e70dbcfbab476f01465d35096b103e53.ppt
- Количество слайдов: 36
 The internet as a means of recruitment, electronic data capture, monitoring, patient education and data management – a regulatory perspective Steve Wilson, Dr. PH (Biostatistics) Deputy Director, Division of Biometrics II, CDER, FDA Session 2: Regulatory Updates and Clinical The 4 th Kitasato University-Harvard School of Public Health Symposium on Advanced and Global Drug Development Techniques: Emerging Trends, Technology Updates, and Novel Paradigms 15: 50 – 18: 50, Tuesday, October 28, 2003 ANA Hotel Tokyo
The internet as a means of recruitment, electronic data capture, monitoring, patient education and data management – a regulatory perspective Steve Wilson, Dr. PH (Biostatistics) Deputy Director, Division of Biometrics II, CDER, FDA Session 2: Regulatory Updates and Clinical The 4 th Kitasato University-Harvard School of Public Health Symposium on Advanced and Global Drug Development Techniques: Emerging Trends, Technology Updates, and Novel Paradigms 15: 50 – 18: 50, Tuesday, October 28, 2003 ANA Hotel Tokyo
 Disclaimer Views expressed in this presentation are those of the speaker and not, necessarily, of the Food and Drug Administration.
Disclaimer Views expressed in this presentation are those of the speaker and not, necessarily, of the Food and Drug Administration.
 Acknowledgements • Frances Cook, CDER/FDA • Rebecca Kush, CDISC • Randy Levin, CDER/FDA • Edward Nevius, CDER/FDA • Robert T. O’Neill, CDER/FDA
Acknowledgements • Frances Cook, CDER/FDA • Rebecca Kush, CDISC • Randy Levin, CDER/FDA • Edward Nevius, CDER/FDA • Robert T. O’Neill, CDER/FDA
 Outline • Internet Applications for Today’s Clinical Trial – – – Recruitment Monitoring Patient Education Electronic Data Capture Data Management • A Regulatory Perspective on Data Quality • Internet Applications for Tomorrow’s Clinical Trial – the “e. Clinical Trial” • The Key Role of Data Standards • Concluding Comments
Outline • Internet Applications for Today’s Clinical Trial – – – Recruitment Monitoring Patient Education Electronic Data Capture Data Management • A Regulatory Perspective on Data Quality • Internet Applications for Tomorrow’s Clinical Trial – the “e. Clinical Trial” • The Key Role of Data Standards • Concluding Comments
 Today’s Clinical Trial • Phase 3, confirmatory study -- large, complex experiment (this is Science!) • Difficult to plan and implement – protocoldriven, yet unpredictable • Expensive (Phase 3, confirmatory clinical trials) • Many people involved – many “players” • May be conducted in a number of locations/countries
Today’s Clinical Trial • Phase 3, confirmatory study -- large, complex experiment (this is Science!) • Difficult to plan and implement – protocoldriven, yet unpredictable • Expensive (Phase 3, confirmatory clinical trials) • Many people involved – many “players” • May be conducted in a number of locations/countries
 Today’s Clinical Trial • Mostly paper-based (90%? )– large paper burden • Must be concerned about regulatory aspects • Based on established “Good Practice” norms • Conservative – reluctance to adopt new technology • Communication is important • Data Quality is important • Did I say Expensive?
Today’s Clinical Trial • Mostly paper-based (90%? )– large paper burden • Must be concerned about regulatory aspects • Based on established “Good Practice” norms • Conservative – reluctance to adopt new technology • Communication is important • Data Quality is important • Did I say Expensive?
