608da3cd9006f8f47f432a5deaa04c02.ppt
- Количество слайдов: 23
 THE FOLLOW-UP STUDY International study of caesarean section surgical techniques
THE FOLLOW-UP STUDY International study of caesarean section surgical techniques
 The CORONIS Trial Follow-up Study is funded by the UK Medical Research Council
The CORONIS Trial Follow-up Study is funded by the UK Medical Research Council
 Study design – CORONIS is a multicentre, fractional factorial randomised controlled trial – The collaborating institutions are centres with experience in conducting trials – These centres also have experience in detailed follow-up of large numbers of women
Study design – CORONIS is a multicentre, fractional factorial randomised controlled trial – The collaborating institutions are centres with experience in conducting trials – These centres also have experience in detailed follow-up of large numbers of women
 Interventions Each woman is allocated to a combination of three of the five interventions 1. 2. 3. 4. 5. Blunt versus sharp abdominal entry Exteriorisation of the uterus for repair versus intraabdominal repair Single versus double layer closure of the uterus Closure versus non-closure of the peritoneum (pelvic and parietal) Chromic catgut versus Polyglactin-910 for uterine repair
Interventions Each woman is allocated to a combination of three of the five interventions 1. 2. 3. 4. 5. Blunt versus sharp abdominal entry Exteriorisation of the uterus for repair versus intraabdominal repair Single versus double layer closure of the uterus Closure versus non-closure of the peritoneum (pelvic and parietal) Chromic catgut versus Polyglactin-910 for uterine repair
 Primary outcome Composite outcome of: Death or maternal infectious morbidity i. e. one or more of the following: antibiotic use for maternal febrile morbidity during postnatal hospital stay, antibiotic use for endometritis, wound infection or peritonitis or further operative procedures or blood transfusion (>I unit of whole blood/packed cells)
Primary outcome Composite outcome of: Death or maternal infectious morbidity i. e. one or more of the following: antibiotic use for maternal febrile morbidity during postnatal hospital stay, antibiotic use for endometritis, wound infection or peritonitis or further operative procedures or blood transfusion (>I unit of whole blood/packed cells)
 Data collection Data collected: • At trial entry • Immediately following delivery • Immediately following surgery • During the postpartum stay in hospital • At 6 weeks after discharge from hospital • 3 or 6 monthly thereafter
Data collection Data collected: • At trial entry • Immediately following delivery • Immediately following surgery • During the postpartum stay in hospital • At 6 weeks after discharge from hospital • 3 or 6 monthly thereafter
 Data received in Oxford Women recruited worldwide 15, 936 Trial Entries logged at RTOs 15, 894 (99. 7%) Complete data collection booklets 15, 894 (99. 7%) Complete 6 week forms 15, 482 (97. 1%)
