7889e828edd7c8080381ae2ff62e6b61.ppt
- Количество слайдов: 57
 The First Law of Thermodynamics Meeting 6 Section 4 -1
The First Law of Thermodynamics Meeting 6 Section 4 -1
 So far we’ve studied two forms of energy transfer Work Energy (W) *Equivalent to raising a weight Heat (Thermal) Energy (Q) *Caused by a temperature difference
So far we’ve studied two forms of energy transfer Work Energy (W) *Equivalent to raising a weight Heat (Thermal) Energy (Q) *Caused by a temperature difference
 A note about work and heat: and Both Q and W are path dependent!
A note about work and heat: and Both Q and W are path dependent!
 • Work and Heat are forms of energy transfers that happens at the boundary of a system. • As Work and Heat cross the boundary, the system Energy changes. • Work and Heat are not stored on the system but the Energy yes.
• Work and Heat are forms of energy transfers that happens at the boundary of a system. • As Work and Heat cross the boundary, the system Energy changes. • Work and Heat are not stored on the system but the Energy yes.
 The Business of the First Law • Energy is not destroyed but it is conserved. • In fact during a thermodynamic process it is transformed in one type to another. • The first law expresses a energy balance of the system. • The energy fluxes in a system (Work and Heat) is equal to the change in the system Energy.
The Business of the First Law • Energy is not destroyed but it is conserved. • In fact during a thermodynamic process it is transformed in one type to another. • The first law expresses a energy balance of the system. • The energy fluxes in a system (Work and Heat) is equal to the change in the system Energy.
 The system energy forms • Prior to state the First Law, is necessary to define the system energy forms.
The system energy forms • Prior to state the First Law, is necessary to define the system energy forms.
 System energy consists of three components: • U is internal energy, • KE is kinetic energy, and • PE is potential energy. NOTE: All values are changes (deltas)
System energy consists of three components: • U is internal energy, • KE is kinetic energy, and • PE is potential energy. NOTE: All values are changes (deltas)
 Internal energy…. . Internal energy is the energy a molecule possesses, mostly as a result of: All these are forms of kinetic energy. We will neglect other forms of molecular energy which exist on the atomic level.
Internal energy…. . Internal energy is the energy a molecule possesses, mostly as a result of: All these are forms of kinetic energy. We will neglect other forms of molecular energy which exist on the atomic level.
 Translation • Kinetic energy is possessed by a molecule as it moves through space. It transfers this energy to other systems by means of collisions in which its linear momentum changes. Collisions with such things as thermometers and thermocouples are the basis for temperature measurement. • It is a characteristic of both polyatomic molecules and atoms.
Translation • Kinetic energy is possessed by a molecule as it moves through space. It transfers this energy to other systems by means of collisions in which its linear momentum changes. Collisions with such things as thermometers and thermocouples are the basis for temperature measurement. • It is a characteristic of both polyatomic molecules and atoms.
 Vibration • Molecules (not atoms) also vibrate along their intermolecular bonds. The molecule has vibrational (kinetic) energy in this mode.
Vibration • Molecules (not atoms) also vibrate along their intermolecular bonds. The molecule has vibrational (kinetic) energy in this mode.
 Rotation • Molecules (and atoms) can also rotate and they possess kinetic energy in this rotational mode. They have angular momentum which can be changed to add or remove energy.
Rotation • Molecules (and atoms) can also rotate and they possess kinetic energy in this rotational mode. They have angular momentum which can be changed to add or remove energy.
 We will not worry about the microscopic details of internal energy Internal energy is a property of the system. Often it shows up as a change in temperature or pressure of the system … but it can also show up as a change in composition if it’s a mixture.
We will not worry about the microscopic details of internal energy Internal energy is a property of the system. Often it shows up as a change in temperature or pressure of the system … but it can also show up as a change in composition if it’s a mixture.
 The kinetic energy is given by:
The kinetic energy is given by:
 The energy change in accelerating a mass of 10 kg from Vi= 0 to Vf = 10 m/s is?
The energy change in accelerating a mass of 10 kg from Vi= 0 to Vf = 10 m/s is?
 Gravity is another force acting on our system. It shows up in the potential energy change. Work can be done by a change in elevation of the system
Gravity is another force acting on our system. It shows up in the potential energy change. Work can be done by a change in elevation of the system
 NOTE THE CONVERSION TO GET FROM m 2/s 2 to k. J/kg REMEMBER IT! YOU WILL NEED IT.
NOTE THE CONVERSION TO GET FROM m 2/s 2 to k. J/kg REMEMBER IT! YOU WILL NEED IT.
