02d459ad8f554dc428339126df4e05ee.ppt
- Количество слайдов: 32
 The Chinese University of Hong Kong Department of Geography and Resource Management Soil Formation Factors with Special Reference to Oxisols and Aridisols Chau Kwai Cheong
The Chinese University of Hong Kong Department of Geography and Resource Management Soil Formation Factors with Special Reference to Oxisols and Aridisols Chau Kwai Cheong
 Outline of Presentation 1. 2. 3. 4. 5. 6. What is soil? What is a true soil? How are soils formed? How are soils distributed spatially? What are oxisols? What are aridisols? Department of Geography and Resource Management 2001
Outline of Presentation 1. 2. 3. 4. 5. 6. What is soil? What is a true soil? How are soils formed? How are soils distributed spatially? What are oxisols? What are aridisols? Department of Geography and Resource Management 2001
 What is Soil? The most basic natural resource of the world • Recycling system for nutrients and organic wastes • Habitat for soil organisms • Engineering medium • Medium for plant growth • System for water supply and purification Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 3. Department of Geography and Resource Management 2001
What is Soil? The most basic natural resource of the world • Recycling system for nutrients and organic wastes • Habitat for soil organisms • Engineering medium • Medium for plant growth • System for water supply and purification Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 3. Department of Geography and Resource Management 2001
 Regolith, soil and bedrock What is Soil? (cont’d. ) • Regolith: The unconsolidated mantle of weathered rock and soil material on the earth’s surface; loose earth materials above solid rock. • Soil: (1) A dynamic natural body composed of mineral and organic materials and living forms in which plants grow. (2) The collection of natural bodies occupying parts of the earth’s surface that support plants and that have properties due to the integrated effect of climate and living matter acting upon parent materials, as conditioned by relief, over periods of time. • Bedrock: The solid rock underlying soils and the regolith in depths ranging from zero (where exposed by erosion) to several hundreds feet. Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 827 -862. Department of Geography and Resource Management 2001
Regolith, soil and bedrock What is Soil? (cont’d. ) • Regolith: The unconsolidated mantle of weathered rock and soil material on the earth’s surface; loose earth materials above solid rock. • Soil: (1) A dynamic natural body composed of mineral and organic materials and living forms in which plants grow. (2) The collection of natural bodies occupying parts of the earth’s surface that support plants and that have properties due to the integrated effect of climate and living matter acting upon parent materials, as conditioned by relief, over periods of time. • Bedrock: The solid rock underlying soils and the regolith in depths ranging from zero (where exposed by erosion) to several hundreds feet. Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 827 -862. Department of Geography and Resource Management 2001