Data received in Oxford Women recruited worldwide 15, 936 Trial Entries logged at RTOs 15, 894 (99. 7%) Complete data collection booklets 15, 894 (99. 7%) Complete 6 week forms 15, 482 (97. 1%)
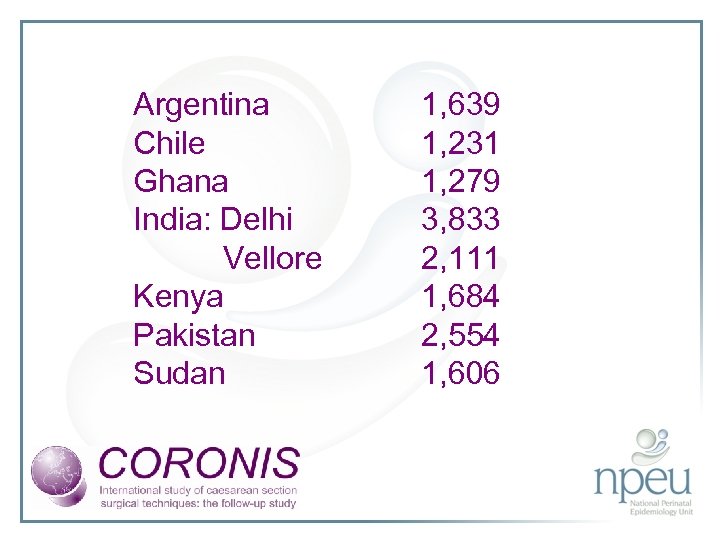 Argentina Chile Ghana India: Delhi Vellore Kenya Pakistan Sudan 1, 639 1, 231 1, 279 3, 833 2, 111 1, 684 2, 554 1, 606
Argentina Chile Ghana India: Delhi Vellore Kenya Pakistan Sudan 1, 639 1, 231 1, 279 3, 833 2, 111 1, 684 2, 554 1, 606
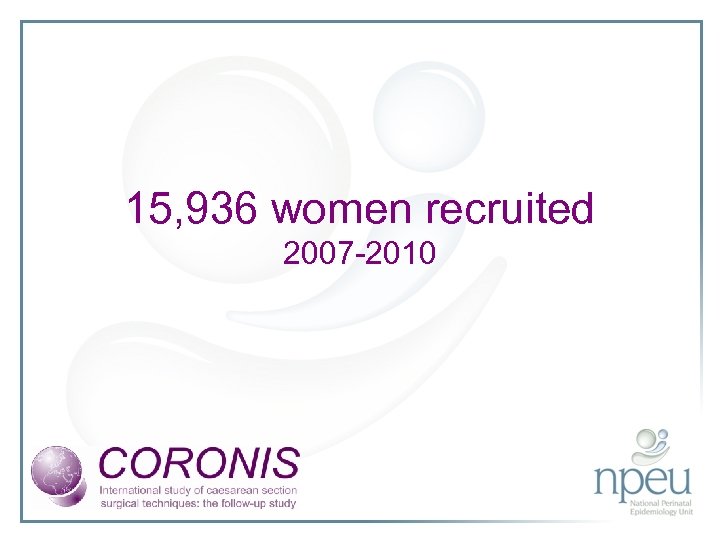 15, 936 women recruited 2007 -2010
15, 936 women recruited 2007 -2010
 Participating countries • Argentina • Chile • Ghana • India: two regions; Delhi and Vellore • Kenya • Pakistan • Sudan • 19 participating hospitals
Participating countries • Argentina • Chile • Ghana • India: two regions; Delhi and Vellore • Kenya • Pakistan • Sudan • 19 participating hospitals
 Investigator Group Chief Investigator Principal Investigators Argentina Chile Ghana India: Delhi India: Vellore Kenya Pakistan Sudan Professor Peter Brocklehurst Dr Edgardo Abalos Dr Enrique Oyarzun Dr Victor Addo Dr Jai Sharma Dr Jiji Mathews Professor James Oyieke Dr Shabeen Mazood Professor Mohamed El. Shiekh
Investigator Group Chief Investigator Principal Investigators Argentina Chile Ghana India: Delhi India: Vellore Kenya Pakistan Sudan Professor Peter Brocklehurst Dr Edgardo Abalos Dr Enrique Oyarzun Dr Victor Addo Dr Jai Sharma Dr Jiji Mathews Professor James Oyieke Dr Shabeen Mazood Professor Mohamed El. Shiekh
 THE FOLLOW-UP STUDY at least three years after having a caesarean section in the CORONIS Trial
THE FOLLOW-UP STUDY at least three years after having a caesarean section in the CORONIS Trial
 Two studies of long-term follow-up 1. Single vs double layer uterine closure – 145 women out of 906 randomised – followed up at time of next pregnancy – no difference found between the groups (not surprisingly) Chapman SJ, Owen J, Hauth JC. One versus two-layer closure of a low transverse cesarean: the next pregnancy. Obstet Gynecol 1997; 89: 16 -18. 2. Non-closure vs closure of peritoneum – 144 women out of 280 randomised – no differences found between the groups Bahmanyar E, Boulvain M, Irion O. Non-closure of the peritoneum during cesarean section: long -term follow-up of a randomized controlled trial. Am J Obstet Gynecol 2001; 185: S 125.
Two studies of long-term follow-up 1. Single vs double layer uterine closure – 145 women out of 906 randomised – followed up at time of next pregnancy – no difference found between the groups (not surprisingly) Chapman SJ, Owen J, Hauth JC. One versus two-layer closure of a low transverse cesarean: the next pregnancy. Obstet Gynecol 1997; 89: 16 -18. 2. Non-closure vs closure of peritoneum – 144 women out of 280 randomised – no differences found between the groups Bahmanyar E, Boulvain M, Irion O. Non-closure of the peritoneum during cesarean section: long -term follow-up of a randomized controlled trial. Am J Obstet Gynecol 2001; 185: S 125.