 TEAMPLAY Let’s say we have a 10 kg mass that we drop 100 m. We also have a device that will convert all the potential energy into kinetic energy of an object. If the object’s mass is 1 kg and it is initially at rest, what would be it’s final velocity from absorbing the potential from a 100 m drop? Assume the object travels horizontally.
TEAMPLAY Let’s say we have a 10 kg mass that we drop 100 m. We also have a device that will convert all the potential energy into kinetic energy of an object. If the object’s mass is 1 kg and it is initially at rest, what would be it’s final velocity from absorbing the potential from a 100 m drop? Assume the object travels horizontally.
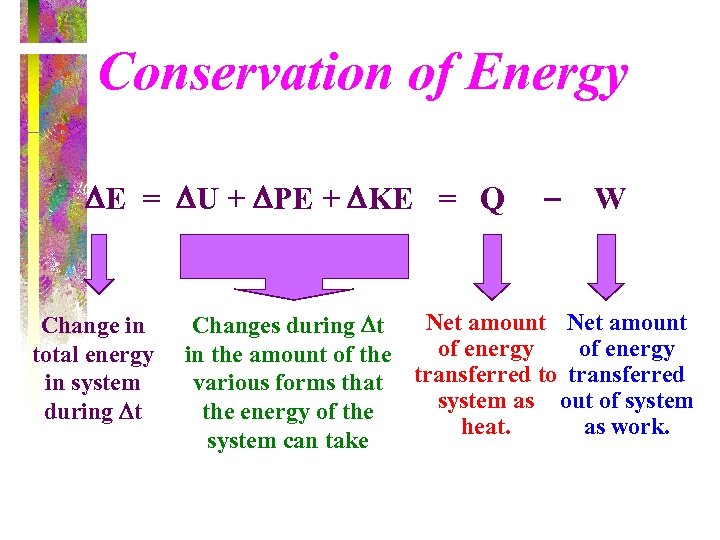 Conservation of Energy E = U + PE + KE = Q Change in total energy in system during t W Net amount Changes during t of energy in the amount of the various forms that transferred to transferred system as out of system the energy of the heat. as work. system can take
Conservation of Energy E = U + PE + KE = Q Change in total energy in system during t W Net amount Changes during t of energy in the amount of the various forms that transferred to transferred system as out of system the energy of the heat. as work. system can take
 Some comments about this statement of the first law Q – W = ΔE • All terms on the left hand side are forms of energy that cross the boundary of the system • Q in is positive, W out is positive • Right hand side is a change in system energy • Algebraic form of first law
Some comments about this statement of the first law Q – W = ΔE • All terms on the left hand side are forms of energy that cross the boundary of the system • Q in is positive, W out is positive • Right hand side is a change in system energy • Algebraic form of first law
 The right hand side of the energy equation consists of three terms: ΔE = ΔU + ΔKE + ΔPE • ΔKE - Motion of the system as a whole with respect to some fixed reference frame. • ΔPE - Position change of the system as a whole in the earth’s gravity field. • ΔU - Internal energy of the molecule-translation, rotation, vibration, [and energy stored in electronic orbital states, nuclear spin, and others].
The right hand side of the energy equation consists of three terms: ΔE = ΔU + ΔKE + ΔPE • ΔKE - Motion of the system as a whole with respect to some fixed reference frame. • ΔPE - Position change of the system as a whole in the earth’s gravity field. • ΔU - Internal energy of the molecule-translation, rotation, vibration, [and energy stored in electronic orbital states, nuclear spin, and others].
 We previously had conservation of energy • E = U + PE + KE • We can change the total energy E of a system by • Changing the internal energy, perhaps best exemplified by heating. • Changing the PE by raising or lowering. • Changing the KE by accelerating or decelerating.
We previously had conservation of energy • E = U + PE + KE • We can change the total energy E of a system by • Changing the internal energy, perhaps best exemplified by heating. • Changing the PE by raising or lowering. • Changing the KE by accelerating or decelerating.
 Conservation of Energy for Stationary System • Stationary means not moving so PE and KE are zero and the first law becomes
Conservation of Energy for Stationary System • Stationary means not moving so PE and KE are zero and the first law becomes
 First Law Forms for Stationary Systems • Differential Form: • Rate Form: • Integrated Form:
First Law Forms for Stationary Systems • Differential Form: • Rate Form: • Integrated Form:
 We can also write the first law in differential terms d. E = Q – W, and d. U + d. PE + d. KE = Change in the amount of energy of the system during some time interval. Q – Differential amount of energy transferred in (+) or out (-) by heat transfer. W Differential amount of energy transferred out (+) or in (-) by work interaction.
We can also write the first law in differential terms d. E = Q – W, and d. U + d. PE + d. KE = Change in the amount of energy of the system during some time interval. Q – Differential amount of energy transferred in (+) or out (-) by heat transfer. W Differential amount of energy transferred out (+) or in (-) by work interaction.
