4d145ee20eb558e75fa29cf03c9a5087.ppt
- Количество слайдов: 48
 The Drug Industry and Clinicians Steven Miles, MD miles 001@umn. edu Slides available
The Drug Industry and Clinicians Steven Miles, MD miles 001@umn. edu Slides available
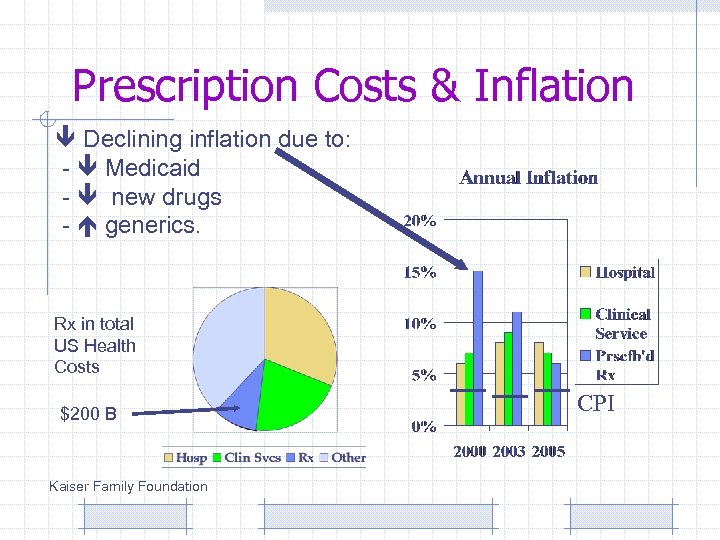 Prescription Costs & Inflation Declining inflation due to: - Medicaid - new drugs - generics. Rx in total US Health Costs $200 B Kaiser Family Foundation CPI
Prescription Costs & Inflation Declining inflation due to: - Medicaid - new drugs - generics. Rx in total US Health Costs $200 B Kaiser Family Foundation CPI
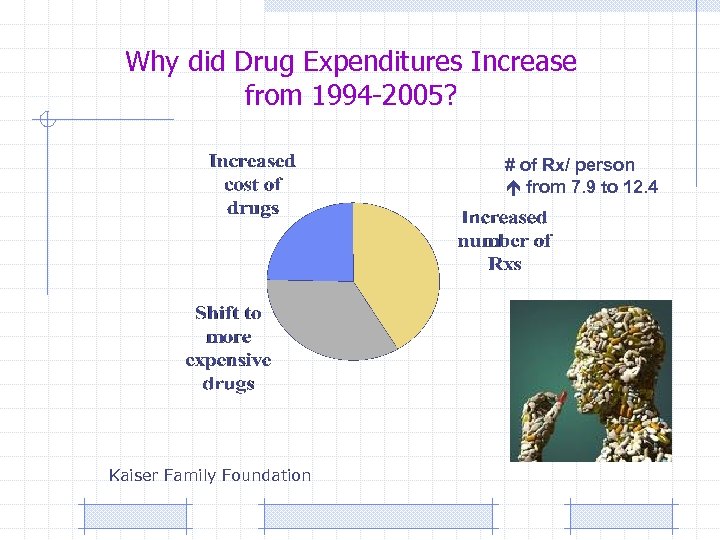 Why did Drug Expenditures Increase from 1994 -2005? # of Rx/ person from 7. 9 to 12. 4 Kaiser Family Foundation
Why did Drug Expenditures Increase from 1994 -2005? # of Rx/ person from 7. 9 to 12. 4 Kaiser Family Foundation
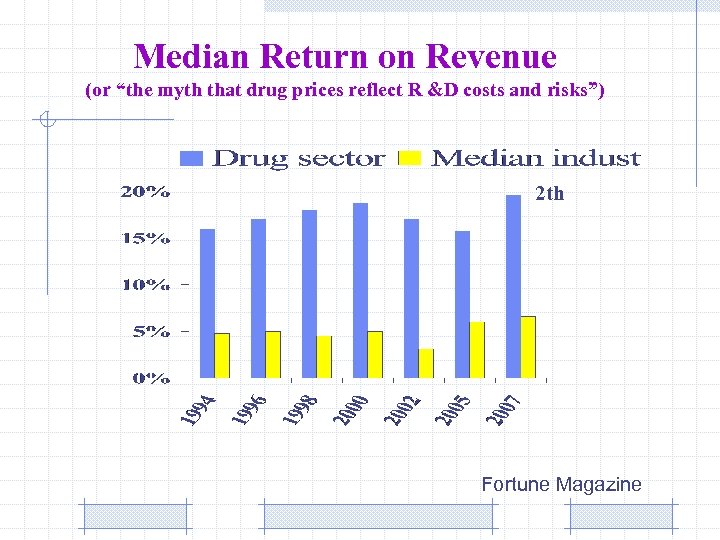 Median Return on Revenue (or “the myth that drug prices reflect R &D costs and risks”) 2 th Fortune Magazine
Median Return on Revenue (or “the myth that drug prices reflect R &D costs and risks”) 2 th Fortune Magazine
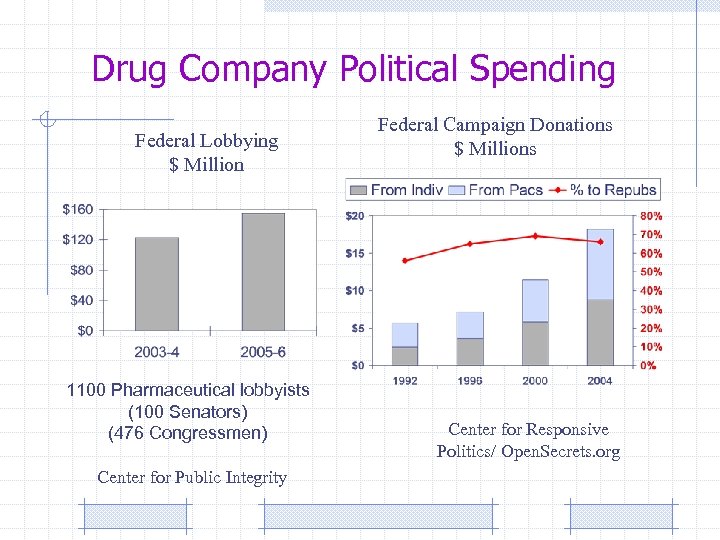 Drug Company Political Spending Federal Lobbying $ Million 1100 Pharmaceutical lobbyists (100 Senators) (476 Congressmen) Center for Public Integrity Federal Campaign Donations $ Millions Center for Responsive Politics/ Open. Secrets. org
Drug Company Political Spending Federal Lobbying $ Million 1100 Pharmaceutical lobbyists (100 Senators) (476 Congressmen) Center for Public Integrity Federal Campaign Donations $ Millions Center for Responsive Politics/ Open. Secrets. org
 Political Priority: Extend patents • 1984: US law extends Rx patents 5 years to allow for R & D time. • 1994: GATT extends drug patents 22 months more. • 1999 Inventors Protection Act • Overall Patents increased 8. 1 to 15 years with longer increases for more recent drugs despite faster FDA approval. – National Institute for Health Care Management, 2002
Political Priority: Extend patents • 1984: US law extends Rx patents 5 years to allow for R & D time. • 1994: GATT extends drug patents 22 months more. • 1999 Inventors Protection Act • Overall Patents increased 8. 1 to 15 years with longer increases for more recent drugs despite faster FDA approval. – National Institute for Health Care Management, 2002
 Best Pharmaceuticals for Children Act 2002 • Extends 188 patents for six months if maker agrees to meet FDA requirement that new drug applications are studied in relevant pediatric groups. • Pediatric tests: $3. 87 M /drug. • FDA: patent extensions worth 40 times that. Coalition for Children s Health, "leading coalition in Washington on children s health policy, " is funded by drug industry and chaired by ex drug lobbyist. It is now closed. • 3 of 4 sponsors in top recipients of drug campaign gifts. • Subcommittee members voting “Aye” received $64, 691 since 1990; “Nays” $25, 493. •