 Today’s Clinical Trial • However, there is growing pressure to do it better! • We need to be more efficient! • We need to overcome our dependence on paper! • Unprecedented attention from regulatory parties (ICH, FDA’s Strategic Plan) • More proof – my participation in this meeting
Today’s Clinical Trial • However, there is growing pressure to do it better! • We need to be more efficient! • We need to overcome our dependence on paper! • Unprecedented attention from regulatory parties (ICH, FDA’s Strategic Plan) • More proof – my participation in this meeting
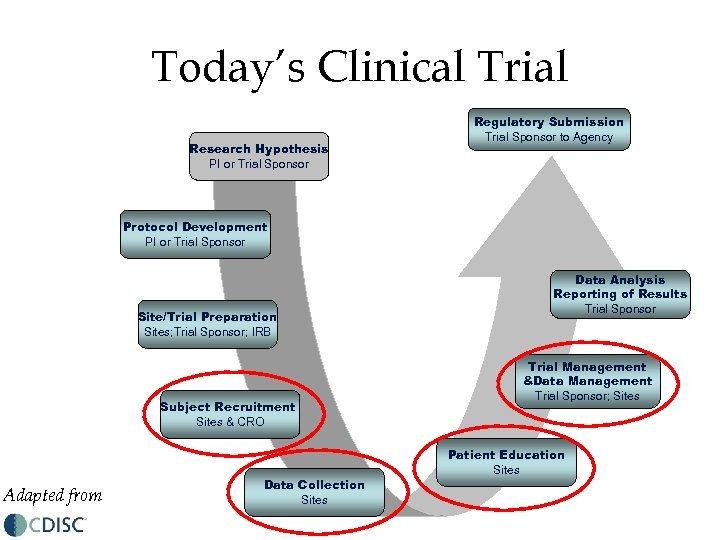 Today’s Clinical Trial Research Hypothesis PI or Trial Sponsor Regulatory Submission Trial Sponsor to Agency Protocol Development PI or Trial Sponsor Site/Trial Preparation Sites; Trial Sponsor; IRB Subject Recruitment Sites & CRO Adapted from Data Collection Sites Data Analysis Reporting of Results Trial Sponsor Trial Management &Data Management Trial Sponsor; Sites Patient Education Sites
Today’s Clinical Trial Research Hypothesis PI or Trial Sponsor Regulatory Submission Trial Sponsor to Agency Protocol Development PI or Trial Sponsor Site/Trial Preparation Sites; Trial Sponsor; IRB Subject Recruitment Sites & CRO Adapted from Data Collection Sites Data Analysis Reporting of Results Trial Sponsor Trial Management &Data Management Trial Sponsor; Sites Patient Education Sites
 Internet Applications for Today’s Clinical Trial Sponsors and vendors have recognized the value of the internet for: • • • Patient Recruitment Monitoring Patient Education Electronic Data Capture Data Management
Internet Applications for Today’s Clinical Trial Sponsors and vendors have recognized the value of the internet for: • • • Patient Recruitment Monitoring Patient Education Electronic Data Capture Data Management
 Why the Internet? • Growing popularity/accessibility • Many uses – Business – Personal • Still a number of concerns – Privacy/security – Data integrity – Bandwidth • Developing new technology -- issues with adoption (generation gap)
Why the Internet? • Growing popularity/accessibility • Many uses – Business – Personal • Still a number of concerns – Privacy/security – Data integrity – Bandwidth • Developing new technology -- issues with adoption (generation gap)
 Patient Recruitment • Competition for participants • Sometimes difficult to find patients -- specific inclusion and exclusion criteria (screening) • Sometimes difficult to enroll • Multi-center, international studies • Evidence requirements (e. g. , heterogeneity) • With it’s ability to reach a large population of potential subjects, the internet is a natural, important tool for recruitment for a number of trials • Large number of websites have been developed
Patient Recruitment • Competition for participants • Sometimes difficult to find patients -- specific inclusion and exclusion criteria (screening) • Sometimes difficult to enroll • Multi-center, international studies • Evidence requirements (e. g. , heterogeneity) • With it’s ability to reach a large population of potential subjects, the internet is a natural, important tool for recruitment for a number of trials • Large number of websites have been developed
 Patient Recruitment & the Internet www. clinicaltrials. gov
Patient Recruitment & the Internet www. clinicaltrials. gov
 Data Monitoring • Quality of the data from clinical trials is the responsibility of the sponsor • US regulation requires that the sponsor monitor clinical trials • No standards – each sponsor determines how they are going to monitor each trial • Process ensures that data are consistent with source data – often many transcriptions, hiddeneffects, etc. • Multi-center trials labor-, cost- and paperintensive (manpower, travel, communication, etc. )