 If we are analyzing a transient process, we’ll need the rate form of the first law Where:
If we are analyzing a transient process, we’ll need the rate form of the first law Where:
 Rate form will allow us to calculate: • • Changes in temperature with time Changes in pressure with time Changes in speed with time Changes in altitude with time
Rate form will allow us to calculate: • • Changes in temperature with time Changes in pressure with time Changes in speed with time Changes in altitude with time
 Hints to set up a problem 1. Define the system carefully indicating clearly its boundaries. 2. Enroll all the simplifying hypothesis to the case. 3. Draw the heat and work fluxes at the boundaries including their signals. 4. Sketch a process representation on a thermodynamic diagram Pv or Tv.
Hints to set up a problem 1. Define the system carefully indicating clearly its boundaries. 2. Enroll all the simplifying hypothesis to the case. 3. Draw the heat and work fluxes at the boundaries including their signals. 4. Sketch a process representation on a thermodynamic diagram Pv or Tv.
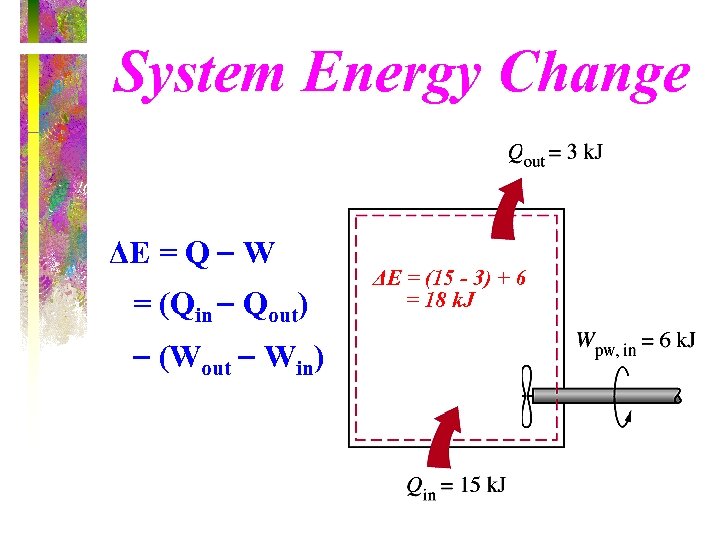 System Energy Change ΔE = Q W = (Qin Qout) (Wout Win) ΔE = (15 - 3) + 6 = 18 k. J
System Energy Change ΔE = Q W = (Qin Qout) (Wout Win) ΔE = (15 - 3) + 6 = 18 k. J
 Example 4 -1 0. 01 kg of air is compressed in a piston-cylinder. Find the rate of temperature rise at an instant of time when T = 400 K. Work is being done at a rate of 8. 165 KW and Heat is being removed at a rate of 1. 0 KW. Solution on the black board
Example 4 -1 0. 01 kg of air is compressed in a piston-cylinder. Find the rate of temperature rise at an instant of time when T = 400 K. Work is being done at a rate of 8. 165 KW and Heat is being removed at a rate of 1. 0 KW. Solution on the black board
 Example 4 -2 Isothermal Process An ideal gas is compressed reversibly and isothermally from a volume of 0. 01 m 3 and a pressure of 0. 1 MPa to a pressure of 1 MPa. How much heat is transferred during this process? Solution on the black board
Example 4 -2 Isothermal Process An ideal gas is compressed reversibly and isothermally from a volume of 0. 01 m 3 and a pressure of 0. 1 MPa to a pressure of 1 MPa. How much heat is transferred during this process? Solution on the black board
 Example 4 -3 Isobaric Process The volume below a weighted piston contains 0. 01 kg of water. The piston area is of 0. 01 m 2 and the piston mass is of 102 kg. The top face of the piston is at atmospheric pressure, 0. 1 MPa. Initially the water is at 25 o. C and the final state is saturated vapor (x=1). How much heat and work are done on or by the water? Solution on the black board
Example 4 -3 Isobaric Process The volume below a weighted piston contains 0. 01 kg of water. The piston area is of 0. 01 m 2 and the piston mass is of 102 kg. The top face of the piston is at atmospheric pressure, 0. 1 MPa. Initially the water is at 25 o. C and the final state is saturated vapor (x=1). How much heat and work are done on or by the water? Solution on the black board
 Isobaric Process For a constant-pressure process, Wb + ΔU = PΔV + ΔU = Δ(U+PV) = ΔH Thus, Q - Wother = Δ H + Δ KE + Δ PE (k. J) Example: Boil water at constant pressure
Isobaric Process For a constant-pressure process, Wb + ΔU = PΔV + ΔU = Δ(U+PV) = ΔH Thus, Q - Wother = Δ H + Δ KE + Δ PE (k. J) Example: Boil water at constant pressure
 Example An insulated tank is divided into two parts by a partition. One part of the tank contains 2. 5 kg of compressed liquid water at 60 o. C and 600 k. Pa while the other part is evacuated. The partition is now removed, and the water expands to fill the entire tank. Determine the final temperature of the water and the volume of the tank for a final pressure of 10 k. Pa.