Best Pharmaceuticals for Children Act 2002 • Extends 188 patents for six months if maker agrees to meet FDA requirement that new drug applications are studied in relevant pediatric groups. • Pediatric tests: $3. 87 M /drug. • FDA: patent extensions worth 40 times that. Coalition for Children s Health, "leading coalition in Washington on children s health policy, " is funded by drug industry and chaired by ex drug lobbyist. It is now closed. • 3 of 4 sponsors in top recipients of drug campaign gifts. • Subcommittee members voting “Aye” received $64, 691 since 1990; “Nays” $25, 493. •
 Medicare Prescription Drug and Modernization Act 2003 Medicaid may not mandate bulk purchasing (has lowered drug prices). • Medicare is not direct purchaser (no price controls) and cannot press for discounts. • No fed funds for drug costeffectiveness studies. • Medicare may not interfere “in any way with negotiations” between insurers and drug corps. • • • $50 million in TV ads $10 million to Chamber of Commerce to run pro drug position ads. “Having both house of Congress Republican-controlled was great. Like in Monopoly, when you get to add hotels. ” Drug lobbyist. NYT 9/5/2003 A 1, C 4
Medicare Prescription Drug and Modernization Act 2003 Medicaid may not mandate bulk purchasing (has lowered drug prices). • Medicare is not direct purchaser (no price controls) and cannot press for discounts. • No fed funds for drug costeffectiveness studies. • Medicare may not interfere “in any way with negotiations” between insurers and drug corps. • • • $50 million in TV ads $10 million to Chamber of Commerce to run pro drug position ads. “Having both house of Congress Republican-controlled was great. Like in Monopoly, when you get to add hotels. ” Drug lobbyist. NYT 9/5/2003 A 1, C 4
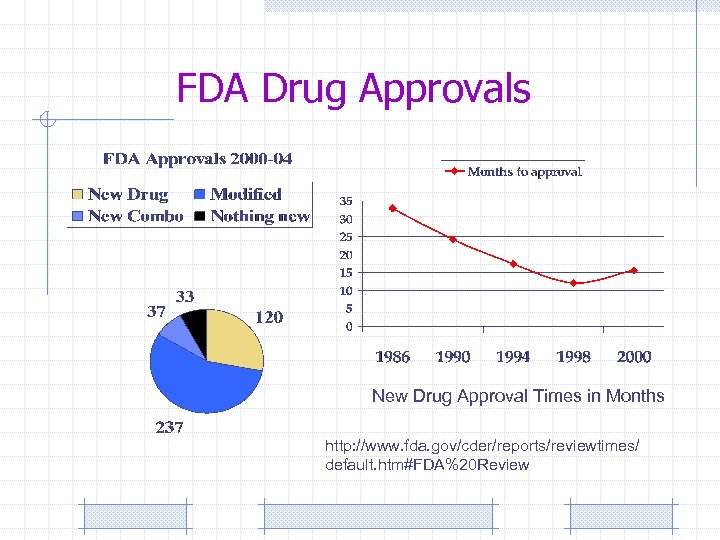 FDA Drug Approvals New Drug Approval Times in Months http: //www. fda. gov/cder/reports/reviewtimes/ default. htm#FDA%20 Review
FDA Drug Approvals New Drug Approval Times in Months http: //www. fda. gov/cder/reports/reviewtimes/ default. htm#FDA%20 Review
 Lawsuits to Delay Generics • Generic prices will be 80% lower. • Claritin’s lawsuit to delay generics by six months cost $5 Million. • Claritin 6 month sales: $1. 3 Billion. – NYT 3/11/2001
Lawsuits to Delay Generics • Generic prices will be 80% lower. • Claritin’s lawsuit to delay generics by six months cost $5 Million. • Claritin 6 month sales: $1. 3 Billion. – NYT 3/11/2001
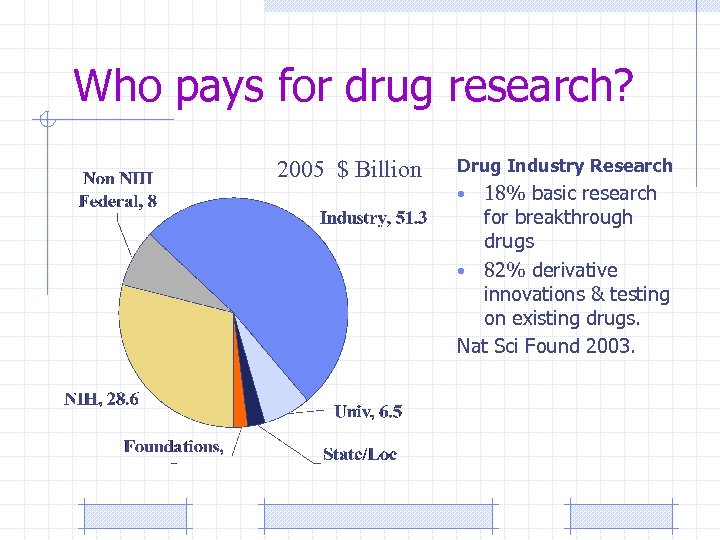 Who pays for drug research? 2005 $ Billion Drug Industry Research 18% basic research for breakthrough drugs • 82% derivative innovations & testing on existing drugs. Nat Sci Found 2003. •
Who pays for drug research? 2005 $ Billion Drug Industry Research 18% basic research for breakthrough drugs • 82% derivative innovations & testing on existing drugs. Nat Sci Found 2003. •
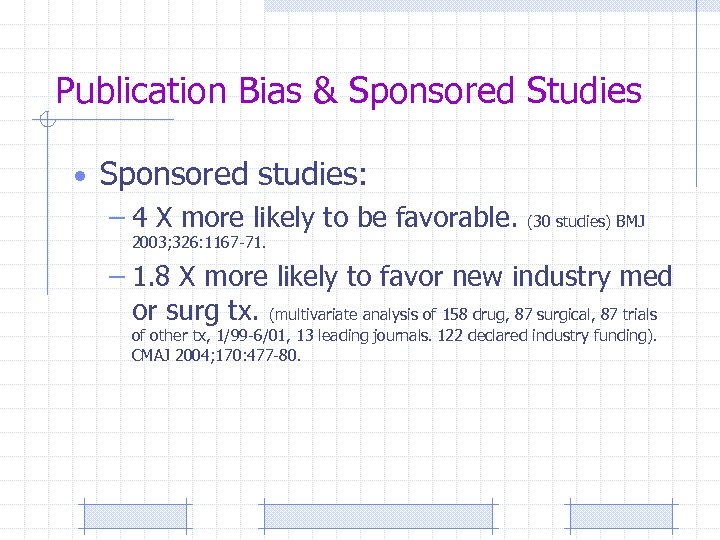 Publication Bias & Sponsored Studies • Sponsored studies: – 4 X more likely to be favorable. (30 studies) BMJ 2003; 326: 1167 -71. – 1. 8 X more likely to favor new industry med or surg tx. (multivariate analysis of 158 drug, 87 surgical, 87 trials of other tx, 1/99 -6/01, 13 leading journals. 122 declared industry funding). CMAJ 2004; 170: 477 -80.