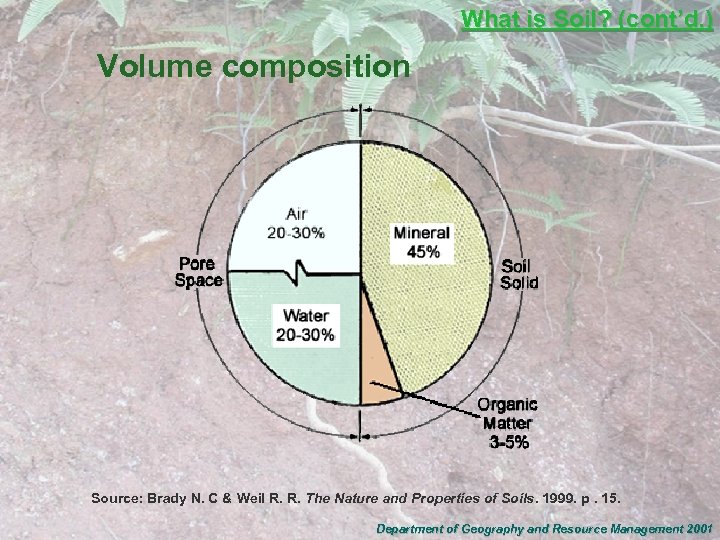 What is Soil? (cont’d. ) Volume composition Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 15. Department of Geography and Resource Management 2001
What is Soil? (cont’d. ) Volume composition Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 15. Department of Geography and Resource Management 2001
 What is a true soil? Uniform weathered parent materials True Soils Department of Geography and Resource Management 2001
What is a true soil? Uniform weathered parent materials True Soils Department of Geography and Resource Management 2001
 What is a true soil? (Cont’d) A true soil has layers or horizons: - Accumulation of organic matter - Incorporation of organic matter (humus) into upper soil by animal, water and gravity - Downward movement of soluble ions (e. g. Ca 2+, SO 42 -) - Formation of clay (secondary aluminosilicate) - Further leaching of soluble ions and translocation of clay downwards - Structural formation Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 3. - Formation of eluvial and illuvial layers Department of Geography and Resource Management 2001
What is a true soil? (Cont’d) A true soil has layers or horizons: - Accumulation of organic matter - Incorporation of organic matter (humus) into upper soil by animal, water and gravity - Downward movement of soluble ions (e. g. Ca 2+, SO 42 -) - Formation of clay (secondary aluminosilicate) - Further leaching of soluble ions and translocation of clay downwards - Structural formation Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 3. - Formation of eluvial and illuvial layers Department of Geography and Resource Management 2001
 How are soils formed? · Weathering (physical, chemical and biological) · Soil genesis (additions, losses, transformation, translocation) Department of Geography and Resource Management 2001
How are soils formed? · Weathering (physical, chemical and biological) · Soil genesis (additions, losses, transformation, translocation) Department of Geography and Resource Management 2001
 Weathering 1. Physical weathering exposes rock surfaces to air, water and carbon dioxide 2. Chemical weathering of solution, oxidation, hydrolysis and carbonation proceed jointly Solution Na. Cl(s) + H 2 O Na+(aq) + OH-(aq) + H+(aq) + Cl-(aq) Oxidation 4 Fe 2+ + 3 O 2 2 Fe 2 O 3(s) Hydration Fe 2 O 3(s) + H 2 O 2 Fe. O. OH(s) Haematite (red) goethite (brown) Department of Geography and Resource Management 2001
Weathering 1. Physical weathering exposes rock surfaces to air, water and carbon dioxide 2. Chemical weathering of solution, oxidation, hydrolysis and carbonation proceed jointly Solution Na. Cl(s) + H 2 O Na+(aq) + OH-(aq) + H+(aq) + Cl-(aq) Oxidation 4 Fe 2+ + 3 O 2 2 Fe 2 O 3(s) Hydration Fe 2 O 3(s) + H 2 O 2 Fe. O. OH(s) Haematite (red) goethite (brown) Department of Geography and Resource Management 2001
