dc6acc3dce1ad494adb6cf05a288b43c.ppt
- Количество слайдов: 18

The Analyses of Statewide Laboratory Surveys for Electronic Laboratory Reporting Hwa-Gan Chang 1 Dave Di. Cesare 1 Kathryn Schmit 1 Jennifer Baumgartner 2 Laura Goodman 2 Charles Di. Donato 3 Perry Smith 1 New York State Department of Health 2 New York City Department of Health 3 Keane, Inc 1

New York ECLRS Development and Implementation Initiation Development Implementation 1999 2000 -01 2001 -05 Maintenance/ Enhancement 2005 -current Mandatory 2007+ ECLRS RFP & a Vendor Selected ECLRS Development ECLRS Training 1 st Lab Roll out Add HIV, Lead NYCDOH, Cancer Report card Audit L aw passed Add Cong Malf, Newborn
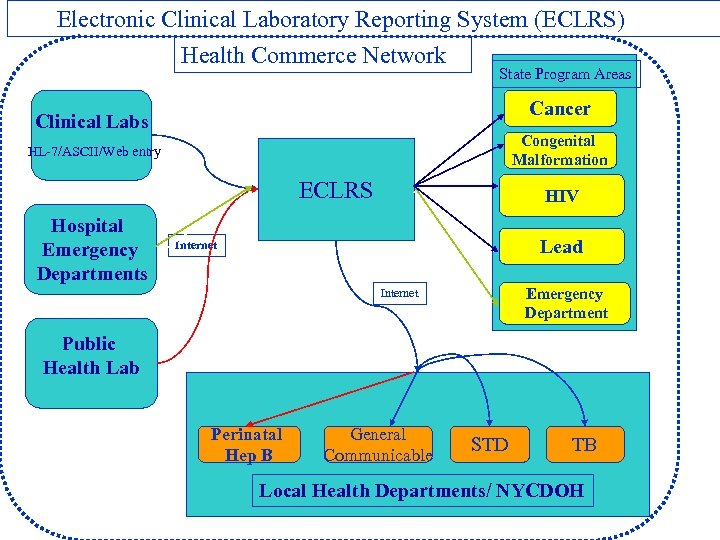
Electronic Clinical Laboratory Reporting System (ECLRS) Health Commerce Network State Program Areas Cancer Clinical Labs Congenital Malformation HL-7/ASCII/Web entry ECLRS Hospital Emergency Departments HIV Lead Internet Emergency Department Internet Public Health Lab Perinatal Hep B General Communicable STD TB Local Health Departments/ NYCDOH

New York Mandatory Electronic Laboratory Reporting • Law was passed on July 18, 2007 and was posted on ECLRS home page • A letter was mailed to laboratory directors (N=927) in February 2008 • A lab survey was developed and posted on ECLRS web page in March 2008 • A reminder was emailed to lab contacts in May 2008

Materials Ø Lab survey #1: Web survey entered by laboratory staff Ø Lab survey #2: Paper survey filled out by DOH investigators

Lab Survey #1 - Objectives Ø To gain knowledge regarding type of tests that labs perform Ø To identify labs that are not reporting via ECLRS Ø To learn which LIS vendors are used Ø To learn which reference labs are used

Survey Questions • Lab location and contact information • Testing of specimens from NYS residents (Y/N) • Types of tests performed for: – – – – General Communicable Diseases (68) Sexually Transmitted Diseases (6) Tuberculosis HIV Heavy metals (lead, arsenic, cadmium, mercury, zinc) Pathology/Cancer Congenital Malformations Hemoglobin A 1 c

