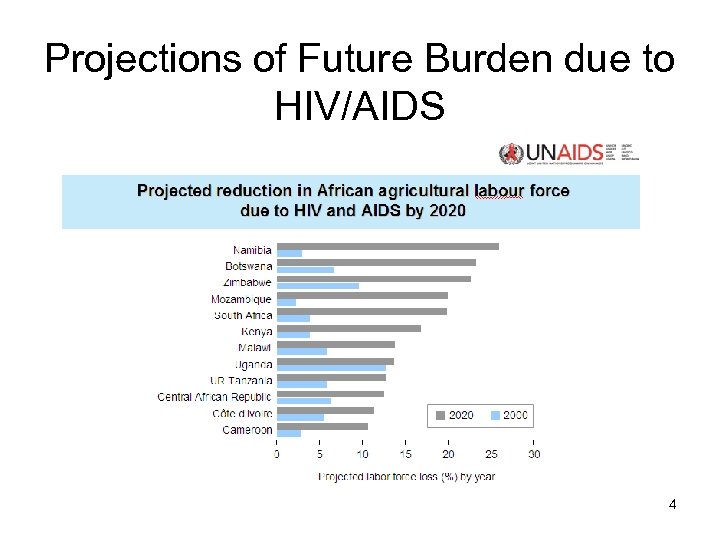
Projections of Future Burden due to HIV/AIDS 4
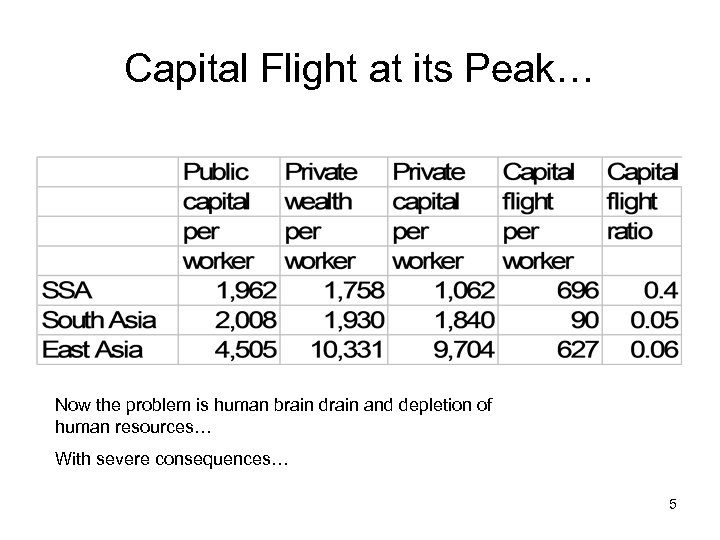
Capital Flight at its Peak… Now the problem is human brain drain and depletion of human resources… With severe consequences… 5
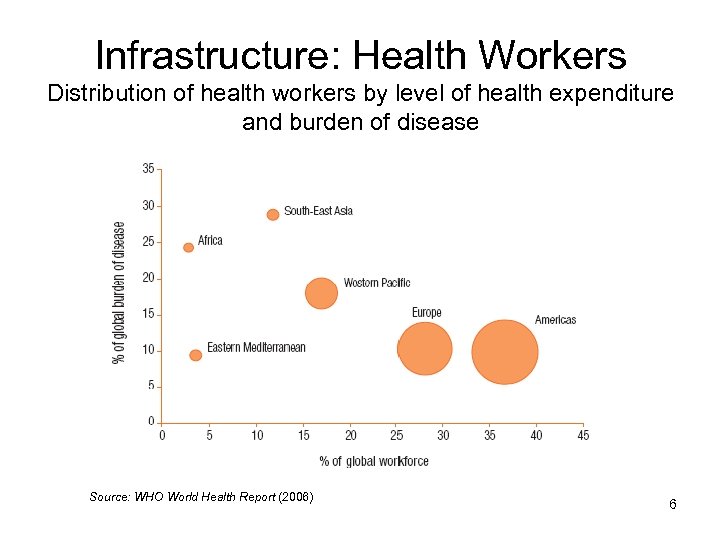
Infrastructure: Health Workers Distribution of health workers by level of health expenditure and burden of disease Source: WHO World Health Report (2006) 6
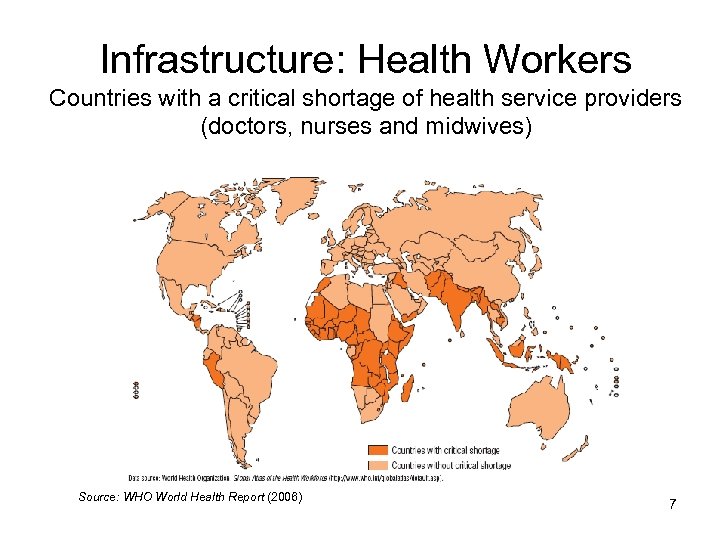
Infrastructure: Health Workers Countries with a critical shortage of health service providers (doctors, nurses and midwives) Source: WHO World Health Report (2006) 7
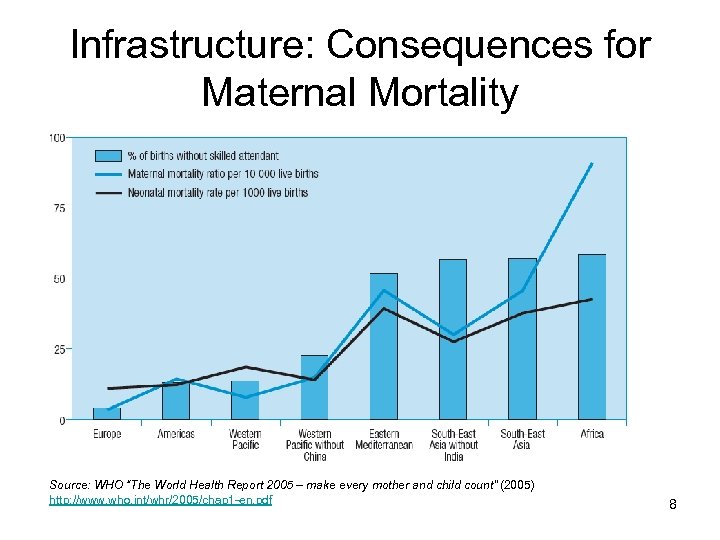
Infrastructure: Consequences for Maternal Mortality Source: WHO “The World Health Report 2005 – make every mother and child count” (2005) http: //www. who. int/whr/2005/chap 1 -en. pdf 8

Maternal Mortality per 100 000 Live Births in 2000 Source: WHO “The World Health Report 2005 – make every mother and child count” (2005) http: //www. who. int/whr/2005/chap 1 -en. pdf 9
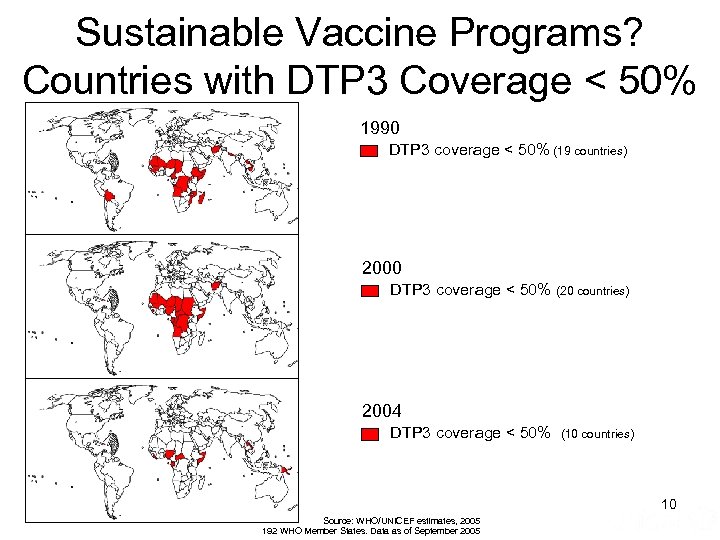