 Weathering (cont’d) Carbonation Ca. CO 3(s) + H 2 O + CO 2 Ca 2+(aq) + 2 HCO 3 -(aq) limestone calcium bicarbonate K 2 O. Al 2 O 3. 6 Si. O 2(s) + 2 H 2 O + CO 2 Al 2 O 3. 2 Si. O 2. 2 H 2 O(s) + K 2 CO 3(aq) + 4 Si. O 2(s) Orthoclase feldspar kaolinite potassium carbonate Hydrolysis K 2 O. Al 2 O 3. 6 Si. O 2(s) + 11 H 2 O + CO 2 Al 2 O 3. 2 Si. O 2. 2 H 2 O(s) + 4 H 4 Si. O 4(aq) + 2 K+(aq) + 2 OH-(aq) Orthoclase feldspar kaolinite silicic acid In the presence of acid rain (e. g. H 2 SO 4; HNO 3; H 2 CO 3) generally: Aluminosilicates(s) + H 2 O + H 2 SO 4 clay mineral(s) + (Ca 2+, K+, Na+)(aq) + OH -(aq) + H 4 Si. O 4(aq) + SO 42 -(aq) Department of Geography and Resource Management 2001
Weathering (cont’d) Carbonation Ca. CO 3(s) + H 2 O + CO 2 Ca 2+(aq) + 2 HCO 3 -(aq) limestone calcium bicarbonate K 2 O. Al 2 O 3. 6 Si. O 2(s) + 2 H 2 O + CO 2 Al 2 O 3. 2 Si. O 2. 2 H 2 O(s) + K 2 CO 3(aq) + 4 Si. O 2(s) Orthoclase feldspar kaolinite potassium carbonate Hydrolysis K 2 O. Al 2 O 3. 6 Si. O 2(s) + 11 H 2 O + CO 2 Al 2 O 3. 2 Si. O 2. 2 H 2 O(s) + 4 H 4 Si. O 4(aq) + 2 K+(aq) + 2 OH-(aq) Orthoclase feldspar kaolinite silicic acid In the presence of acid rain (e. g. H 2 SO 4; HNO 3; H 2 CO 3) generally: Aluminosilicates(s) + H 2 O + H 2 SO 4 clay mineral(s) + (Ca 2+, K+, Na+)(aq) + OH -(aq) + H 4 Si. O 4(aq) + SO 42 -(aq) Department of Geography and Resource Management 2001
 Weathering (cont’d) 3. Biological weathering (Root growth and exudates) - Root pressure - Carbonation - Organic acids Department of Geography and Resource Management 2001
Weathering (cont’d) 3. Biological weathering (Root growth and exudates) - Root pressure - Carbonation - Organic acids Department of Geography and Resource Management 2001
 Soil genesis 1. Additions ---- Organic matter, gases 2. Losses ---- salts, carbonate, bicarbonate, ammonia 3. Transformation ---- organic matter to humus, primary mineral to secondary mineral 4. Translocation ---- humus, clay, sesquioxides Department of Geography and Resource Management 2001
Soil genesis 1. Additions ---- Organic matter, gases 2. Losses ---- salts, carbonate, bicarbonate, ammonia 3. Transformation ---- organic matter to humus, primary mineral to secondary mineral 4. Translocation ---- humus, clay, sesquioxides Department of Geography and Resource Management 2001
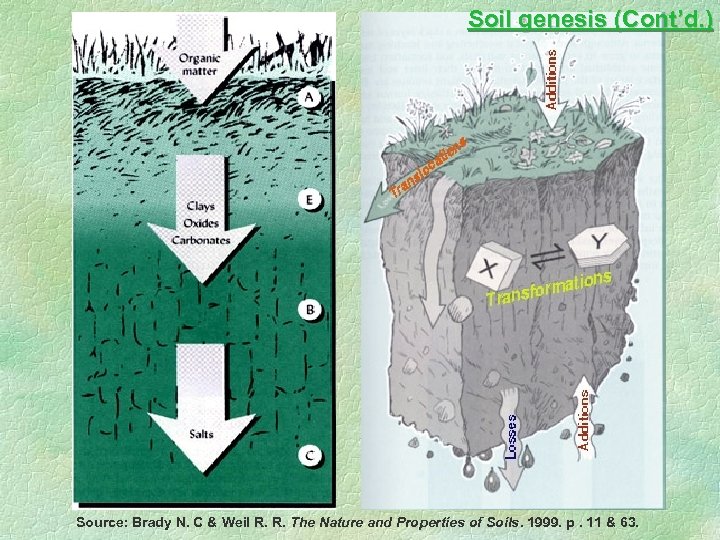 Soil genesis (Cont’d. ) Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 11 & 63.
Soil genesis (Cont’d. ) Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 11 & 63.
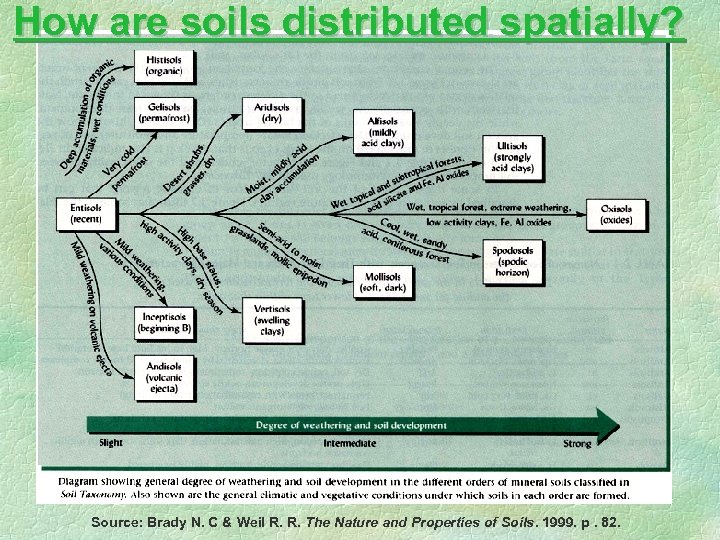 How are soils distributed spatially? Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 82.
How are soils distributed spatially? Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 82.