Example An insulated tank is divided into two parts by a partition. One part of the tank contains 2. 5 kg of compressed liquid water at 60 o. C and 600 k. Pa while the other part is evacuated. The partition is now removed, and the water expands to fill the entire tank. Determine the final temperature of the water and the volume of the tank for a final pressure of 10 k. Pa.
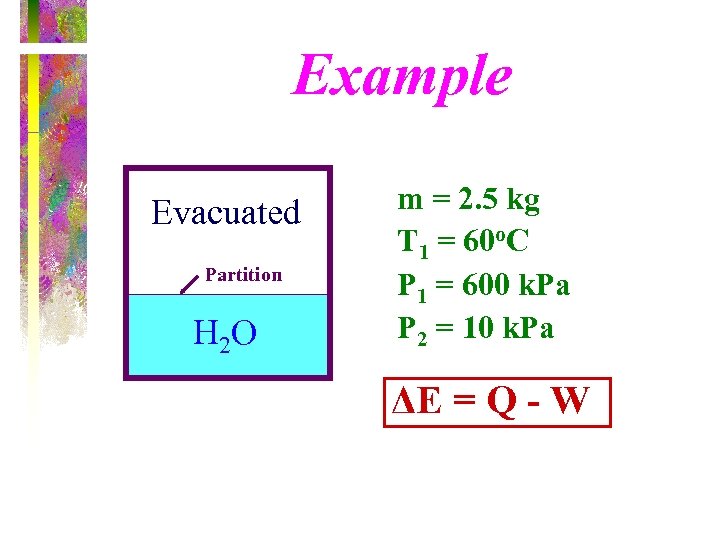 Example Evacuated Partition H 2 O m = 2. 5 kg T 1 = 60 o. C P 1 = 600 k. Pa P 2 = 10 k. Pa ΔE = Q - W
Example Evacuated Partition H 2 O m = 2. 5 kg T 1 = 60 o. C P 1 = 600 k. Pa P 2 = 10 k. Pa ΔE = Q - W
 Solution - page 1 First Law: Q - W = ΔE Q = W = ΔKE = ΔPE = 0 ΔE = ΔU = m(u 2 - u 1) = 0 u 1= u 2 No Work and no Heat therefore the internal Energy is kept constant
Solution - page 1 First Law: Q - W = ΔE Q = W = ΔKE = ΔPE = 0 ΔE = ΔU = m(u 2 - u 1) = 0 u 1= u 2 No Work and no Heat therefore the internal Energy is kept constant
 Solution - page 2 State 1: compressed liquid P 1 = 600 k. Pa, T 1 = 60 o. C vf = vf@60 o. C = 0. 001017 m 3/kg uf = uf@60 o. C = 251. 11 k. J/kg State 2: saturated liquid-vapor mixture P 2 = 10 k. Pa, u 2 = u 1 = 251. 11 k. J/kg uf = 191. 82 k. J/Kg, ufg = 2246. 1 k. J/kg
Solution - page 2 State 1: compressed liquid P 1 = 600 k. Pa, T 1 = 60 o. C vf = vf@60 o. C = 0. 001017 m 3/kg uf = uf@60 o. C = 251. 11 k. J/kg State 2: saturated liquid-vapor mixture P 2 = 10 k. Pa, u 2 = u 1 = 251. 11 k. J/kg uf = 191. 82 k. J/Kg, ufg = 2246. 1 k. J/kg
 Solution - page 3 Thus, T 2 = Tsat@10 k. Pa = 45. 81 o. C v 2 = vf + x 2 vg = [0. 00101+0. 0264*(14. 67 - 0. 00101)] m 3/kg = 0. 388 m 3/kg V 2 = mv 2 = (2. 5 kg)(0. 388 m 3/kg) = 0. 97 m 3
Solution - page 3 Thus, T 2 = Tsat@10 k. Pa = 45. 81 o. C v 2 = vf + x 2 vg = [0. 00101+0. 0264*(14. 67 - 0. 00101)] m 3/kg = 0. 388 m 3/kg V 2 = mv 2 = (2. 5 kg)(0. 388 m 3/kg) = 0. 97 m 3
 Example One kilogram of water is contained in a pistoncylinder device at 100 o. C. The piston rests on lower stops such that the volume occupied by the water is 0. 835 m 3. The cylinder is fitted with an upper set of stops. The volume enclosed by the piston-cylinder device is 0. 841 m 3 when the piston rests against the upper stops. A pressure of 200 k. Pa is required to support the piston. Heat is added to the water until the water exists as a saturated vapor. How much work does the water do on the piston?