Data Monitoring • Quality of the data from clinical trials is the responsibility of the sponsor • US regulation requires that the sponsor monitor clinical trials • No standards – each sponsor determines how they are going to monitor each trial • Process ensures that data are consistent with source data – often many transcriptions, hiddeneffects, etc. • Multi-center trials labor-, cost- and paperintensive (manpower, travel, communication, etc. )
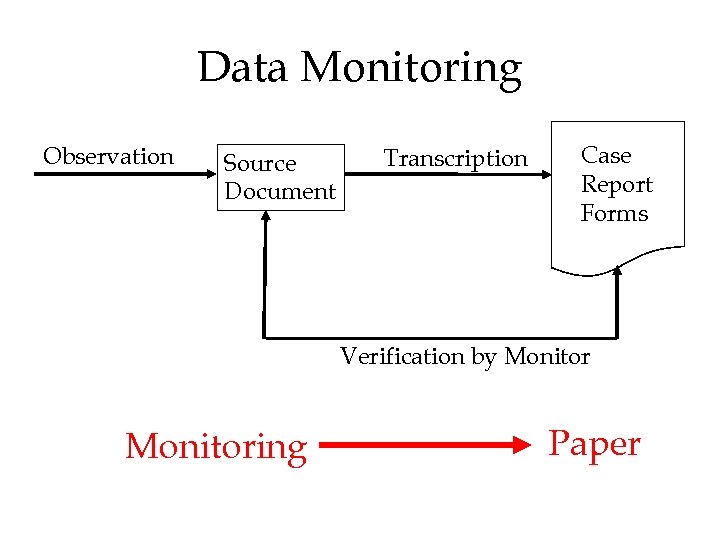 Data Monitoring Observation Source Document Transcription Case Report Forms Verification by Monitoring Paper
Data Monitoring Observation Source Document Transcription Case Report Forms Verification by Monitoring Paper
 Data Monitoring & the Internet • Growing number of vendor and in-house sponsor applications • Great potential for gain in efficiency, but slow to realize and accommodate • Need for better description of process, metrics and “use cases” • Need to work on GDMPs (Good Data Monitoring Practices) in order to develop more consistent technology solutions • There is a potential that standards for collection of “operational data” will enhance solutions
Data Monitoring & the Internet • Growing number of vendor and in-house sponsor applications • Great potential for gain in efficiency, but slow to realize and accommodate • Need for better description of process, metrics and “use cases” • Need to work on GDMPs (Good Data Monitoring Practices) in order to develop more consistent technology solutions • There is a potential that standards for collection of “operational data” will enhance solutions
 Patient Education • Patients are an important • For the experiment to work – participants must be motivated and informed (drop-outs and issues with missing data may affect the interpretability of trial results) – better trial design! • It is important that patients understand follow protocol instructions • Need for communication with investigator • Ethical responsibility – connection to IRB • With the growing dependence on the internet for medical advice, this is another natural for the internet – an open field for innovation in applications to clinical trials
Patient Education • Patients are an important • For the experiment to work – participants must be motivated and informed (drop-outs and issues with missing data may affect the interpretability of trial results) – better trial design! • It is important that patients understand follow protocol instructions • Need for communication with investigator • Ethical responsibility – connection to IRB • With the growing dependence on the internet for medical advice, this is another natural for the internet – an open field for innovation in applications to clinical trials
 Patient Education & the Internet http: //www. nlm. nih. gov/medlineplus/druginformation. html
Patient Education & the Internet http: //www. nlm. nih. gov/medlineplus/druginformation. html
 Electronic Data Capture (EDC) • Long history of systems to capture and transmit data electronically (e. g. , “remote data entry”) • Large number of sponsor/vendor solutions • To be successful – need attention to sites, “e. Source” data, standards, regulatory data quality/privacy conformance, trusted third parties, more technology, etc. • Tremendous potential for efficiency and quality improvements
Electronic Data Capture (EDC) • Long history of systems to capture and transmit data electronically (e. g. , “remote data entry”) • Large number of sponsor/vendor solutions • To be successful – need attention to sites, “e. Source” data, standards, regulatory data quality/privacy conformance, trusted third parties, more technology, etc. • Tremendous potential for efficiency and quality improvements
 e. Source Data • Source data (as described by ICH) captured initially into a permanent record. • Note: “Permanent” implies that any changes made to the electronic data are recorded via a “protected” audit trail.