 Conclusion (1) “Soil formation is stimulated by climate and living organisms acting on parent materials over periods of time and under the modifying influence of topography” (Brady and Weil, 1996) Soil Formation = f ( C, R, P, O, T ) (Jenny 1941) Where, S = Soil formation / Soil properties C = Climate R = Relief P = Parent material O = Organism T = Time (2) “The great diversity of soils in the world results not from the operation of many different processes, but rather from variations in the intensity and length of time the processes have operated” (Brady 1974) Department of Geography and Resource Management 2001
Conclusion (1) “Soil formation is stimulated by climate and living organisms acting on parent materials over periods of time and under the modifying influence of topography” (Brady and Weil, 1996) Soil Formation = f ( C, R, P, O, T ) (Jenny 1941) Where, S = Soil formation / Soil properties C = Climate R = Relief P = Parent material O = Organism T = Time (2) “The great diversity of soils in the world results not from the operation of many different processes, but rather from variations in the intensity and length of time the processes have operated” (Brady 1974) Department of Geography and Resource Management 2001
 Oxisols Properties and Distribution ¨ Most highly weathered soils found mostly in tropical areas (16 -80 m thick) ¨ Occupy nearly 9% of the world’s land ¨ Characterized by a deep oxic subsurface horizon ¨ The oxic horizon is dominated by clay-size particles of the hydrous oxides of iron (Fe 2 O 3) and aluminum (Al 2 O 3. 3 H 2 O), with a low silicasesquioxide ratio of around 1. 5. ¨ Deep red in color due to the presence of iron oxide ¨ High clay content, but the clays are of low-activity, nonsticky type (e. g. kaolinite) ¨ Good physical property, easy to till and drains extremely well ¨ Acidic in reaction, low levels of humus and base content, infertile ¨ Formed under rainforest vegetation in the tropics, hence most of the nutrients are stored in the overstorey layer Department of Geography and Resource Management 2001
Oxisols Properties and Distribution ¨ Most highly weathered soils found mostly in tropical areas (16 -80 m thick) ¨ Occupy nearly 9% of the world’s land ¨ Characterized by a deep oxic subsurface horizon ¨ The oxic horizon is dominated by clay-size particles of the hydrous oxides of iron (Fe 2 O 3) and aluminum (Al 2 O 3. 3 H 2 O), with a low silicasesquioxide ratio of around 1. 5. ¨ Deep red in color due to the presence of iron oxide ¨ High clay content, but the clays are of low-activity, nonsticky type (e. g. kaolinite) ¨ Good physical property, easy to till and drains extremely well ¨ Acidic in reaction, low levels of humus and base content, infertile ¨ Formed under rainforest vegetation in the tropics, hence most of the nutrients are stored in the overstorey layer Department of Geography and Resource Management 2001
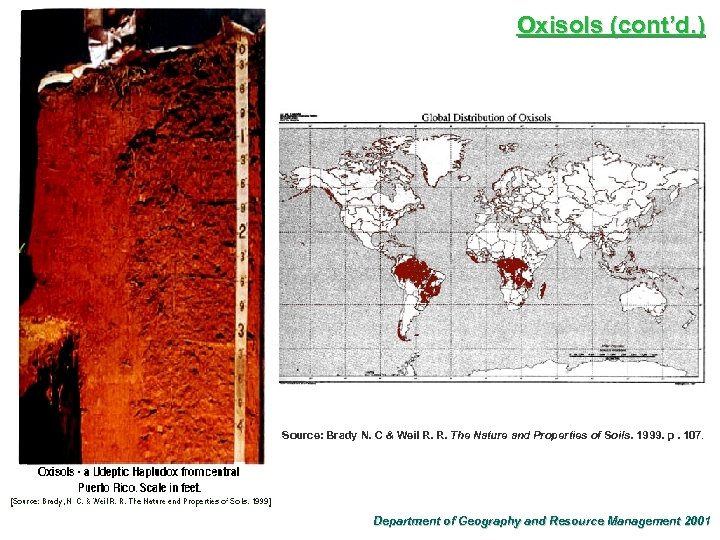 Oxisols (cont’d. ) Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 107. Department of Geography and Resource Management 2001
Oxisols (cont’d. ) Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 107. Department of Geography and Resource Management 2001
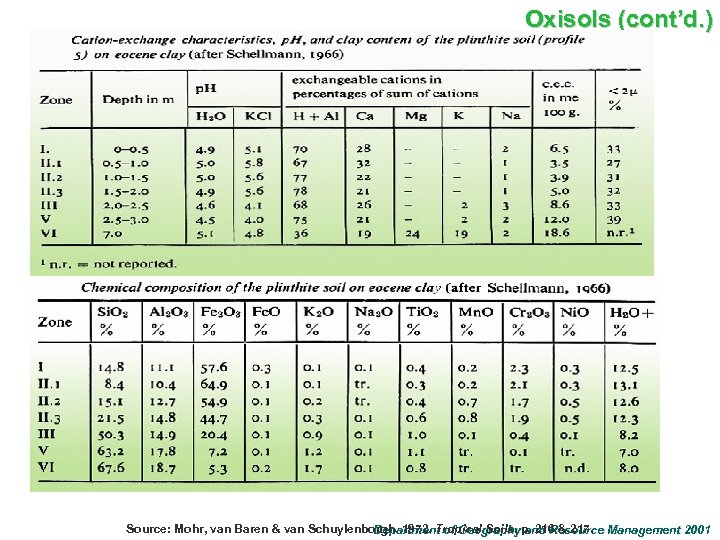 Oxisols (cont’d. ) Source: Mohr, van Baren & van Schuylenborgh. 1972. Tropical Soils. p. 216 & 217. Management 2001 Department of Geography and Resource
Oxisols (cont’d. ) Source: Mohr, van Baren & van Schuylenborgh. 1972. Tropical Soils. p. 216 & 217. Management 2001 Department of Geography and Resource