 Why are we doing the Follow-up Study? • Lack of evidence of long-term outcomes for women and their families • Unique opportunity to evaluate women following a caesarean section • Biggest cohort • Reassurance for women • Inform clinicians and policy makers
Why are we doing the Follow-up Study? • Lack of evidence of long-term outcomes for women and their families • Unique opportunity to evaluate women following a caesarean section • Biggest cohort • Reassurance for women • Inform clinicians and policy makers
 What are we looking for? Undesirable outcomes for the woman and/or her child • Problems with fertility • Problems with obstetric health • Problems in subsequent pregnancies • Child’s health
What are we looking for? Undesirable outcomes for the woman and/or her child • Problems with fertility • Problems with obstetric health • Problems in subsequent pregnancies • Child’s health
 How will the follow-up be done? • The follow-up study will be co-ordinated by the Regional Trial Office team • The team will organise appointments with all women recruited to the CORONIS Trial • The follow-up interview will take place at least three years after the woman joined the trial
How will the follow-up be done? • The follow-up study will be co-ordinated by the Regional Trial Office team • The team will organise appointments with all women recruited to the CORONIS Trial • The follow-up interview will take place at least three years after the woman joined the trial
 The Study Team Each Regional Trial Office will have a: • Study Co-ordinator • Data Manager • Follow-up Assistant • Data Assistant • Dedicated assessment doctor(s)
The Study Team Each Regional Trial Office will have a: • Study Co-ordinator • Data Manager • Follow-up Assistant • Data Assistant • Dedicated assessment doctor(s)
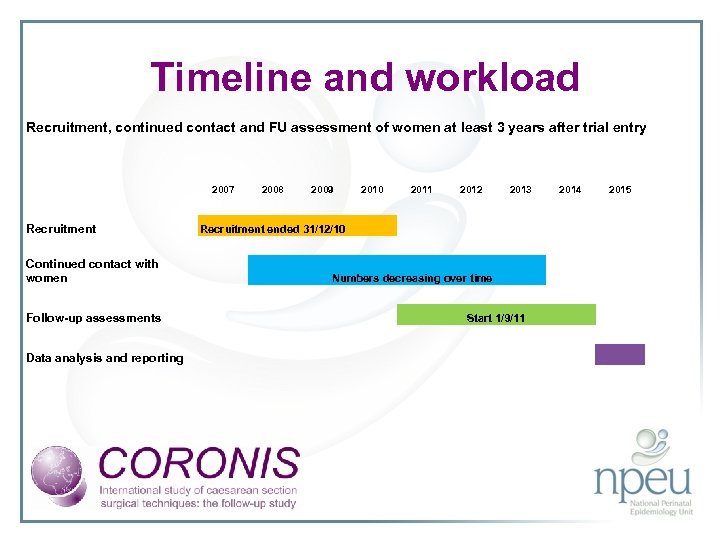 Timeline and workload Recruitment, continued contact and FU assessment of women at least 3 years after trial entry 2007 Recruitment Continued contact with women Follow-up assessments 2008 2009 2010 2011 2012 2013 2014 2015 Recruitment ended 31/12/10 Numbers decreasing over time Start 1/9/11 Data analysis and reporting
Timeline and workload Recruitment, continued contact and FU assessment of women at least 3 years after trial entry 2007 Recruitment Continued contact with women Follow-up assessments 2008 2009 2010 2011 2012 2013 2014 2015 Recruitment ended 31/12/10 Numbers decreasing over time Start 1/9/11 Data analysis and reporting
 Follow-up health assessments • Face-to-face interview with a dedicated assessment doctor • Doctor will take a medical history, guided by the data required to complete the Health Assessment Questionnaire
Follow-up health assessments • Face-to-face interview with a dedicated assessment doctor • Doctor will take a medical history, guided by the data required to complete the Health Assessment Questionnaire

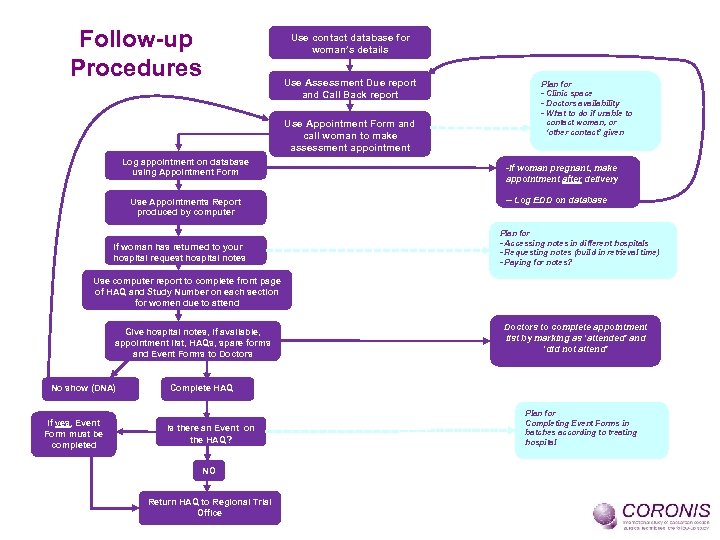 Follow-up Procedures Use contact database for woman’s details Use Assessment Due report and Call Back report Use Appointment Form and call woman to make assessment appointment Log appointment on database using Appointment Form Use Appointments Report produced by computer If woman has returned to your hospital request hospital notes Plan for - Clinic space - Doctors availability - What to do if unable to contact woman, or ‘other contact’ given -If woman pregnant, make appointment after delivery -- Log EDD on database Plan for - Accessing notes in different hospitals - Requesting notes (build in retrieval time) - Paying for notes? Use computer report to complete front page of HAQ and Study Number on each section for women due to attend Give hospital notes, if available, appointment list, HAQs, spare forms and Event Forms to Doctors No show (DNA) If yes, Event Form must be completed Doctors to complete appointment list by marking as ‘attended’ and ‘did not attend’ Complete HAQ Is there an Event on the HAQ? NO Return HAQ to Regional Trial Office Plan for Completing Event Forms in batches according to treating hospital
