6eaa52e6d3f36d246bc3c31d05bacf72.ppt
- Количество слайдов: 27

Tech. Lab Diarrheal Diseases in Underdeveloped Countries • Worldwide Problem • More than 10, 000 deaths per day • On average, about 18 diarrheal episodes per year • Primarily in children

Tech. Lab Diarrheal Diseases in Developed Countries • Continues to be a problem • 25 million enteric infections per year • 10, 000 deaths per year in the U. S. A • Highest rate in children under 5

Tech. Lab Immunodiagnostics for Diarrheal Disease Clostridium difficile Entamoeba histolytica/dispar Giardia Cryptosporidium Clostridium perfringens Fecal Leukocytes Inflammatory Bowel/Irritable Bowel

Tech. Lab Clostridium difficile

Tech. Lab • Clostridium difficile is a gram positive, spore forming bacillus • Now known to be the cause of pseudomembranous colitis (PMC)and antibiotic associated diarrhea(AAD) • Causes almost all cases of PMC but only 25% of AAD. The most common cause of nosocomial diarrhea

Tech. Lab • Organism first described in 1935 isolated from feces of normal infants and named Bacillus difficilis because of difficulty in isolation • Only recognized as a pathogen in late 1970 s • Diarrhea is efficient method of spreading spores

Tech. Lab In early ‘ 80 s Dr Wilkins and his research team at the Anaerobe Lab at Virginia Polytechnic Institute (VPI) isolated and characterized toxin A and toxin B of Clostridium difficile. Dr Bartlett at Johns Hopkins University had recognized that C. difficile produces two toxins in 1980

Tech. Lab • Dr Wilkins’ research group also developed the first diagnostic test for the disease and developed and patented the antibodies subsequently used by all other companies for the development of their competitive EIAs. • The names Dr Wilkins and/or Dr Lyerly (the founders and co-owners of Tech. Lab Inc) are to be found in the reference list of ALL competitive companies kit inserts

Tech. Lab Clinical Terms • Antibiotic Associated Diarrhea • Antibiotic Associated Colitis • Pseudomembranous Colitis

Tech. Lab Sequence of events leading to C. difficile infection • Alteration of normal gut flora • Nosocomial infection by C. difficile • Growth and production of toxins • Tissue damage by toxin A, exacerbated by toxin B • Diarrhea and colitis due to tissue damage and influx of fluid

Tech. Lab Biological Activities of Toxin A and B of C. difficile

Tech. Lab Are there unusual strains that may be missed with an ELISA that detects only toxin A?

Tech. Lab YES! Multicenter Evaluation of the Clostridium difficile TOX A/B TEST Journal of Clinical Microbiology, Jan 1998, p. 184 -190 An Outbreak of Toxin A negative, Toxin B positive Clostridium difficile Associated Diarrhea in a Canadian Tertiary Care Hospital. Canada Communicable Disease Report, April 1999 Characterization of a Toxin A negative, Toxin B positive Strain of Clostridium difficile Responsible for a Nosocomial Outbreak of Clostridium difficile Associated Diarrhea Journal of Clinical Microbiology, July 2000, p. 2706 -2714 Pseudomembranous Colitis Caused by a Toxin A- Toxin B+ strain of Clostridium difficile Journal of Clinical Microbiology, April 2000, p. 1696 -1697

Tech. Lab • These strains have caused fatalities in Europe and North America • Actual incidence (at this time) is unknown - we believe it is around 10%, however, a study in Israel showed an incidence of more than 66% in one hospital setting