 Formation l Intense weathering of rock minerals results in the release of silica (desilication), alkali and alkaline earths from the primary aluminosilicates. K 2 O. Al 2 O 3. 6 Si. O 2(s) +11 H 2 O + CO 2 Al 2 O 3. 2 Si. O 2. 2 H 2 O(s) + 4 H 4 Si. O 4(aq)+ 2 K+(aq) + 2 OH-(aq) Orthoclase feldspar kaolinite silicic acid = Alkali and alkaline earths are completely leached out of the profile. = The released silica (Si. O 2) either combines with alumina (Al 2 O 3) to form kaolinite (Al 2 O 3. 2 Si. O 2. 2 H 2 O) or is leached by the percolating water. The soil is thus impoverished in silica, but enriched with iron oxides (Fe 2 O 3. n. H 2 O) and hydroxides (Fe. O. OH). Department of Geography and Resource Management 2001
Formation l Intense weathering of rock minerals results in the release of silica (desilication), alkali and alkaline earths from the primary aluminosilicates. K 2 O. Al 2 O 3. 6 Si. O 2(s) +11 H 2 O + CO 2 Al 2 O 3. 2 Si. O 2. 2 H 2 O(s) + 4 H 4 Si. O 4(aq)+ 2 K+(aq) + 2 OH-(aq) Orthoclase feldspar kaolinite silicic acid = Alkali and alkaline earths are completely leached out of the profile. = The released silica (Si. O 2) either combines with alumina (Al 2 O 3) to form kaolinite (Al 2 O 3. 2 Si. O 2. 2 H 2 O) or is leached by the percolating water. The soil is thus impoverished in silica, but enriched with iron oxides (Fe 2 O 3. n. H 2 O) and hydroxides (Fe. O. OH). Department of Geography and Resource Management 2001
 Formation (cont’d. ) = Alternatively, the decrease in silica can also be relative, and caused by the deposition of sesquioxides (R 2 O 3) transported laterally with groundwater from the higher surroundings. = Desilication results in the formation of the oxic mineral horizon underneath the surface. It consists of weathered mixtures of sesquioxides, clay and quartz sand. It is poor in humus and is at least 30 cm thick. = Under high rainfall conditions and where there is no erosion of the land, this oxic horizon remains soft all the time. Conversely, it hardens irreversibly to form laterite when exposed to air. Department of Geography and Resource Management 2001
Formation (cont’d. ) = Alternatively, the decrease in silica can also be relative, and caused by the deposition of sesquioxides (R 2 O 3) transported laterally with groundwater from the higher surroundings. = Desilication results in the formation of the oxic mineral horizon underneath the surface. It consists of weathered mixtures of sesquioxides, clay and quartz sand. It is poor in humus and is at least 30 cm thick. = Under high rainfall conditions and where there is no erosion of the land, this oxic horizon remains soft all the time. Conversely, it hardens irreversibly to form laterite when exposed to air. Department of Geography and Resource Management 2001
 Use and Problems = Supports 300 -400 million shifting cultivators in the humid tropics = Under undisturbed conditions, deep-rooted trees can pump nutrients from the subsurface layers and recycle them effectively forming a tight nutrient cycle. = Deforestation (excessive logging, conversion to ranches and tropical plantations, fire) disrupts the nutrient cycling process and upsets the ecological balance = Phosphorus deficiency due to fixation by iron and aluminum oxides = Intensive leaching of soluble ions and plant nutrients under high rainfall conditions = When the oxic horizon is exposed to air as a result of deforestation and erosion of the surface soil, it hardens irreversibly to form the so-called laterite (Mekong Project) Department of Geography and Resource Management 2001