Publication Bias & Sponsored Studies • Sponsored studies: – 4 X more likely to be favorable. (30 studies) BMJ 2003; 326: 1167 -71. – 1. 8 X more likely to favor new industry med or surg tx. (multivariate analysis of 158 drug, 87 surgical, 87 trials of other tx, 1/99 -6/01, 13 leading journals. 122 declared industry funding). CMAJ 2004; 170: 477 -80.
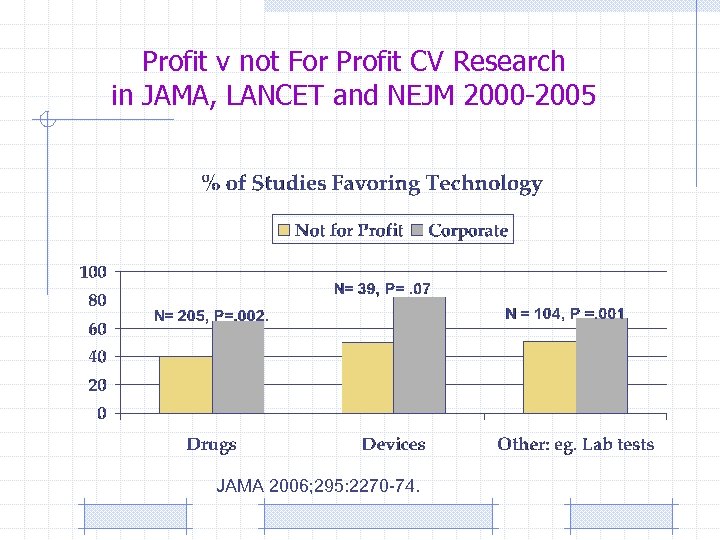 Profit v not For Profit CV Research in JAMA, LANCET and NEJM 2000 -2005 JAMA 2006; 295: 2270 -74.
Profit v not For Profit CV Research in JAMA, LANCET and NEJM 2000 -2005 JAMA 2006; 295: 2270 -74.
 RCTs comparing a statin drug to another statin or non-statin drug • 50% 95/192 of RCTs were funded by industry • 20. 2: Odds ratio of results favoring corporate sponsor v non corporate drug [CI 4. 4 -93], p < 0. 001). • 34. 6: Odds ratio of conclusions favoring corporate sponsor v non corporate drug [CI 7. 1168], p < 0. 001). PLo. S Medicine 2007; 4(6): e 184. Cross-sectional, multivariate.
RCTs comparing a statin drug to another statin or non-statin drug • 50% 95/192 of RCTs were funded by industry • 20. 2: Odds ratio of results favoring corporate sponsor v non corporate drug [CI 4. 4 -93], p < 0. 001). • 34. 6: Odds ratio of conclusions favoring corporate sponsor v non corporate drug [CI 7. 1168], p < 0. 001). PLo. S Medicine 2007; 4(6): e 184. Cross-sectional, multivariate.
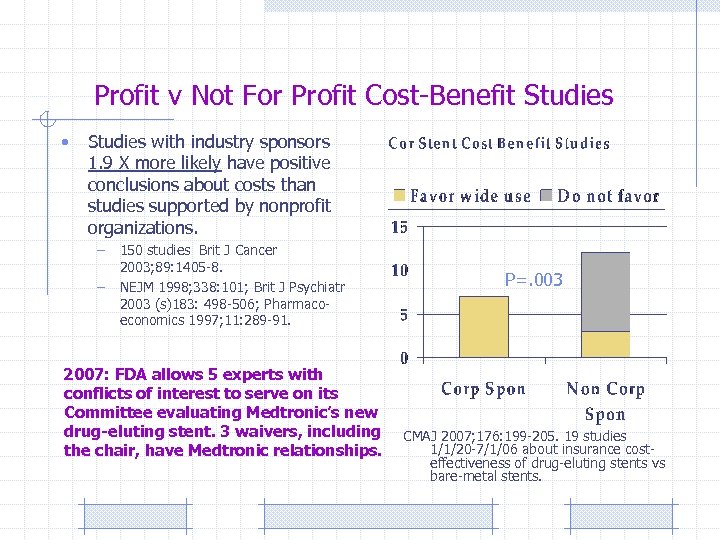 Profit v Not For Profit Cost-Benefit Studies • Studies with industry sponsors 1. 9 X more likely have positive conclusions about costs than studies supported by nonprofit organizations. – – 150 studies Brit J Cancer 2003; 89: 1405 -8. NEJM 1998; 338: 101; Brit J Psychiatr 2003 (s)183: 498 -506; Pharmaco- economics 1997; 11: 289 -91. FDA allows 5 experts with conflicts of interest to serve on its Committee evaluating Medtronic’s new drug-eluting stent. 3 waivers, including the chair, have Medtronic relationships. P=. 003 2007: CMAJ 2007; 176: 199 -205. 19 studies 1/1/20 -7/1/06 about insurance costeffectiveness of drug-eluting stents vs bare-metal stents.
Profit v Not For Profit Cost-Benefit Studies • Studies with industry sponsors 1. 9 X more likely have positive conclusions about costs than studies supported by nonprofit organizations. – – 150 studies Brit J Cancer 2003; 89: 1405 -8. NEJM 1998; 338: 101; Brit J Psychiatr 2003 (s)183: 498 -506; Pharmaco- economics 1997; 11: 289 -91. FDA allows 5 experts with conflicts of interest to serve on its Committee evaluating Medtronic’s new drug-eluting stent. 3 waivers, including the chair, have Medtronic relationships. P=. 003 2007: CMAJ 2007; 176: 199 -205. 19 studies 1/1/20 -7/1/06 about insurance costeffectiveness of drug-eluting stents vs bare-metal stents.