Example One kilogram of water is contained in a pistoncylinder device at 100 o. C. The piston rests on lower stops such that the volume occupied by the water is 0. 835 m 3. The cylinder is fitted with an upper set of stops. The volume enclosed by the piston-cylinder device is 0. 841 m 3 when the piston rests against the upper stops. A pressure of 200 k. Pa is required to support the piston. Heat is added to the water until the water exists as a saturated vapor. How much work does the water do on the piston?
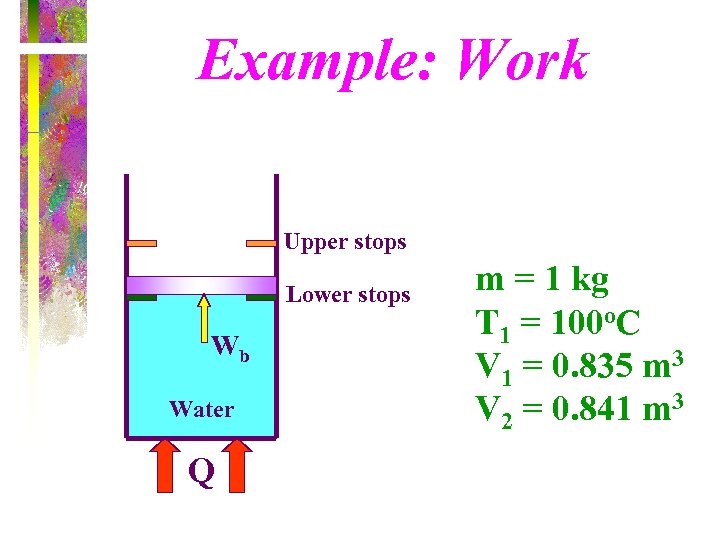 Example: Work Upper stops Lower stops Wb Water Q m = 1 kg T 1 = 100 o. C V 1 = 0. 835 m 3 V 2 = 0. 841 m 3
Example: Work Upper stops Lower stops Wb Water Q m = 1 kg T 1 = 100 o. C V 1 = 0. 835 m 3 V 2 = 0. 841 m 3
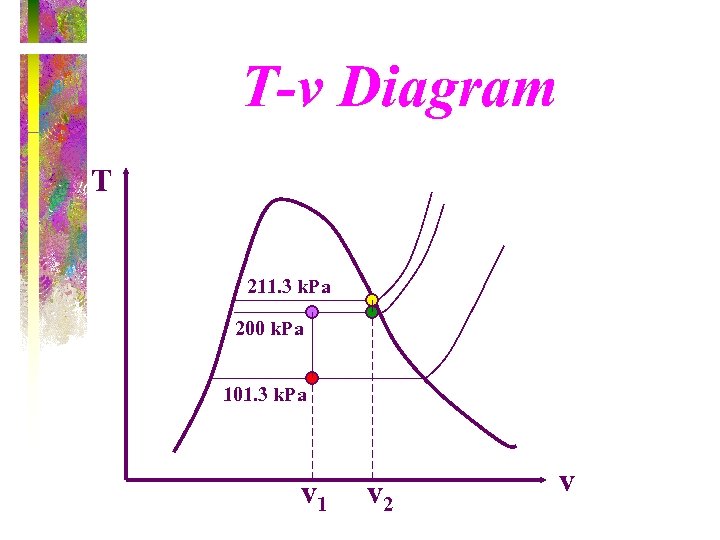 T-v Diagram T 211. 3 k. Pa 200 k. Pa 101. 3 k. Pa v 1 v 2 v
T-v Diagram T 211. 3 k. Pa 200 k. Pa 101. 3 k. Pa v 1 v 2 v
 Solution - page 1 State 1: saturated liquid-vapor mixture T 1 = 100 o. C, vf=0. 001044 m 3/kg , vg=1. 6729 m 3/kg vf < vg ==> saturation P 1=101. 35 k. Pa
Solution - page 1 State 1: saturated liquid-vapor mixture T 1 = 100 o. C, vf=0. 001044 m 3/kg , vg=1. 6729 m 3/kg vf < vg ==> saturation P 1=101. 35 k. Pa
 Solution - page 2 Process 1 -2: The volume stay constant until the pressure increases to 200 k. Pa. Then the piston will move. Process 2 -3: Piston lifts off the bottom stop while the pressure stays constant. State 2: saturated liquid-vapor mixture P 2= 200 k. Pa , v 2 = v 1 = 0. 835 m 3 Does the piston hit upper stops before or after reaching the saturated vapor state?