e. Source Data • Source data (as described by ICH) captured initially into a permanent record. • Note: “Permanent” implies that any changes made to the electronic data are recorded via a “protected” audit trail.
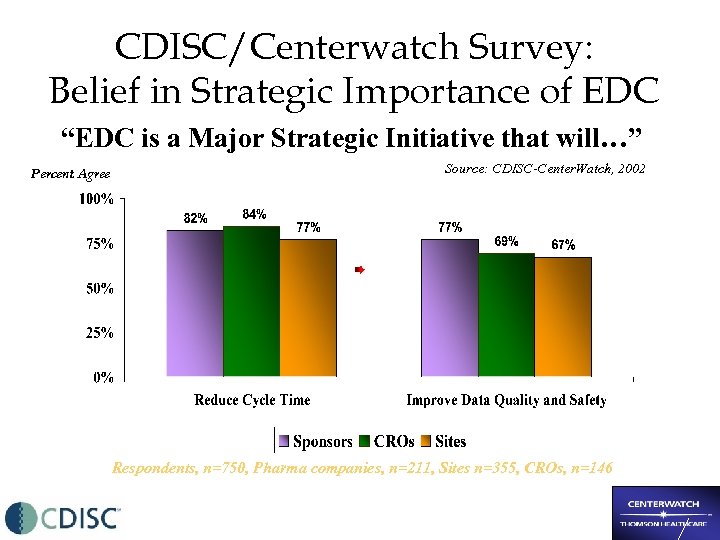 CDISC/Centerwatch Survey: Belief in Strategic Importance of EDC “EDC is a Major Strategic Initiative that will…” Percent Agree Source: CDISC-Center. Watch, 2002 Respondents, n=750, Pharma companies, n=211, Sites n=355, CROs, n=146
CDISC/Centerwatch Survey: Belief in Strategic Importance of EDC “EDC is a Major Strategic Initiative that will…” Percent Agree Source: CDISC-Center. Watch, 2002 Respondents, n=750, Pharma companies, n=211, Sites n=355, CROs, n=146
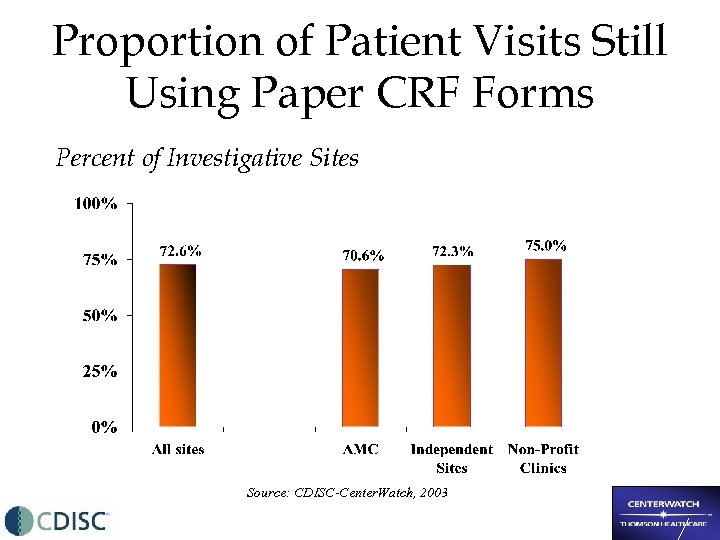 Proportion of Patient Visits Still Using Paper CRF Forms Percent of Investigative Sites Source: CDISC-Center. Watch, 2003
Proportion of Patient Visits Still Using Paper CRF Forms Percent of Investigative Sites Source: CDISC-Center. Watch, 2003
 Tech Providers CROs Bio. Pharma Top 5 Reasons for EDC Adoption Delays 1 2 3 4 5 Regulatory concerns 53. 3% Lack of clear leader ECT/EDC application space 50. 9% Cost or perceived cost 49. 7% Inertia or concern with changing current process 49. 4% Insufficient implementation of common data standards 48. 5% Regulatory concerns 59. 6% Inertia or concern with changing current process 56. 9% Cost or perceived cost 54. 5% Lack of clear leader ECT/EDC application space 50. 9% Insufficient implementation of common data standards 50. 4% Inertia or concern with changing current process Resistance from internal users at sponsors Cost or perceived cost Resistance from management Company is undecided as to importance of EDC 76. 3% 65. 8% 63. 2% 60. 5%