 Publication Bias & Sponsored Studies • Duplicate publication of the same studies resulted in meta-analyses overstating ondansetron’s antiemetic efficacy by 23%. – BMJ 1997; 315: 635 -40. • When results are positive, sponsors more likely to publish multiple papers on preliminary and final results. – BMJ 2003; 326: 1171 -6. (42 studies)
Publication Bias & Sponsored Studies • Duplicate publication of the same studies resulted in meta-analyses overstating ondansetron’s antiemetic efficacy by 23%. – BMJ 1997; 315: 635 -40. • When results are positive, sponsors more likely to publish multiple papers on preliminary and final results. – BMJ 2003; 326: 1171 -6. (42 studies)
 Drug Company Meta-Analysis • Of 124 meta-analysis, – 40% were supported by a drug company. – Corporate sponsored were 5. 1 (OR 1. 5 - 16. 9) times more likely to report favorable conclusions. • BMJ 2007; 335: 1202 -05 (regression analysis) Meta-analysis: the statistical compilation of several studies for finding out their larger conclusion. (Glass, 1976)
Drug Company Meta-Analysis • Of 124 meta-analysis, – 40% were supported by a drug company. – Corporate sponsored were 5. 1 (OR 1. 5 - 16. 9) times more likely to report favorable conclusions. • BMJ 2007; 335: 1202 -05 (regression analysis) Meta-analysis: the statistical compilation of several studies for finding out their larger conclusion. (Glass, 1976)
 Corporate Ghostwriters • 75% in industry-sponsored trials • JAMA 2008; 299 1800 -12 • 11 -16% of papers • JAMA 1998; 280: 222 -24. • See also JAMA 1994; 271: 469 -71. • Refoxecoxib (Vioxx) – 92% of clinical trials ghost authors disclosed industry support. – 50% (36/72) of review ghost authors disclosed support. – JAMA 2008; 299 -1800 -12.
Corporate Ghostwriters • 75% in industry-sponsored trials • JAMA 2008; 299 1800 -12 • 11 -16% of papers • JAMA 1998; 280: 222 -24. • See also JAMA 1994; 271: 469 -71. • Refoxecoxib (Vioxx) – 92% of clinical trials ghost authors disclosed industry support. – 50% (36/72) of review ghost authors disclosed support. – JAMA 2008; 299 -1800 -12.
 Corporate Ghostwriters • Wyeth paid Excerpta Medical (medical communications company) $200, 000 for 10 articles. – “I was given explicit instructions about what to play up and what to play down—whether to enhance broader off label use of the pain product or go strictly by the FDA. ” --RS – “I was given a list of drug company approved phrases. I was pressured to revise my drafts to position the product more favorably. “ –ML Lancet 1999; 354: 136. • After controlling for size of benefit, sponsored papers are 5. 3 X more likely to recommend experimental drug as “drug of choice. ” JAMA 2003; 290: 921 -928 370 RCTs from a random sample of 167 Cochrane reviews.
Corporate Ghostwriters • Wyeth paid Excerpta Medical (medical communications company) $200, 000 for 10 articles. – “I was given explicit instructions about what to play up and what to play down—whether to enhance broader off label use of the pain product or go strictly by the FDA. ” --RS – “I was given a list of drug company approved phrases. I was pressured to revise my drafts to position the product more favorably. “ –ML Lancet 1999; 354: 136. • After controlling for size of benefit, sponsored papers are 5. 3 X more likely to recommend experimental drug as “drug of choice. ” JAMA 2003; 290: 921 -928 370 RCTs from a random sample of 167 Cochrane reviews.
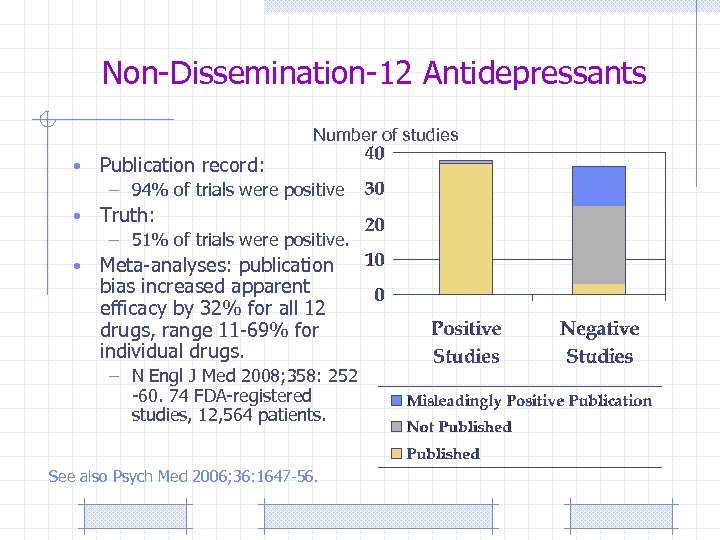 Non-Dissemination-12 Antidepressants Number of studies • Publication record: – 94% of trials were positive • Truth: – 51% of trials were positive. • Meta-analyses: publication bias increased apparent efficacy by 32% for all 12 drugs, range 11 -69% for individual drugs. – N Engl J Med 2008; 358: 252 -60. 74 FDA-registered studies, 12, 564 patients. See also Psych Med 2006; 36: 1647 -56.
Non-Dissemination-12 Antidepressants Number of studies • Publication record: – 94% of trials were positive • Truth: – 51% of trials were positive. • Meta-analyses: publication bias increased apparent efficacy by 32% for all 12 drugs, range 11 -69% for individual drugs. – N Engl J Med 2008; 358: 252 -60. 74 FDA-registered studies, 12, 564 patients. See also Psych Med 2006; 36: 1647 -56.
 Corporate Non-Dissemination • Terminating study in progress that – Doesn’t favor the sponsor. Lancet 1987 5/9/87 p 1091 – No longer serve a marketing priority because of new drug entering market. BMJ; 2001; 322: 603 -6 • Threatening to end relationships with researchers who do not deliver favorable findings. • Threaten to sue researchers or journals if unfavorable, albeit accurate, data is published. – NEJM 1998; 338: 101; 2000; 342: 1539 -44 JAMA. 1999: 1453 -7
Corporate Non-Dissemination • Terminating study in progress that – Doesn’t favor the sponsor. Lancet 1987 5/9/87 p 1091 – No longer serve a marketing priority because of new drug entering market. BMJ; 2001; 322: 603 -6 • Threatening to end relationships with researchers who do not deliver favorable findings. • Threaten to sue researchers or journals if unfavorable, albeit accurate, data is published. – NEJM 1998; 338: 101; 2000; 342: 1539 -44 JAMA. 1999: 1453 -7
 Delay Outcome Research and Results Publication: Zetia and Vytorin • Tx: 800, 000 Americans/week • Earn $4 billion/yr. – Generic Zo. Cor costs a third as much. • 20% of US anti- cholesterol drug market. 2002 FDA approval 2004 Makers begin events trial. 2006 April: Trial Completed. 2008 Results leak: No benefit Possible Harm. • 2005: Ad budget $155 Million Zetia and Vytorin -Forbes
Delay Outcome Research and Results Publication: Zetia and Vytorin • Tx: 800, 000 Americans/week • Earn $4 billion/yr. – Generic Zo. Cor costs a third as much. • 20% of US anti- cholesterol drug market. 2002 FDA approval 2004 Makers begin events trial. 2006 April: Trial Completed. 2008 Results leak: No benefit Possible Harm. • 2005: Ad budget $155 Million Zetia and Vytorin -Forbes
 Relationships between Authors of Clinical Practice Guidelines and Pharmaceutical Industry • • Researchers accepting sponsor funds • • 2. 6 x as likely to favor a controversial Ca++ channel blocker than those without funding. P. <. 0001 • • NEJM 1998; 338: 101 -6. • 4 x as likely to favor a controversial diet aid (Olestra) than those without Proctor and Gamble funding. P<. 0001 • • Am J Pub Health 2003; 93: 664 -9. • See also BMJ 2002; 325: 249; JAMA 2000; 342: 1539 -44 Votes on FDA Vioxx panel. Non industry connected panelists voted 14 to 8 to keep Vioxx off the market. Panelists with company ties voted 9 to 1 to bring Vioxx back to the market. P<. 0001 {miles}
Relationships between Authors of Clinical Practice Guidelines and Pharmaceutical Industry • • Researchers accepting sponsor funds • • 2. 6 x as likely to favor a controversial Ca++ channel blocker than those without funding. P. <. 0001 • • NEJM 1998; 338: 101 -6. • 4 x as likely to favor a controversial diet aid (Olestra) than those without Proctor and Gamble funding. P<. 0001 • • Am J Pub Health 2003; 93: 664 -9. • See also BMJ 2002; 325: 249; JAMA 2000; 342: 1539 -44 Votes on FDA Vioxx panel. Non industry connected panelists voted 14 to 8 to keep Vioxx off the market. Panelists with company ties voted 9 to 1 to bring Vioxx back to the market. P<. 0001 {miles}
 PR and Medical Education • Omnicom, Interpublic, and WPP (Madison Avenue’s 3 largest PR firms) have spent $10 s of millions buying companies to perform drug clinical trials. “We provide services that go • The PR companies from the beginning of drug – Write clinical papers development all the way to – Offer CME the launch of your product. ” – Recruit peer MDs to promote drugs. CEO • NYT 9/22/02 C 1, 4 • 42% of news reports of corporate drug studies disclose funding; 67% refer to drug by brand name. – JAMA 2008; 300: 1544 -50.
