 Technology Update and Status of Current Clinical Trials Rhonda Robb, VP Catheter Based Therapies, Medtronic, Inc.
Technology Update and Status of Current Clinical Trials Rhonda Robb, VP Catheter Based Therapies, Medtronic, Inc.
 Conflict of Interest Employee and Shareholder of Medtronic, Inc.
Conflict of Interest Employee and Shareholder of Medtronic, Inc.
 Caution Statement The Medtronic Core. Valve® System is not currently available in the USA for clinical trials or for commercialization. Not available for sale in the US. Core. Valve is a registered trademark of Medtronic CV Luxembourg S. a. r. l.
Caution Statement The Medtronic Core. Valve® System is not currently available in the USA for clinical trials or for commercialization. Not available for sale in the US. Core. Valve is a registered trademark of Medtronic CV Luxembourg S. a. r. l.
 History Core. Valve Design Strategic Intent: • Treat severe aortic stenosis, requiring valve replacement, with a truly percutaneous system • Design must accommodate an inelastic, often irregularly shaped (oval) annulus • Minimize trauma during deployment • Maximize hemodynamic performance • Durability = Design & Proper Placement (including ability to reposition) 1 st Clinical Implants Initiated in Summer 2004 Gen 1 25 F Gen 2 21 F Gen 3 18 F 2004 2005 2006 2007