Use and Problems = Supports 300 -400 million shifting cultivators in the humid tropics = Under undisturbed conditions, deep-rooted trees can pump nutrients from the subsurface layers and recycle them effectively forming a tight nutrient cycle. = Deforestation (excessive logging, conversion to ranches and tropical plantations, fire) disrupts the nutrient cycling process and upsets the ecological balance = Phosphorus deficiency due to fixation by iron and aluminum oxides = Intensive leaching of soluble ions and plant nutrients under high rainfall conditions = When the oxic horizon is exposed to air as a result of deforestation and erosion of the surface soil, it hardens irreversibly to form the so-called laterite (Mekong Project) Department of Geography and Resource Management 2001
 Management = Avoid removal of the natural vegetation to protect the soil and preserve the nutrients = Shorten the period of farming after slash and burn (sustainable shifting cultivation) = Growth of perennial tree crops, such as rubber, coconut and pepper to restore the nutrient cycling system = Inclusion of legumes in crop rotation (e. g. peanut) to provide additional nitrogen = Restore degraded soils (abandoned pastures, mines) with native tree species Department of Geography and Resource Management 2001
Management = Avoid removal of the natural vegetation to protect the soil and preserve the nutrients = Shorten the period of farming after slash and burn (sustainable shifting cultivation) = Growth of perennial tree crops, such as rubber, coconut and pepper to restore the nutrient cycling system = Inclusion of legumes in crop rotation (e. g. peanut) to provide additional nitrogen = Restore degraded soils (abandoned pastures, mines) with native tree species Department of Geography and Resource Management 2001
 Aridisols (Dry Soils) Properties and Distribution · Dry soils in arid region · Occupy 12% of all global soils · Soil moisture supports plant growth for no longer than 90 consecutive days · Associated with scattered desert shrubs and short bunchgrasses · Horizon of accumulation of calcium carbonate (calcic), gypsum (gypsic), soluble salts (salic) or sodium (natric) · Erosion of fine particles leaves behind windrounded pebbles known as desert pavement or Gobi desert Department of Geography and Resource Management 2001
Aridisols (Dry Soils) Properties and Distribution · Dry soils in arid region · Occupy 12% of all global soils · Soil moisture supports plant growth for no longer than 90 consecutive days · Associated with scattered desert shrubs and short bunchgrasses · Horizon of accumulation of calcium carbonate (calcic), gypsum (gypsic), soluble salts (salic) or sodium (natric) · Erosion of fine particles leaves behind windrounded pebbles known as desert pavement or Gobi desert Department of Geography and Resource Management 2001
 Aridisols (cont’d. ) (Sahara Desert, Gobi and Taklamakan Deserts in China, Turkestan Desert, southern and central Australia, SW Africa and southern Argentina) Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 94. Department of Geography and Resource Management 2001
Aridisols (cont’d. ) (Sahara Desert, Gobi and Taklamakan Deserts in China, Turkestan Desert, southern and central Australia, SW Africa and southern Argentina) Source: Brady N. C & Weil R. R. The Nature and Properties of Soils. 1999. p. 94. Department of Geography and Resource Management 2001
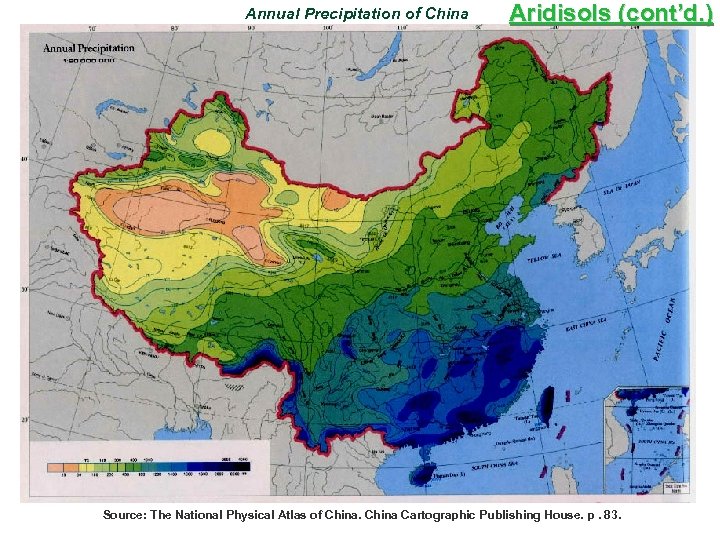 Annual Precipitation of China Aridisols (cont’d. ) Source: The National Physical Atlas of China Cartographic Publishing House. p. 83.