PR and Medical Education • Omnicom, Interpublic, and WPP (Madison Avenue’s 3 largest PR firms) have spent $10 s of millions buying companies to perform drug clinical trials. “We provide services that go • The PR companies from the beginning of drug – Write clinical papers development all the way to – Offer CME the launch of your product. ” – Recruit peer MDs to promote drugs. CEO • NYT 9/22/02 C 1, 4 • 42% of news reports of corporate drug studies disclose funding; 67% refer to drug by brand name. – JAMA 2008; 300: 1544 -50.
 Journals Strike Back: 9/2001 • Reserve right to refuse corporate sponsored studies unless researchers are guaranteed independence. – – • New England J Med Lancet Annals of Inter Med JAMA Ann Int Med 2001; 135: 463 -5. • “patently absurd… The journals are becoming more and more antithetical to even considering an industry perspective. ” – Senior VP, Ph. RMA
Journals Strike Back: 9/2001 • Reserve right to refuse corporate sponsored studies unless researchers are guaranteed independence. – – • New England J Med Lancet Annals of Inter Med JAMA Ann Int Med 2001; 135: 463 -5. • “patently absurd… The journals are becoming more and more antithetical to even considering an industry perspective. ” – Senior VP, Ph. RMA
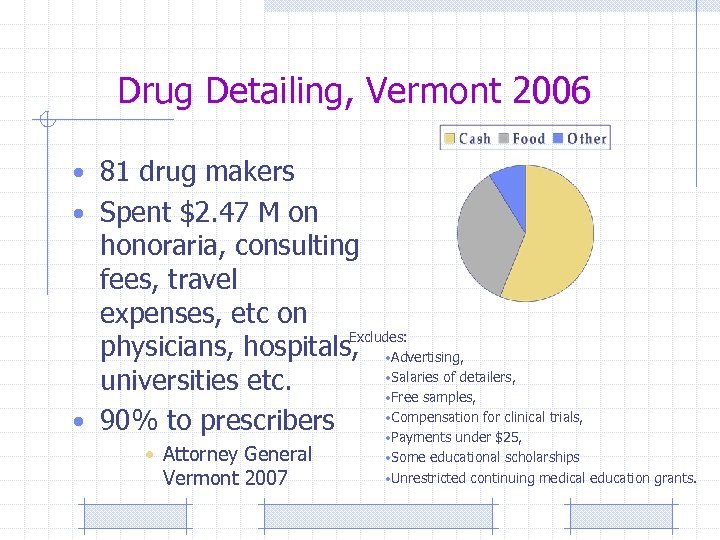 Drug Detailing, Vermont 2006 • 81 drug makers • Spent $2. 47 M on honoraria, consulting fees, travel expenses, etc on Excludes: physicians, hospitals, • Advertising, • Salaries of detailers, universities etc. • Free samples, • Compensation for clinical trials, • 90% to prescribers • Payments under $25, • Attorney General Vermont 2007 • Some educational scholarships • Unrestricted continuing medical education grants.
Drug Detailing, Vermont 2006 • 81 drug makers • Spent $2. 47 M on honoraria, consulting fees, travel expenses, etc on Excludes: physicians, hospitals, • Advertising, • Salaries of detailers, universities etc. • Free samples, • Compensation for clinical trials, • 90% to prescribers • Payments under $25, • Attorney General Vermont 2007 • Some educational scholarships • Unrestricted continuing medical education grants.
 American Psychiatric Assn • Pharmaceutical Revenue – 28% ($14 M) of 38, 000 member org budget. • Amednews. com 2008 Aug 18.
American Psychiatric Assn • Pharmaceutical Revenue – 28% ($14 M) of 38, 000 member org budget. • Amednews. com 2008 Aug 18.
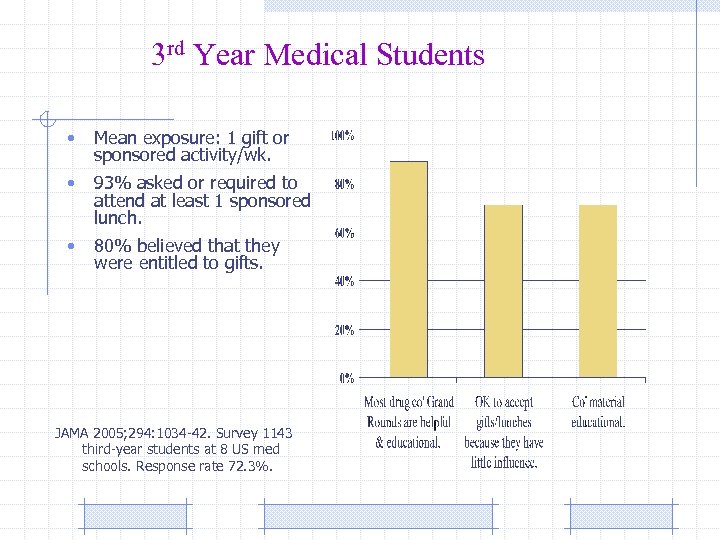 3 rd Year Medical Students • Mean exposure: 1 gift or sponsored activity/wk. • 93% asked or required to attend at least 1 sponsored lunch. • 80% believed that they were entitled to gifts. JAMA 2005; 294: 1034 -42. Survey 1143 third-year students at 8 US med schools. Response rate 72. 3%.
3 rd Year Medical Students • Mean exposure: 1 gift or sponsored activity/wk. • 93% asked or required to attend at least 1 sponsored lunch. • 80% believed that they were entitled to gifts. JAMA 2005; 294: 1034 -42. Survey 1143 third-year students at 8 US med schools. Response rate 72. 3%.