History Core. Valve Design Strategic Intent: • Treat severe aortic stenosis, requiring valve replacement, with a truly percutaneous system • Design must accommodate an inelastic, often irregularly shaped (oval) annulus • Minimize trauma during deployment • Maximize hemodynamic performance • Durability = Design & Proper Placement (including ability to reposition) 1 st Clinical Implants Initiated in Summer 2004 Gen 1 25 F Gen 2 21 F Gen 3 18 F 2004 2005 2006 2007
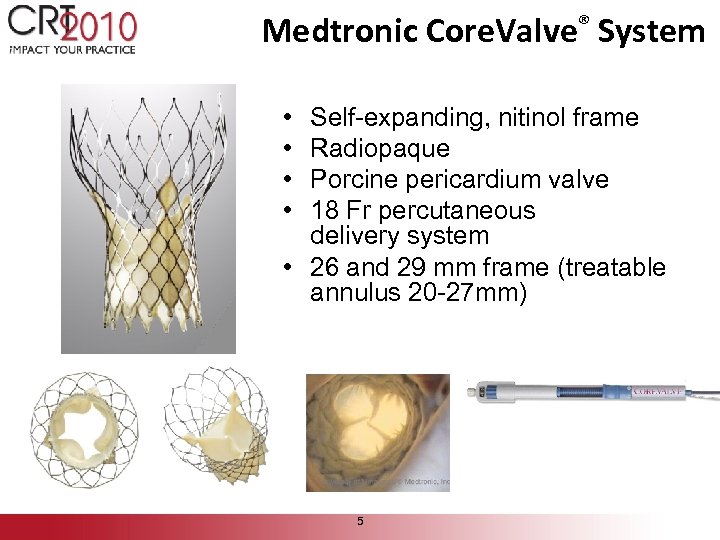 Medtronic Core. Valve® System • • Self-expanding, nitinol frame Radiopaque Porcine pericardium valve 18 Fr percutaneous delivery system • 26 and 29 mm frame (treatable annulus 20 -27 mm) 5
Medtronic Core. Valve® System • • Self-expanding, nitinol frame Radiopaque Porcine pericardium valve 18 Fr percutaneous delivery system • 26 and 29 mm frame (treatable annulus 20 -27 mm) 5
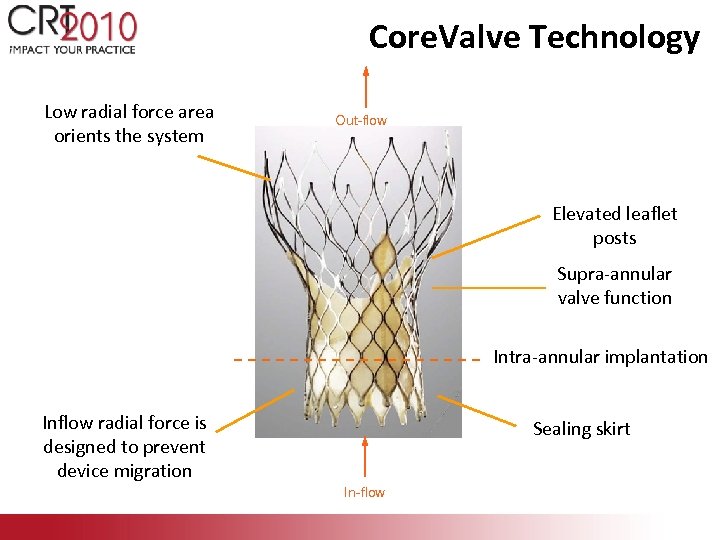 Core. Valve Technology Low radial force area orients the system Out-flow Elevated leaflet posts Supra-annular valve function Intra-annular implantation Inflow radial force is designed to prevent device migration Sealing skirt In-flow