Solution - page 2 Process 1 -2: The volume stay constant until the pressure increases to 200 k. Pa. Then the piston will move. Process 2 -3: Piston lifts off the bottom stop while the pressure stays constant. State 2: saturated liquid-vapor mixture P 2= 200 k. Pa , v 2 = v 1 = 0. 835 m 3 Does the piston hit upper stops before or after reaching the saturated vapor state?
 Solution - page 3 State 3: Saturated liquid-vapor mixture P 3 = P 2 = 200 k. Pa vf = 0. 001061 m 3/kg , vg = 0. 8857 m 3/kg vf < v 3 < vg ==> piston hit the upper stops before water reaches the saturated vapor state.
Solution - page 3 State 3: Saturated liquid-vapor mixture P 3 = P 2 = 200 k. Pa vf = 0. 001061 m 3/kg , vg = 0. 8857 m 3/kg vf < v 3 < vg ==> piston hit the upper stops before water reaches the saturated vapor state.
 Solution - page 4 Process 3 -4 : With the piston against the upper stops, the volume remains constant during the final heating to the saturated vapor state and the pressure increases. State 4: Saturated vapor state v 4 = v 3 = 0. 841 m 3/kg = vg P 4 = 211. 3 k. Pa , T 4 = 122 o. C
Solution - page 4 Process 3 -4 : With the piston against the upper stops, the volume remains constant during the final heating to the saturated vapor state and the pressure increases. State 4: Saturated vapor state v 4 = v 3 = 0. 841 m 3/kg = vg P 4 = 211. 3 k. Pa , T 4 = 122 o. C
 Solution - page 5 (> 0, done by the system)
Solution - page 5 (> 0, done by the system)
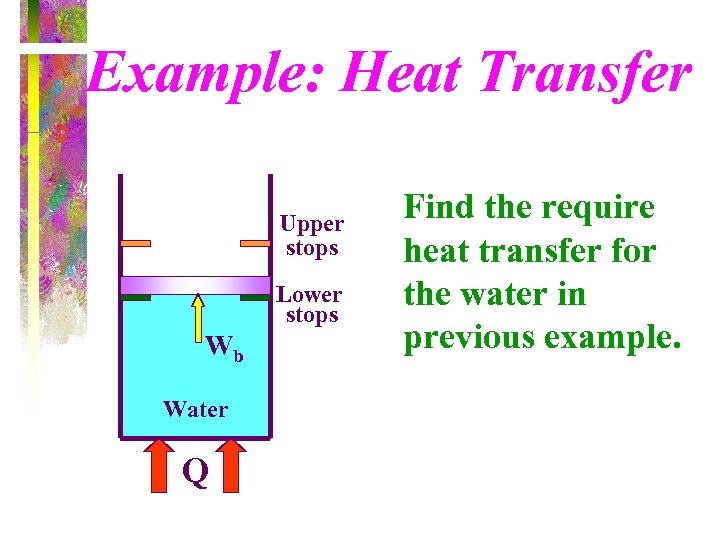 Example: Heat Transfer Upper stops Lower stops Wb Water Q Find the require heat transfer for the water in previous example.
Example: Heat Transfer Upper stops Lower stops Wb Water Q Find the require heat transfer for the water in previous example.
 Solution - page 1 First Law: Conservation of Energy Q - W = ΔE = ΔU + ΔKE + ΔPE Q 14 = Wb, 14 + ΔU 14 = m(u 4 - u 1)
Solution - page 1 First Law: Conservation of Energy Q - W = ΔE = ΔU + ΔKE + ΔPE Q 14 = Wb, 14 + ΔU 14 = m(u 4 - u 1)
 Solution - page 2 State 1: saturated liquid-vapor mixture
Solution - page 2 State 1: saturated liquid-vapor mixture
 Solution - page 3 State 4: saturated vapor state v 4 = 0. 841 m 3/kg = vg u 4 = 2531. 48 k. J/kg (interpolation) (> 0, added to the water)
Solution - page 3 State 4: saturated vapor state v 4 = 0. 841 m 3/kg = vg u 4 = 2531. 48 k. J/kg (interpolation) (> 0, added to the water)
 TEAMPLAY EX. 4 -6 A pressure cooker with volume of 2 liters operates at 0. 2 MPa with water at x = 0. 5. After operation the pressure cooker is left aside allowing its contents to cool. The heat loss is 50 watts, how long does it take for the pressure drop to 0. 1 MPa? What is the state of the water at this point? Indicate the process on a T-v diagram.