Sustainable Vaccine Programs? Countries with DTP 3 Coverage < 50% 1990 DTP 3 coverage < 50% (19 countries) 2000 DTP 3 coverage < 50% (20 countries) 2004 DTP 3 coverage < 50% (10 countries) 10 Source: WHO/UNICEF estimates, 2005 192 WHO Member States. Data as of September 2005
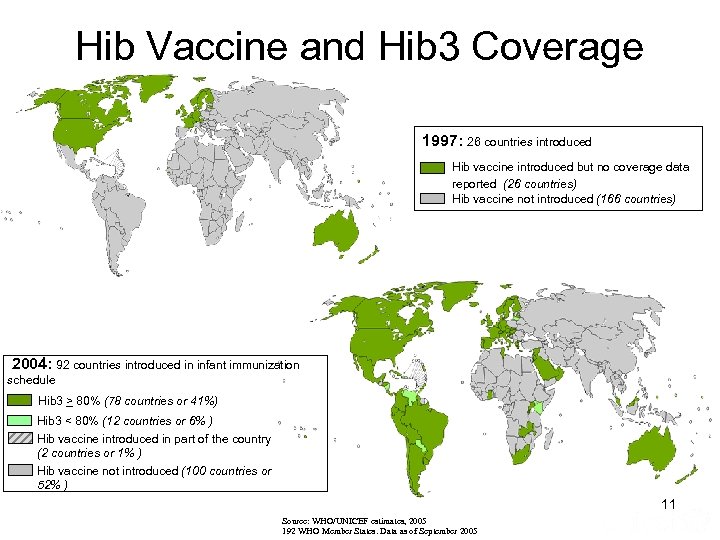
Hib Vaccine and Hib 3 Coverage 1997: 26 countries introduced Hib vaccine introduced but no coverage data reported (26 countries) Hib vaccine not introduced (166 countries) 2004: 92 countries introduced in infant immunization schedule Hib 3 > 80% (78 countries or 41%) Hib 3 < 80% (12 countries or 6% ) Hib vaccine introduced in part of the country (2 countries or 1% ) Hib vaccine not introduced (100 countries or 52% ) 11 Source: WHO/UNICEF estimates, 2005 192 WHO Member States. Data as of September 2005
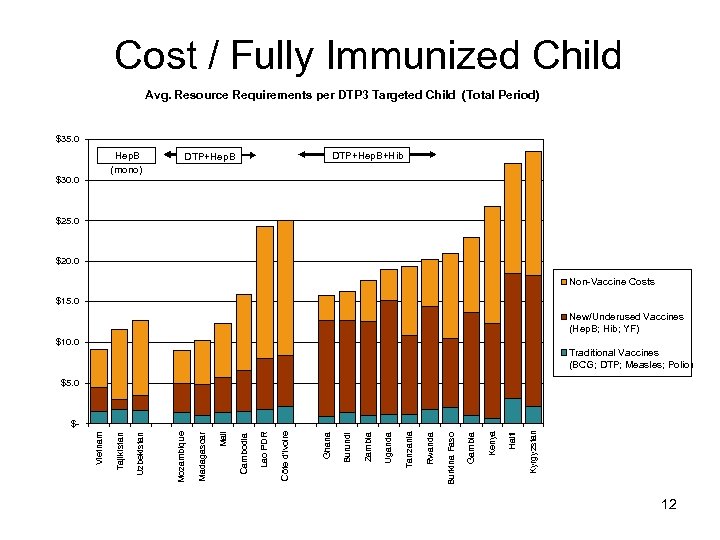
Cost / Fully Immunized Child Avg. Resource Requirements per DTP 3 Targeted Child (Total Period) $35. 0 Hep. B (mono) $30. 0 DTP+Hep. B+Hib DTP+Hep. B $25. 0 $20. 0 Non-Vaccine Costs $15. 0 New/Underused Vaccines (Hep. B; Hib; YF) $10. 0 Traditional Vaccines (BCG; DTP; Measles; Polio) $5. 0 Kyrgyzstan Haiti Kenya Gambia Burkina Faso Rwanda Tanzania Uganda Zambia Burundi Ghana Côte d'Ivoire Lao PDR Cambodia Mali Madagascar Mozambique Uzbekistan Tajikistan Vietnam $- 12