Core. Valve Technology Low radial force area orients the system Out-flow Elevated leaflet posts Supra-annular valve function Intra-annular implantation Inflow radial force is designed to prevent device migration Sealing skirt In-flow
 Valve Assembly Tissue Preparation Valve Sub-Assembly Valve Final Assembly
Valve Assembly Tissue Preparation Valve Sub-Assembly Valve Final Assembly
 Medtronic Core. Valve® Technology
Medtronic Core. Valve® Technology
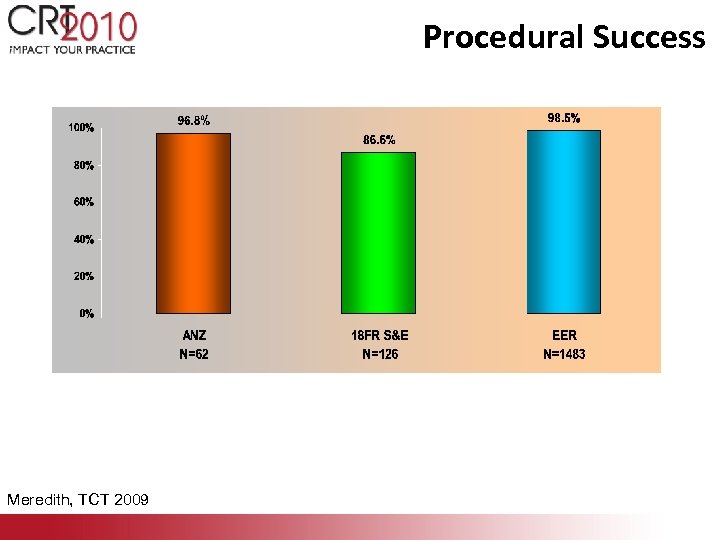 Procedural Success Meredith, TCT 2009
Procedural Success Meredith, TCT 2009
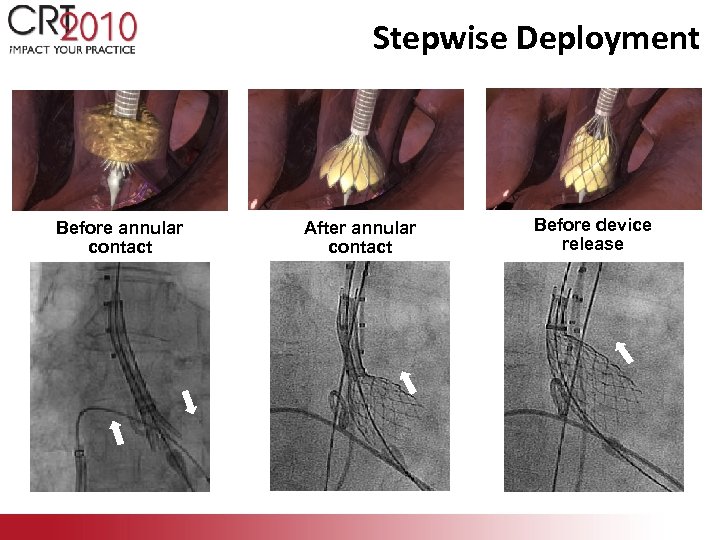 Stepwise Deployment Before annular contact After annular contact Before device release
Stepwise Deployment Before annular contact After annular contact Before device release
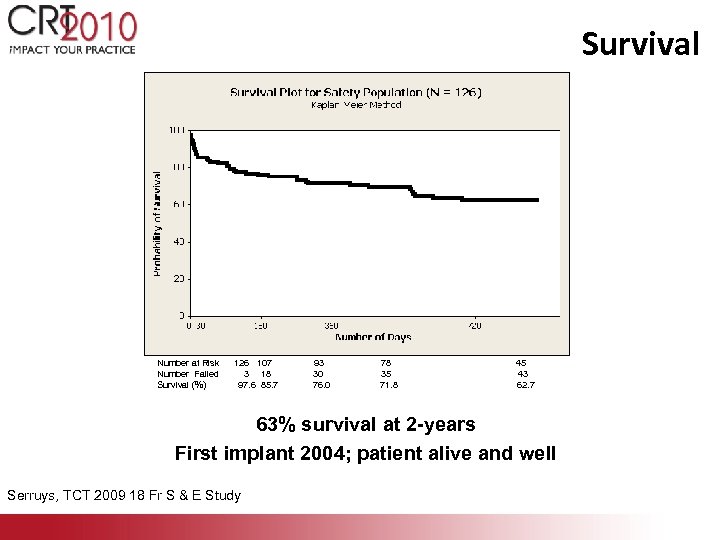 Survival Number at Risk Number Failed Survival (%) 126 107 3 18 97. 6 85. 7 93 30 76. 0 78 35 71. 8 45 43 62. 7 63% survival at 2 -years First implant 2004; patient alive and well Serruys, TCT 2009 18 Fr S & E Study
Survival Number at Risk Number Failed Survival (%) 126 107 3 18 97. 6 85. 7 93 30 76. 0 78 35 71. 8 45 43 62. 7 63% survival at 2 -years First implant 2004; patient alive and well Serruys, TCT 2009 18 Fr S & E Study
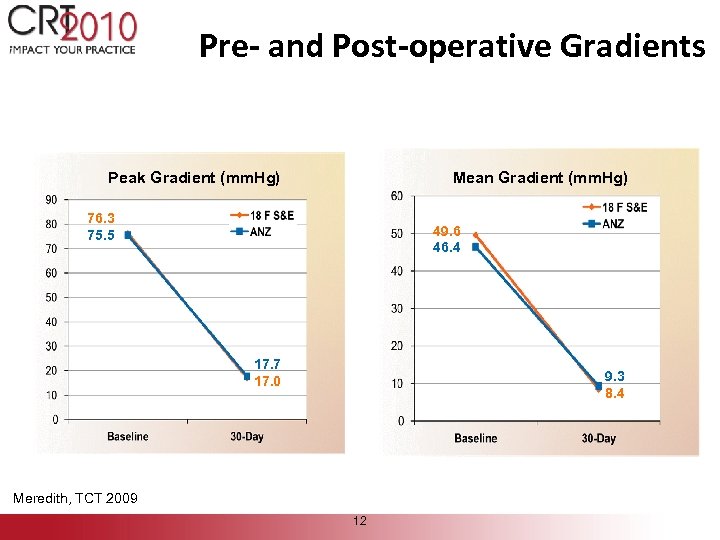 Pre- and Post-operative Gradients Peak Gradient (mm. Hg) Mean Gradient (mm. Hg) 76. 3 75. 5 49. 6 46. 4 17. 7 17. 0 9. 3 8. 4 Meredith, TCT 2009 12