TEAMPLAY EX. 4 -6 A pressure cooker with volume of 2 liters operates at 0. 2 MPa with water at x = 0. 5. After operation the pressure cooker is left aside allowing its contents to cool. The heat loss is 50 watts, how long does it take for the pressure drop to 0. 1 MPa? What is the state of the water at this point? Indicate the process on a T-v diagram.
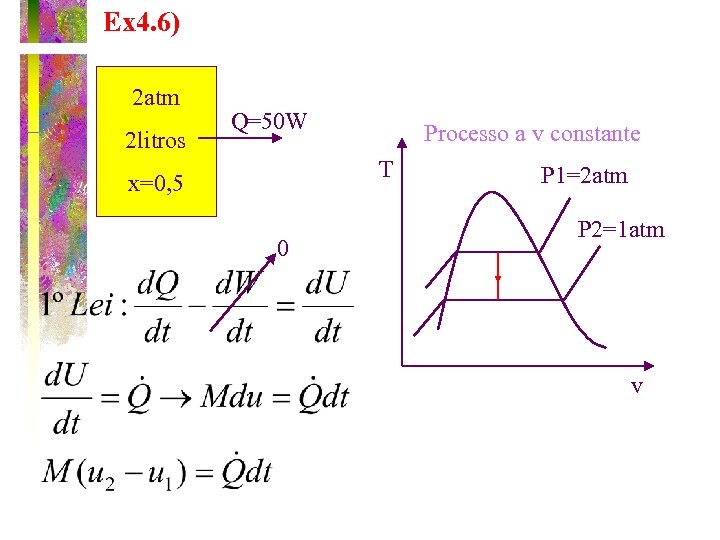 Ex 4. 6) 2 atm 2 litros Q=50 W Processo a v constante T x=0, 5 0 P 1=2 atm P 2=1 atm v
Ex 4. 6) 2 atm 2 litros Q=50 W Processo a v constante T x=0, 5 0 P 1=2 atm P 2=1 atm v
 P 1=0, 2 MPa Tsat=120ºC v 1 L=0, 001 m 3/kg v 1 G=0, 8919 m 3/kg u 1 L=503, 5 KJ/kg u 1 G=2025, 8 KJ/kg x=0, 5 P 2=0, 1 MPa Tsat=100ºC v 2 L=0, 001 m 3/kg v 2 G=1, 6729 m 3/kg u 2 L=418, 9 KJ/kg u 2 G=2087, 5 KJ/kg Vol. Estado 1 = Vol. Estado 2 v=(1 -x)v. L+xv. G v=0, 5*0, 001+0, 5*0, 8919 v 1=v 2=0, 446 m 3/kg Título estado 2: Energia interna estado 2: u 2=(1 -x)u 2 L+xu 2 G u 2=0, 734*418+0, 266*2087, 5 u 2=862 KJ/kg Energia interna estado 1: u 1=(1 -x)u 1 L+xu 1 G u 1=0, 5*503+0, 5*2025, 8 u 1=1264 KJ/kg
P 1=0, 2 MPa Tsat=120ºC v 1 L=0, 001 m 3/kg v 1 G=0, 8919 m 3/kg u 1 L=503, 5 KJ/kg u 1 G=2025, 8 KJ/kg x=0, 5 P 2=0, 1 MPa Tsat=100ºC v 2 L=0, 001 m 3/kg v 2 G=1, 6729 m 3/kg u 2 L=418, 9 KJ/kg u 2 G=2087, 5 KJ/kg Vol. Estado 1 = Vol. Estado 2 v=(1 -x)v. L+xv. G v=0, 5*0, 001+0, 5*0, 8919 v 1=v 2=0, 446 m 3/kg Título estado 2: Energia interna estado 2: u 2=(1 -x)u 2 L+xu 2 G u 2=0, 734*418+0, 266*2087, 5 u 2=862 KJ/kg Energia interna estado 1: u 1=(1 -x)u 1 L+xu 1 G u 1=0, 5*503+0, 5*2025, 8 u 1=1264 KJ/kg
 Massa de Água v=V/M M=V/v M=2*10 -3/0, 446 M=0, 004 kg Aplicando na 1ºLei:
Massa de Água v=V/M M=V/v M=2*10 -3/0, 446 M=0, 004 kg Aplicando na 1ºLei:
 TEAMPLAY EX. 4 -7 A powerful 847 W blender is used to raise 1. 36 kg of water from a temperature of 20 o. C to 70 o. C. If the water loses heat to the surroundings at the rate of 0. 176 W, how much time will the process take?
TEAMPLAY EX. 4 -7 A powerful 847 W blender is used to raise 1. 36 kg of water from a temperature of 20 o. C to 70 o. C. If the water loses heat to the surroundings at the rate of 0. 176 W, how much time will the process take?