Survey Questions • Reference labs used • Laboratory Information System vendors • Program area specific information – NYCDOH: susceptibility testing, Hemoglobin A 1 c – HIV: type of testing, kit – Cancer: facilities served – Heavy Metals: type of Metals – Congenital Malformations: type of cytogenetic testing
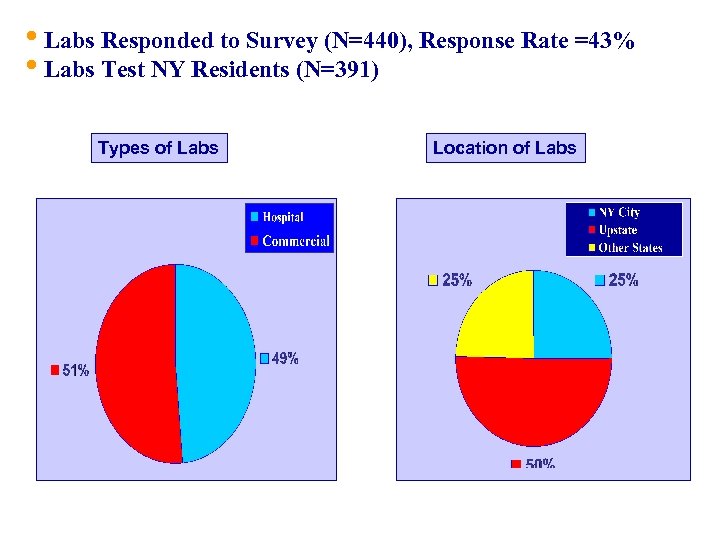
• Labs Responded to Survey (N=440), Response Rate =43% • Labs Test NY Residents (N=391) Types of Labs Location of Labs

Labs Testing NYS Residents • 213 labs: General Communicable Diseases – – – Less than 10 diseases tested for: 10 – 30 diseases tested for: 31 – 60 diseases tested for: 59 71 83 • 202 labs: Sexually Transmitted Diseases • 100 labs: Tuberculosis • 46 labs: HIV • 54 labs: Heavy metals • 24 labs: Congenital Malformations • 161 labs: Pathology / Cancer

Use of Reference Labs • Yes: 159 hospital labs, 83 commercial labs • No: 31 hospital labs, 118 commercial labs • Most frequently used reference labs: – – – – QUEST NYSDOH public health lab LABCORP MAYO SPECIALTY NYCDOHMH LAB ARUP 114 64 62 17 28 21 13

Lab Information System Vendors • Hospital labs: – – – – MEDITECH SOFT CERNER MYSIS SUNQUEST IN-HOUSE Other 190 47 28 25 23 18 6 29 • Commercial labs: 201 – – – – MEDITECH SOFT CERNER MYSIS SUNQUEST IN-HOUSE Other 3 8 11 8 1 49 79

Number of Labs Reporting via ECLRS by Reportable Conditions Labs that responded General Communicable STDs, TB HIV 224 Labs that report to ECLRS 195 19 in progress Labs that do not report to ECLRS 10 46 46 0 161 36 4 in progress 1 Heavy Metals 54 48 4 in progress 2 Congenital Malformations 24 1 2 in progress 21 Cancer

Lab Audit Survey #2 - Objectives • To validate lab contact information while visiting labs for audit • To randomly select 8 general communicable, 8 STD, and 8 HIV tests from lab log for validation • To evaluate labs’ completeness of reports if reporting via ECLRS

Survey Questions • Lab contact information • Surveyor information • Type of tests certified by State • Name, location, and test type of reference labs • A representative sample of 8 positive test results by test type with specimen accession number, collection date, and reportable condition

Lab Survey Results for Communicable Diseases as of July 31, 2008 No. of Laboratories Surveyed N=380 No. of Labs Reporting via ECLRS N=251 No. of Labs Not Reporting via ECLRS N=129 100% Reporting Partial Reporting List not matched Enrolled / Just certified N=150 N=11 N=14 N=76 Do not test for reportable diseases Test for reportable diseases N=125 N=4

Summary • • Form builder utility can be easily applied to any web survey The low lab web survey response rate indicated the difficulty of obtaining information, but the responses were helpful in identifying labs not reporting via ECLRS The investigators visiting labs were valuable for evaluating the completeness of reports via ECLRS For communicable disease lab reporting, virtually 98% labs are reporting electronically
dc6acc3dce1ad494adb6cf05a288b43c.ppt