Follow-up Procedures Use contact database for woman’s details Use Assessment Due report and Call Back report Use Appointment Form and call woman to make assessment appointment Log appointment on database using Appointment Form Use Appointments Report produced by computer If woman has returned to your hospital request hospital notes Plan for - Clinic space - Doctors availability - What to do if unable to contact woman, or ‘other contact’ given -If woman pregnant, make appointment after delivery -- Log EDD on database Plan for - Accessing notes in different hospitals - Requesting notes (build in retrieval time) - Paying for notes? Use computer report to complete front page of HAQ and Study Number on each section for women due to attend Give hospital notes, if available, appointment list, HAQs, spare forms and Event Forms to Doctors No show (DNA) If yes, Event Form must be completed Doctors to complete appointment list by marking as ‘attended’ and ‘did not attend’ Complete HAQ Is there an Event on the HAQ? NO Return HAQ to Regional Trial Office Plan for Completing Event Forms in batches according to treating hospital
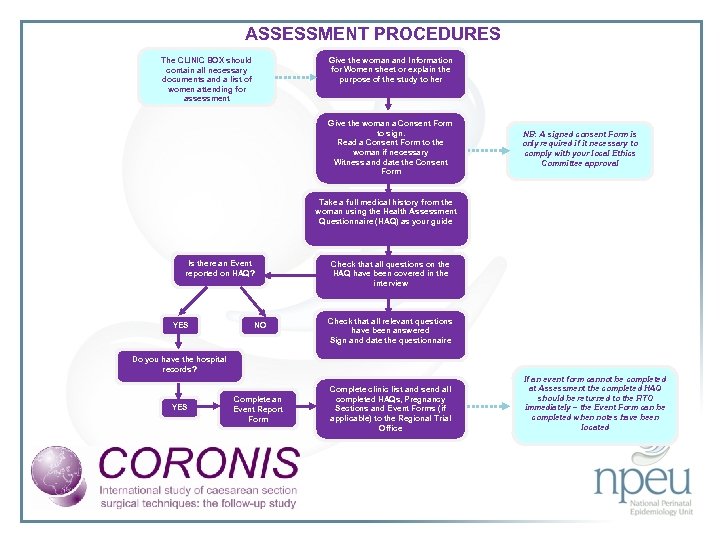 ASSESSMENT PROCEDURES The CLINIC BOX should contain all necessary documents and a list of women attending for assessment Give the woman and Information for Women sheet or explain the purpose of the study to her Give the woman a Consent Form to sign. Read a Consent Form to the woman if necessary Witness and date the Consent Form NB: A signed consent Form is only required if it necessary to comply with your local Ethics Committee approval Take a full medical history from the woman using the Health Assessment Questionnaire (HAQ) as your guide Is there an Event reported on HAQ? YES NO Check that all questions on the HAQ have been covered in the interview Check that all relevant questions have been answered Sign and date the questionnaire Do you have the hospital records? YES Complete an Event Report Form Complete clinic list and send all completed HAQs, Pregnancy Sections and Event Forms (if applicable) to the Regional Trial Office If an event form cannot be completed at Assessment the completed HAQ should be returned to the RTO immediately – the Event Form can be completed when notes have been located
ASSESSMENT PROCEDURES The CLINIC BOX should contain all necessary documents and a list of women attending for assessment Give the woman and Information for Women sheet or explain the purpose of the study to her Give the woman a Consent Form to sign. Read a Consent Form to the woman if necessary Witness and date the Consent Form NB: A signed consent Form is only required if it necessary to comply with your local Ethics Committee approval Take a full medical history from the woman using the Health Assessment Questionnaire (HAQ) as your guide Is there an Event reported on HAQ? YES NO Check that all questions on the HAQ have been covered in the interview Check that all relevant questions have been answered Sign and date the questionnaire Do you have the hospital records? YES Complete an Event Report Form Complete clinic list and send all completed HAQs, Pregnancy Sections and Event Forms (if applicable) to the Regional Trial Office If an event form cannot be completed at Assessment the completed HAQ should be returned to the RTO immediately – the Event Form can be completed when notes have been located
 Shan Rich Patsy Spark Barbara Farrell Study Co-ordinator IT Co-ordinator Trial Director Thank you
Shan Rich Patsy Spark Barbara Farrell Study Co-ordinator IT Co-ordinator Trial Director Thank you