Annual Precipitation of China Aridisols (cont’d. ) Source: The National Physical Atlas of China Cartographic Publishing House. p. 83.
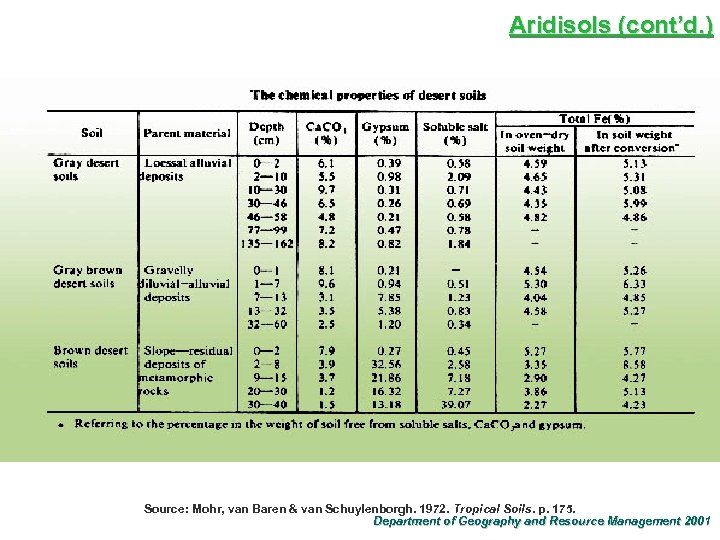 Aridisols (cont’d. ) Source: Mohr, van Baren & van Schuylenborgh. 1972. Tropical Soils. p. 175. Department of Geography and Resource Management 2001
Aridisols (cont’d. ) Source: Mohr, van Baren & van Schuylenborgh. 1972. Tropical Soils. p. 175. Department of Geography and Resource Management 2001
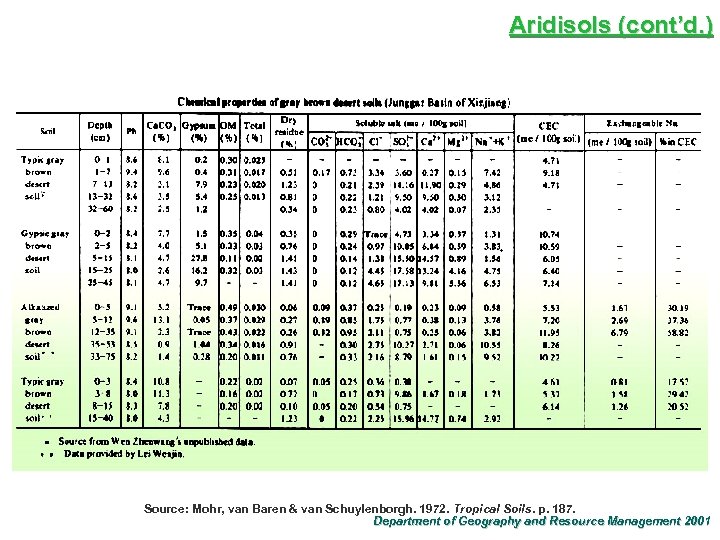 Aridisols (cont’d. ) Source: Mohr, van Baren & van Schuylenborgh. 1972. Tropical Soils. p. 187. Department of Geography and Resource Management 2001
Aridisols (cont’d. ) Source: Mohr, van Baren & van Schuylenborgh. 1972. Tropical Soils. p. 187. Department of Geography and Resource Management 2001
 Formation · Occur in poorly drained valley floors, flats and basins in the continental interior · Weathering of parent materials or deposit of secondary aluminosilicates · Surface runoff evaporates, leaving behind large amounts of carbonates (CO 32 -), bicarbonates (HCO 3 -), sulfates (SO 42 -) and chlorides (Cl-) of sodium, calcium, magnesium and potassium · Sources of the salts - weathering of rocks and minerals - brought to the surface through rainfall, irrigation, capillarity - salt deposits during geological time in the bottom of now extinct lakes or oceans Department of Geography and Resource Management 2001
Formation · Occur in poorly drained valley floors, flats and basins in the continental interior · Weathering of parent materials or deposit of secondary aluminosilicates · Surface runoff evaporates, leaving behind large amounts of carbonates (CO 32 -), bicarbonates (HCO 3 -), sulfates (SO 42 -) and chlorides (Cl-) of sodium, calcium, magnesium and potassium · Sources of the salts - weathering of rocks and minerals - brought to the surface through rainfall, irrigation, capillarity - salt deposits during geological time in the bottom of now extinct lakes or oceans Department of Geography and Resource Management 2001
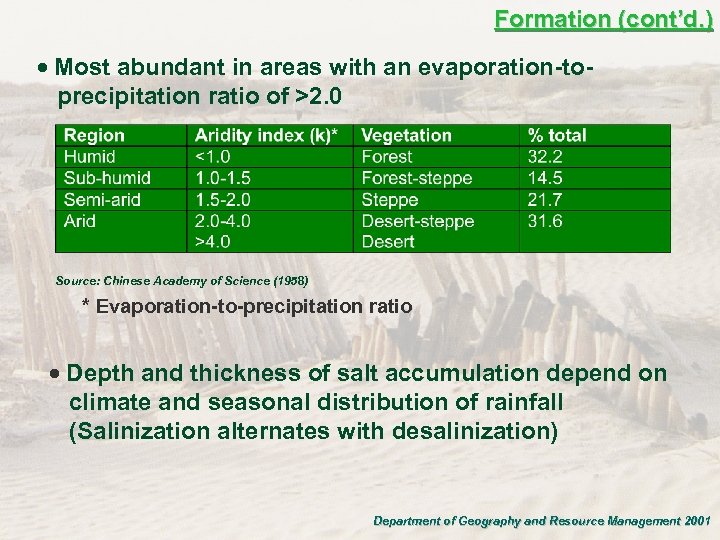 Formation (cont’d. ) · Most abundant in areas with an evaporation-toprecipitation ratio of >2. 0 Source: Chinese Academy of Science (1958) * Evaporation-to-precipitation ratio · Depth and thickness of salt accumulation depend on climate and seasonal distribution of rainfall (Salinization alternates with desalinization) Department of Geography and Resource Management 2001