Pre- and Post-operative Gradients Peak Gradient (mm. Hg) Mean Gradient (mm. Hg) 76. 3 75. 5 49. 6 46. 4 17. 7 17. 0 9. 3 8. 4 Meredith, TCT 2009 12
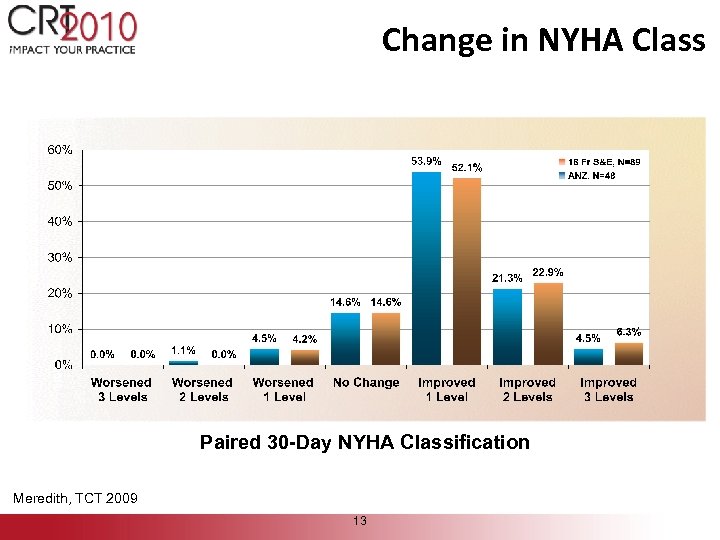 Change in NYHA Class Paired 30 -Day NYHA Classification Meredith, TCT 2009 13
Change in NYHA Class Paired 30 -Day NYHA Classification Meredith, TCT 2009 13
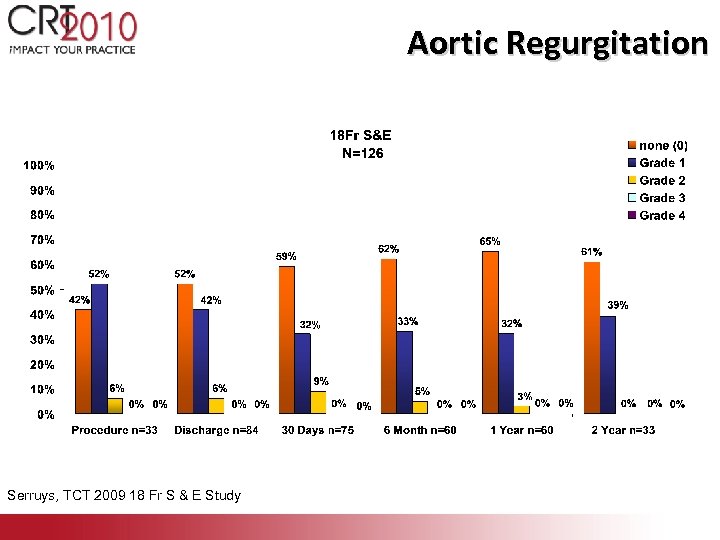 Aortic Regurgitation Serruys, TCT 2009 18 Fr S & E Study
Aortic Regurgitation Serruys, TCT 2009 18 Fr S & E Study
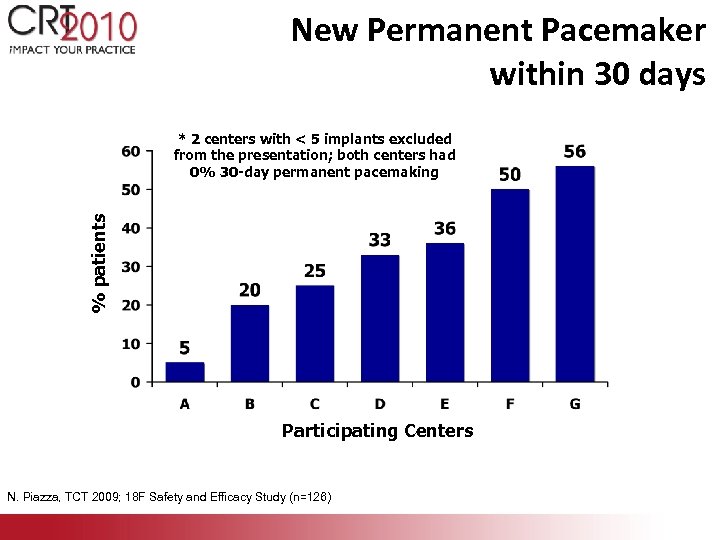 New Permanent Pacemaker within 30 days % patients * 2 centers with < 5 implants excluded from the presentation; both centers had 0% 30 -day permanent pacemaking Participating Centers N. Piazza, TCT 2009; 18 F Safety and Efficacy Study (n=126)
New Permanent Pacemaker within 30 days % patients * 2 centers with < 5 implants excluded from the presentation; both centers had 0% 30 -day permanent pacemaking Participating Centers N. Piazza, TCT 2009; 18 F Safety and Efficacy Study (n=126)
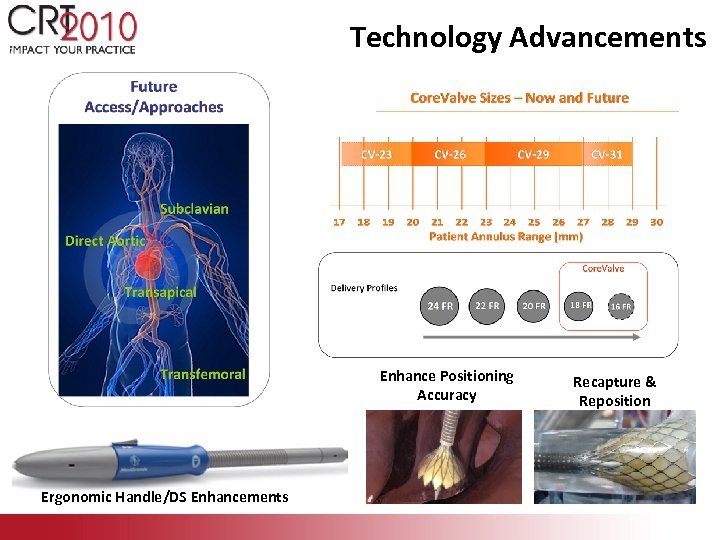 Technology Advancements Enhance Positioning Accuracy Ergonomic Handle/DS Enhancements Recapture & Reposition
Technology Advancements Enhance Positioning Accuracy Ergonomic Handle/DS Enhancements Recapture & Reposition