Tech Providers CROs Bio. Pharma Top 5 Reasons for EDC Adoption Delays 1 2 3 4 5 Regulatory concerns 53. 3% Lack of clear leader ECT/EDC application space 50. 9% Cost or perceived cost 49. 7% Inertia or concern with changing current process 49. 4% Insufficient implementation of common data standards 48. 5% Regulatory concerns 59. 6% Inertia or concern with changing current process 56. 9% Cost or perceived cost 54. 5% Lack of clear leader ECT/EDC application space 50. 9% Insufficient implementation of common data standards 50. 4% Inertia or concern with changing current process Resistance from internal users at sponsors Cost or perceived cost Resistance from management Company is undecided as to importance of EDC 76. 3% 65. 8% 63. 2% 60. 5%
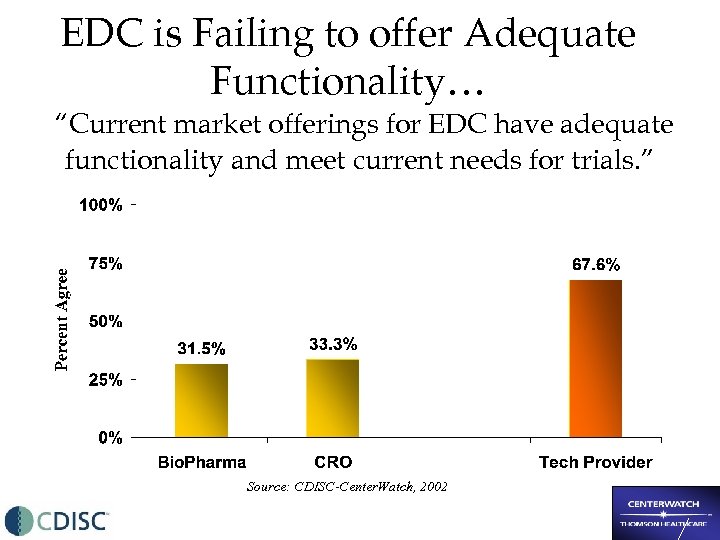 EDC is Failing to offer Adequate Functionality… “Current market offerings for EDC have adequate functionality and meet current needs for trials. ” Source: CDISC-Center. Watch, 2002
EDC is Failing to offer Adequate Functionality… “Current market offerings for EDC have adequate functionality and meet current needs for trials. ” Source: CDISC-Center. Watch, 2002
 Data Management • Large, expensive operation • Includes large number of operations, from case report form (CRF) design to production of reports • Large portion of the cost of a trial. • Software support (e. g. , Oracle Clinical, SAS) • Each sponsor develops own system • Described “Good Practice” – Society for Clinical Data Management (SCDM)
Data Management • Large, expensive operation • Includes large number of operations, from case report form (CRF) design to production of reports • Large portion of the cost of a trial. • Software support (e. g. , Oracle Clinical, SAS) • Each sponsor develops own system • Described “Good Practice” – Society for Clinical Data Management (SCDM)
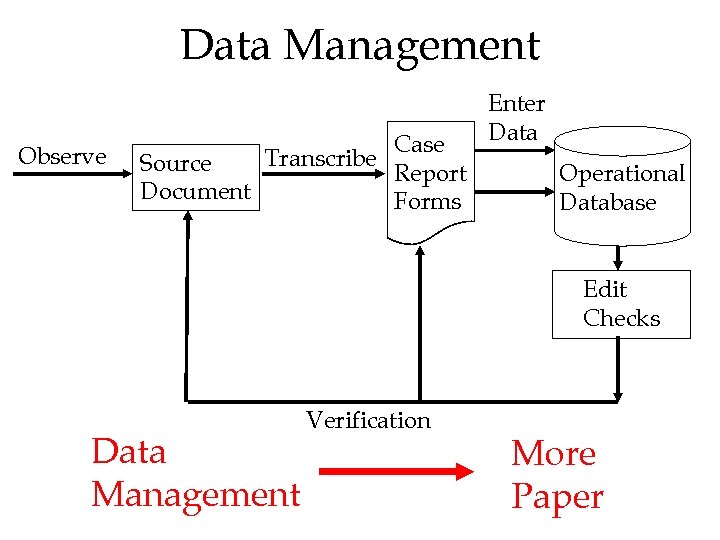 Data Management Observe Case Transcribe Source Report Document Forms Enter Data Operational Database Edit Checks Data Management Verification More Paper
Data Management Observe Case Transcribe Source Report Document Forms Enter Data Operational Database Edit Checks Data Management Verification More Paper