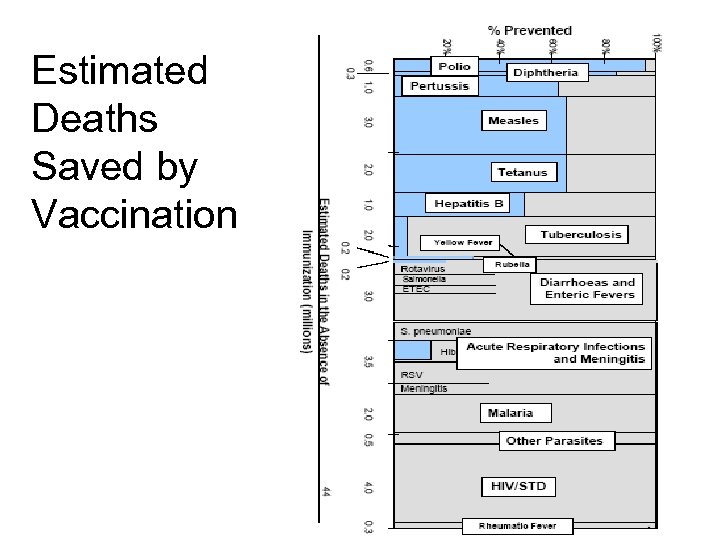
Estimated Deaths Saved by Vaccination 14
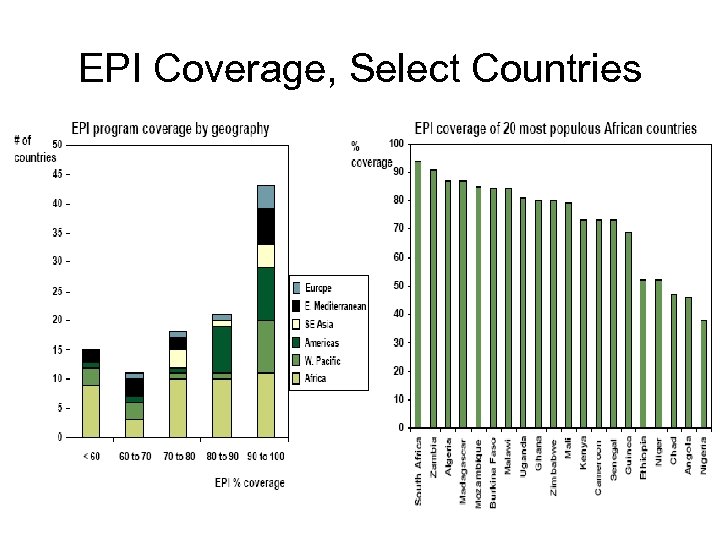
EPI Coverage, Select Countries 15
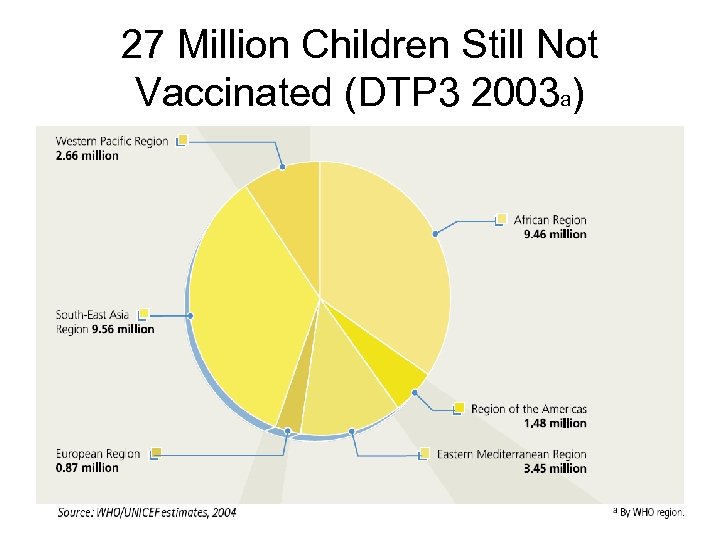
27 Million Children Still Not Vaccinated (DTP 3 2003 a) 16
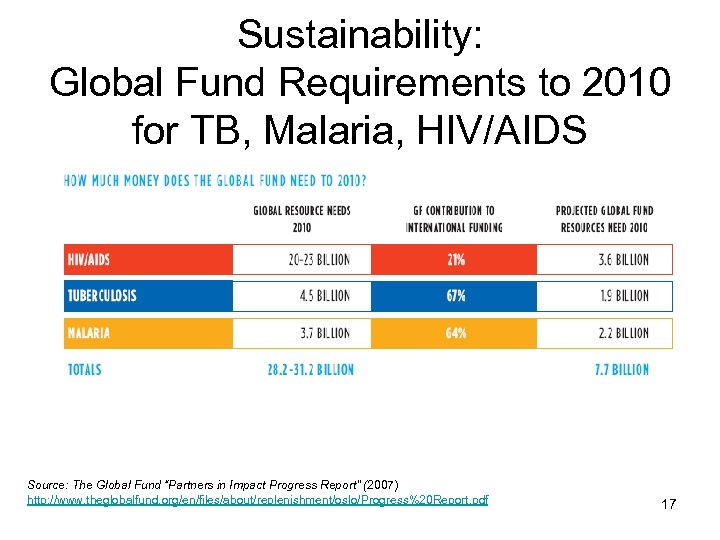
Sustainability: Global Fund Requirements to 2010 for TB, Malaria, HIV/AIDS Source: The Global Fund “Partners in Impact Progress Report” (2007) http: //www. theglobalfund. org/en/files/about/replenishment/oslo/Progress%20 Report. pdf 17
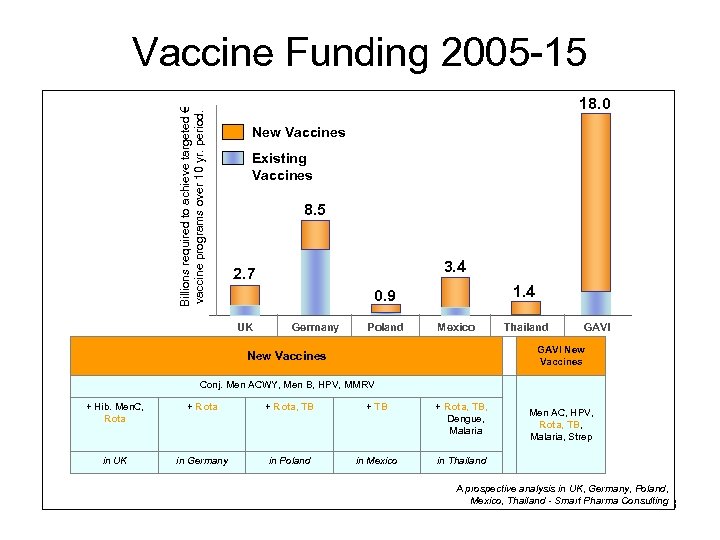
Billions required to achieve targeted € vaccine programs over 10 yr. period. Vaccine Funding 2005 -15 18. 0 New Vaccines Existing Vaccines 8. 5 3. 4 2. 7 1. 4 0. 9 UK Germany Poland Mexico Thailand GAVI New Vaccines Conj. Men ACWY, Men B, HPV, MMRV + Hib. Men. C, Rota + Rota, TB, Dengue, Malaria in UK in Germany in Poland in Mexico in Thailand Men AC, HPV, Rota, TB, Malaria, Strep A prospective analysis in UK, Germany, Poland, Mexico, Thailand - Smart Pharma Consulting 18