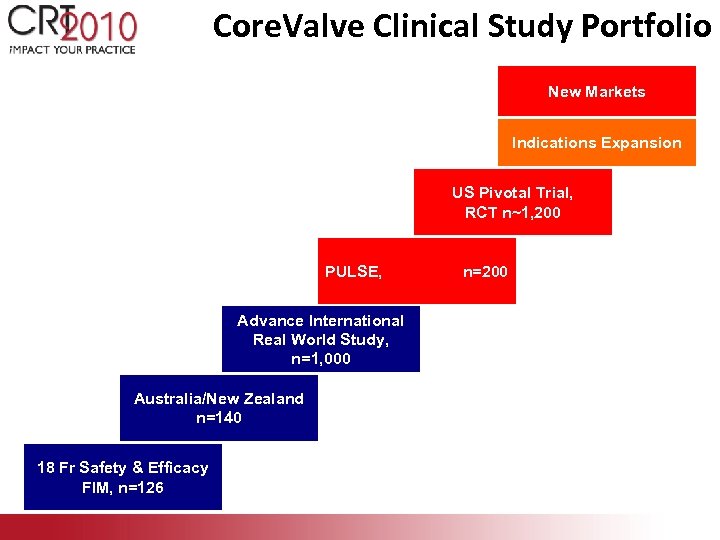 Core. Valve Clinical Study Portfolio New Markets Indications Expansion US Pivotal Trial, RCT n~1, 200 PULSE, Advance International Real World Study, n=1, 000 Australia/New Zealand n=140 18 Fr Safety & Efficacy FIM, n=126 n=200
Core. Valve Clinical Study Portfolio New Markets Indications Expansion US Pivotal Trial, RCT n~1, 200 PULSE, Advance International Real World Study, n=1, 000 Australia/New Zealand n=140 18 Fr Safety & Efficacy FIM, n=126 n=200
 Conclusions • Early experience with Core. Valve is promising • Continued growth confirms a transferable learning curve • Future technology focus will further ease of use, outcomes improvement and serving more patients who can benefit • Ongoing clinical trials are required to further prove therapy and enable growth with current and future patient populations
Conclusions • Early experience with Core. Valve is promising • Continued growth confirms a transferable learning curve • Future technology focus will further ease of use, outcomes improvement and serving more patients who can benefit • Ongoing clinical trials are required to further prove therapy and enable growth with current and future patient populations
 Thank You
Thank You