Formation (cont’d. ) · Most abundant in areas with an evaporation-toprecipitation ratio of >2. 0 Source: Chinese Academy of Science (1958) * Evaporation-to-precipitation ratio · Depth and thickness of salt accumulation depend on climate and seasonal distribution of rainfall (Salinization alternates with desalinization) Department of Geography and Resource Management 2001
 Use and Problems · Salt accumulation (up to 10% in NW China) decreases the osmotic water potential which, in turn, reduces the rate of water uptake by roots and germinating seeds. Plants die off as a result of wilting. · Water deficiency · Where salts accumulate as crust on the surface, the soil is vulnerable to dessication and wind erosion, resulting in the decline of biological productivity. · Extreme temperatures and strong winds detrimental to plant growth · Coarse texture/stoniness not favourable to crop growth · Phosphorus deficiency due to fixation by calcium · High sodium content destabilizes soil structure Department of Geography and Resource Management 2001
Use and Problems · Salt accumulation (up to 10% in NW China) decreases the osmotic water potential which, in turn, reduces the rate of water uptake by roots and germinating seeds. Plants die off as a result of wilting. · Water deficiency · Where salts accumulate as crust on the surface, the soil is vulnerable to dessication and wind erosion, resulting in the decline of biological productivity. · Extreme temperatures and strong winds detrimental to plant growth · Coarse texture/stoniness not favourable to crop growth · Phosphorus deficiency due to fixation by calcium · High sodium content destabilizes soil structure Department of Geography and Resource Management 2001
 Management · Minimize irrigation with salt-laden water and practise drip irrigation · Avoid over-grazing, overcultivation and excessive cutting of vegetation · Avoid construction of reservoir dam upstream of inland rivers (e. g. Tarim River) · Recycle organic matter · Reclamation with salt-tolerant grasses, shrubs and trees · Increase surface roughness to halt the advance of moving sand dunes (Phragmites spp. ) Department of Geography and Resource Management 2001
Management · Minimize irrigation with salt-laden water and practise drip irrigation · Avoid over-grazing, overcultivation and excessive cutting of vegetation · Avoid construction of reservoir dam upstream of inland rivers (e. g. Tarim River) · Recycle organic matter · Reclamation with salt-tolerant grasses, shrubs and trees · Increase surface roughness to halt the advance of moving sand dunes (Phragmites spp. ) Department of Geography and Resource Management 2001
 Thank you ! Department of Geography and Resource Management 2001
Thank you ! Department of Geography and Resource Management 2001