 Data Management & the Internet • Growing number of applications and enhancements to current systems using internet browser access to process • XML offers potential for dramatic improvements • CDISC ODM (Operational Data Model) provides a common path to describing systems • To be successful – attention to sites, “e. Source, ” data standards, regulatory data quality/privacy conformance, trusted third parties, better technology, etc. • Maturing -- large potential for improvement
Data Management & the Internet • Growing number of applications and enhancements to current systems using internet browser access to process • XML offers potential for dramatic improvements • CDISC ODM (Operational Data Model) provides a common path to describing systems • To be successful – attention to sites, “e. Source, ” data standards, regulatory data quality/privacy conformance, trusted third parties, better technology, etc. • Maturing -- large potential for improvement
 A Regulatory Perspective on Data Quality • GCP: Regulations and Guidance (ICH) • 21 CFR 11, 1997 • FDA Guidance on Computerized Systems Used in Clinical Trials • Health Insurance Portability and Accountability Act, HIPAA, 1996
A Regulatory Perspective on Data Quality • GCP: Regulations and Guidance (ICH) • 21 CFR 11, 1997 • FDA Guidance on Computerized Systems Used in Clinical Trials • Health Insurance Portability and Accountability Act, HIPAA, 1996
 A Regulatory Perspective on Data Quality • ALCOA – Attributable – Legible – Contemporaneous – Original – Accurate • We know what we do with paper, need to be comfortable that there are guarantees with electronic (example: corrections to CRF) • Impasse -- Trusted Third Party Solution? ?
A Regulatory Perspective on Data Quality • ALCOA – Attributable – Legible – Contemporaneous – Original – Accurate • We know what we do with paper, need to be comfortable that there are guarantees with electronic (example: corrections to CRF) • Impasse -- Trusted Third Party Solution? ?
