bc516b60d0ab88ed00bdb584b91469e8.ppt
- Количество слайдов: 12

TB/HIV Core Group meeting Geneva, November 3 -4, 2009 Assisting countries to implement the WHA resolution on MDR-TB The Global Laboratory Initiative Karin Weyer, WHO-STB, GLI secretariat John Ridderhof, CDC, GLI chair

Global Laboratory Initiative Platform of coordination and communication, providing the required infrastructure, focused on TB laboratory strengthening, in the areas of: • Global policy guidance (norms, standards, best practices) • Laboratory capacity development • Interface with other laboratory networks, enabling integration • Standardised laboratory quality assurance • Coordination of technical assistance • Effective knowledge sharing • Advocacy and resource mobilisation
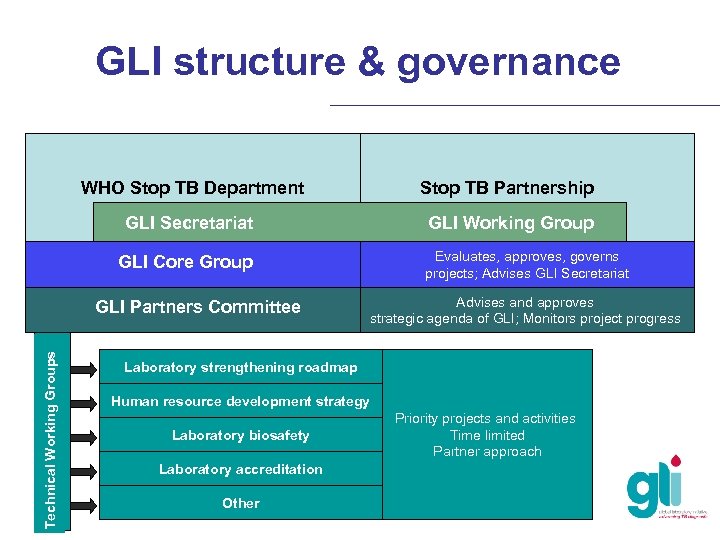
GLI structure & governance WHO Stop TB Department GLI Secretariat Stop TB Partnership GLI Working Group Evaluates, approves, governs projects; Advises GLI Secretariat GLI Core Group Technical Working Groups GLI Partners Committee Advises and approves strategic agenda of GLI; Monitors project progress Laboratory strengthening roadmap Human resource development strategy Laboratory biosafety Laboratory accreditation Other Priority projects and activities Time limited Partner approach
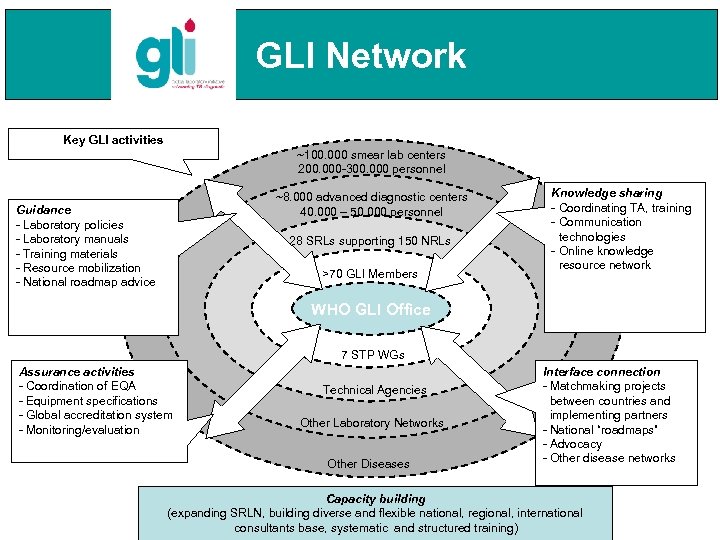
GLI Network Key GLI activities ~100. 000 smear lab centers 200. 000 -300. 000 personnel ~8. 000 advanced diagnostic centers 40. 000 – 50. 000 personnel Guidance - Laboratory policies - Laboratory manuals - Training materials - Resource mobilization - National roadmap advice 28 SRLs supporting 150 NRLs >70 GLI Members Knowledge sharing - Coordinating TA, training - Communication technologies - Online knowledge resource network WHO GLI Office 7 STP WGs Assurance activities - Coordination of EQA - Equipment specifications - Global accreditation system - Monitoring/evaluation Technical Agencies Other Laboratory Networks Other Diseases Interface connection - Matchmaking projects between countries and implementing partners - National “roadmaps” - Advocacy - Other disease networks Capacity building (expanding SRLN, building diverse and flexible national, regional, international consultants base, systematic and structured training)

GLI strategic priorities • Accelerating evidence-based policy development on diagnostics and laboratory practices • Promoting a structured framework/roadmap for TB laboratory strengthening within the context of national laboratory plans at country level • Developing a comprehensive set of tools, norms and standards based on international standards and bestpractice • Advancing laboratory strengthening through global, regional and local partnerships • Developing multi-level laboratory human resource strategies to address the capacity crisis • Accelerating new diagnostics into countries