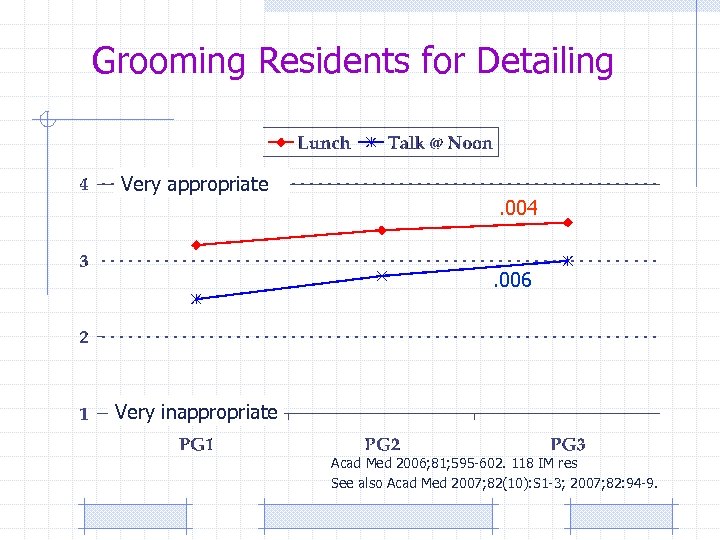 Grooming Residents for Detailing Very appropriate . 004. 006 Very inappropriate Acad Med 2006; 81; 595 -602. 118 IM res See also Acad Med 2007; 82(10): S 1 -3; 2007; 82: 94 -9.
Grooming Residents for Detailing Very appropriate . 004. 006 Very inappropriate Acad Med 2006; 81; 595 -602. 118 IM res See also Acad Med 2007; 82(10): S 1 -3; 2007; 82: 94 -9.
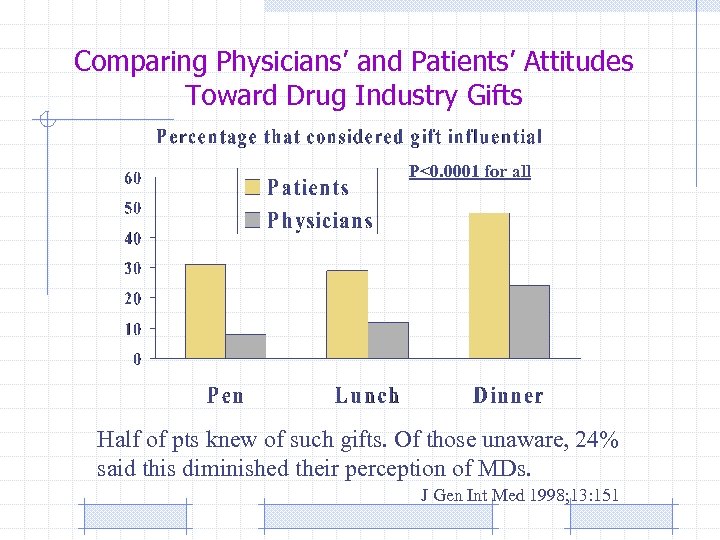 Comparing Physicians’ and Patients’ Attitudes Toward Drug Industry Gifts P<0. 0001 for all Half of pts knew of such gifts. Of those unaware, 24% said this diminished their perception of MDs. J Gen Int Med 1998; 13: 151
Comparing Physicians’ and Patients’ Attitudes Toward Drug Industry Gifts P<0. 0001 for all Half of pts knew of such gifts. Of those unaware, 24% said this diminished their perception of MDs. J Gen Int Med 1998; 13: 151
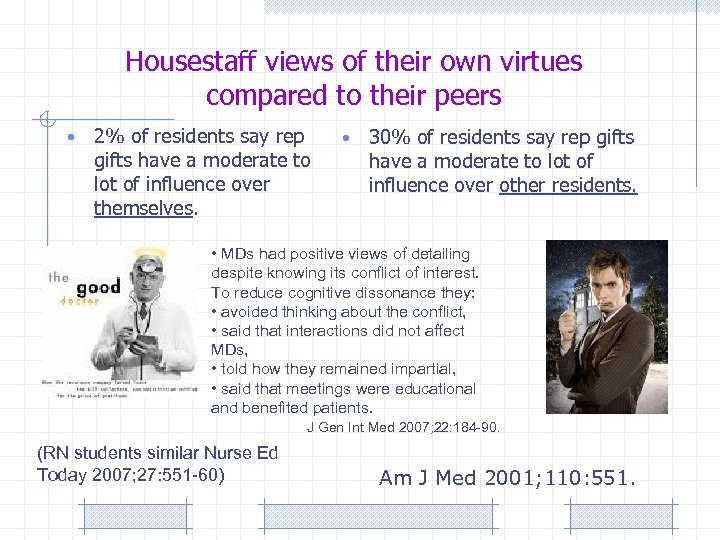 Housestaff views of their own virtues compared to their peers • 2% of residents say rep gifts have a moderate to lot of influence over themselves. • 30% of residents say rep gifts have a moderate to lot of influence over other residents. • MDs had positive views of detailing despite knowing its conflict of interest. To reduce cognitive dissonance they: • avoided thinking about the conflict, • said that interactions did not affect MDs, • told how they remained impartial, • said that meetings were educational and benefited patients. J Gen Int Med 2007; 22: 184 -90. (RN students similar Nurse Ed Today 2007; 27: 551 -60) Am J Med 2001; 110: 551.
Housestaff views of their own virtues compared to their peers • 2% of residents say rep gifts have a moderate to lot of influence over themselves. • 30% of residents say rep gifts have a moderate to lot of influence over other residents. • MDs had positive views of detailing despite knowing its conflict of interest. To reduce cognitive dissonance they: • avoided thinking about the conflict, • said that interactions did not affect MDs, • told how they remained impartial, • said that meetings were educational and benefited patients. J Gen Int Med 2007; 22: 184 -90. (RN students similar Nurse Ed Today 2007; 27: 551 -60) Am J Med 2001; 110: 551.
 Sales Reps’ Information • 11% of statements were inaccurate; all errors favored promoted drug. • No statements about competitors' were favorable, but all were accurate. • 37% of attendees said rep info influenced prescribing. • One University teaching hospital. • JAMA 1995; 273: 1296 -8.
Sales Reps’ Information • 11% of statements were inaccurate; all errors favored promoted drug. • No statements about competitors' were favorable, but all were accurate. • 37% of attendees said rep info influenced prescribing. • One University teaching hospital. • JAMA 1995; 273: 1296 -8.
 What Do Residents Remember After Sales Rep’s Talk? • 8. 4 x more likely to correctly choose rep’s product when it was the first-line agent. • 7. 8 x more likely to incorrectly choose rep’s drug when it was a second line agent. • No attendees, unlike those not exposed to rep’ presentations, knew proper, cheaper drug for the 2 nd indication. (p<. 05). Academic Medicine. 1996; 71: 86 -8
What Do Residents Remember After Sales Rep’s Talk? • 8. 4 x more likely to correctly choose rep’s product when it was the first-line agent. • 7. 8 x more likely to incorrectly choose rep’s drug when it was a second line agent. • No attendees, unlike those not exposed to rep’ presentations, knew proper, cheaper drug for the 2 nd indication. (p<. 05). Academic Medicine. 1996; 71: 86 -8
 Drug Rep Course and Prescribing Invitation Course Months Chest 1992; 102: 270 -73
Drug Rep Course and Prescribing Invitation Course Months Chest 1992; 102: 270 -73
 Off-Label Marketing: Neurontin • Pfizer paid academic MDs $1, 000 to $300, 000/year to promote non-FDA approved uses. • Corporate notes (obtained at trial) – 65% of MDs valued sales rep’ info. – 38% of visits: main message promoted off label use. • ‘This illegal and fraudulent promotion corrupted the information relied up by doctors in their decisions thereby putting patients at risk. ‘ • 5/04/03 Pfizer pled guilty to illegally marketing; paid $430 M from set aside funds. • PLo. S 2007; 40743 -50. In 2003, Neurontin sales were $2. 7 Billion; 90% of prescriptions were for off label use. NYT 5/30/03 p C 1, 2; NYT 5/14/04 C 1.