Financial Sustainability • GAVI: “Although self-sufficiency is the ultimate goal, in the nearer term, sustainable financing is the ability of a country to mobilize and efficiently use domestic and supplementary external resources on a reliable basis to achieve target levels of immunization performance. ” 19
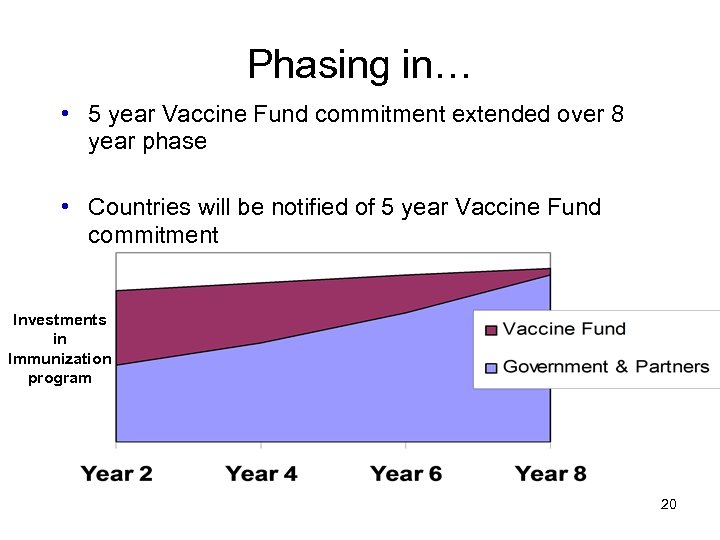
Phasing in… • 5 year Vaccine Fund commitment extended over 8 year phase • Countries will be notified of 5 year Vaccine Fund commitment Investments in Immunization program 20
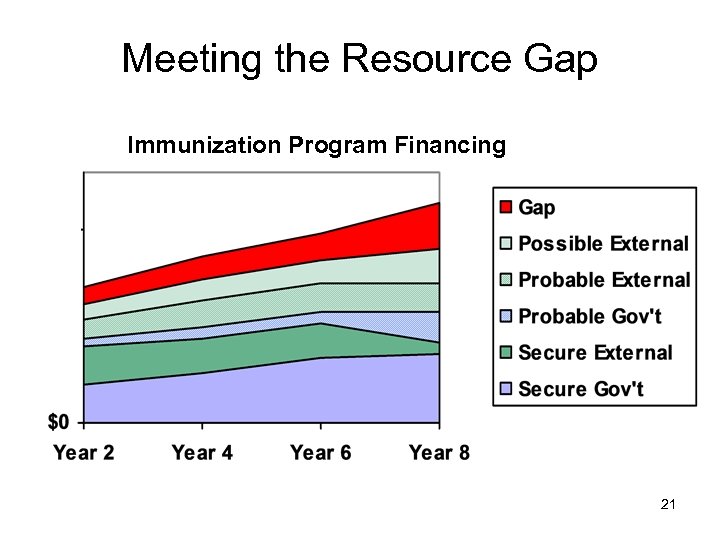
Meeting the Resource Gap Immunization Program Financing 21
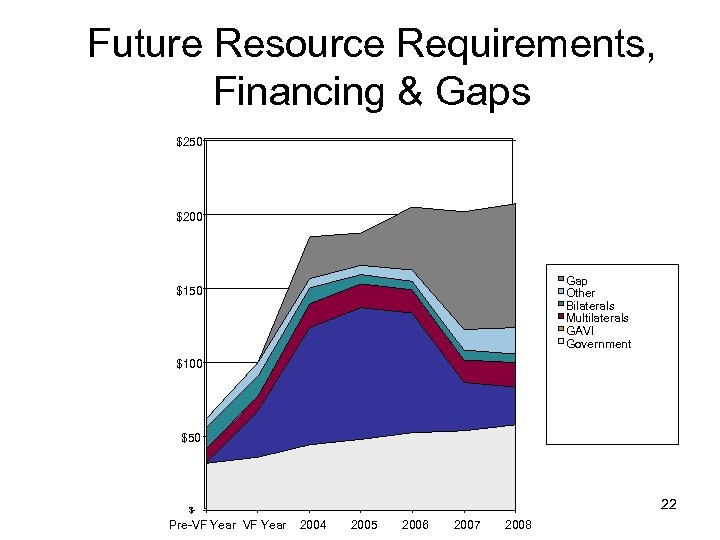
Future Resource Requirements, Financing & Gaps $250 $200 Gap Other Bilaterals Multilaterals GAVI Government $150 $100 $50 22 $- Pre-VF Year 2004 2005 2006 2007 2008

Financial Sustainability ‘Innovative’ Financing Mechanisms – Global Alliance for Vaccines & Immunization • The Vaccine Fund • Advanced Development & Introduction Plans • International Finance Facility for Immunization – Other Funding Mechanisms • PAHO Revolving Fund • Vaccine Independence Initiative • ARIVAS (Appui au Renforcement de l’independence Vaccinal en Afrique Sub-Saharien ) • ‘Advance Market Commitments’/prize funds GAVI, IFFIm, and prize funds $5 bn-$10 bn 20062010 23