 Internet Applications for Tomorrow’s Clinical Trial – The “e. Clinical Trial”
Internet Applications for Tomorrow’s Clinical Trial – The “e. Clinical Trial”
 Internet Applications for Tomorrow’s Clinical Trials – The “e. Clinical Trial” Research Hypothesis PI or Trial Sponsor Regulatory Submission Trial Sponsor to Agency Protocol Development PI or Trial Sponsor Site/Trial Preparation Sites; Trial Sponsor; IRB Subject Recruitment Sites & CRO Adapted from Data Collection Sites Data Analysis Reporting of Results Trial Sponsor Trial Management &Data Management Trial Sponsor; Sites Patient Education Sites
Internet Applications for Tomorrow’s Clinical Trials – The “e. Clinical Trial” Research Hypothesis PI or Trial Sponsor Regulatory Submission Trial Sponsor to Agency Protocol Development PI or Trial Sponsor Site/Trial Preparation Sites; Trial Sponsor; IRB Subject Recruitment Sites & CRO Adapted from Data Collection Sites Data Analysis Reporting of Results Trial Sponsor Trial Management &Data Management Trial Sponsor; Sites Patient Education Sites
 e. Clinical “A clinical trial in which primarily electronic processes are used to collect (acquire), access, exchange and archive data required for conduct, management, analysis and reporting of the trial. “ Ref. : Rebecca Kush, et al. , e. Clinical Trials: Planning and Implementation, Thomson/Centerwatch, Boston, MA, 2003
e. Clinical “A clinical trial in which primarily electronic processes are used to collect (acquire), access, exchange and archive data required for conduct, management, analysis and reporting of the trial. “ Ref. : Rebecca Kush, et al. , e. Clinical Trials: Planning and Implementation, Thomson/Centerwatch, Boston, MA, 2003
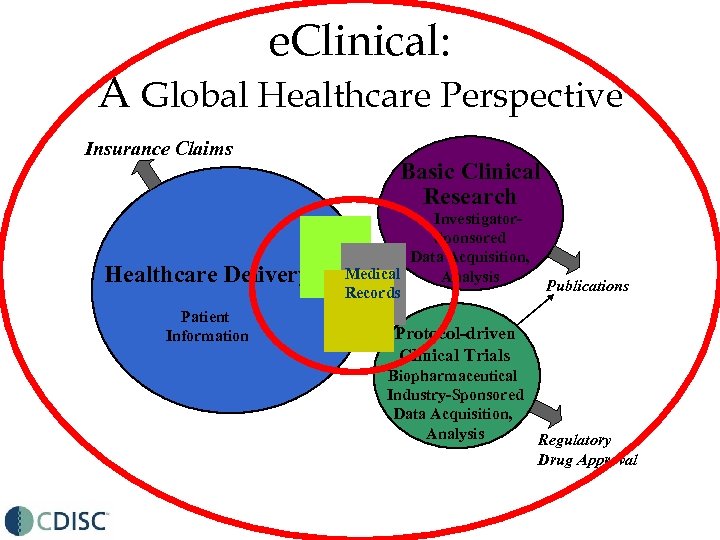 e. Clinical: A Global Healthcare Perspective Insurance Claims Healthcare Delivery Patient Information Basic Clinical Research Investigator. Sponsored Data Acquisition, Medical Analysis Records Publications Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Regulatory Drug Approval
e. Clinical: A Global Healthcare Perspective Insurance Claims Healthcare Delivery Patient Information Basic Clinical Research Investigator. Sponsored Data Acquisition, Medical Analysis Records Publications Protocol-driven Clinical Trials Biopharmaceutical Industry-Sponsored Data Acquisition, Analysis Regulatory Drug Approval
 The Key Role of Data Standards • Increase efficiencies of performing clinical trials • Facilitate ‘business’ processes among investigators, pharma companies, CROs, vendors, laboratories • Streamline data collection at investigator sites • Higher quality data due to consistency of requirements • Allow software development against a common standard • Provide long-term means for electronic data archive • Facilitate FDA review of submissions • Improve links between healthcare delivery and clinical research/trials