Off-Label Marketing: Neurontin • Pfizer paid academic MDs $1, 000 to $300, 000/year to promote non-FDA approved uses. • Corporate notes (obtained at trial) – 65% of MDs valued sales rep’ info. – 38% of visits: main message promoted off label use. • ‘This illegal and fraudulent promotion corrupted the information relied up by doctors in their decisions thereby putting patients at risk. ‘ • 5/04/03 Pfizer pled guilty to illegally marketing; paid $430 M from set aside funds. • PLo. S 2007; 40743 -50. In 2003, Neurontin sales were $2. 7 Billion; 90% of prescriptions were for off label use. NYT 5/30/03 p C 1, 2; NYT 5/14/04 C 1.
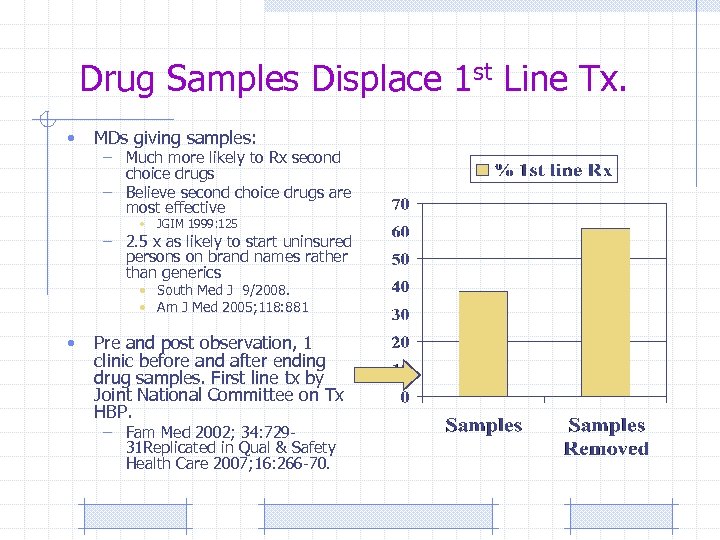 Drug Samples Displace 1 st Line Tx. • MDs giving samples: – Much more likely to Rx second choice drugs – Believe second choice drugs are most effective • JGIM 1999: 125 – 2. 5 x as likely to start uninsured persons on brand names rather than generics • South Med J 9/2008. • Am J Med 2005; 118: 881 • Pre and post observation, 1 clinic before and after ending drug samples. First line tx by Joint National Committee on Tx HBP. – Fam Med 2002; 34: 72931 Replicated in Qual & Safety Health Care 2007; 16: 266 -70.
Drug Samples Displace 1 st Line Tx. • MDs giving samples: – Much more likely to Rx second choice drugs – Believe second choice drugs are most effective • JGIM 1999: 125 – 2. 5 x as likely to start uninsured persons on brand names rather than generics • South Med J 9/2008. • Am J Med 2005; 118: 881 • Pre and post observation, 1 clinic before and after ending drug samples. First line tx by Joint National Committee on Tx HBP. – Fam Med 2002; 34: 72931 Replicated in Qual & Safety Health Care 2007; 16: 266 -70.
 Predictors of Internal Medicine Residency Board (ABIM) Program Pass Rates (3 year running average) Positively Related • Number of faculty Inversely Related • Financial support from drug companies • Clinical duties of residency director (less time for residents) Acad Med 2002; 77: 50
Predictors of Internal Medicine Residency Board (ABIM) Program Pass Rates (3 year running average) Positively Related • Number of faculty Inversely Related • Financial support from drug companies • Clinical duties of residency director (less time for residents) Acad Med 2002; 77: 50
 Pharmacy Residents • 89%: pharmaceutical company-sponsored educational events enhance knowledge. • 43%: information influences therapies. • 26% trained in pharmacist-industry interactions – Am J Health Sys Pharm 2007; 64: 1724 -31. 496 pharmacy residents • See Am J Pharm Ed 2007; 71: Article 68 for a study of Pharmacy Deans views of the potential influence of industry on Pharm D career choices or school policy.
Pharmacy Residents • 89%: pharmaceutical company-sponsored educational events enhance knowledge. • 43%: information influences therapies. • 26% trained in pharmacist-industry interactions – Am J Health Sys Pharm 2007; 64: 1724 -31. 496 pharmacy residents • See Am J Pharm Ed 2007; 71: Article 68 for a study of Pharmacy Deans views of the potential influence of industry on Pharm D career choices or school policy.
 Outline • Industry Snapshot • Political Power • Sponsored Research • Drug Detailing and Clinicians • Drug Detailing and Medical Education • Consumer Marketing
Outline • Industry Snapshot • Political Power • Sponsored Research • Drug Detailing and Clinicians • Drug Detailing and Medical Education • Consumer Marketing
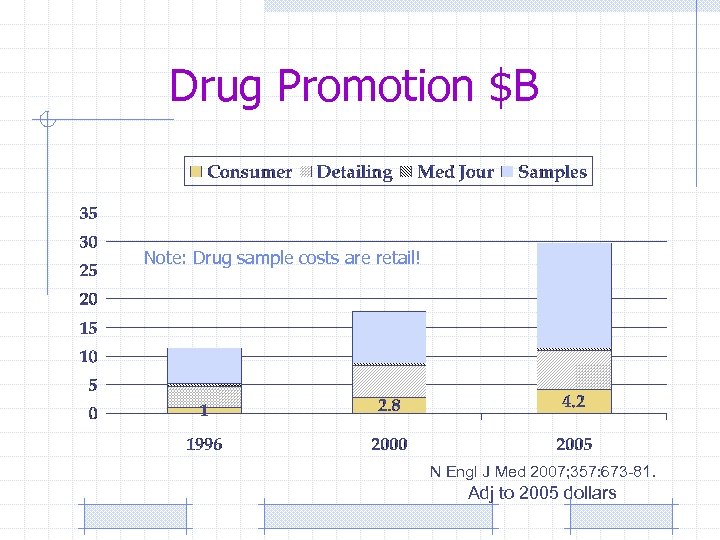 Drug Promotion $B Note: Drug sample costs are retail! N Engl J Med 2007; 357: 673 -81. Adj to 2005 dollars
Drug Promotion $B Note: Drug sample costs are retail! N Engl J Med 2007; 357: 673 -81. Adj to 2005 dollars
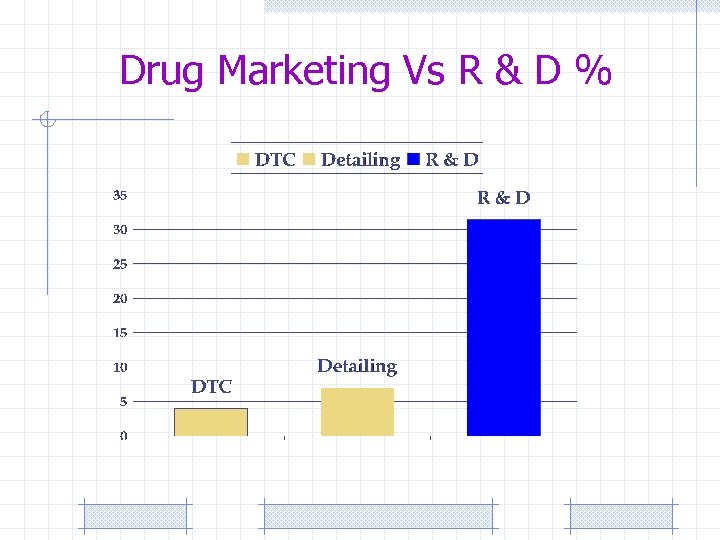 Drug Marketing Vs R & D %
Drug Marketing Vs R & D %
 FDA and deceptive advertising. • • • Bush administration bars FDA from telling a drug company to stop running a deceptive ad until it had run for 11 weeks (earlier limit was 2 weeks). Drug ad life is ~ 12 wks. • US Congress 1/2004 (Waxman). “Promotional spending is an effective method of sharing information and encouraging patients to talk to their doctors. ” --Glaxo spokesperson 50% of public thinks ads are approved by FDA. J Gen Med 1999; 14 651 -657. N Engl J Med 2007; 357: 673 -81.