1. What is the IFFIm? • An IFF for immunization (IFFIm) has been proposed as a pilot for the IFF mechanism in general – IFF a large-scale US$50 -75 billion per year mechanism to double global aid and help meet the MDGs – On September 9 th 2006 the IFFIm was launched in London with the five donors - UK, France, Italy, Spain, and Sweden: now Norway and Brazil have announced contribution as well; South Africa is considering a contribution – Estimated disbursable of $3. 2 billion before 2015 – Ongoing effort to secure resources from additional donors to reach $4 (now $6) billion resource goal • First bond issuance took place late 2006 24
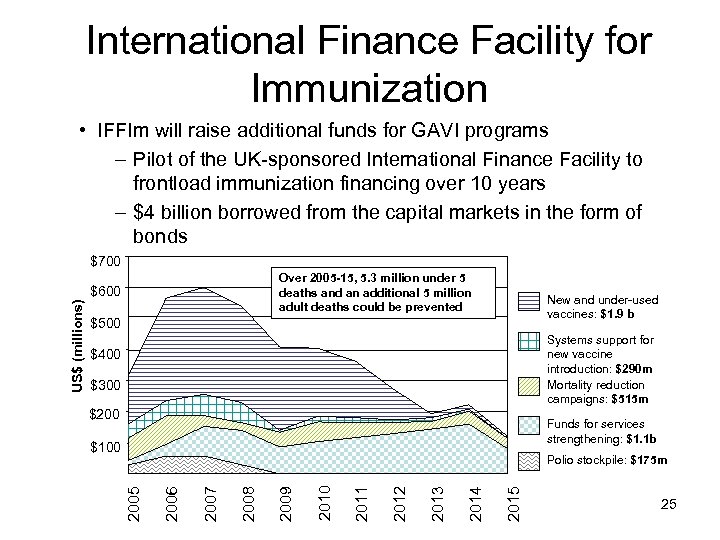
International Finance Facility for Immunization • IFFIm will raise additional funds for GAVI programs – Pilot of the UK-sponsored International Finance Facility to frontload immunization financing over 10 years – $4 billion borrowed from the capital markets in the form of bonds $700 Over 2005 -15, 5. 3 million under 5 deaths and an additional 5 million adult deaths could be prevented New and under-used vaccines: $1. 