Tech. Lab • These strains do not lack toxin A - they only lack the region of the toxin A gene that codes for the antibody binding site -known as the receptor region.
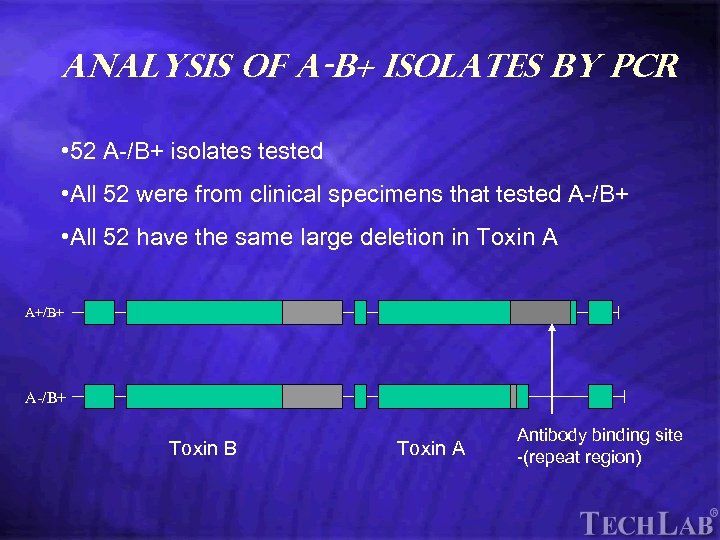
Tech. Lab Analysis of A-B+ isolates by PCR • 52 A-/B+ isolates tested • All 52 were from clinical specimens that tested A-/B+ • All 52 have the same large deletion in Toxin A A+/B+ A-/B+ Toxin B Toxin A Antibody binding site -(repeat region)

Tech. Lab All Broad Spectrum Antibiotics cause C. difficile Infections • Not due to resistance of C. difficile • Due to susceptibility of normal flora • Diarrhea sometimes can be stopped by stopping the inciting agent

Tech. Lab Epidemiology of C. difficile Disease • <3% normal carrier rate in healthy adults • 50% or higher in infants usually asymptomatic • Outbreaks occur in hospitals and medical centers

Tech. Lab Transmission of C. difficile • Primarily a nosocomial pathogen - however can be present in the community (mainly a disease of the aged) • Spore former - study showed that spores spread on floor of clean hospital room could be isolated and cultured months later • Has been isolated from hands of health care workers, library books, medical equipment, cords used to summon nurses, carpet etc.

Tech. Lab Benefits of Tech. Lab TOX A/B II TEST • Has highest correlation with tissue culture (gold standard), highly sensitive and specific • Detects both toxins • Diluent stabilizes toxin for transport or storage

Tech. Lab Benefits of Tech. Lab TOX A/B II TEST • Can be performed in as little as 40 minutes (rapid format) • Developed by the pioneers of C. difficile testing (Competitors use the Tech. Lab patented monoclonal antibody) • No indeterminates, no repeat testing • Can be used with the Tech. Lab stool preparation device to cut down on technician time and stabilize the toxin for transport

Tech. Lab Clostridium difficile Testing • Culture • Latex agglutination • Tissue Culture • ELISA

Tech. Lab Culture • Culturing is not standardized • Requires anaerobic techniques, special selective media and expertise not readily available in may labs • A toxin test has to be performed on the cultured isolates to determine whether the organisms is toxigenic or nontoxigenic • Organisms from asymptomatic patients can be isolated these patients do not require antibiotic therapy • Organisms present in very low numbers can also be cultured - these may or may not cause disease

Tech. Lab Latex Agglutination • Detects glutamate dehydrogenase, an enzyme produced by both toxigenic and nontoxigenic C. difficile and some other Clostridia • Very low sensitivity and specificity

Tech. Lab Tissue Culture (Cytotoxicity Test) • Gold standard as it was the first test available and can detect picogram quantities of toxin B (remember, toxin B is the most potent cytotoxin known to man - toxin A is also cytotoxic but at the dilutions used in this test it has no cytotoxic effect). • Test requires that sample be diluted, centrifuged, filter sterilized and dispensed into the wells of a microtiter plate containing a confluent sheet of mammalian cells • Labor intensive and takes 24 -48 hours for a result • Requires considerable expertise and specialized equipment

Tech. Lab ELISA Clostridium difficile Tests that are toxin A specific • Tech. Lab TOX A TEST • Meridian Premier Toxin A • Bartels Prima (recently purchased by Trinity) • Alexon Prospec. T Toxin A • Vidas CDA • Becton Dickinson Culturette CD • Biosite Triage • Becton Dickinson Color. Pac Toxin A

Tech. Lab EIA TESTS that detect both toxin A and toxin B • Tech. Lab TOX A/B TEST • Meridian Premier A+B
6eaa52e6d3f36d246bc3c31d05bacf72.ppt