• Accelerated uptake of new MDR-TB diagnostics in 27 countries, 2009 - 2013 (additional 15 planned by 2012) • State-of the-art commodities (instruments, tests, reagents) funded by UNITAID • Leverage other local partners to address non-commodity components (infrastructure, training, etc. ) • Long-term mentoring and TA: in-country hands-on support to optimise technology and knowledge transfer, closely linked to capacity building • Full ownership of MOH, NTP, Laboratory • Integrated laboratory approach (notably TB and HIV) • Adjustment based on growing evidence ('learning by doing')

Funding for essential instruments, reagents, supplies • Coordinate and manage • Policies, norms • Negotiate with procurement and international partners to ensure delivery standards lowest prices • With FIND, engage • Participate in lab • Ensure customer industry to ensure assessments support in place • Provide long-term, on Share know-how from affordability and • sustained price -site monitoring product development decreases • Develop indicators process and tools for M&E • Provide long-term, on • Collaborate with WHO Pre-qualification to -site mentoring for include diagnostics technology transfer
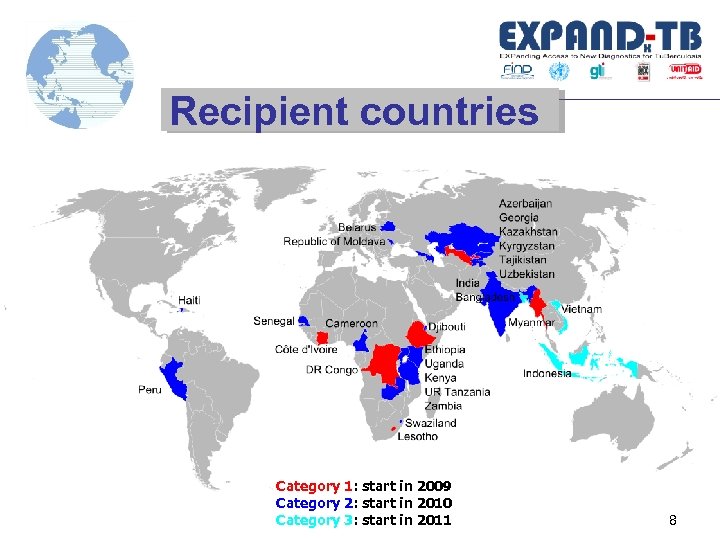
Recipient countries Category 1: start in 2009 Category 2: start in 2010 Category 3: start in 2011 8
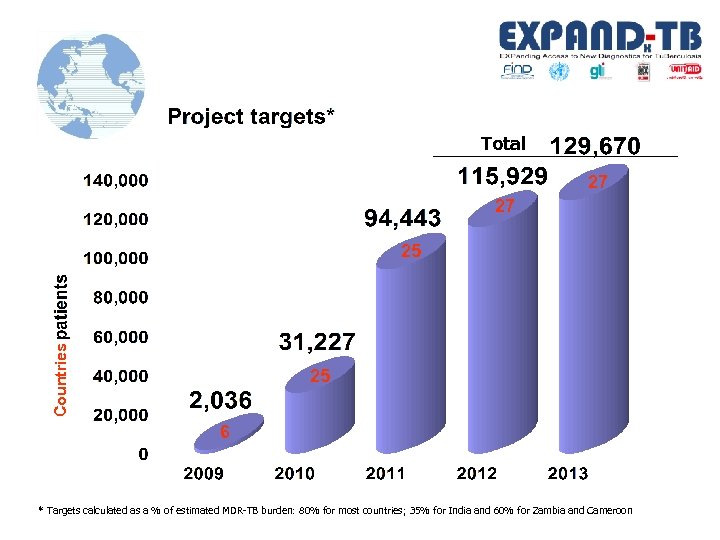
Total 27 27 Countries 25 25 6 * Targets calculated as a % of estimated MDR-TB burden: 80% for most countries; 35% for India and 60% for Zambia and Cameroon

Country partners • • • • • American Society for Microbiology Centers for Disease Control and Prevention Johns Hopkins University GHESKIO Global AIDS Programme GTZ Kf. W Bank, Germany KNCV Ministeries of Health, through NTPs Partners in Health PEPFAR OGAC Project Hope ICRC The Global Fund The Union USAID WHO Supranational Reference Laboratory Network …

Future scale-up • EXPAND-TB – 15 additional countries by 2012 • Other low-income, high-endemic countries – Adoption of EXPAND-TB model with other funding sources and other laboratory initiatives (eg. HIV, Malaria, H 1 N 1) • Middle-income, high-endemic countries – Two-pronged approach (manufacturers and laboratory networks) in innovative PPM models • Acceleration of GLI human resource development plan – – At least 50 global consultants At least 50 long-term, on-site mentors Up to 20, 000 laboratory technicians with advance skills MOH responsibilities in revised human resource plans & strategies

Global targets met for MDR-TB patients diagnosed and provided with appropriate treatment under international standards of care Patient care Drugs Diagnostics
bc516b60d0ab88ed00bdb584b91469e8.ppt