9 b $500 Systems support for new vaccine introduction: $290 m Mortality reduction campaigns: $515 m $400 $300 $200 Funds for services strengthening: $1. 1 b $100 2015 2014 2013 2012 2011 2010 2009 2008 2007 2006 Polio stockpile: $175 m 2005 US$ (millions) $600 25
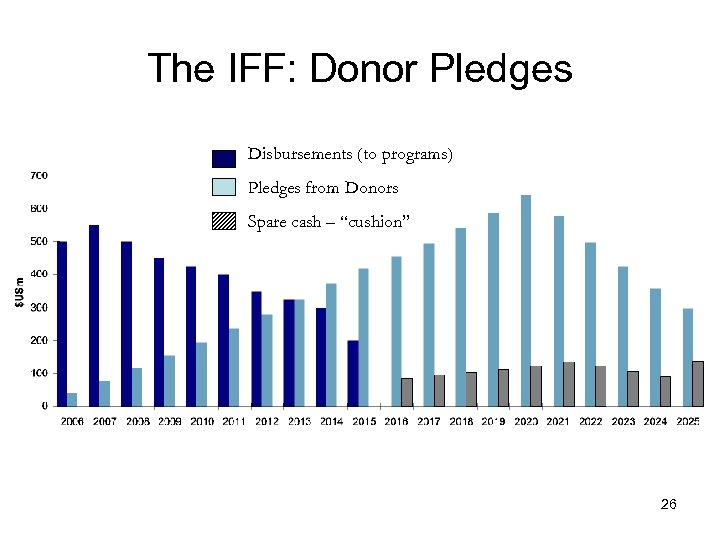
The IFF: Donor Pledges Disbursements (to programs) Pledges from Donors Spare cash – “cushion” 26

Implications of the IFFIm • Influencing the market – Long-term predictable commitments allow longerterm planning for supply strategy – Increased industry capacity and lower vaccine prices • Better planning and sustainability for countries – Commitments can be made to countries over longer -term allowing for better integration within national planning cycles and longer lead time to plan for country financing and eventual sustainability 27

Implications of the IFFIm • Additional financing & donors – Countries not previously contributing to GAVI attracted • Accelerating coverage of immunization with traditional and new and under-used vaccines • But: • Transaction costs have proved much higher than expected (not per se negative, but must be factored in) • It has to be repaid, and will phase out at a later date • How will funding be sustained if still needed? 28