The Key Role of Data Standards • Increase efficiencies of performing clinical trials • Facilitate ‘business’ processes among investigators, pharma companies, CROs, vendors, laboratories • Streamline data collection at investigator sites • Higher quality data due to consistency of requirements • Allow software development against a common standard • Provide long-term means for electronic data archive • Facilitate FDA review of submissions • Improve links between healthcare delivery and clinical research/trials
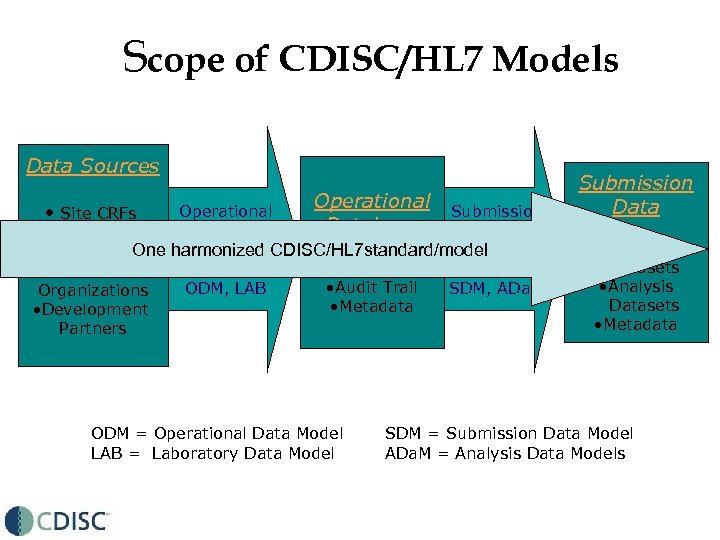 Scope of CDISC/HL 7 Models Data Sources • Site CRFs Operational Submission Database Data • Laboratories Interchange • Contract One harmonized CDISC/HL 7 standard/model • Study Data & Archive: Research • Audit Trail ODM, LAB SDM, ADa. M Organizations • Metadata • Development Partners ODM = Operational Data Model LAB = Laboratory Data Model Submission Data • CRT/Domain Datasets • Analysis Datasets • Metadata SDM = Submission Data Model ADa. M = Analysis Data Models
Scope of CDISC/HL 7 Models Data Sources • Site CRFs Operational Submission Database Data • Laboratories Interchange • Contract One harmonized CDISC/HL 7 standard/model • Study Data & Archive: Research • Audit Trail ODM, LAB SDM, ADa. M Organizations • Metadata • Development Partners ODM = Operational Data Model LAB = Laboratory Data Model Submission Data • CRT/Domain Datasets • Analysis Datasets • Metadata SDM = Submission Data Model ADa. M = Analysis Data Models
 Concluding Comments • As illustrated by the development of recruitment, electronic data capture, monitoring, patient education and data management applications, the internet is becoming an important feature of today’s clinical trial paradigm • For the internet’s full potential of efficiency and quality improvement to be realized, there is a need for: – a more comprehensive appreciation of clinical trial activities (i. e. , the “e. Clinical” trial with a focus on the global healthcare enterprise) – a commitment to data quality/regulatory requirements (and the potential role of TTPs? ) – universal attention (devotion? ) to the development of standards (ICH/CDISC/HL 7)
Concluding Comments • As illustrated by the development of recruitment, electronic data capture, monitoring, patient education and data management applications, the internet is becoming an important feature of today’s clinical trial paradigm • For the internet’s full potential of efficiency and quality improvement to be realized, there is a need for: – a more comprehensive appreciation of clinical trial activities (i. e. , the “e. Clinical” trial with a focus on the global healthcare enterprise) – a commitment to data quality/regulatory requirements (and the potential role of TTPs? ) – universal attention (devotion? ) to the development of standards (ICH/CDISC/HL 7)
 THANK YOU Any Questions? wilsons@cder. fda. gov 301 -827 -5583
THANK YOU Any Questions? wilsons@cder. fda. gov 301 -827 -5583