FDA and deceptive advertising. • • • Bush administration bars FDA from telling a drug company to stop running a deceptive ad until it had run for 11 weeks (earlier limit was 2 weeks). Drug ad life is ~ 12 wks. • US Congress 1/2004 (Waxman). “Promotional spending is an effective method of sharing information and encouraging patients to talk to their doctors. ” --Glaxo spokesperson 50% of public thinks ads are approved by FDA. J Gen Med 1999; 14 651 -657. N Engl J Med 2007; 357: 673 -81.
 Vioxx Advertising 2000 • Merck internal documents obtained at trial show that it was editing out data on excess deaths in 2001. • JAMA 2008; 299: 1813 -17. Meta-analysis: risk x 2. 38 heart attack, unstable angina, sudden cardiac death, stroke, TIA with COX-2 inhibitors. JAMA 2001; 286: 954 -59. September 30, 2004: Merck withdraws Vioxx because research confirms that it risk of heart attack and stroke.
Vioxx Advertising 2000 • Merck internal documents obtained at trial show that it was editing out data on excess deaths in 2001. • JAMA 2008; 299: 1813 -17. Meta-analysis: risk x 2. 38 heart attack, unstable angina, sudden cardiac death, stroke, TIA with COX-2 inhibitors. JAMA 2001; 286: 954 -59. September 30, 2004: Merck withdraws Vioxx because research confirms that it risk of heart attack and stroke.
 Ads’ Effects on Patients with more self-reported ad exposure, conditions treatable by advertised drugs, or more reliant on advertising ask for more advertised drugs. • (12) [7] % of patients request an advertised drug. • Pts asking for advertised drug are (9) [17] times as likely to get it. • MDs are more ambivalent (40 vs 30%) [50 V 12%] about value of a requested advertised drug. • (BMJ 2002; 324: 278 -9. 500 visits, 78 MDs, 1431 patients. ) [CMAJ 2003; 169: 40512 78 MDs, 1431 patients. ] See also: JAMA 2005; 293): 1995 -02.
Ads’ Effects on Patients with more self-reported ad exposure, conditions treatable by advertised drugs, or more reliant on advertising ask for more advertised drugs. • (12) [7] % of patients request an advertised drug. • Pts asking for advertised drug are (9) [17] times as likely to get it. • MDs are more ambivalent (40 vs 30%) [50 V 12%] about value of a requested advertised drug. • (BMJ 2002; 324: 278 -9. 500 visits, 78 MDs, 1431 patients. ) [CMAJ 2003; 169: 40512 78 MDs, 1431 patients. ] See also: JAMA 2005; 293): 1995 -02.
 Pharmacists and Pediatric Off Label Prescriptions • • 70% familiar with off-label prescribing. 40% recalled dispensing pediatric off-label Rx in past month. – – younger than recommended (85%, 297/351), antihistamines, analgesics and beta(2)-agonists, higher dose than recommended (74%, 229/310) lower dose than recommended (41%, 103/258) 60% had been asked by public to sell OTCs (antihist, analges, steroid preps) for off-label pediatric use. • 78% believed they had a duty to inform prescriber • 66% believed that they had a duty to inform parents. • • Brit J Clin Pharm 2007; 64: 90 -5. 1500 randomly selected community pharmacies 32. 1% return.
Pharmacists and Pediatric Off Label Prescriptions • • 70% familiar with off-label prescribing. 40% recalled dispensing pediatric off-label Rx in past month. – – younger than recommended (85%, 297/351), antihistamines, analgesics and beta(2)-agonists, higher dose than recommended (74%, 229/310) lower dose than recommended (41%, 103/258) 60% had been asked by public to sell OTCs (antihist, analges, steroid preps) for off-label pediatric use. • 78% believed they had a duty to inform prescriber • 66% believed that they had a duty to inform parents. • • Brit J Clin Pharm 2007; 64: 90 -5. 1500 randomly selected community pharmacies 32. 1% return.
 Evening News and Prime Time Drug Ads 95%: Emotional appeals 82%: Made factual claims. 86%: Rational arguments. 58%: Framed need in terms of losing control over an aspect of life. • 85%: Framed as regaining control over an aspect of life. • 78%: Showed med use as engendering social approval. • 58%: Portrayed product as a medical breakthrough. • • – Ann Fam Med 2007; 5: 6 -13.
Evening News and Prime Time Drug Ads 95%: Emotional appeals 82%: Made factual claims. 86%: Rational arguments. 58%: Framed need in terms of losing control over an aspect of life. • 85%: Framed as regaining control over an aspect of life. • 78%: Showed med use as engendering social approval. • 58%: Portrayed product as a medical breakthrough. • • – Ann Fam Med 2007; 5: 6 -13.
 Dr. Jarvik: The Rest of the Story • 1976: received MD in from University of Utah. • No internship or residency. • Never licensed to practice. • 2006: Lipitor sales $13. 6 Billion. • “I’m glad I take Lipitor, as a doctor and as a dad. ” 2010: Generic Atorvastatin comes out, about 90% cheaper. – Pfizer lost battle to extend patent; now fighting generic “For me, there is no substitute. ” manufacturer in court.
Dr. Jarvik: The Rest of the Story • 1976: received MD in from University of Utah. • No internship or residency. • Never licensed to practice. • 2006: Lipitor sales $13. 6 Billion. • “I’m glad I take Lipitor, as a doctor and as a dad. ” 2010: Generic Atorvastatin comes out, about 90% cheaper. – Pfizer lost battle to extend patent; now fighting generic “For me, there is no substitute. ” manufacturer in court.
 Steven Miles, MD miles 001@umn. edu
Steven Miles, MD miles 001@umn. edu