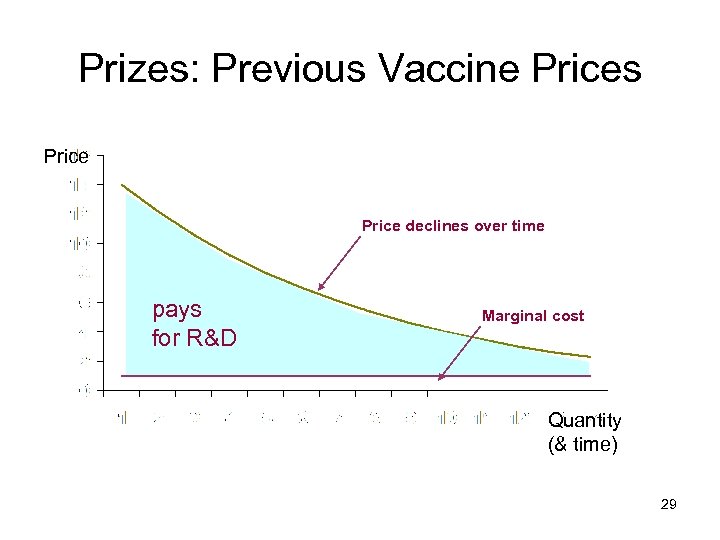
Prizes: Previous Vaccine Prices Price declines over time pays for R&D Marginal cost Quantity (& time) 29
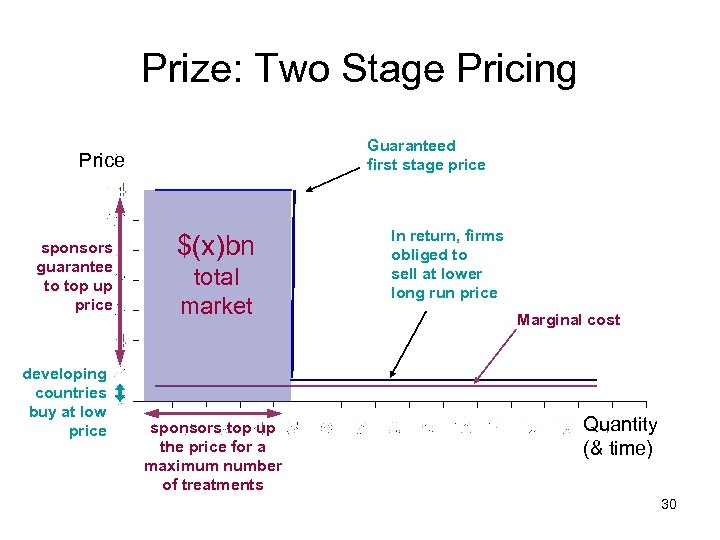
Prize: Two Stage Pricing Guaranteed first stage price Price sponsors guarantee to top up price developing countries buy at low price $(x)bn total market sponsors top up the price for a maximum number of treatments In return, firms obliged to sell at lower long run price Marginal cost Quantity (& time) 30

Some Issues Though • No Simple one-off vaccine solution, – Can’t have a quantity guarantee – Must allow less exhaustive technical standards – Firms must face demand risk? • How to set right? • How to make credible and avoid time inconsistency • Still need to keep pressure on affordability • If a package of measures, how to use a ‘prize’ for one of them? • What about all those ‘on-the-ground’ infrastructure failures? • How to fit in with the typical ‘philosophy’ of PDPs? 31
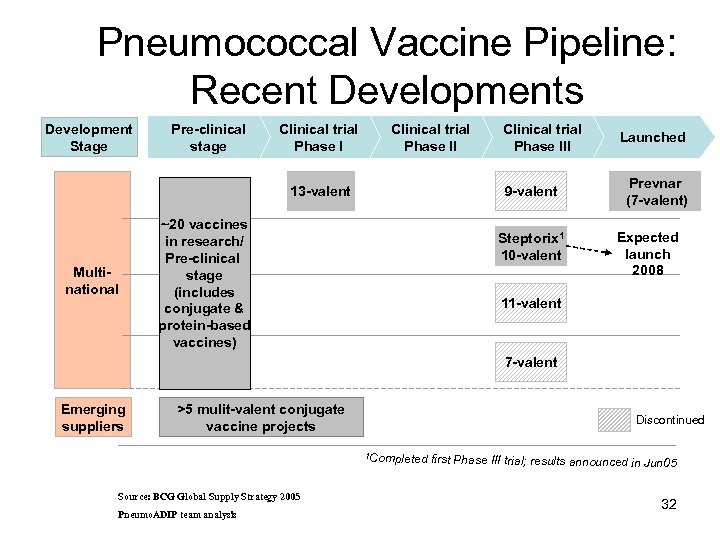