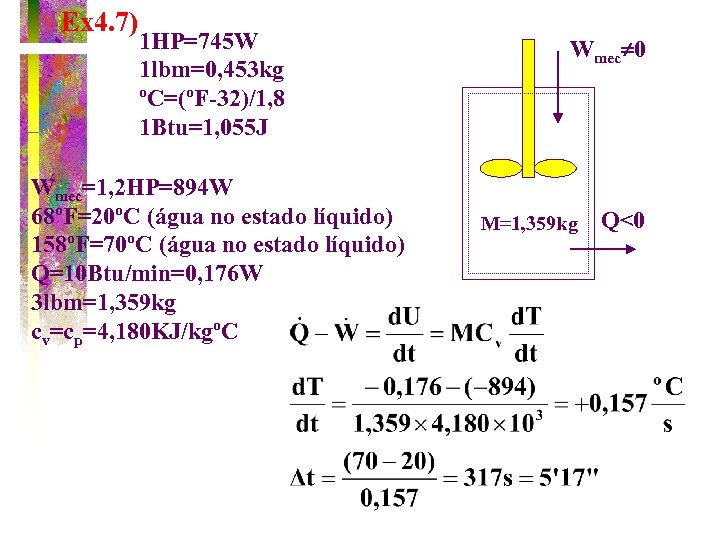 Ex 4. 7) 1 HP=745 W 1 lbm=0, 453 kg ºC=(ºF-32)/1, 8 1 Btu=1, 055 J Wmec=1, 2 HP=894 W 68ºF=20ºC (água no estado líquido) 158ºF=70ºC (água no estado líquido) Q=10 Btu/min=0, 176 W 3 lbm=1, 359 kg cv=cp=4, 180 KJ/kgºC Wmec 0 M=1, 359 kg Q<0
Ex 4. 7) 1 HP=745 W 1 lbm=0, 453 kg ºC=(ºF-32)/1, 8 1 Btu=1, 055 J Wmec=1, 2 HP=894 W 68ºF=20ºC (água no estado líquido) 158ºF=70ºC (água no estado líquido) Q=10 Btu/min=0, 176 W 3 lbm=1, 359 kg cv=cp=4, 180 KJ/kgºC Wmec 0 M=1, 359 kg Q<0
 TEAMPLAY EX. 4 -10 Air, assumed to be ideal gas with constant specific heats, is compressed in a closed piston-cylinder device in a reversible polytropic process with n = 1. 27. The air temperature before compression is 30 o. C and after compression is 130 o. C. Compute the heat transferred on the compression process.
TEAMPLAY EX. 4 -10 Air, assumed to be ideal gas with constant specific heats, is compressed in a closed piston-cylinder device in a reversible polytropic process with n = 1. 27. The air temperature before compression is 30 o. C and after compression is 130 o. C. Compute the heat transferred on the compression process.
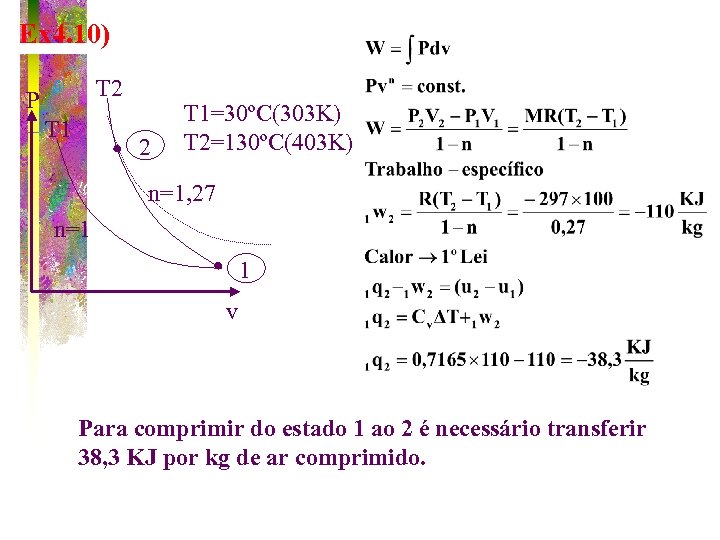 Ex 4. 10) T 2 P T 1 2 T 1=30ºC(303 K) T 2=130ºC(403 K) n=1, 27 n=1 1 v Para comprimir do estado 1 ao 2 é necessário transferir 38, 3 KJ por kg de ar comprimido.
Ex 4. 10) T 2 P T 1 2 T 1=30ºC(303 K) T 2=130ºC(403 K) n=1, 27 n=1 1 v Para comprimir do estado 1 ao 2 é necessário transferir 38, 3 KJ por kg de ar comprimido.