Pneumococcal Vaccine Pipeline: Recent Developments Development Stage Pre-clinical stage Clinical trial Phase II 13 -valent Multinational Clinical trial Phase III 9 -valent ~20 vaccines in research/ Pre-clinical stage (includes conjugate & protein-based vaccines) Steptorix 1 10 -valent Launched Prevnar (7 -valent) Expected launch 2008 11 -valent 7 -valent Emerging suppliers >5 mulit-valent conjugate vaccine projects Discontinued 1 Comp leted first Phase III trial; results announced in Jun 05 Source: BCG Global Supply Strategy 2005 Pneumo. ADIP team analysis 32
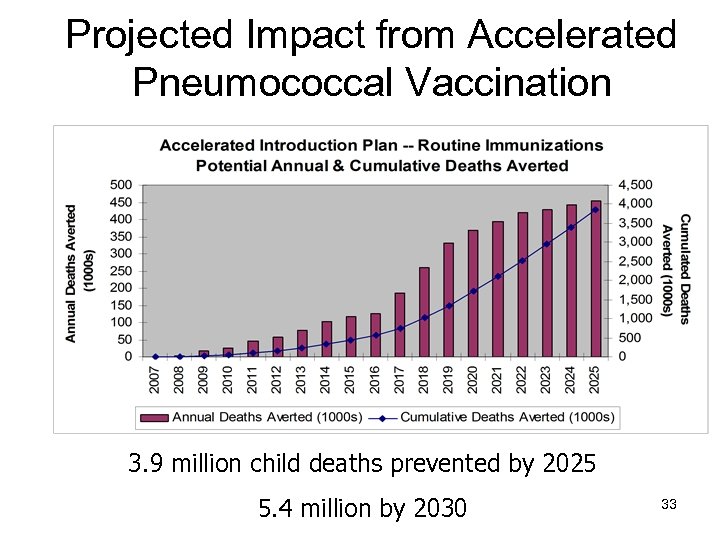
Projected Impact from Accelerated Pneumococcal Vaccination 3. 9 million child deaths prevented by 2025 5. 4 million by 2030 33
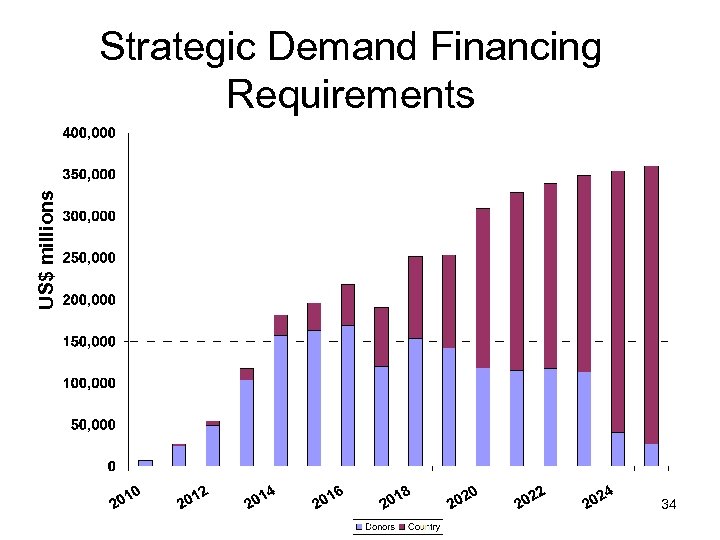
US$ millions Strategic Demand Financing Requirements 34

However… • According to key sponsor files, most resources are gone by 2015 – Leaving 98% of total burden out to 2030 – Follow on vaccines – Capacity risks – Cost of goods – Packaging issues in first round countries – Costs of sustaining first round countries 35

THANK YOU Comments and feedback always welcome: andrew. farlow@sbs. ox. ac. uk 36
40335874605cbe587a720e39f5f07c8d.ppt